Latest Developments in Insect Sex Pheromone Research and Its Application in Agricultural Pest Management
Abstract
Simple Summary
Abstract
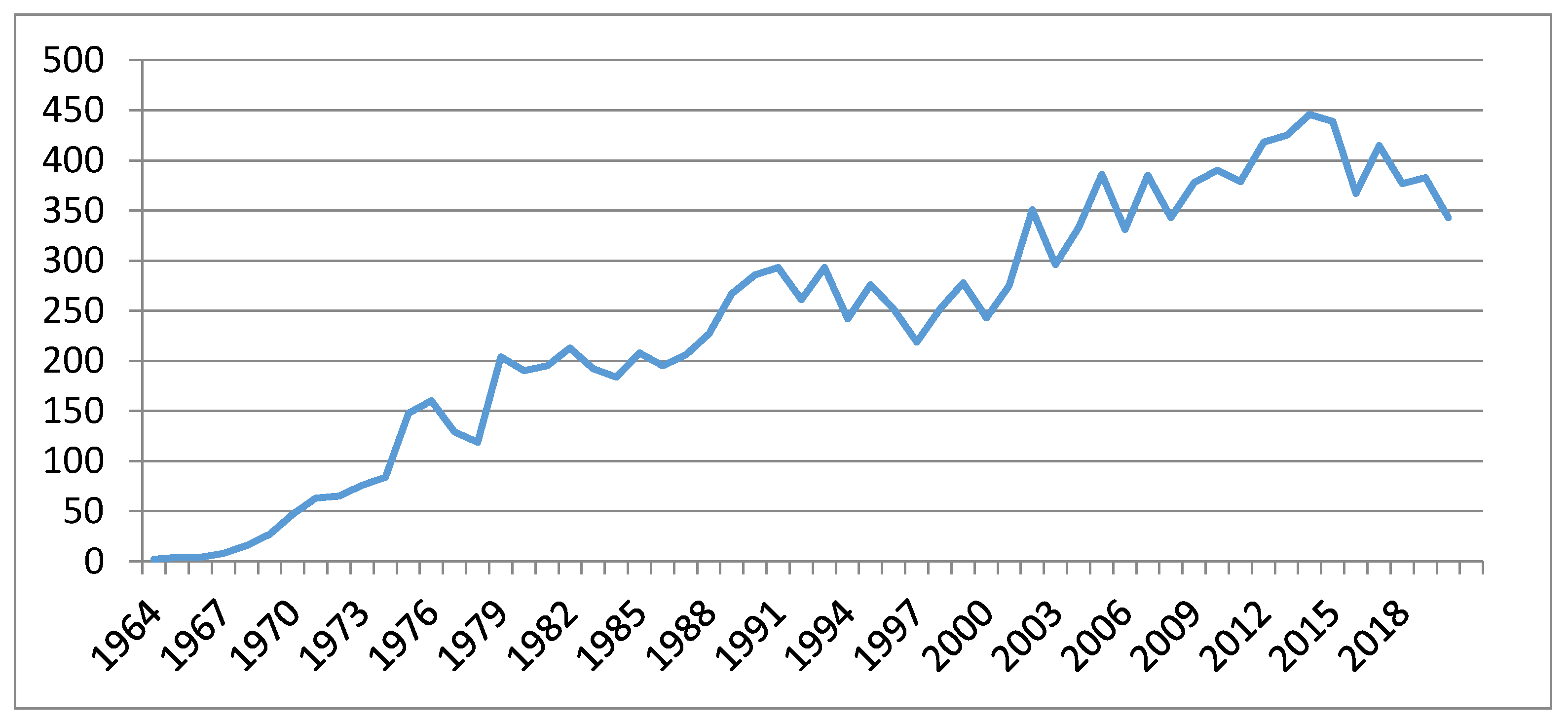
1. Introduction
2. Sex Pheromone Biosynthesis
3. Mechanisms of Insect Sex Pheromone Perception
4. Evolutionary Aspects of Olfactory Receptors
5. Sex Pheromone Autodetection
6. Resistance of Insects to Sex Pheromones
7. Application of Insect Sex Pheromones
7.1. Interactions between Pheromones and Insects Biological Control Agents
7.1.1. Pheromones vs Entomopathogenic Fungi
7.1.2. Pheromones and Bacteria
7.2. Monitoring
7.3. Mass Trapping
7.4. Mating Disruption
7.5. Push-Pull Strategy
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Seybold, S.J.; Bentz, B.J.; Fettig, C.J.; Lundquist, J.E.; Progar, R.A.; Gillette, N.E. Management of western North American bark beetles with semiochemicals. Annu. Rev. Entomol. 2018, 63, 407–432. [Google Scholar] [CrossRef] [PubMed]
- Smart, L.; Aradottir, G.; Bruce, T. Role of semiochemicals in integrated pest management. In Integrated Pest Management; Abrol, D.P., Ed.; Academic Press: Amsterdam, The Netherlands, 2014; pp. 93–109. [Google Scholar] [CrossRef]
- Butenandt, V.A. Uber den sexsual-lockstoff des seidenspinners Bombyx mori. Reindarstellung und Konstitution. Z. Naturforsch. B 1959, 14, 283. [Google Scholar]
- Karlson, P.; Lüscher, M. ‘Pheromones’: A new term for a class of biologically active substances. Nature 1959, 183, 55–56. [Google Scholar] [CrossRef]
- Petkevicius, K.; Löfstedt, C.; Borodina, I. Insect sex pheromone production in yeasts and plants. Curr. Opin. Biotechnol. 2020, 65, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.C. Pheromones and other semiochemicals for monitoring rare and endangered species. J. Chem. Ecol. 2016, 42, 853–868. [Google Scholar] [CrossRef] [PubMed]
- Tewari, S.; Leskey, T.C.; Nielsen, A.L.; Piñero, J.C.; Rodriguez-Saona, C.R. Use of pheromones in insect pest management, with special attention to weevil pheromones. In Integrated Pest Management; Abrol, D.P., Ed.; Academic Press: Amsterdam, The Netherlands, 2014; pp. 141–168. [Google Scholar] [CrossRef]
- Yew, J.Y.; Chung, H. Insect pheromones: An overview of function, form, and discovery. Prog. Lipid Res. 2015, 59, 88–105. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.V.; Guerrero, A. New pheromones and insect control strategies. Vitam. Horm. 2010, 83, 493–519. [Google Scholar] [CrossRef] [PubMed]
- Trematerra, P. Advances in the use of pheromones for stored-product protection. J. Pest Sci. 2012, 85, 285–299. [Google Scholar] [CrossRef]
- Witzgall, P.; Kirsch, P.; Cork, A. Sex pheromones and their impact on pest management. J. Chem. Ecol. 2010, 36, 80–100. [Google Scholar] [CrossRef] [PubMed]
- Hostachy, C.; Couzi, P.; Portemer, G.; Hanafi-Portier, M.; Murmu, M.; Deisig, N.; Dacher, M. Exposure to conspecific and heterospecific sex-pheromones modulates gustatory habituation in the moth Agrotis ipsilon. Front. Physiol. 2019, 10, 1518. [Google Scholar] [CrossRef] [PubMed]
- Molander, M.A.; Eriksson, B.; Winde, I.B.; Zou, Y.; Millar, J.G.; Larsson, M.C. The aggregation-sex pheromones of the cerambycid beetles Anaglyptus mysticus and Xylotrechus antilope ssp. antilope: New model species for insect conservation through pheromone-based monitoring. Chemoecology 2019, 29, 111–124. [Google Scholar] [CrossRef]
- El-Sayed, A.M. The Pherobase: Database of Pheromones and Semiochemicals. 2021. Available online: https://www.pherobase.com (accessed on 19 January 2021).
- Ma, T.; Liu, Z.T.; Zhang, Y.Y.; Sun, Z.H.; Li, Y.Z.; Wen, X.J.; Chen, X.Y. Electrophysiological and behavioral responses of Diaphania glauculalis males to female sex pheromone. Environ. Sci. Pollut. Res. Int. 2015, 22, 15046–15054. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Gibb, A.R.; Mitchell, V.J.; Lee-Anne, M.; Manning, L.-A.M.; Revell, J.; Thistleton, B.; Suckling, D.M. Identification of the sex pheromone of Conogethes pluto: A pest of Alpinia. Chemoecology 2013, 23, 93–101. [Google Scholar] [CrossRef]
- Peng, C.L.; Gu, P.; Li, J.; Chen, Q.Y.; Feng, C.H.; Luo, H.H.; Du, Y.J. Identification and field bioassay of the sex pheromone of Trichophysetis cretacea (Lepidoptera: Crambidae). J. Econ. Entomol. 2012, 105, 1566–1572. [Google Scholar] [CrossRef]
- Symmes, E.J.; Dewhirst, S.Y.; Birkett, M.A.; Campbell, C.A.; Chamberlain, K.; Pickett, J.A.; Zalom, F.G. The sex pheromones of mealy plum (Hyalopterus pruni) and leaf-curl plum (Brachycaudus helichrysi) aphids: Identification and field trapping of male and gynoparous aphids in prune orchards. J. Chem. Ecol. 2012, 38, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Levi-Zada, A.; Ben-Yehuda, S.; Dunkelblum, E.; Gindin, G.; Fefer, D.; Protasov, A.; Kuznetsowa, T.; Manulis-Sasson, S.; Mendel, Z. Identification and field bioassays of the sex pheromone of the yellow-legged clearwing Synanthedon vespiformis (Lepidoptera: Sesiidae). Chemoecology 2011, 21, 227–233. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Mitchell, V.J.; Manning, L.A.; Suckling, D.M. New sex pheromone blend for the lightbrown apple moth, Epiphyas postvittana. J. Chem. Ecol. 2011, 37, 640–646. [Google Scholar] [CrossRef]
- Acín, P.; Rosell, G.; Guerrero, A.; Quero, C. Sex pheromone of the Spanish population of the beet armyworm Spodoptera exigua. J. Chem. Ecol. 2010, 36, 778–786. [Google Scholar] [CrossRef]
- Eliyahu, D.; Nojima, S.; Santangelo, R.G.; Carpenter, S.; Webster, F.X.; Kiemle, D.J.; Gemeno, C.; Leal, W.S.; Schal, C. Unusual macrocyclic lactone sex pheromone of Parcoblatta lata, a primary food source of the endangered red-cockaded woodpecker. Proc. Natl. Acad. Sci. USA 2012, 109, E490–E496. [Google Scholar] [CrossRef]
- Guerrero, A.; Ramos, V.E.; López, S.; Alvarez, J.M.; Domínguez, A.; Coca-Abia, M.M.; Bosch, M.P.; Quero, C. Enantioselective synthesis and activity of all diastereoisomers of (E)-phytal, a pheromone component of the Moroccan locust, Dociostaurus maroccanus. J. Agric. Food Chem. 2019, 67, 72–80. [Google Scholar] [CrossRef]
- Vacas, S.; Navarro, I.; Marzo, J.; Navarro-Llopis, V.; Primo, J. Sex pheromone of the invasive mealybug citrus pest, Delottococcus aberiae (Hemiptera: Pseudococcidae). A new monoterpenoid with a necrodane skeleton. J. Agric. Food Chem. 2019, 67, 9441–9449. [Google Scholar] [CrossRef]
- Bouwer, M.C.; Slippers, B.; Degefu, D.; Wingfield, M.J.; Lawson, S.; Rohwer, E.R. Identification of the sex pheromone of the tree infesting cossid moth Coryphodema tristis (Lepidoptera: Cossidae). PLoS ONE 2015, 10, e0118575. [Google Scholar] [CrossRef]
- Būda, V.; Blažytė-Čereškienė, L.; Radžiutė, S.; Apšegaitė, V.; Stamm, P.; Schulz, S.; Aleknavičius, D.; Mozūraitis, R. Male-produced (−)-δ-heptalactone, pheromone of fruit fly Rhagoletis batava (Diptera: Tephritidae), a sea buckthorn berries pest. Insects 2020, 11, 138. [Google Scholar] [CrossRef]
- Gago, R.; Allyson, J.D.; McElfresh, J.S.; Haynes, K.F.; McKenney, J.; Guerrero, A.; Millar, J. A tetraene aldehyde as the major sex pheromone component of the promethea moth (Callosamia promethea (Drury)). J. Chem. Ecol. 2013, 39, 1263–1272. [Google Scholar] [CrossRef]
- Weiss, I.; Rössler, T.; Hofferberth, J.; Brummer, M.; Ruther, J.; Stökl, J. A nonspecific defensive compound evolves into a competition avoidance cue and a female sex pheromone. Nat. Commun. 2013, 4, 2767. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.B.; Liu, K.W.; Wang, H.B.; Zhang, S.F.; Zhang, Z. Identification and behavioral evaluation of sex pheromone components of the Chinese pine caterpillar moth, Dendrolimus tabulaeformis. PLoS ONE 2012, 7, e33381. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, H.; Yamamoto, M.; Yan, Q.; Naka, H.; Suzuki, T.; Ando, T. Identification of the sex pheromone secreted by a nettle moth, Monema flavescens, using gas chromatography/fourier transform infrared spectroscopy. J. Chem. Ecol. 2013, 39, 350–357. [Google Scholar] [CrossRef]
- De Oliveira, M.W.; Borges, M.; Andrade, C.K.; Laumann, R.A.; Barrigossi, J.A.; Blassioli-Moraes, M.C. Zingiberenol, (1R,4R,1’S)-4-(1′,5′-dimethylhex-4′-enyl)-1-methylcyclohex-2-en-1-ol, identified as the sex pheromone produced by males of the rice stink bug Oebalus poecilus (Heteroptera: Pentatomidae). J. Agric. Food Chem. 2013, 61, 7777–7785. [Google Scholar] [CrossRef] [PubMed]
- Blassioli-Moraes, M.C.; Khrimian, A.; Michereff, M.F.F.; Magalhães, D.M.; Hickel, E.; de Freitas, T.F.S.; Barrigossi, J.A.F.; Laumann, R.A.; Silva, A.T.; Guggilapu, S.D.; et al. Male-produced sex pheromone of Tibraca limbativentris revisited: Absolute configurations of zingiberenol stereoisomers and their influence on chemotaxis behavior of conspecific females. J. Chem. Ecol. 2020, 46, 1–9. [Google Scholar] [CrossRef]
- Millar, J.G.; McElfresh, J.S.; Romero, C.; Vila, M.; Marí-Mena, N.; Lopez-Vaamonde, C. Identification of the sex pheromone of a protected species, the Spanish moon moth Graellsia isabellae. J. Chem. Ecol. 2010, 36, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Welter, S.; Pickel, C.; Millar, J.; Cave, F.; Van Steenwyk, R.; Dunley, J. Pheromone mating disruption offers selective management options for key pests. Calif. Agric. 2005, 59, 16–22. [Google Scholar] [CrossRef]
- Löfstedt, C.; Wahlberg, N.; Millar, J.M. Evolutionary patterns of pheromone diversity in Lepidoptera. In Pheromone Communication in Moths: Evolution, Behavior and Application; Allison, J.D., Cardé, R.T., Eds.; University of California Press: Berkeley, CA, USA, 2016; pp. 43–78. [Google Scholar]
- Ando, T.; Inomata, S.I.; Yamamoto, M. Lepidopteran sex pheromones. In The Chemistry of Pheromones and Other Semiochemicals I, Topics in Current Chemistry; Schulz, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; Volume 239, pp. 51–96. [Google Scholar]
- Naka, H.; Fujii, T. Chemical divergences in the sex pheromone communication systems in moths. In Insect Sex Pheromone Research and Beyond; Ishikawa, Y., Ed.; Springer: Singapore, 2020; pp. 3–17. [Google Scholar] [CrossRef]
- Allison, J.D.; Cardé, R.T. Pheromone Communication in Moths: Evolution, Behavior, and Application, 1st ed.; University of California Press: Oakland, CA, USA, 2016; p. 416. [Google Scholar]
- Jurenka, R. Insect pheromone biosynthesis. Top. Curr. Chem. 2004, 239, 97–132. [Google Scholar] [PubMed]
- Jurenka, R. Regulation of pheromone biosynthesis in moths. Curr. Opin. Insect Sci. 2017, 24, 29–35. [Google Scholar] [CrossRef]
- Tillman, J.A.; Seybold, S.J.; Jurenka, R.A.; Blomquist, G.J. Insect pheromones--an overview of biosynthesis and endocrine regulation. Insect Biochem. Mol. Biol. 1999, 29, 481–514. [Google Scholar] [CrossRef]
- Blomquist, G.J.; Jurenka, R.; Schal, C.; Tittiger, C. Pheromone production: Biochemistry and molecular biology. In Insect Endocrinology, Gilbert, L., Ed.; Academic London: London, UK, 2012. [Google Scholar] [CrossRef]
- Park, H.Y.; Kim, M.S.; Paek, A.; Jeong, S.E.; Knipple, D.C. An abundant acyl-CoA (Delta9) desaturase transcript in pheromone glands of the cabbage moth, Mamestra brassicae, encodes a catalytically inactive protein. Insect Biochem. Mol. Biol. 2008, 38, 581–595. [Google Scholar] [CrossRef]
- Ding, B.J.; Löfstedt, C. Analysis of the Agrotis segetum pheromone gland transcriptome in the light of sex pheromone biosynthesis. BMC Genom. 2015, 16, 711. [Google Scholar] [CrossRef]
- Moto, K.I.; Suzuki, M.G.; Hull, J.J.; Kurata, R.; Takahashi, S.; Yamamoto, M.; Okano, K.; Imai, K.; Ando, T.; Matsumoto, S. Involvement of a bifunctional fatty-acyl desaturase in the biosynthesis of the silkmoth, Bombyx mori, sex pheromone. Proc. Natl. Acad. Sci. USA 2004, 101, 8631–8636. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.J.; Hofvander, P.; Wang, H.L.; Durrett, T.P.; Stymne, S.; Löfstedt, C. A plant factory for moth pheromone production. Nat. Commun. 2014, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Shen, G.; Guo, D.; Wang, S.; Ma, X.; Xiao, H.; Liu, J.; Zhang, Z.; Liu, Y.; Zhang, Y.; et al. InsectBase: A resource for insect genomes and transcriptomes. Nucleic Acids Res. 2016, 44, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Tupec, M.; Buček, A.; Valterová, I.; Pichová, I. Biotechnological potential of insect fatty acid-modifying enzymes. Z. Naturforsch. C 2017, 72, 387–403. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, P.W.; Marsella-Herrick, P.; Rosenfield, C.L.; Knipple, D.C.; Roelofs, W. Cloning and functional expression of a cDNA encoding a metabolic acyl-CoA delta 9-desaturase of the cabbage looper moth, Trichoplusia ni. Insect Biochem. Mol. Biol. 1999, 29, 435–443. [Google Scholar] [CrossRef]
- Zhang, Y.N.; Zhang, X.Q.; Zhu, G.H.; Zheng, M.Y.; Yan, Q.; Zhu, X.Y.; Xu, J.W.; Zhang, Y.Y.; He, P.; Sun, L.; et al. A Δ9 desaturase (SlitDes11) is associated with the biosynthesis of ester sex pheromone components in Spodoptera litura. Pest. Biochem. Physiol. 2019, 156, 152–159. [Google Scholar] [CrossRef]
- Fujii, T.; Yasukochi, Y.; Rong, Y.; Matsuo, T.; Ishikawa, Y. Multiple Δ11-desaturase genes selectively used for sex pheromone biosynthesis are conserved in Ostrinia moth genomes. Insect Biochem. Mol. Biol. 2015, 61, 62–68. [Google Scholar] [CrossRef]
- Hagström, Å.K.; Albre, J.; Tooman, L.K.; Thirmawithana, A.H.; Corcoran, J.; Löfstedt, C.; Newcomb, R.D. A novel fatty acyl desaturase from the pheromone glands of Ctenopseustis obliquana and C. herana with specific Z5-desaturase activity on myristic acid. J. Chem. Ecol. 2014, 40, 63–70. [Google Scholar] [CrossRef]
- Wang, H.L.; Liénard, M.A.; Zhao, C.H.; Wang, C.Z.; Löfstedt, C. Neofunctionalization in an ancestral insect desaturase lineage led to rare Δ6 pheromone signals in the Chinese tussah silkworm. Insect Biochem. Mol. Biol. 2010, 40, 742–751. [Google Scholar] [CrossRef] [PubMed]
- Liénard, M.A.; Lassance, J.M.; Wang, H.L.; Zhao, C.H.; Piskur, J.; Johansson, T.; Löfstedt, C. Elucidation of the sex pheromone biosynthesis producing 5,7-dodecadienes in Dendrolimus punctatus (Lepidoptera: Lasiocampidae) reveals Delta 11- and Delta 9-desaturases with unusual catalytic properties. Insect Biochem. Mol. Biol. 2010, 40, 440–452. [Google Scholar] [CrossRef]
- Hao, G.; Liu, W.; O’Connor, M.; Roelofs, W. Acyl-CoA Z9- and Z10-desaturase genes from a New Zealand leafroller moth species, Planotortrix octo. Insect Biochem. Mol. Biol. 2002, 32, 961–966. [Google Scholar] [CrossRef]
- Roelofs, W.L.; Rooney, A.P. Molecular genetics and evolution of pheromone biosynthesis in Lepidoptera. Proc. Natl. Acad. Sci. USA 2003, 100, 9179–9184. [Google Scholar] [CrossRef]
- Ding, B.J.; Liénard, M.A.; Wang, H.L.; Zhao, C.H.; Löfstedt, C. Terminal fatty-acyl-CoA desaturase involved in sex pheromone biosynthesis in the winter moth (Operophtera brumata). Insect Biochem. Mol. Biol. 2011, 41, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.H.; Zhang, Y.N.; Ding, B.J.; Wang, H.L.; Löfstedt, C. Multi-functional desaturases in two Spodoptera moths with Δ11 and Δ12 desaturation activities. J. Chem. Ecol. 2019, 45, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Serra, M.; Piña, B.; Abad, J.L.; Camps, F.; Fabriàs, G. A multifunctional desaturase involved in the biosynthesis of the processionary moth sex pheromone. Proc. Natl. Acad. Sci. USA 2007, 104, 16444–16449. [Google Scholar] [CrossRef]
- Zhang, Y.N.; Zhang, L.W.; Chen, D.S.; Sun, L.; Li, Z.Q.; Ye, Z.F.; Zheng, M.Y.; Li, J.B.; Zhu, X.Y. Molecular identification of differential expression genes associated with sex pheromone biosynthesis in Spodoptera exigua. Mol. Genet. Genom. 2017, 292, 795–809. [Google Scholar] [CrossRef]
- Bai, Y.; McCoy, J.G.; Levin, E.J.; Sobrado, P.; Rajashankar, K.R.; Fox, B.G.; Zhou, M. X-ray structure of a mammalian stearoyl-CoA desaturase. Nature 2015, 524, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, S. Molecular mechanisms underlying sex pheromone production in moths. Biosci. Biotechnol. Biochem. 2010, 74, 223–231. [Google Scholar] [CrossRef]
- Hagström, A.K.; Liénard, M.A.; Groot, A.T.; Hedenström, E.; Löfstedt, C. Semi-selective fatty acyl reductases from four heliothine moths influence the specific pheromone composition. PLoS ONE 2012, 7, e37230. [Google Scholar] [CrossRef] [PubMed]
- Antony, B.; Fujii, T.; Moto, K.; Matsumoto, S.; Fukuzawa, M.; Nakano, R.; Tatsuki, S.; Ishikawa, Y. Pheromone-gland-specific fatty-acyl reductase in the adzuki bean borer, Ostrinia scapulalis (Lepidoptera: Crambidae). Insect Biochem. Mol. Biol. 2009, 39, 90–95. [Google Scholar] [CrossRef]
- Antony, B.; Ding, B.J.; Moto, K.; Aldosari, S.A.; Aldawood, A.S. Two fatty acyl reductases involved in moth pheromone biosynthesis. Sci. Rep. 2016, 6, 29927. [Google Scholar] [CrossRef] [PubMed]
- Liénard, M.A.; Hagstroem, A.K.; Lassance, J.-M.; Löfstedt, C. Evolution of multicomponent pheromone signals in small ermine moths involves a single fatty-acyl reductase gene. Proc. Natl. Acad. Sci. USA 2010, 107, 10955–10960. [Google Scholar] [CrossRef] [PubMed]
- Günther, C.S.; Chervin, C.; Marsh, K.B.; Newcomb, R.D.; Souleyre, E.J. Characterisation of two alcohol acyltransferases from kiwifruit (Actinidia spp.) reveals distinct substrate preferences. Phytochemistry 2011, 72, 700–710. [Google Scholar] [CrossRef]
- Shalit, M.; Katzir, N.; Tadmor, Y.; Larkov, O.; Burger, Y.; Shalekhet, F.; Lastochkin, E.; Ravid, U.; Amar, O.; Edelstein, M.; et al. Acetyl-CoA: Alcohol acetyltransferase activity and aroma formation in ripening melon fruits. J. Agric. Food Chem. 2001, 49, 794–799. [Google Scholar] [CrossRef]
- Ding, B.J.; Lager, I.; Bansal, S.; Durrett, T.P.; Stymne, S.; Löfstedt, C. The yeast ATF1 acetyltransferase efficiently acetylates insect pheromone alcohols: Implications for the biological production of moth pheromones. Lipids 2016, 51, 469–475. [Google Scholar] [CrossRef]
- Yang, C.Y.; Lee, S.; Choi, K.S.; Jeon, H.Y.; Boo, K.S. Sex pheromone production and response in Korean populations of the diamondback moth, Plutella xylostella. Entomol. Exp. Appl. 2007, 124, 293–298. [Google Scholar] [CrossRef]
- Chen, D.S.; Dai, J.Q.; Han, S.C. Identification of the pheromone biosynthesis genes from the sex pheromone gland transcriptome of the diamondback moth, Plutella xylostella. Sci. Rep. 2017, 7, 16255. [Google Scholar] [CrossRef] [PubMed]
- Vogel, H.; Heidel, A.J.; Heckel, D.G.; Groot, A.T. Transcriptome analysis of the sex pheromone gland of the noctuid moth Heliothis virescens. BMC Genom. 2010, 11, 29. [Google Scholar] [CrossRef]
- Hansson, B.S.; Stensmyr, M.C. Evolution of insect olfaction. Neuron 2011, 72, 698–711. [Google Scholar] [CrossRef] [PubMed]
- Hansson, B.S. Insect Olfaction; Springer: Berlin/Heidelberg, Germany, 2013; p. 458. [Google Scholar] [CrossRef]
- Leal, W.S. Odorant reception in insects: Roles of receptors, binding proteins, and degrading enzymes. Annu. Rev. Entomol. 2013, 58, 373–391. [Google Scholar] [CrossRef]
- Cardé, R.T.; Willis, M.A. Navigational strategies used by insects to find distant, wind-borne sources of odor. J. Chem. Ecol. 2008, 34, 854–866. [Google Scholar] [CrossRef] [PubMed]
- De Bruyne, M.; Baker, T.C. Odor detection in insects: Volatile codes. J. Chem. Ecol. 2008, 34, 882–897. [Google Scholar] [CrossRef]
- Fleischer, J.; Pregitzer, P.; Breer, H.; Krieger, J. Access to the odor world: Olfactory receptors and their role for signal transduction in insects. Cell. Mol. Life Sci. 2018, 75, 485–508. [Google Scholar] [CrossRef] [PubMed]
- Montagné, N.; de Fouchier, A.; Newcomb, R.D.; Jacquin-Joly, E. Advances in the identification and characterization of olfactory receptors in insects. Prog. Mol. Biol. Transl. Sci. 2015, 130, 55–80. [Google Scholar] [CrossRef]
- Jeanne, J.M.; Fişek, M.; Wilson, R.I. The organization of projections from olfactory glomeruli onto higher-order neurons. Neuron 2018, 98, 1198–1213.e6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; McBride, C.S. Evolution of olfactory circuits in insects. J. Comp. Physiol. A 2020, 206, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Fialho, M.D.C.Q.; Guss-Matiello, C.P.; Zanuncio, J.C.; Campos, L.A.O.; Serrão, J.E. A comparative study of the antennal sensilla in corbiculate bees. J. Apic. Res. 2014, 53, 392–403. [Google Scholar] [CrossRef]
- Tsang, T.K.; Bushong, E.A.; Boassa, D.; Hu, J.; Romoli, B.; Phan, S.; Dulcis, D.; Su, C.Y.; Ellisman, M.H. High-quality ultrastructural preservation using cryofixation for 3D electron microscopy of genetically labeled tissues. eLife 2018, 7. [Google Scholar] [CrossRef]
- Zhang, Q.; Lee, W.A.; Paul, D.L.; Ginty, D.D. Multiplexed peroxidase-based electron microscopy labeling enables simultaneous visualization of multiple cell types. Nat. Neurosci. 2019, 22, 828–839. [Google Scholar] [CrossRef]
- Schmidt, H.R.; Benton, R. Molecular mechanisms of olfactory detection in insects: Beyond receptors. Open Biol. 2020, 10, 200252. [Google Scholar] [CrossRef]
- Wang, G.; Carey, A.F.; Carlson, J.R.; Zwiebel, L.J. Molecular basis of odor coding in the malaria vector mosquito Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2010, 107, 4418–4423. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Y.; Walker, W.B.; Dong, S.L.; Wang, G.R. Identification and localization of two sensory neuron membrane proteins from Spodoptera litura (Lepidoptera: Noctuidae). Insect Sci. 2015, 22, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Pellegrino, M.; Nakagawa, T.; Nakagawa, T.; Vosshall, L.B.; Touhara, K. Insect olfactory receptors are heteromeric ligand-gated ion channels. Nature 2008, 452, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Wicher, D.; Schäfer, R.; Bauernfeind, R.; Stensmyr, M.C.; Heller, R.; Heinemann, S.H.; Hansson, B.S. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 2008, 452, 1007–1011. [Google Scholar] [CrossRef]
- Missbach, C.; Dweck, H.K.; Vogel, H.; Vilcinskas, A.; Stensmyr, M.C.; Hansson, B.S.; Grosse-Wilde, E. Evolution of insect olfactory receptors. eLife 2014, 3, e02115. [Google Scholar] [CrossRef]
- Sakurai, T.; Namiki, S.; Kanzaki, R. Molecular and neural mechanisms of sex pheromone reception and processing in the silkmoth Bombyx mori. Front. Physiol. 2014, 5, 125. [Google Scholar] [CrossRef] [PubMed]
- Brito, N.F.; Moreira, M.F.; Melo, A.C. A look inside odorant-binding proteins in insect chemoreception. J. Insect Physiol. 2016, 95, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.J.; Guo, H.; Di, C.; Yu, S.; Zhu, L.; Huang, L.Q.; Wang, C.Z. Sequence similarity and functional comparisons of pheromone receptor orthologs in two closely related Helicoverpa species. Insect Biochem. Mol. Biol. 2014, 48, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Cassau, S.; Krieger, J. The role of SNMPs in insect olfaction. Cell Tissue Res. 2021, 383, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Diaz, C.; Bargeton, B.; Abuin, L.; Bukar, N.; Reina, J.H.; Bartoi, T.; Graf, M.; Ong, H.; Ulbrich, M.H.; Masson, J.F.; et al. A CD36 ectodomain mediates insect pheromone detection via a putative tunnelling mechanism. Nat. Commun. 2016, 7, 11866. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Chang, H.; Liu, W.; Cui, W.; Liu, Y.; Wang, Y.; Ren, B.; Wang, G. Essential role for SNMP1 in detection of sex pheromones in Helicoverpa armigera. Insect Biochem. Mol. Biol. 2020, 127, 103485. [Google Scholar] [CrossRef]
- Zhao, Y.; Ding, J.; Zhang, Z.; Liu, F.; Zhou, C.; Mu, W. Sex- and tissue-specific expression profiles of odorant binding protein and chemosensory protein genes in Bradysia odoriphaga (Diptera: Sciaridae). Front. Physiol. 2018, 9, 107. [Google Scholar] [CrossRef]
- Zhang, Y.N.; Qian, J.L.; Xu, J.W.; Zhu, X.Y.; Li, M.Y.; Xu, X.X.; Liu, C.X.; Xue, T.; Sun, L. Identification of chemosensory genes based on the transcriptomic analysis of six different chemosensory organs in Spodoptera exigua. Front. Physiol. 2018, 9, 432. [Google Scholar] [CrossRef]
- Laughlin, J.D.; Ha, T.S.; Jones, D.N.M.; Smith, D.P. Activation of pheromone-sensitive neurons is mediated by conformational activation of pheromone-binding protein. Cell 2008, 133, 1255–1265. [Google Scholar] [CrossRef]
- Tian, Z.; Zhang, Y. Molecular characterization and functional analysis of pheromone binding protein 1 from Cydia pomonella (L.). Insect Mol. Biol. 2016, 25, 769–777. [Google Scholar] [CrossRef]
- Oliveira, D.S.; Brito, N.F.; Franco, T.A.; Moreira, M.F.; Leal, W.S.; Melo, A.C.A. Functional characterization of odorant binding protein 27 (RproOBP27) from Rhodnius prolixus antennae. Front. Physiol. 2018, 9, 1175. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Liu, Y.; Yang, T. Pheromone binding proteins enhance the sensitivity of olfactory receptors to sex pheromones in Chilo suppressalis. Sci. Rep. 2015, 5, 13093. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.; Li, E.; Li, J.; Chen, Q.; Zhou, H.; Zhang, S.; Li, K.; Ren, B.; Wang, Y.; Yin, J. Molecular characterization and key binding sites of sex pheromone-binding proteins from the meadow moth, Loxostege sticticalis. J. Agric. Food Chem. 2019, 67, 12685–12695. [Google Scholar] [CrossRef] [PubMed]
- Jing, D.; Zhang, T.; Bai, S.; He, K.; Prabu, S.; Luan, J.; Wang, Z. Sexual-biased gene expression of olfactory-related genes in the antennae of Conogethes pinicolalis (Lepidoptera: Crambidae). BMC Genom. 2020, 21, 244. [Google Scholar] [CrossRef]
- Liu, N.Y.; Yang, F.; Yang, K.; He, P.; Niu, X.H.; Xu, W.; Anderson, A.; Dong, S.L. Two subclasses of odorant-binding proteins in Spodoptera exigua display structural conservation and functional divergence. Insect Mol. Biol. 2015, 24, 167–182. [Google Scholar] [CrossRef]
- Yang, H.; Su, T.; Yang, W.; Yang, C.-P.; Chen, Z.-M.; Lu, L.; Liu, Y.-L.; Tao, Y.-Y. Molecular characterization, expression pattern and ligand-binding properties of the pheromone-binding protein gene from Cyrtotrachelus buqueti. Physiol. Entomol. 2017, 42, 369–378. [Google Scholar] [CrossRef]
- Chen, X.L.; Li, G.W.; Xu, X.L.; Wu, J.X. Molecular and functional characterization of odorant binding protein 7 from the oriental fruit moth Grapholita molesta (Busck) (Lepidoptera: Tortricidae). Front. Physiol. 2018, 9, 1762. [Google Scholar] [CrossRef]
- Barrozo, R.B.; Gadenne, C.; Anton, S. Switching attraction to inhibition: Mating-induced reversed role of sex pheromone in an insect. J. Exp. Biol. 2010, 213, 2933–2939. [Google Scholar] [CrossRef]
- Kromann, S.H.; Saveer, A.M.; Binyameen, M.; Bengtsson, M.; Birgersson, G.; Hansson, B.S.; Schlyter, F.; Witzgall, P.; Ignell, R.; Becher, P.G. Concurrent modulation of neuronal and behavioural olfactory responses to sex and host plant cues in a male moth. Proc. Biol. Sci. 2015, 282, 20141884. [Google Scholar] [CrossRef]
- Hatano, E.; Saveer, A.M.; Borrero-Echeverry, F.; Strauch, M.; Zakir, A.; Bengtsson, M.; Ignell, R.; Anderson, P.; Becher, P.G.; Witzgall, P.; et al. Herbivore-induced plant volatile interferes with host plant and mate location in moths through suppression of olfactory signalling pathways. BMC Biol. 2015, 13, 75. [Google Scholar] [CrossRef]
- Jarriault, D.; Gadenne, C.; Rospars, J.P.; Anton, S. Quantitative analysis of sex-pheromone coding in the antennal lobe of the moth Agrotis ipsilon: A tool to study network plasticity. J. Exp. Biol. 2009, 212, 1191–1201. [Google Scholar] [CrossRef]
- Barrozo, R.B.; Jarriault, D.; Simeone, X.; Gaertner, C.; Gadenne, C.; Anton, S. Mating-induced transient inhibition of responses to sex pheromone in a male moth is not mediated by octopamine or serotonin. J. Exp. Biol. 2010, 213, 1100–1106. [Google Scholar] [CrossRef]
- Deisig, N.; Kropf, J.; Vitecek, S.; Pevergne, D.; Rouyar, A.; Sandoz, J.C.; Lucas, P.; Gadenne, C.; Anton, S.; Barrozo, R. Differential interactions of sex pheromone and plant odour in the olfactory pathway of a male moth. PLoS ONE 2012, 7, e33159. [Google Scholar] [CrossRef] [PubMed]
- Chaffiol, A.; Dupuy, F.; Barrozo, R.B.; Kropf, J.; Renou, M.; Rospars, J.-P.; Anton, S. Pheromone modulates plant odor responses in the antennal lobe of a moth. Chem. Senses 2014, 39, 451–463. [Google Scholar] [CrossRef]
- Hoffmann, A.; Bourgeois, T.; Munoz, A.; Anton, S.; Gevar, J.; Dacher, M.; Renou, M. Plant volatile alters the perception of sex pheromone blend ratios in a moth. J. Comp. Physiol. A 2020, 206, 553–570. [Google Scholar] [CrossRef]
- Ian, E.; Kirkerud, N.H.; Galizia, C.G.; Berg, B.G. Coincidence of pheromone and plant odor leads to sensory plasticity in the heliothine olfactory system. PLoS ONE 2017, 12, e0175513. [Google Scholar] [CrossRef] [PubMed]
- Borrero-Echeverry, F.; Bengtsson, M.; Nakamuta, K.; Witzgall, P. Plant odor and sex pheromone are integral elements of specific mate recognition in an insect herbivore. Evolution 2018, 72, 2225–2233. [Google Scholar] [CrossRef]
- Ammagarahalli, B.; Gemeno, C. Interference of plant volatiles on pheromone receptor neurons of male Grapholita molesta (Lepidoptera: Tortricidae). J. Insect Physiol. 2015, 81, 118–128. [Google Scholar] [CrossRef]
- Renou, M.; Party, V.; Rouyar, A.; Anton, S. Olfactory signal coding in an odor background. BioSystems 2015, 136, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Trona, F.; Anfora, G.; Balkenius, A.; Bengtsson, M.; Tasin, M.; Knight, A.; Janz, N.; Witzgall, P.; Ignell, R. Neural coding merges sex and habitat chemosensory signals in an insect herbivore. Proc. R. Soc. B 2013, 280, 20130267. [Google Scholar] [CrossRef] [PubMed]
- Galizia, C.G.; Rössler, W. Parallel olfactory system in insects: Anatomy and function. Annu. Rev. Entomol. 2010, 55, 399–420. [Google Scholar] [CrossRef] [PubMed]
- Varela, N.; Avilla, J.; Gemeno, C.; Anton, S. Ordinary glomeruli in the antennal lobe of male and female tortricid moth Grapholita molesta (Busck) (Lepidoptera: Tortricidae) process sex pheromone and host-plant volatiles. J. Exp. Biol. 2011, 214, 637–645. [Google Scholar] [CrossRef]
- Trona, F.; Anfora, G.; Bengtsson, M.; Witzgall, P.; Ignell, R. Coding and interaction of sex pheromone and plant volatile signals in the antennal lobe of the codling moth Cydia pomonella. J. Exp. Biol. 2010, 213, 4291–4303. [Google Scholar] [CrossRef]
- Anton, S.; Rössler, W. Plasticity and modulation of olfactory circuits in insects. Cell Tissue Res. 2021, 383, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Renou, M.; Anton, S. Insect olfactory communication in a complex and changing world. Curr. Opin. Insect Sci. 2020, 42, 1–7. [Google Scholar] [CrossRef]
- Murmu, M.S.; Hanoune, J.; Choi, A.; Bureau, V.; Renou, M.; Dacher, M.; Deisig, N. Modulatory effects of pheromone on olfactory learning and memory in moths. J. Insect Physiol. 2020, 127, 104159. [Google Scholar] [CrossRef]
- De Fouchier, A.; Walker III, W.B.; Montagné, N.; Steiner, C.; Binyameen, M.; Schlyter, F.; Chertemps, T.; Maria, A.; François, M.C.; Monsempes, C.; et al. Functional evolution of Lepidoptera olfactory receptors revealed by deorphanization of a moth repertoire. Nat. Commun. 2017, 8, 15709. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.C.; Domingos, A.I.; Jones, W.D.; Chiappe, M.E.; Amrein, H.; Vosshall, L.B. Or83b encodes a broadly expressed odorant receptor essential for Drosophila olfaction. Neuron 2004, 43, 703–714. [Google Scholar] [CrossRef]
- Hallem, E.A.; Ho, M.G.; Carlson, J.R. The molecular basis of odor coding in the Drosophila antenna. Cell 2004, 117, 965–979. [Google Scholar] [CrossRef]
- Legeai, F.; Malpel, S.; Montagné, N.; Monsempes, C.; Cousserans, F.; Merlin, C.; François, M.-C.; Maïbèche-Coisné, M.; Gavory, F.; Poulain, J.; et al. An expressed sequence tag collection from the male antennae of the noctuid moth Spodoptera littoralis: A resource for olfactory and pheromone detection research. BMC Genom. 2011, 12, 86. [Google Scholar] [CrossRef]
- Ochieng, S.A.; Anderson, P.; Hansson, B.S. Antennal lobe projection patterns of olfactory receptor neurons involved in sex pheromone detection in Spodoptera littoralis (Lepidoptera: Noctuidae). Tissue Cell 1995, 27, 221–232. [Google Scholar] [CrossRef]
- Schneider, D.; Schulz, S.; Priesner, E.; Ziesmann, J.; Francke, W. Autodetection and chemistry of female and male pheromone in both sexes of the tiger moth Panaxia quadripunctaria. J. Comp. Physiol. A 1998, 182, 153–161. [Google Scholar] [CrossRef]
- Holdcraft, R.; Rodriguez-Saona, C.; Stelinski, L.L. Pheromone autodetection: Evidence and implications. Insects 2016, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Bakthavatsalam, N.; Vinutha, J.; Ramakrishna, P.; Raghavendra, A.; Ravindra, K.; Verghese, A. Autodetection in Helicoverpa armigera (Hubner). Curr. Sci. 2016, 2261–2267. [Google Scholar] [CrossRef]
- Hillier, N.; Kleineidam, C.; Vickers, N.J. Physiology and glomerular projections of olfactory receptor neurons on the antenna of female Heliothis virescens (Lepidoptera: Noctuidae) responsive to behaviorally relevant odors. J. Comp. Physiol. A 2006, 192, 199–219. [Google Scholar] [CrossRef] [PubMed]
- Kuhns, E.H.; Pelz-Stelinski, K.; Stelinski, L.L. Reduced mating success of female tortricid moths following intense pheromone auto-exposure varies with sophistication of mating system. J. Chem. Ecol. 2012, 38, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Stelinski, L.; Holdcraft, R.; Rodriguez-Saona, C. Female moth calling and flight behavior are altered hours following pheromone autodetection: Possible implications for practical management with mating disruption. Insects 2014, 5, 459–473. [Google Scholar] [CrossRef]
- Stelinski, L.; Il’Ichev, A.; Gut, L. Antennal and behavioral responses of virgin and mated oriental fruit moth (Lepidoptera: Tortricidae) females to their sex pheromone. Ann. Entomol. Soc. Am. 2006, 99, 898–904. [Google Scholar] [CrossRef]
- Domínguez, A.; López, S.; Bernabé, A.; Guerrero, A.; Quero, C. Influence of age, host plant and mating status in pheromone production and new insights on perception plasticity in Tuta absoluta. Insects 2019, 10, 256. [Google Scholar] [CrossRef] [PubMed]
- Hodgdon, E.A.; Hallett, R.H.; Heal, J.D.; Swan, A.E.; Chen, Y.H. Synthetic pheromone exposure increases calling and reduces subsequent mating in female Contarinia nasturtii (Diptera: Cecidomyiidae). Pest Manag. Sci. 2021, 77, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Poivet, E.; Rharrabe, K.; Monsempes, C.; Glaser, N.; Rochat, D.; Renou, M.; Marion-Poll, F.; Jacquin-Joly, E. The use of the sex pheromone as an evolutionary solution to food source selection in caterpillars. Nat. Commun. 2012, 3, 1047. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, M.; Gehrke, P.; Badeke, E.; Sachse, S.; Breer, H.; Krieger, J. Larval sensilla of the moth Heliothis virescens respond to sex pheromone components. Insect Mol. Biol. 2016, 25, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Haynes, K.; Gaston, L.; Pope, M.M.; Baker, T.C. Potential for evolution of resistance to pheromones: Interindividual and interpopulational variation in chemical communication system of pink bollworm moth. J. Chem. Ecol. 1984, 10, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- Haynes, K.F.; Baker, T.C. Potential for evolution of resistance to pheromones. J. Chem. Ecol. 1988, 14, 1547–1560. [Google Scholar] [CrossRef]
- Doane, C.C.; Brooks, T.W. Research and development of pheromones for insect control with emphasis on the pink bollworm. In Management of Insect Pests with Semiochemicals; Mitchell, E.R., Ed.; Springer: Boston, MA, USA, 1981; pp. 285–303. [Google Scholar] [CrossRef]
- Collins, R.; Cardé, R. Variation in and heritability of aspects of pheromone production in the pink bollworm moth, Pectinophora gossypiella (Lepidoptera: Gelechiidae). Ann. Entomol. Soc. Am. 1985, 78, 229–234. [Google Scholar] [CrossRef]
- Evenden, M.; Haynes, K. Potential for the evolution of resistance to pheromone-based mating disruption tested using two pheromone strains of the cabbage looper, Trichoplusia ni. Entomol. Exp. Appl. 2001, 100, 131–134. [Google Scholar] [CrossRef]
- Löfstedt, C. Moth pheromone genetics and evolution. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1993, 340, 167–177. [Google Scholar]
- Phelan, P. Evolution of sex pheromones and the role of asymmetric tracking. In Insect Chemical Ecology: An Evolutionary Approach; Roitberg, B.D., Isman, M.B., Eds.; Chapman and Hall: New York, NY, USA, 1992; pp. 265–314. [Google Scholar]
- Mochizuki, F.; Fukumoto, T.; Noguchi, H.; Sugie, H.; Morimoto, T.; Ohtani, K. Resistance to a mating disruptant composed of (Z)-11-tetradecenyl acetate in the smaller tea tortrix, Adoxophyes honmai (Yasuda) (Lepidoptera: Tortricidae). Appl. Entomol. Zool. 2002, 37, 299–304. [Google Scholar] [CrossRef]
- Mochizuki, F.; Noguchi, H.; Sugie, H.; Tabata, J.; Kainoh, Y. Sex pheromone communication from a population resistant to mating disruptant of the smaller tea tortrix, Adoxophyes honmai Yasuda (Lepidoptera: Tortricidae). Appl. Entomol. Zool. 2008, 43, 293–298. [Google Scholar] [CrossRef]
- Tabata, J.; Noguchi, H.; Kainoh, Y.; Mochizuki, F.; Sugie, H. Behavioral response to sex pheromone-component blends in the mating disruption-resistant strain of the smaller tea tortrix, Adoxophyes honmai Yasuda (Lepidoptera: Tortricidae), and its mode of inheritance. Appl. Entomol. Zool. 2007, 42, 675–683. [Google Scholar] [CrossRef]
- Tabata, J.; Noguchi, H.; Kainoh, Y.; Mochizuki, F.; Sugie, H. Sex pheromone production and perception in the mating disruption-resistant strain of the smaller tea leafroller moth, Adoxophyes honmai. Entomol. Exp. Appl. 2007, 122, 145–153. [Google Scholar] [CrossRef]
- Stelinski, L.L.; Vogel, K.J.; Gut, L.J.; Miller, J.R. Seconds-long pre-exposures to pheromone from rubber septum or polyethelene tube dispensers alters subsequent behavioral responses of male Grapholita molesta (Lepidoptera:Tortricidae) in a sustained-flight tunnel. Environ. Entomol. 2005, 34, 696–704. [Google Scholar] [CrossRef]
- Miller, J.R.; Gut, L.J. Mating disruption for the 21st century: Matching technology with mechanism. Environ. Entomol. 2015, 44, 427–453. [Google Scholar] [CrossRef]
- Judd, G.J.R.; Gardiner, M.G.T.; DeLury, N.C.; Karg, G. Reduced antennal sensitivity, behavioural response, and attraction of male codling moths, Cydia pomonella, to their pheromone (E,E)-8,10-dodecadien-1-ol following various pre-exposure regimes. Entomol. Exp. Appl. 2005, 114, 65–78. [Google Scholar] [CrossRef]
- Suckling, D.M.; Stringer, L.D.; Jiménez-Pérez, A.; Walter, G.H.; Sullivan, N.; El-Sayed, A.M. With or without pheromone habituation: Possible differences between insect orders? Pest Manag. Sci. 2018, 74, 1259–1264. [Google Scholar] [CrossRef] [PubMed]
- Bartell, R.J.; Lawrence, L.A. Reduction in sexual responsiveness of male light-brown apple moth [Epiphyas postvittana (Wlk.)] following previous brief pheromonal exposure is concentration dependent. J. Aust. Entomol. Soc. 1976, 15, 236. [Google Scholar] [CrossRef]
- Sarles, L.; Verhaeghe, A.; Francis, F.; Verheggen, F.J. Semiochemicals of Rhagoletis fruit flies: Potential for integrated pest management. Crop Prot. 2015, 78, 114–118. [Google Scholar] [CrossRef]
- Sharma, A.; Sandhi, R.K.; Reddy, G.V. A review of interactions between insect biological control agents and semiochemicals. Insects 2019, 10, 439. [Google Scholar] [CrossRef] [PubMed]
- Bitsadze, N.; Jaronski, S.; Khasdan, V.; Abashidze, E.; Abashidze, M.; Latchininsky, A.; Samadashvili, D.; Sokhadze, I.; Rippa, M.; Ishaaya, I. Joint action of Beauveria bassiana and the insect growth regulators diflubenzuron and novaluron, on the migratory locust, Locusta migratoria. J. Pest Sci. 2013, 86, 293–300. [Google Scholar] [CrossRef]
- Reddy, G.V.; Zhao, Z.; Humber, R.A. Laboratory and field efficacy of entomopathogenic fungi for the management of the sweetpotato weevil, Cylas formicarius (Coleoptera: Brentidae). J. Invertebr. Pathol. 2014, 122, 10–15. [Google Scholar] [CrossRef]
- Abdellaoui, K.; Miladi, M.; Mkhinini, M.; Boughattas, I.; Hamouda, A.B.; Hajji-Hedfi, L.; Tlili, H.; Acheuk, F. The aggregation pheromone phenylacetonitrile: Joint action with the entomopathogenic fungus Metarhizium anisopliae var. acridum and physiological and transcriptomic effects on Schistocerca gregaria nymphs. Pest. Biochem. Physiol. 2020, 167, 104594. [Google Scholar] [CrossRef]
- Gutiérrez-Cárdenas, O.G.; Cortez-Madrigal, H.; Malo, E.A.; Gómez-Ruíz, J.; Nord, R. Physiological and pathogenical characterization of Beauveria bassiana and Metarhizium anisopliae isolates for management of adult Spodoptera frugiperda. Southwest. Entomol. 2019, 44, 409–421. [Google Scholar] [CrossRef]
- Akutse, K.S.; Khamis, F.M.; Ambele, F.C.; Kimemia, J.W.; Ekesi, S.; Subramanian, S. Combining insect pathogenic fungi and a pheromone trap for sustainable management of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Invertebr. Pathol. 2020, 177, 107477. [Google Scholar] [CrossRef]
- Akutse, K.S.; Subramanian, S.; Khamis, F.M.; Ekesi, S.; Mohamed, S.A. Entomopathogenic fungus isolates for adult Tuta absoluta (Lepidoptera: Gelechiidae) management and their compatibility with Tuta pheromone. J. Appl. Entomol. 2020, 144, 777–787. [Google Scholar] [CrossRef]
- Sammani, A.M.; Dissanayaka, D.M.; Wijayaratne, L.K.; Bamunuarachchige, T.C.; Morrison, W.R. Effect of pheromones, plant volatiles and spinosad on mating, male attraction and burrowing of Cadra cautella (Walk.) (Lepidoptera: Pyralidae). Insects 2020, 11, 845. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.; Yingao, M.; Xie, M.; Lu, Y.; Cheng, D. Rectal bacteria produce sex pheromones in the male oriental fruit fly. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mori, B.A.; Evenden, M.L. Factors affecting pheromone-baited trap capture of male Coleophora deauratella, an invasive pest of clover in Canada. J. Econ. Entomol. 2013, 106, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.V.; Cruz, Z.T.; Guerrero, A. Development of an efficient pheromone-based trapping method for the banana root borer Cosmopolites sordidus. J. Chem. Ecol. 2009, 35, 111–117. [Google Scholar] [CrossRef]
- Reddy, G.V.; Gadi, N.; Taianao, A.J. Efficient sex pheromone trapping: Catching the sweetpotato weevil, Cylas formicarius. J. Chem. Ecol. 2012, 38, 846–853. [Google Scholar] [CrossRef]
- Reddy, G.V.; Shrestha, G.; Miller, D.A.; Oehlschlager, A.C. Pheromone-trap monitoring system for pea leaf weevil, Sitona lineatus: Effects of trap type, lure type and trap placement within fields. Insects 2018, 9, 75. [Google Scholar] [CrossRef]
- Athanassiou, C.G.; Kavallieratos, N.G.; Gakis, S.F.; Kyrtsa, L.A.; Mazomenos, B.E.; Gravanis, F.T. Influence of trap type, trap colour, and trapping location on the capture of the pine moth, Thaumetopoea pityocampa. Entomol. Exp. Appl. 2007, 122, 117–123. [Google Scholar] [CrossRef]
- Jactel, H.; Menassieu, P.; Vétillard, F.; Barthélémy, B.; Piou, D.; Frérot, B.; Rousselet, J.; Goussard, F.; Branco, M.; Battisti, A. Population monitoring of the pine processionary moth (Lepidoptera: Thaumetopoeidae) with pheromone-baited traps. For. Ecol. Manag. 2006, 235, 96–106. [Google Scholar] [CrossRef]
- Jones, O.T. Practical applications of pheromones and other semiochemicals. In Insect Pheromones and Their Use in Pest Management; Howse, P., Stevens, I., Jones, O., Eds.; Chapman and Hall: London, UK, 1998; pp. 261–355. [Google Scholar]
- Trematerra, P.; Colacci, M. Recent advances in management by pheromones of Thaumetopoea moths in urban parks and woodland recreational areas. Insects 2019, 10, 395. [Google Scholar] [CrossRef]
- Byers, J.A. Modelling female mating success during mass trapping and natural competitive attraction of searching males or females. Entomol. Exp. Appl. 2012, 145, 228–237. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Suckling, D.M.; Wearing, C.H.; Byers, J.A. Potential of mass trapping for long-term pest management and eradication of invasive species. J. Econ. Entomol. 2006, 99, 1550–1564. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.; Serrano, J.M.; Johnson, P.J.; Millar, J.G. 13-Tetradecenyl acetate, a female-produced sex pheromone component of the economically important click beetle Melanotus communis (Gyllenhal) (Coleoptera: Elateridae). Sci. Rep. 2019, 9, 16197. [Google Scholar] [CrossRef] [PubMed]
- Hegazi, E.; Khafagi, W.E.; Konstantopoulou, M.; Raptopoulos, D.; Tawfik, H.; El-Aziz, G.M.A.; El-Rahman, S.M.A.; Atwa, A.; Aggamy, E.; Showeil, S. Efficient Mass-trapping method as an alternative tactic for suppressing populations of leopard moth (Lepidoptera: Cossidae). Ann. Entomol. Soc. Am. 2009, 102, 809–818. [Google Scholar] [CrossRef]
- Abbes, K.; Chermiti, B. Comparison of two marks of sex pheromone dispensers commercialized in Tunisia for their efficiency to monitor and to control by mass-trapping Tuta absoluta under greenhouses. Tunis. J. Plant Prot. 2011, 6, 133–148. [Google Scholar]
- Lobos, E.; Occhionero, M.; Werenitzky, D.; Fernandez, J.; Gonzalez, L.M.; Rodriguez, C.; Calvo, C.; Lopez, G.; Oehlschlager, A.C. Optimization of a trap for Tuta absoluta Meyrick (Lepidoptera: Gelechiidae) and trials to determine the effectiveness of mass trapping. Neotrop. Entomol. 2013, 42, 448–457. [Google Scholar] [CrossRef]
- Alpizar, D.; Fallas, M.; Oehlschlager, A.C.; Gonzalez, L.M. Management of Cosmopolites sordidus and Metamasius hemipterus in banana by pheromone-based mass trapping. J. Chem. Ecol. 2012, 38, 245–252. [Google Scholar] [CrossRef]
- Martin, J.C. Development of environment-friendly strategies in the management of processionary moths. In Processionary Moths and Climate Change: An Update; Roques, A., Ed.; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Trematerra, P.; Colacci, M.; Sciarretta, A. Mass-trapping trials for the control of pine processionary moth in a pine woodland recreational area. J. Appl. Entomol. 2019, 143, 129–136. [Google Scholar] [CrossRef]
- Lance, D.R.; Leonard, D.S.; Mastro, V.C.; Walters, M.L. Mating disruption as a suppression tactic in programs targeting regulated lepidopteran pests in US. J. Chem. Ecol. 2016, 42, 590–605. [Google Scholar] [CrossRef] [PubMed]
- Hummel, H.E.; Langner, S.S.; Eisinger, M.T. Pheromone dispensers, including organic polymer fibers, described in the crop protection literature: Comparison of their innovation potential. Commun. Agric. Appl. Biol. Sci. 2013, 78, 233–252. [Google Scholar] [PubMed]
- Ioriatti, C.; Lucchi, A. Semiochemical strategies for tortricid moth control in apple orchards and vineyards in Italy. J. Chem. Ecol. 2016, 42, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Witzgall, P.; Stelinski, L.; Gut, L.; Thomson, D. Codling moth management and chemical ecology. Annu. Rev. Entomol. 2008, 53, 503–522. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.; Zahavi, T.; Anshelevich, L.; Harel, M.; Ovadia, S.; Dunkelblum, E.; Harari, A.R. Mating disruption of Lobesia botrana (Lepidoptera: Tortricidae): Effect of pheromone formulations and concentrations. J. Econ. Entomol. 2005, 98, 135–142. [Google Scholar] [CrossRef]
- Stelinski, L.L.; Miller, J.R.; Ledebuhr, R.; Siegert, P.; Gut, L.J. Season-long mating disruption of Grapholita molesta (Lepidoptera: Tortricidae) by one machine application of pheromone in wax drops (SPLAT-OFM). J. Pest Sci. 2007, 80, 109–117. [Google Scholar] [CrossRef]
- Trematerra, P.; Athanassiou, C.; Stejskal, V.; Sciarretta, A.; Kavallieratos, N.; Palyvos, N. Large-scale mating disruption of Ephestia spp. and Plodia interpunctella in Czech Republic, Greece and Italy. J. Appl. Entomol. 2011, 135, 749–762. [Google Scholar] [CrossRef]
- Hoshi, H.; Takabe, M.; Nakamuta, K. Mating disruption of a carpenter moth, Cossus insularis (Lepidoptera: Cossidae) in apple orchards with synthetic sex pheromone, and registration of the pheromone as an agrochemical. J. Chem. Ecol. 2016, 42, 606–611. [Google Scholar] [CrossRef]
- Cocco, A.; Deliperi, S.; Delrio, G. Control of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) in greenhouse tomato crops using the mating disruption technique. J. Appl. Entomol. 2013, 137, 16–28. [Google Scholar] [CrossRef]
- Vacas, S.; Alfaro, C.; Primo, J.; Navarro-Llopis, V. Studies on the development of a mating disruption system to control the tomato leafminer, Tuta absoluta Povolny (Lepidoptera: Gelechiidae). Pest Manag. Sci. 2011, 67, 1473–1480. [Google Scholar] [CrossRef] [PubMed]
- Hummel, H.E.; Langner, S.S.; Breuer, M. Electrospun mesofibers, a novel biodegradable pheromone dispenser technology, are combined with mechanical deployment for efficient IPM of Lobesia botrana in vineyards. Commun. Agric. Appl. Biol. Sci. 2015, 80, 331–341. [Google Scholar] [PubMed]
- Lucchi, A.; Ladurner, E.; Iodice, A.; Savino, F.; Ricciardi, R.; Cosci, F.; Conte, G.; Benelli, G. Eco-friendly pheromone dispensers-a green route to manage the European grapevine moth? Environ. Sci. Pollut. Res. Int. 2018, 25, 9426–9442. [Google Scholar] [CrossRef] [PubMed]
- Mori, B.A.; Evenden, M.L. Challenges of mating disruption using aerosol-emitting pheromone puffers in red clover seed production fields to control Coleophora deauratella (Lepidoptera: Coleophoridae). Environ. Entomol. 2015, 44, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Ferracini, C.; Saitta, V.; Pogolotti, C.; Rollet, I.; Vertui, F.; Dovigo, L. Monitoring and management of the pine processionary moth in the North-Western Italian Alps. Forests 2020, 11, 1253. [Google Scholar] [CrossRef]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of push-pull strategies in integrated pest management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef]
- Dickens, J. Plant volatiles moderate response to aggregation pheromone in Colorado potato beetle. J. Appl. Entomol. 2006, 130, 26–31. [Google Scholar] [CrossRef]
- Reddy, G.V.; Guerrero, A. Interactions of insect pheromones and plant semiochemicals. Trends Plant Sci. 2004, 9, 253–261. [Google Scholar] [CrossRef]
- Powell, W.; Pickett, J.A. Manipulation of parasitoids for aphid pest management: Progress and prospects. Pest Manag. Sci. 2003, 59, 149–155. [Google Scholar] [CrossRef]
- Nakashima, Y.; Birkett, M.A.; Pye, B.J.; Pickett, J.A.; Powell, W. The role of semiochemicals in the avoidance of the seven-spot ladybird, Coccinella septempunctata, by the aphid parasitoid, Aphidius ervi. J. Chem. Ecol. 2004, 30, 1103–1116. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, Y.; Ida, T.Y.; Powell, W.; Pickett, J.A.; Birkett, M.A.; Taki, H.; Takabayashi, J. Field evaluation of synthetic aphid sex pheromone in enhancing suppression of aphid abundance by their natural enemies. BioControl 2016, 61, 485–496. [Google Scholar] [CrossRef][Green Version]
- Miller, J.R.; Gut, L.J.; de Lame, F.M.; Stelinski, L.L. Differentiation of competitive vs. non-competitive mechanisms mediating disruption of moth sexual communication by point sources of sex pheromone (part I): Theory. J. Chem. Ecol. 2006, 10, 2089–2114. [Google Scholar] [CrossRef] [PubMed]
| Order | Family | Genus | Publications |
|---|---|---|---|
| Lepidoptera | Noctuidae | Helicoverpa | 194 |
| Spodoptera | 173 | ||
| Tortricidae | Grapholita | 137 | |
| Cydia | 128 | ||
| Plutellidae | Plutella | 72 | |
| Crambidae | Ostrinia | 63 | |
| Bombycidae | Bombyx | 61 | |
| Diptera | Drosophilidae | Drosophila | 49 |
| Lepidoptera | Pyralidae | Chilo | 42 |
| Tortricidae | Lobesia | 41 | |
| Noctuidae | Agrotis | 40 | |
| Diptera | Tephritidae | Bactrocera | 37 |
| Hemiptera | Pseudococcidae | Planococcus | 36 |
| Lepidoptera | Lymantriidae | Lymantria | 36 |
| Coleoptera | Scarabaeidae | Holotrichia | 35 |
| Lepidoptera | Gelechiidae | Tuta | 33 |
| Geometridae | Ectropis | 30 | |
| Lasiocampidae | Dendrolimus | 29 | |
| Diptera | Psychodidae | Lutzomyia | 26 |
| Lepidoptera | Sesiidae | Synanthedon | 25 |
| Blattodea | Blatteidae | Blattella | 25 |
| Coleoptera | Buprestidae | Agrilus | 25 |
| Lepidoptera | Crambidae | Pyrausta | 24 |
| Gracillariidae | Phyllocnistis | 23 | |
| Erebidae | Hyphantria | 22 | |
| Hemiptera | Pseudococcidae | Pseudococcus | 21 |
| Lepidoptera | Pyralidae | Plodia | 21 |
| Crambidae | Cnaphalocrocis | 18 | |
| Tortricidae | Choristoneura | 18 | |
| Hemiptera | Miridae | Apolygus | 18 |
| Pheromone/Pheromone Components | Insect | References |
|---|---|---|
| (E)-11-hexadecenal, (E,E)-10,12-hexadecadienal | Diaphania glauculalis (Lepidoptera: Crambidae) | Ma et al. [15] |
| (E)-10-hexadecenal, (Z)-10-hexadecenal, (E)-10-hexadecenol, (E,E)-10,12-hexadecadienal, (Z,Z,Z)-3,6,9-tricosatriene | Conogethes pluto (Lepidoptera: Crambidae) | El Sayed et al. [16] |
| (Z)-11-hexadecenyl acetate, (Z)-11-hexadecenal, (Z)-11-hexadecenol | Trichophysetis cretacea (Lepidoptera: Crambidae) | Pong et al. [17] |
| (4aS,7S,7aR)-nepetalactone, (1R,4aS,7S,7aR)-nepetalactol | Hyalopterus pruni, Brachycaudus helichrysi (Hemiptera: Aphididae) | Symmes et al. [18] |
| (E,Z)-3,13-octadecadienyl acetate, (Z,Z)-3,13-octadecadienyl acetate | Synanthedon vespiformis (Lepidoptera: Sesiidae) | Levi-Zada et al. [19] |
| (E)-11-tetradecenyl acetate, (E,E)-9,11-tetradecadienyl acetate, (E)-11-tetradecenol, (E)-11-hexadecenyl acetate | Epiphyas postvittana (Lepidoptera: Tortricidae) | El Sayed et al. [20] |
| (Z,E)-9,12-tetradecadienyl acetate, (Z)-9-tetradecenyl acetate, (Z)-11-hexadecenyl acetate, (Z,E)-9,12-tetradecadienol, (Z)-9-tetradecenol, (Z)-11-hexadecenol | Spodoptera exigua (Lepidoptera: Noctuidae) | Acín et al. [21] |
| (Z,Z)-3,13-dodecadienolide | Parcoblatta lata (broad wood cochroach) | Eliyahu et al. [22] |
| (R,R)-(Z)-3,7,11,15-tetramethyl hexadec-2-enal, (R,R)-(E)-3,7,11,15-tetramethyl hexadec-2-enal | Dociostaurus maroccanus (Moroccan locust) | Guerrero et al. [23] |
| (4,5,5)-(trimethyl-3-methylenecyclopent-1-en-1-yl)methyl acetate | Delottococcus aberiae (Hemiptera: Pseudococcidae) | Vacas et al. [24] |
| (Z)-9-tetradecenyl acetate, (Z)-9-tetradecenol, tetradecyl acetate | Coryphodema tristis (Lepidoptera: Cossidae) | Bouwer et al. [25] |
| (-)-δ-heptalactone | Rhagoletis batava (Diptera: Tephritidae) | Büda et al. [26] |
| (E,E,Z,Z)-4,6,11,13-hexadecatetraenal | Callosamia promethea (Lepidoptera: Saturniidae) | Gago et al. [27] |
| (-)-iridomyrmecin | Leptopilina heterotoma (Hymenoptera: Figitidae) | Weiss et al. [28] |
| (Z,E)-5,7-dodecadienyl acetate, (Z,E)-5,7-dodecadienol, (Z,E)-5,7-dodecadienyl propionate | Dendrolimus tabulaeformis (Lepidoptera: Lasiocampidae) | Kong et al. [29] |
| (E)-7,9-decadienol, (E)-8-decenol | Monema flavescens (Lepidoptera: Limacodidae) | Shibasaki et al. [30] |
| (1S,4R,1′S)-4-(1′,5′-dimethylhex-4′-enyl)-1-methylcyclohex-2-en-1-ol | Oebalus poecilus (Heteroptera: Pentatomidae) | Oliveira et al. [31] |
| (3S,6S,7R)-1,10-bisaboladien-3-ol, (3R,6S,7R)-1,10-bisaboladien-3-ol | Tibraca limbativentris (Hemiptera: Pentatomidae) | Blassioli-Moraes et al. [32] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizvi, S.A.H.; George, J.; Reddy, G.V.P.; Zeng, X.; Guerrero, A. Latest Developments in Insect Sex Pheromone Research and Its Application in Agricultural Pest Management. Insects 2021, 12, 484. https://doi.org/10.3390/insects12060484
Rizvi SAH, George J, Reddy GVP, Zeng X, Guerrero A. Latest Developments in Insect Sex Pheromone Research and Its Application in Agricultural Pest Management. Insects. 2021; 12(6):484. https://doi.org/10.3390/insects12060484
Chicago/Turabian StyleRizvi, Syed Arif Hussain, Justin George, Gadi V. P. Reddy, Xinnian Zeng, and Angel Guerrero. 2021. "Latest Developments in Insect Sex Pheromone Research and Its Application in Agricultural Pest Management" Insects 12, no. 6: 484. https://doi.org/10.3390/insects12060484
APA StyleRizvi, S. A. H., George, J., Reddy, G. V. P., Zeng, X., & Guerrero, A. (2021). Latest Developments in Insect Sex Pheromone Research and Its Application in Agricultural Pest Management. Insects, 12(6), 484. https://doi.org/10.3390/insects12060484









