Habituation of the Light-Startle Response of Orange Head Cockroaches (Eublaberus posticus): Effects of Acclimation, Stimulus Duration, Presence of Food, and Intertrial Interval
Simple Summary
Abstract
1. Introduction
2. Experiment 1
2.1. Methods
2.1.1. Subjects
2.1.2. Procedure
2.1.3. Analysis
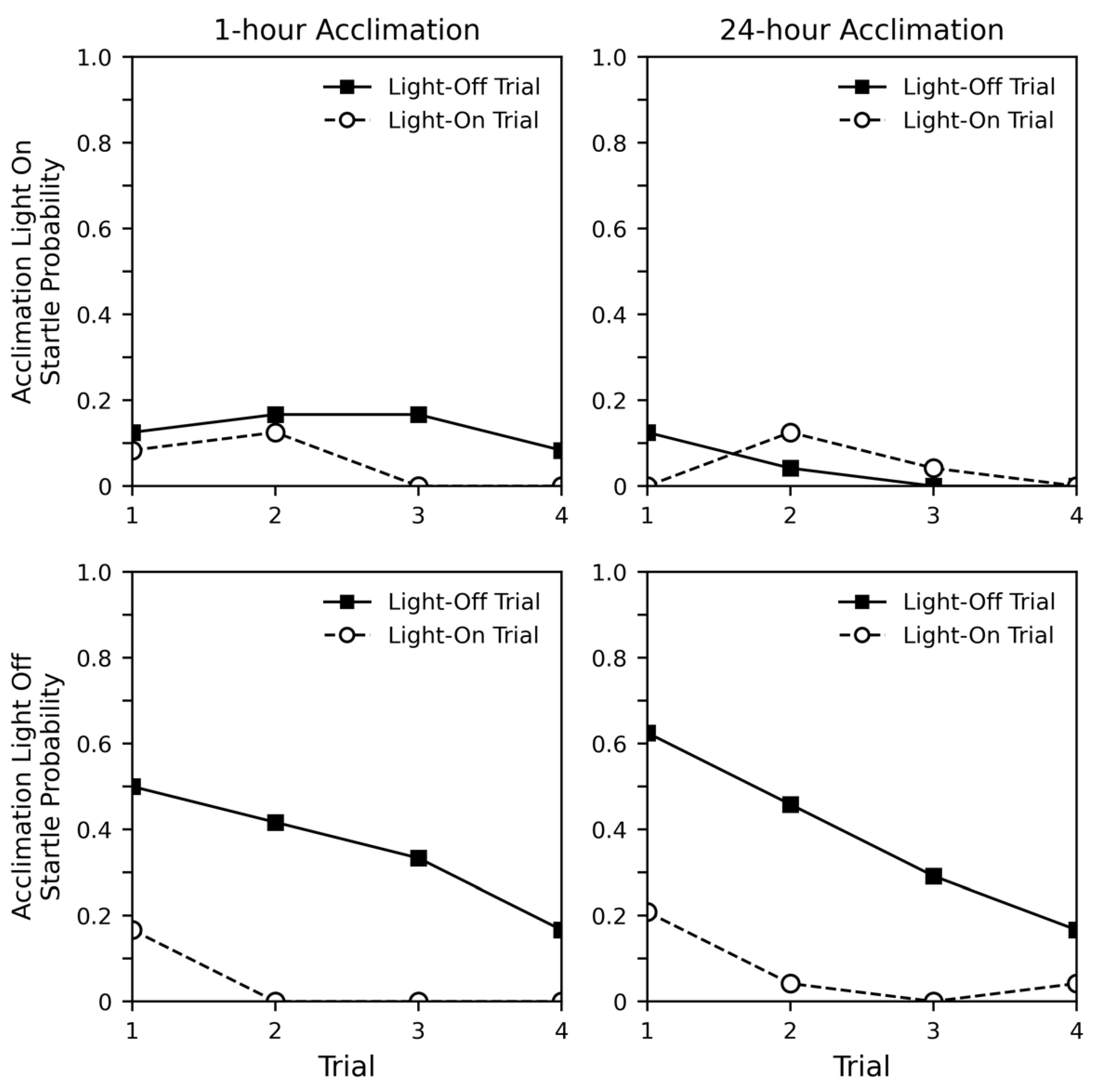
2.2. Results
2.3. Discussion
3. Experiment 2
3.1. Methods
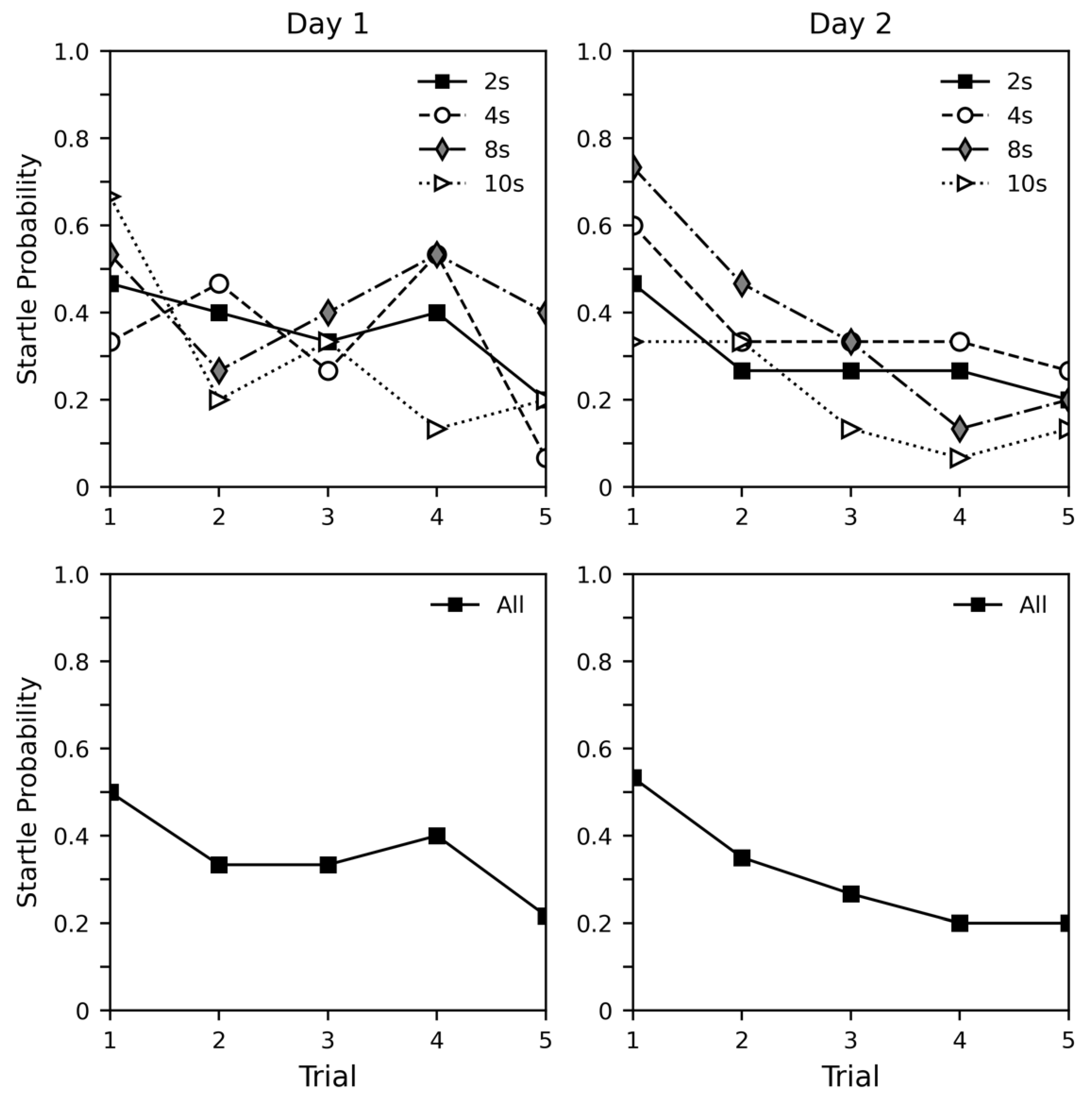
3.2. Results
3.3. Discussion
4. Experiment 3
4.1. Methods
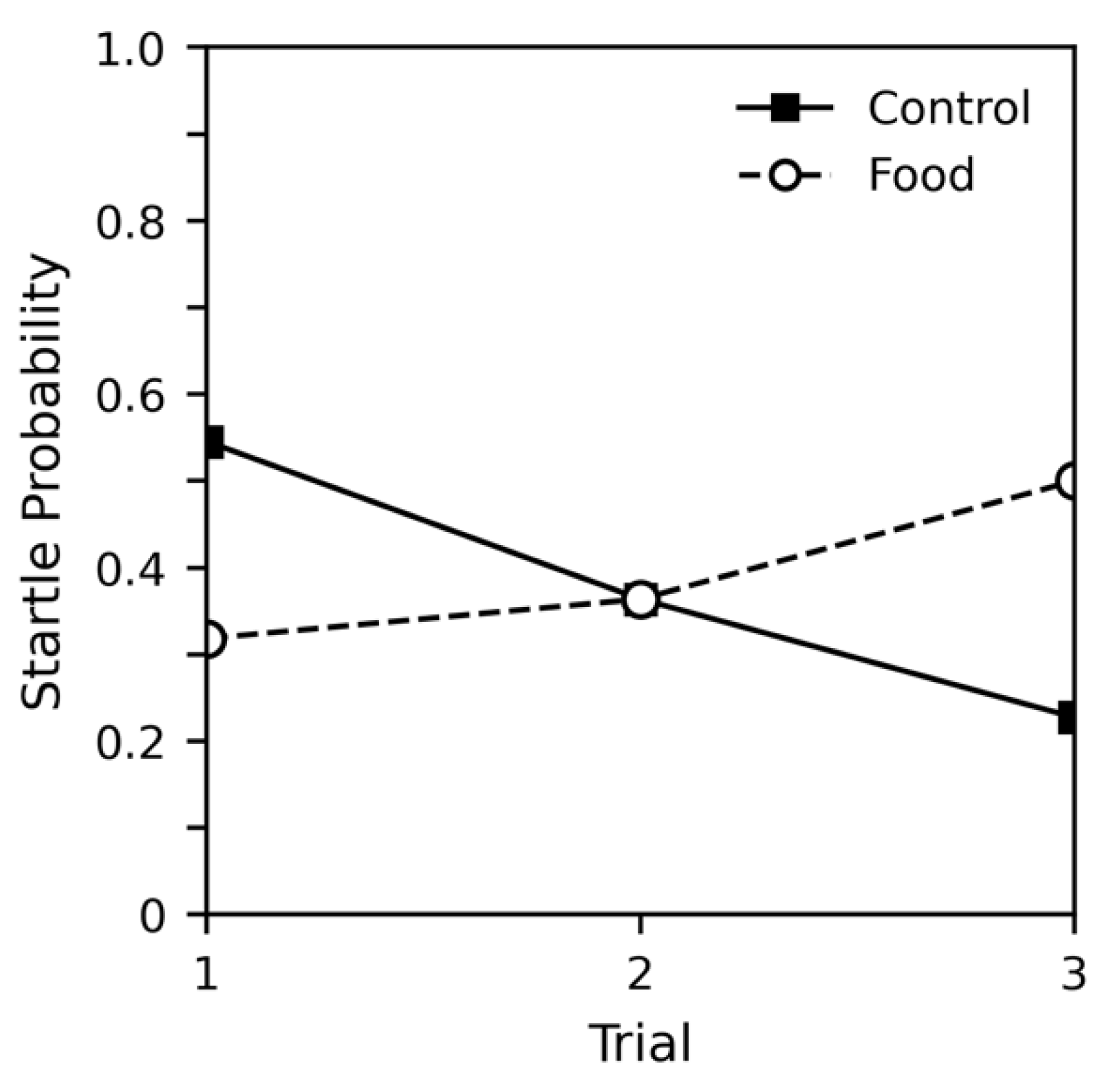
4.2. Results
4.3. Discussion
5. Experiment 4
5.1. Methods
5.2. Results
5.3. Discussion
6. General Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Burghardt, G.M. Darwin’s legacy to comparative psychology and ethology. Am. Psychol. 2009, 64, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Dewsbury, D. Comparative Psychology in the Twentieth Century; Hutchinson Ross Inc.: Stroudsburg, PA, USA, 1984. [Google Scholar]
- Jaynes, J. The historical origins of ‘ethology’ and ‘comparative psychology’. Anim. Behav. 1969, 17, 601–606. [Google Scholar] [CrossRef]
- Lockard, R.B. Reflections on the rise and fall of comparative psychology: Is there a lesson for us all? Am. Psychol. 1971, 26, 168–179. [Google Scholar] [CrossRef]
- Papini, M.R. Comparative psychology. In Handbook of Research Methods in Experimental Psychology; Davis, S.F., Ed.; Blackwell Publishing: Malden, MA, USA, 2003; pp. 211–240. [Google Scholar]
- Tinbergen, N. On aims and methods of ethology. Z. Tierpsychol. 1963, 20, 410–443. [Google Scholar] [CrossRef]
- Gallup, G.G., Jr. Editorial. J. Comp. Psychol. 1989, 103, 3. [Google Scholar] [CrossRef]
- Logan, C.A. The altered rationale for the choice of a standard animal in experimental psychology: Henry H. Donaldson, Adolf Meyer, and “the albino rat”. Hist. Psychol. 1999, 2, 3–24. [Google Scholar] [CrossRef] [PubMed]
- Beran, M.J.; Parrish, A.E.; Perdue, B.M.; Washburn, D.A. Comparative cognition: Past, Present, and Future. Int. J. Comp. Psychol. 2014, 27, 3–30. [Google Scholar]
- Varnon, C.A.; Lang, H.; Abramson, C.I. Automated research in comparative psychology: Limitations and new directions. Int. J. Comp. Psychol. 2018, 31, 1–17. [Google Scholar] [CrossRef]
- Abramson, C.I.; Wells, H. An inconvenient truth: Some neglected issues in invertebrate learning. Perspect. Behav. Sci. 2018, 41, 395–416. [Google Scholar] [CrossRef]
- McMonigle, O. For the Love of Cockroaches: Husbandry, Biology and History of Pet and Feeder Blattodea; Coachwhip Publications: Darke County, OH, USA, 2016; p. 354. [Google Scholar]
- Gorton, R.E.; Julmer, J., Jr.; Bell, W.J. Spacing patterns and dominance in the cockroach, Eublaberus posticus (Dictyoptera: Blaberidae). J. Kans. Entomol. Soc. 1979, 52, 334–343. [Google Scholar]
- Bell, W.J.; Robinson, S.; Tourtellot, M.K.; Breed, M.D. An ethometric analysis of agonistic behavior and social hierarchies in the cockroach, Eublaberus posticus. Z. Tierpsychol. 1978, 48, 203–218. [Google Scholar] [CrossRef]
- Thompson, R.F.; Spencer, W.A. Habituation: A model phenomenon for the study of neuronal substrates of behavior. Psychol. Rev. 1966, 73, 16–43. [Google Scholar] [CrossRef]
- Rankin, C.H.; Abrams, T.; Barry, R.J.; Bhatnager, S.; Claytong, D.; Colombo, J.; Thompson, R.F. Habituation revisited: An updated and revised description of the behavioral characteristics of habituation. Neurobiol. Learn. Mem. 2009, 92, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Varnon, C.A.; Vallely, N.; Beheler, C.; Coffin, C. The disturbance leg-lift response (DLR): An undescribed behavior in bumble bees. PeerJ 2021, 9, e10997. [Google Scholar] [CrossRef]
- Place, A.J.; Abramson, C.I. Habituation of the rattle response in western diamondback rattlesnakes, Crotalus atrox. Copeia 2008, 2008, 835–843. [Google Scholar] [CrossRef]
- Geyer, M.A.; Braff, D.L. Startle habituation and sensorimotor gating in schizophrenia and related animal models. Schizophr. Bull. 1987, 13, 643–668. [Google Scholar] [CrossRef] [PubMed]
- Fourtner, C.R.; Drewes, C.D. Excitation of the Common Inhibitory Motor Neuron: A Possible Role in the Startle Reflex of the Cockroach, Periplaneta americana. J. Neurobiol. 1977, 8, 477–489. [Google Scholar] [CrossRef]
- Camhi, J.M. Escape behavior in the cockroach: Distributed neural processing. Experientia. 1988, 44, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Camhi, J.M.; Nolen, T.G. Properties of the escape system of cockroaches during walking. J. Comp. Physiol. 1981, 142, 339–346. [Google Scholar] [CrossRef]
- Comer, C.M.; Mara, E.; Murphy, K.A.; Getman, M.; Mungy, M.C. Multisensory control of escape in the cockroach Periplaneta americana. I. Initial evidence from patterns of wind-evoked behavior. J. Comp. Physiol. A 1994, 174, 1–11. [Google Scholar] [CrossRef]
- Comer, C.M.; Mara, E.; Murphy, K.A.; Getman, M.; Mungy, M.C. Multisensory control of escape in the cockroach Periplaneta americana. II. Patterns of touch-evoked behavior. J. Comp. Physiol. A 1994, 174, 13–26. [Google Scholar] [CrossRef]
- Zilber-Gachelin, N.F.; Chartier, M.P. Modification of the motor reflex responses due to repetition of the peripheral stimulus in the cockroach. I. Habituation at the level of an isolated abdominal ganglia. J. Exp. Biol. 1973, 59, 359–381. [Google Scholar]
- Zilber-Gachelin, N.F.; Chartier, M.P. Modification of the motor reflex responses due to repetition of the peripheral stimulus in the cockroach. II. Conditions of activations of the motoneurons. J. Exp. Biol. 1973, 59, 359–381. [Google Scholar] [PubMed]
- Davis, H.; Heslop, E. Habituation of hissing by Madagascar hissing cockroaches (Gromphadorhina portentosa): Evidence of discrimination between humans? Behav. Process. 2004, 67, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.M.; Mote, M.I. Avoidance of monochromatic light by the cockroach Periplanta americana. J. Insect Physiol. 1990, 36, 287–291. [Google Scholar] [CrossRef]
- Harris, C.L. Learning of leg position by cockroaches in response to light. Physiol. Behav. 1993, 53, 313–316. [Google Scholar] [CrossRef]
- Lovell, K.L.; Eisenstien, E.M. Dark avoidance learning and memory disruption by carbon dioxide in cockroaches. Physiol. Behav. 1973, 10, 835–840. [Google Scholar] [CrossRef]
- Brown, S.; Strausfeld, N. The effect of age on a visual learning task in the American cockroach. Learn. Mem. 2009, 16, 210–223. [Google Scholar] [CrossRef]
- Gautier, J. Étude comparée de la distribution spatiale et temporelle des adultes de Blaberus atropos et B. colosseus (Dictyoptères) dans cinq grottes de l’île de Trinidad. Rev. Comport. Anim. 1974, 9, 237–258. [Google Scholar]
- StatsModels. Version 0.9.0. Available online: http://www.statsmodels.org (accessed on 8 April 2021).
- Hardin, J.; Hilbe, J.M. Generalized Estimating Equations; Chapman & Hall: Boca Raton, FL, USA, 2003. [Google Scholar]
- Clogg, C.C.; Petkova, E.; Haritou, A. Statistical methods for comparing regression coefficients between models. Am. J. Sociol. 1995, 100, 1261–1293. [Google Scholar] [CrossRef]
- Paternoster, R.; Brame, R.; Mazerolle, P.; Piquero, A. Using the correct statistical test for the equality of regression coefficients. Criminology 1998, 36, 859–866. [Google Scholar] [CrossRef]
- Varnon, C.A.; Dinges, C.W.; Black, T.E.; Wells, H.; Abramson, C.I. Failure to find ethanol-induced conditioned taste aversion in honey bees (Apis mellifera L.). Alcohol Clin. Exp. Res. 2018, 42, 1260–1270. [Google Scholar] [CrossRef]
- Gunn, D.L. The daily rhythm of activity of the cockroach, Blatta orientalis L. J. Exp. Biol. 1940, 17, 267–277. [Google Scholar]
- Harker, J.E. Factors controlled the diurnal rhythm of activity of Periplaneta americana L. J. Exp. Biol. 1956, 33, 224–234. [Google Scholar]
- Eiserer, L.A.; Ramsay, D.S. Priming of darkness-rewarded runway responses in the American cockroach (Periplaneta americana). J. Gen. Psychol. 1981, 104, 213–221. [Google Scholar] [CrossRef]
- Decker, S.; McConnaughey, S.; Page, T.L. Circadian regulation of insect olfactory learning. Proc. Natl. Acad. Sci. USA. 2007, 104, 15905–15910. [Google Scholar] [CrossRef] [PubMed]
- Garren, M.V.; Sexauer, S.B.; Page, T.L. Effect of circadian phase on memory acquisition and recall: Operant conditioning vs. classical conditioning. PLoS ONE 2013, 8, e58693. [Google Scholar] [CrossRef] [PubMed]
- Lubinski, A.J.; Page, T.L. The optic lobes regulate circadian rhythms of olfactory learning and memory in the cockroach. J. Biol. Rhythms 2016, 31, 161–169. [Google Scholar] [CrossRef]
- Wantanabe, H.; Kobayashi, Y.; Saura, M.; Matsumoto, Y.; Mizunami, M. Classical olfactory conditioning in the cockroach Periplaneta americana. Zool. Sci. 2003, 20, 1147–1454. [Google Scholar] [CrossRef]
- Lent, D.D.; Kwon, H. Antennal movements reveal associative learning in the American cockroach Periplaneta americana. J. Exp. Biol. 2004, 207, 369–375. [Google Scholar] [CrossRef]
- Deng, Z.; Lubinski, A.J.; Page, T.L. Zeta inhibitory peptide (ZIP) erases long-term memories in a cockroach. Neurobiol. Learn. Mem. 2015, 118, 89–95. [Google Scholar] [CrossRef]
- Davis, M. Effects of interstimulus interval length and variability on startle-response habituation in the rat. J. Comp. Psychol. 1970, 72, 177–192. [Google Scholar] [CrossRef]
- Beck, C.D.O.; Rankin, C.H. Long-term habituation is produced by distributed training at long ISIs and not by massed training or short ISIs in Caenorhabditis elegans. Learn. Behav. 1997, 25, 446–457. [Google Scholar] [CrossRef]
- Carew, T.J.; Pinsker, H.M.; Kandel, E.R. Long-term habituation of a defensive withdrawal reflex in Aplysia. Science 1972, 175, 451–454. [Google Scholar] [CrossRef]
- Pedreira, M.E.; Romano, A.; Romsic, D.; Lozada, M.; Maldonado, H. Massed and space training build up different components of long-term habituation in the crab Chasmagnathus. Anim. Learn. Behav. 1998, 26, 34–45. [Google Scholar] [CrossRef]
- Tomsic, D.; Berón de Astrada, M.; Sztarker, J.; Maldonado, H. Behavioral and neuronal attributes of short- and long-term habituation in the crab Chasmagnathus. Neurobiol. Learn. Mem. 2009, 92, 176–182. [Google Scholar] [CrossRef]
- Anderson, M.J.; Jablonski, S.A.; Klimas, D.B. Spaced initial stimulus familiarization enhances novelty preference in Long-Evans rats. Behav. Process. 2008, 78, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, D.J.; Bannerman, D.M. Competitive short-term and long-term memory processes in spatial habituation. J. Exp. Psychol. Animal Learn. Cogn. 2011, 37, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Gatchel, R.J. Effects of interstimulus interval length on short-term and long-term habituation of autonomic components of orienting response. Psychobiology 1975, 3, 133–136. [Google Scholar] [CrossRef]
- Wagner, A.R. SOP: A model of automatic memory processing in animal behavior. In Information Processing in Animals: Memory Mechanisms; Spear, N.E., Miller, R.R., Eds.; Erlbaum Inc.: Hillsdale, NJ, USA, 1981; pp. 5–47. [Google Scholar]
- Anderson, D.C.; Sergio, J.P.; Ewing, M. Food deprivation and startle magnitude: Inhibition, potentiation, or neither. Bull. Psychon. Soc. 1982, 19, 165–168. [Google Scholar] [CrossRef]
- Schmid, A.; Koch, M.; Schnitzler, H. Conditioned pleasure attenuates the startle response in rats. Neurobiol. Learn. Mem. 1995, 64, 1–3. [Google Scholar] [CrossRef]
- Blumenthal, T.D. Extraversion, attention, and startle response reactivity. Pers. Individ. Differ. 2001, 30, 495–503. [Google Scholar] [CrossRef]
- Levenson, R.W.; Ekman, P.; Ricard, M. Meditation and the startle response: A case study. Emotion 2012, 12, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Reynierse, J.H.; Manning, A.; Cafferty, D. The effects of hunger and thrust on body weight and activity in the cockroach (Nauphoeta cinerea). Anim. Behav. 1972, 20, 751–757. [Google Scholar] [CrossRef]
- Barcay, S.J.; Bennett, G.W. Influence of Starvation and Lighting on the Movement Behavior of the German Cockroach (Blattodea: Blattellidae). J. Econ. Entomol. 1991, 84, 1520–1524. [Google Scholar] [CrossRef] [PubMed]
- Toda, N.R.T.; Song, J.; Nieh, J.C. Bumblebees exhibit the memory spacing effect. Sci. Nat. 2009, 96, 1185–1191. [Google Scholar] [CrossRef][Green Version]
- Hosono, S.; Matusmoto, Y.; Mizunami, M. Interaction of inhibitory and facilitatory effects of conditioning trials on long-term memory formation. Learn. Mem. 2016, 23, 669–678. [Google Scholar] [CrossRef]
- Thon, B. Acquisition and retention of habituation as a function of intertrial interval duration during training in the blowfly. Behav. Process. 1987, 15, 45–57. [Google Scholar] [CrossRef]
- Groves, P.M.; Thompson, R.F. Habituation: A dual-process theory. Psychol. Rev. 1970, 77, 419–450. [Google Scholar] [CrossRef]
- Abramson, C.I.; Craig, D.P.A.; Varnon, C.A.; Wells, H. The effect of ethanol on reversal learning in honey bees (Apis mellifera anatolica): Response inhibition in a social insect model. Alcohol 2015, 49, 245–258. [Google Scholar] [CrossRef]
- Agarwal, M.; Giannoni Guzmán, M.; Morales-Matos, C.; Del Valle Díaz, R.A.; Abramson, C.I.; Giray, T. Dopamine and Octopamine Influence Avoidance Learning of Honey Bees in a Place Preference Assay. PLoS ONE 2011, 6, e25371. [Google Scholar] [CrossRef]
- Abramson, C.I. Invertebrates in the Classroom. Teach. Psychol. 1986, 13, 24–29. [Google Scholar] [CrossRef]
- Dixon, M.; Daar, J.H.; Gunnarsson, K.F.; Johnson, M.L.; Shayter, A. Stimulus preference and reinforcement effects of the madagascar hissing cockroach (Gromphadorhina portentosa): A case of reverse translational research. Psychol. Rec. 2015, 66, 41–51. [Google Scholar] [CrossRef]
- Proctor, D.; Jones, M. Cockroaches to the rescue: A new approach to reintroduce animal labs to the psychology undergraduate curriculum. Scholarsh. Teach. Learn. Psychol. 2020. [Google Scholar] [CrossRef]
- Tomsic, D.; Massoni, V.; Maldonado, H. Habituation to a danger stimulus in two semiterrestrial crabs: Ontogenic, ecological and opioid modulation correlates. J. Comp. Physiol. A 1993, 173, 621–633. [Google Scholar] [CrossRef]
- Rosenthal, R. The file drawer problem and tolerance for null results. Psychol. Bull. 1979, 86, 638–641. [Google Scholar] [CrossRef]
- Scargle, J.R. Publication bias: The “file-drawer” problem in scientific inference. J. Sci. Explor. 2000, 14, 91–106. [Google Scholar]

| Parameter | Estimate | Standard Error | p-Value |
|---|---|---|---|
| Intercept | 0.117 | 0.473 | 0.805 |
| Light-On Trial | −1.918 | 0.689 | 0.005 |
| Acclimation Light On | −2.839 | 0.642 | 0.000 |
| Light-On Trial × Acclimation Light On | 1.821 | 1.049 | 0.083 |
| Trial | −0.309 | 0.060 | 0.000 |
| Trial × Light-On Trial | −0.380 | 0.332 | 0.252 |
| Trial × Acclimation Light On | 0.136 | 0.136 | 0.319 |
| Trial × Light-On Trial × Acclimation Light On | 0.276 | 0.370 | 0.455 |
| Weight (g) | 0.352 | 0.137 | 0.010 |
| Parameter | Estimate | Standard Error | 95% Confidence Intervals | p-Value | |
|---|---|---|---|---|---|
| Intercept | −0.057 | 0.512 | −1.062 | 0.947 | 0.911 |
| Trial | −0.222 | 0.078 | −0.375 | −0.069 | 0.004 |
| Day | 0.288 | 0.342 | −0.382 | 0.957 | 0.400 |
| Trial × Day | −0.180 | 0.104 | −0.384 | 0.024 | 0.083 |
| Light duration (s) | −0.014 | 0.048 | −0.108 | 0.080 | 0.771 |
| Weight (g) | 0.063 | 0.064 | −0.063 | 0.189 | 0.328 |
| Parameter | Estimate | Standard Error | 95% Confidence Intervals | p-Value | |
|---|---|---|---|---|---|
| Control | 1.121 | 1.126 | −1.085 | 3.328 | 0.319 |
| Food | −0.929 | 1.298 | −3.473 | 1.615 | 0.474 |
| Control × Trial | −0.704 | 0.273 | −1.239 | −0.170 | 0.010 |
| Food × Trial | 0.387 | 0.264 | −0.130 | 0.904 | 0.142 |
| Weight (g) | 1.121 | 1.126 | −1.085 | 3.328 | 0.319 |
| Parameter | Estimate | Standard Error | 95% Confidence Intervals | p-Value | |
|---|---|---|---|---|---|
| Intercept | 2.995 | 0.760 | 1.506 | 4.485 | 0.000 |
| Trial | −0.838 | 0.154 | −1.139 | −0.536 | 0.000 |
| ITI | −0.001 | 0.001 | −0.002 | 0.001 | 0.260 |
| Trial × ITI | 0.001 | 0.000 | 0.000 | 0.001 | 0.004 |
| Weight (g) | −0.353 | 0.183 | −0.713 | 0.006 | 0.054 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varnon, C.A.; Adams, A.T. Habituation of the Light-Startle Response of Orange Head Cockroaches (Eublaberus posticus): Effects of Acclimation, Stimulus Duration, Presence of Food, and Intertrial Interval. Insects 2021, 12, 339. https://doi.org/10.3390/insects12040339
Varnon CA, Adams AT. Habituation of the Light-Startle Response of Orange Head Cockroaches (Eublaberus posticus): Effects of Acclimation, Stimulus Duration, Presence of Food, and Intertrial Interval. Insects. 2021; 12(4):339. https://doi.org/10.3390/insects12040339
Chicago/Turabian StyleVarnon, Christopher A., and Ann Taylor Adams. 2021. "Habituation of the Light-Startle Response of Orange Head Cockroaches (Eublaberus posticus): Effects of Acclimation, Stimulus Duration, Presence of Food, and Intertrial Interval" Insects 12, no. 4: 339. https://doi.org/10.3390/insects12040339
APA StyleVarnon, C. A., & Adams, A. T. (2021). Habituation of the Light-Startle Response of Orange Head Cockroaches (Eublaberus posticus): Effects of Acclimation, Stimulus Duration, Presence of Food, and Intertrial Interval. Insects, 12(4), 339. https://doi.org/10.3390/insects12040339





