Ecosystem Birth near Melting Glaciers: A Review on the Pioneer Role of Ground-Dwelling Arthropods
Simple Summary
Abstract
1. The Questions
- A.
- How “flexible” are arthropod communities in the early phase of primary succession?
- B.
- Do pioneer arthropod communities under different conditions still have certain basic traits in common?
2. Conquering Newly Deglaciated Ground: A Passage through Ecological Filters
2.1. Ability to Disperse
2.2. Ability to Find Acceptable Habitat Qualities
2.3. Ability to Find Food
2.4. Ability to Reproduce
2.5. Different Filters for Different Successional Stages
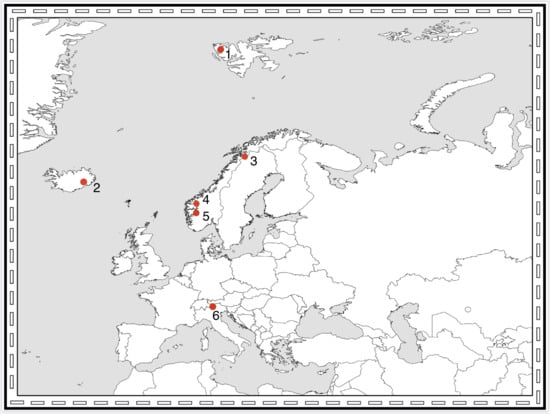
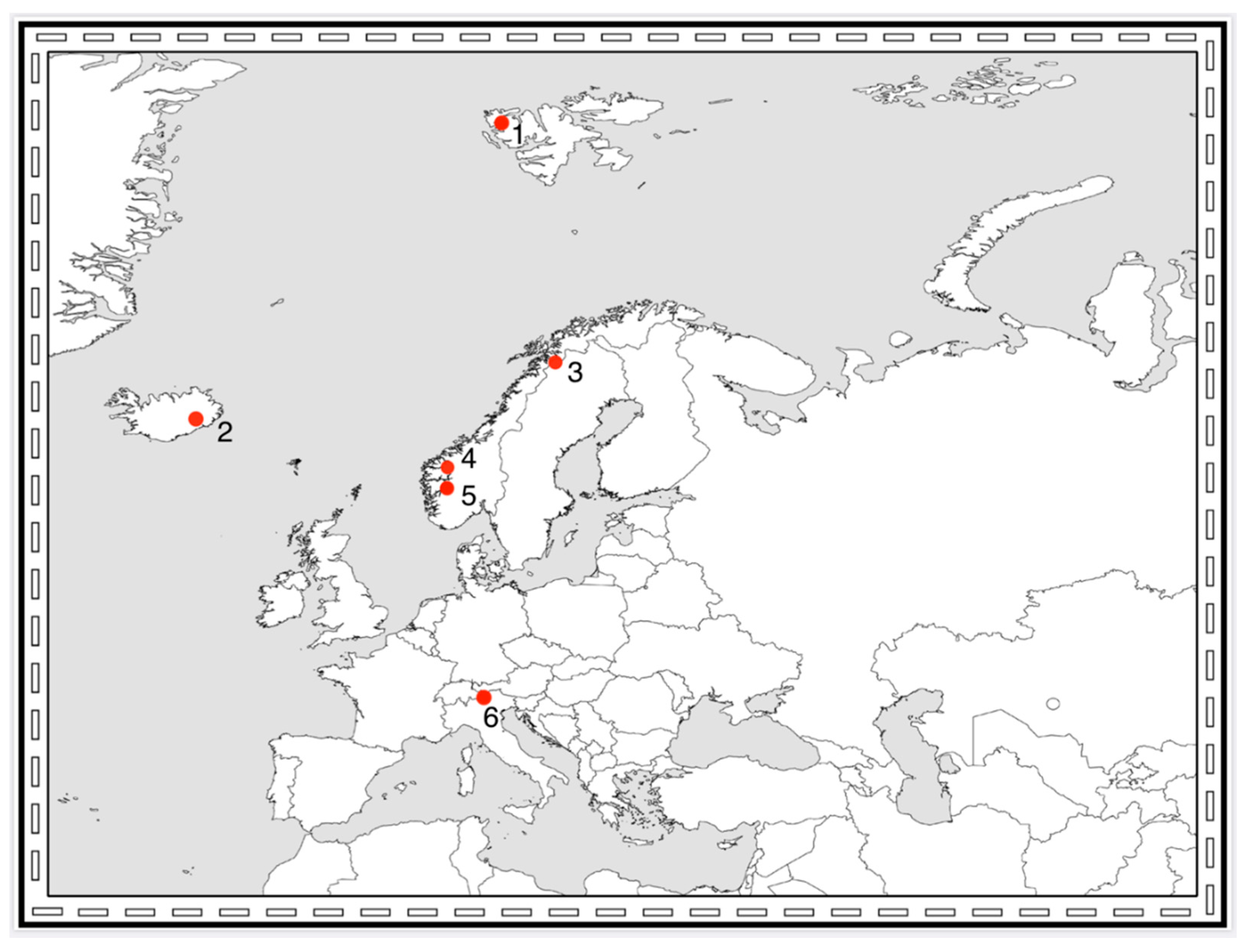
3. Which Arthropod Taxa are Present on Pioneer Ground? A Comparison between Northern Europe and the Alps
3.1. Microarthropods: The Super-Pioneers
3.1.1. Northern Europe
3.1.2. The Alps
3.2. Macroarthropods: The Early Predators
3.2.1. Northern Europe
3.2.2. The Alps
3.3. Comparing Pioneer Communities in Northern Europe and the Alps
3.4. Effects of Altitude and Latitude on Pioneer Fauna
4. From Structure to Function: A Comparison between Two Well-studied Pioneer Food Webs
- -
- If the two food webs are different: why are they different, and can the difference illustrate flexibility in pioneer arthropod communities?
- -
- Can these well-studied pioneer food webs shed new light on the “predator first paradox”?
4.1. Study Sites and Methods
4.2. Main Similarities and Differences
- Typical macroarthropod predators in the pioneer community were Carabidae beetles, Opiliones and Lycosidae, and Linyphiidae among spiders.
- The main food for pioneer predators was considered to be produced locally and not transported to the pioneer ground by air.
- Collembola seemed to be among the very first animal colonizers of newly exposed ground.
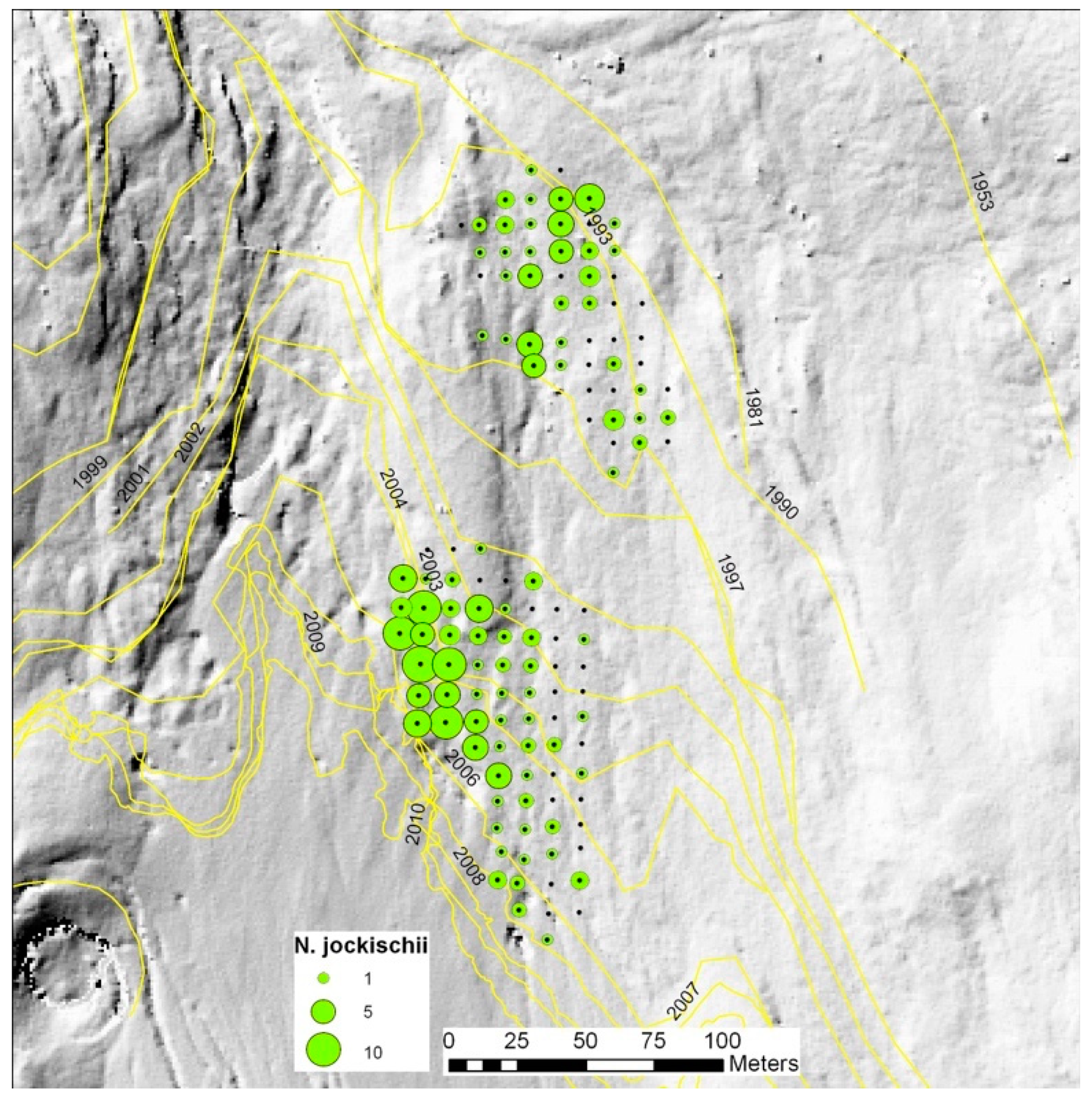
4.3. Special Observations in the Austrian Case
4.4. Special Observations in the Norwegian Case
4.5. Discussion of the Two Studies
4.5.1. Flexibility in Pioneer Food Webs
4.5.2. The Predator first Paradox Resolved?
5. General Discussion
5.1. Driving Forces in Early Succession: A Geoecological View
5.2. How to Quantify Food Choice?
5.3. Pristine Ground—Both a Sink and a New Ecosystem
5.4. The Pioneer Community Enigma: A Mix of Different Life Forms
5.5. Nature Conservation Aspects
6. Conclusions
6.1. General Findings
- Arthropods colonize deglaciated ground in a rapid succession and may develop food webs within a few years, before higher plants establish or only occur sporadically.
- Pioneer species are fewer, and succession is slower at high altitudes and latitudes.
- Each pioneer species has to overcome certain obstacles, or “ecological filters”: It must travel, it must find an acceptable habitat with tolerable climate and shelter to avoid hostile conditions, it must be able to find food, and it must be able to reproduce.
- Microarthropods, especially Collembola, are “super-pioneers”. They colonize during the very first years, even in high alpine or high arctic conditions.
- Macroarthropod predators follow closely and are well represented after just a few years (1–5 years). Typical taxa in Europe are Carabidae among Coleoptera, Lycosidae, and Linyphiidae among Aranea, and Mitopus sp. among Opiliones.
- On species level, pioneer arthropods differ between northern Europe and the Alps, due to different glaciation and speciation history. However, certain genera are common to both.
- A pioneer arthropod community is a mixture of specialists and generalists.
- The pioneer community may include both parthenogenetic and sexually reproducing species, species with short or long life cycle, and small as well as large species.
- Certain specialists may disappear after a few decades (30–50 years). Examples are cold-adapted species and species preferring open ground.
- Melting of glaciers threaten several cold-adapted species within various groups as Carabidae, Araneae, and Collembola.
- Recent studies challenge the classic “predator first paradox”, which describe a heterotrophic pioneer community depending on airborne prey for predators and airborne dead organic material for decomposing Collembola. It has been shown that predators can be fed by locally produced food, and Collembola can be herbivores.
- Certain “invisible” carbon sources can contribute to the pioneer community: terrestrial biofilm with diatom algae or cyanobacteria, tiny pioneer mosses, pollen, and bioavailable ancient carbon released by the glacier.
- Two well-studied pioneer food webs, one from Norway and one from Austria, revealed similarities in arthropod families and genera, but the structure and function of the food webs differed.
- The pioneer ground can be surprisingly rich in microhabitats and food sources.
- For many arthropods, pioneer ground is a sink, but dead animals can contribute as prey or soil fertilizers.
- Definition of succession is a matter of scale. For instance, a small pioneer moss turf may harbor moss-eating Byrrhidae beetles. Moreover, patches with different microclimate or substrate may have different succession pathways.
- Terrestrial and aquatic environments can be connected in early succession. For instance, young ponds and glacial stream rivers may produce chironomid midges that serve as prey for terrestrial predators.
6.2. The Birth of an Ecosystem: Short Answers to the Two Main Questions
- A.
- How “flexible” are arthropod communities in the early phase of primary succession?
- B.
- Do pioneer arthropod communities under different conditions still have certain basic traits in common?
7. Suggestions for Further Studies
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oerlemans, J. Extracting a climate signal from 169 glacier records. Science 2005, 308, 675–677. [Google Scholar] [CrossRef]
- Jomelli, V.; Khodri, M.; Favier, V.; Brunstein, D.; Ledru, M.-P.; Wagnon, P.; Blard, P.-H.; Sicar, J.-E.; Braucher, R.; Grancher, D.; et al. Irregular tropical glacier retreat over the Holocene epoch driven by progressive warming. Nature 2011, 474, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Malcomb, N.L.; Wiles, G.C. Tree-ring-based reconstructions of North American glacier mass balance through the Little Ice Age—Contemporary warming transition. Quat. Res. 2013, 79, 123–137. [Google Scholar] [CrossRef]
- Elven, R.; Ryvarden, L. Dispersal and primary establishment of vegetation. In Fennoscandian Tundra Ecosystems, Part I; Wielgolaski, F.E., Ed.; Springer: Berlin/Heidelberg, Germany, 1975; pp. 81–85. [Google Scholar]
- Connell, J.H.; Slatyer., R.O. Mechanisms of succession in natural communities and their role in community stability and organization. Am. Nat. 1977, 111, 1119–1144. [Google Scholar] [CrossRef]
- Elven, R. Association analysis of moraine vegetation at the glacier Hardangerjøkulen, Finse, South Norway. Norweg. J. Bot. 1978, 25, 171–191. [Google Scholar]
- Elven, R. The Omnsbreen glacier nunataks—A case study of plant immigration. Norweg. J. Bot. 1980, 27, 1–16. [Google Scholar]
- Matthews, J.A. The Ecology of Recently-Deglaciated Terrain: A Geoecological Approach to Glacier Forelands and Primary Succession; Cambridge University Press: Cambridge, UK, 1992; 386p. [Google Scholar]
- Kaufmann, R.; Raffl, C. Diversity in primary succession: The chronosequence of a glacier foreland. In Global Mountain Biodiversity: A Global Assessment; Körner, C., Spehn, E.M., Eds.; Parthenon: London, UK, 2002; pp. 177–190. [Google Scholar]
- Tampucci, D.; Gobbi, M.; Boracchi, P.; Cabrini, E.; Compostella, C.; Mangili., F.; Marano, G.; Pantini, P.; Caccianiga, M. Plant and arthropod colonisation of a glacier foreland in a peripheral mountain range. Biodiversity 2015, 16, 213–223. [Google Scholar] [CrossRef]
- Hågvar, S.; Pedersen, A. Food choice of invertebrates during early glacier foreland succession. Arct. Antarct. Alp. Res. 2015, 47, 561–572. [Google Scholar] [CrossRef]
- Vater, A.E.; Matthews, J.A. Testing the “addition and persistence” model of invertebrate succession in a subalpine glacier-foreland chronosequence: Fåbergstølsbreen, southern Norway. Holocene 2013, 23, 1151–1162. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Webb, N.R.; Coulson, S.J. Primary community assembly on land—the missing stages: Why are the heterotrophic organisms always there first? J. Ecol. 2002, 90, 569–577. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Coulson, S.J.; Webb, N.R. Invertebrate community assembly along proglacial chronosequences in the high Arctic. J. Anim. Ecol. 2004, 73, 556–568. [Google Scholar] [CrossRef]
- Ingimarsdóttir, M.; Ripa, J.; Magnúsdóttir, Ó.B.; Hedlund, K. Food web assembly in isolated habitats: A study from recently emerged nunataks, Iceland. Basic Appl. Ecol. 2013, 14, 174–183. [Google Scholar] [CrossRef]
- Ingimarsdóttir, M.; Ripa, J.; Hedlund, K. Corridor or drift fence? The role of medial moraines for fly dispersal over glacier. Polar Biol. 2013, 36, 925–932. [Google Scholar] [CrossRef]
- Sint, D.; Kaufmann, R.; Mayer, R.; Traugott, M. Resolving the predator first paradox: Arthropod predator food webs in pioneer sites of glacier forelands. Mol. Ecol. 2019, 28, 336–347. [Google Scholar] [CrossRef]
- Schallhart, N. Das Beutespektrum der räuberdominierten Kolonisatorengemeinschaften im Gletschervorfeld des Rotmoosferners (Ötztaler Alpen, Tirol). Diploma Thesis, Leopold-Franzens-Universität Innsbruck, Innsbruck, Austria, 2005; 104p. [Google Scholar]
- Ingimarsdóttir, M.; Caruso, T.; Ripa, J.; Magnúsdottir, O.B.; Migliorini, M.; Hedlund, K. Primary assembly of soil communities: Disentangling the effect of dispersal and local environment. Oecologia 2012, 170, 745–754. [Google Scholar] [CrossRef]
- Flø, D.; Hågvar, S. Aerial dispersal of invertebrates and mosses close to a receding alpine glacier in Southern Norway. Arct. Antarct. Alp. Res. 2013, 45, 481–490. [Google Scholar] [CrossRef]
- Coulson, S.J.; Hodkinson, I.D.; Webb, N.R. Aerial dispersal of invertebrates over a high-Arctic glacier foreland: Midtre Lovénbreen, Svalbard. Polar Biol. 2003, 26, 530–537. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Coulson, S.J.; Harrison, J.; Webb, N.R. What a wonderful web they weave: Spiders, nutrient capture and early ecosystem development in the high Arctic—Some counter-intuitive ideas on community assembly. Oikos 2001, 95, 349–352. [Google Scholar] [CrossRef]
- Hågvar, S. Primary succession of springtails (Collembola) in a Norwegian glacier foreland. Arct. Antarct. Alp. Res. 2010, 42, 422–429. [Google Scholar] [CrossRef]
- Gobbi, M.; Isaia, M.; De Bernardi, F. Arthropod colonisation of a debris-covered glacier. Holocene 2011, 21, 343–349. [Google Scholar] [CrossRef]
- Hittorf, M. Collembola of the glacier foreland—Pioneers colonizing moraines. Diploma Thesis, Leopold-Franzens-Universität Innsbruck, Innsbruck, Austria, 2017; 45p. [Google Scholar]
- Hågvar, S.; Flø, D. Succession and phenology of the generalist predator Mitopus morio (Fabricius, 1799) (Opiliones) in a glacier foreland. Nor. J. Entomol. 2015, 62, 210–215. [Google Scholar]
- Hågvar, S.; Ohlson, M.; Brittain, J.E. A melting glacier feeds aquatic and terrestrial invertebrates with ancient carbon and supports early succession. Arct. Antarct. Alp. Res. 2016, 48, 551–562. [Google Scholar] [CrossRef]
- Gobbi, M.; Lencioni, V. Glacial Biodiversity: Lessons from ground-dwelling and aquatic insects. In Glaciers; Godone, D., Ed.; Intech Open Access Publisher: London, UK, 2020. [Google Scholar] [CrossRef]
- Tenan, S.; Maffioletti, C.; Caccianiga, M.; Compostella, C.; Seppi, R.; Gobbi, M. Hierarchical models for describing space-for-time variations in insect population size and sex-ratio along a primary succession. Ecol. Model. 2016, 329, 18–28. [Google Scholar] [CrossRef]
- Hågvar, S. Primary succession in glacier forelands: How small animals conquer new land around melting glaciers. In International Perspectives on Global Environmental Change; Young, S.S., Silvern, S.E., Eds.; Intech Open Access Publisher: London, UK, 2012; pp. 151–172. Available online: www.intechopen.com (accessed on 18 September 2020).
- Kaufmann, R. Invertebrate succession on an Alpine glacier foreland. Ecology 2001, 82, 2261–2278. [Google Scholar] [CrossRef]
- Kaufmann, R. Glacier foreland colonisation: Distinguishing between short-term and long-term effects of climate change. Oecologia 2002, 130, 470–475. [Google Scholar] [CrossRef]
- Kaufmann, R.; Juen, A. Habitat use and niche segregation of the genus Nebria (Coleoptera: Carabidae) in the Austrian Alps. Mitt. Schweiz. Entomol. Ges. 2001, 74, 237–254. [Google Scholar]
- Hågvar, S.; Ohlson, M.; Flø, D. Animal successional pathways for about 200 years near a melting glacier: A Norwegian case study. In Glacier Evolution in a Changing World; Godone, D., Ed.; Intech Open Access Publisher: London, UK, 2017; pp. 147–176. Available online: www.intechopen.com (accessed on 18 September 2020).
- Raso, L.; Sint, D.; Mayer, R.; Plangg, S.; Recheis, T.; Brunner, S.; Kaufmann, R.; Traugott, M. Intraguild predation in pioneer predator communities of alpine glacier forelands. Mol. Ecol. 2014, 23, 3744–3754. [Google Scholar] [CrossRef]
- Bråten, A.T.; Flø, D.; Hågvar, S.; Hanssen, O.; Mong, C.E.; Aakra, K. Primary succession of surface active beetles and spiders in an alpine glacier foreland, central south Norway. Arct. Antarct. Alp. Res. 2012, 44, 2–15. [Google Scholar] [CrossRef]
- Sota, T. Altitudinal variation in life cycles of Carabid beetles: Life-cycle strategy and colonization in alpine zones. Arct. Alp. Res. 1996, 28, 441–447. [Google Scholar] [CrossRef]
- Gobbi, M.; Rossaro, B.; Vater, A.; De Bernardi, F.; Pelfini, M.; Brandmayr, P. Environmental features influencing Carabid beetle (Coleoptera) assemblages along a recently deglaciated area in the Alpine region. Ecol. Entomol. 2007, 32, 682–689. [Google Scholar] [CrossRef]
- Gobbi, M.; Ballarin, F.; Brambilla, M.; Compostella, C.; Isaia, M.; Losapio, G.; Maffioletti, C.; Seppi, R.; Tampucci, D.; Caccianiga, M. Life in harsh environments: Carabid and spider trait types and functional diversity on a debris-covered glacier and along its foreland. Ecol. Entomol. 2017, 42, 838–848. [Google Scholar] [CrossRef]
- Valle, B.; Ambrosini, R.; Caccianiga, M.; Gobbi, M. Nebria germari Heer, 1837—Insight about the distribution and ecology of a climate-sensitive species of the Eastern Alps. ARPHA Conf. Abstr. 2019, 2, e37074. [Google Scholar] [CrossRef]
- Gereben-Krenn, B.-A.; Krenn, H.W.; Strodl, M.A. Initial Colonization of New Terrain in an Alpine Glacier Foreland by Carabid Beetles (Carabidae, Coleoptera). Arct. Antarct. Alp. Res. 2011, 43, 397–403. [Google Scholar] [CrossRef]
- Janetschek, H. Tierische Successionen auf Hochalpinem Neuland. 1949. Available online: https://www.zobodat.at/pdf/BERI_48_49_0001-0215.pdf (accessed on 18 September 2020).
- Bernasconi, M.G.; Borgatti, M.S.; Tognetti, M.; Valle, B.; Caccianiga, M.; Casarotto, C.; Ballarin, F.; Gobbi, M. Checklist Ragionata Della Flora e Degli Artropodi (Coleoptera: Carabidae e Arachnida: Aranae) dei Ghiacciai Centrale e Occidentale del Sorapiss (Dolomiti d’Ampezzo). Frammenti Conoscere e Tutelare la Natura Bellunese 2019, 9, 49–65. [Google Scholar]
- Gobbi, M.; Brambilla, M. Patterns of spatial autocorrelation in the distribution and diversity of carabid beetles and spiders along Alpine glacier forelands. Ital. J. Zool. 2016, 83, 600–605. [Google Scholar] [CrossRef]
- Hågvar, S.; Solhøy, T.; Mong, C. Primary succession of soil mites (Acari) in a Norwegian glacier foreland, with emphasis on Oribatid species. Arct. Antarct. Alp. Res. 2009, 41, 219–227. [Google Scholar] [CrossRef]
- Gwiazdowicz, D.J.; Zawieja, B.; Olejniczak, I.; Skubała, P.; Gdula, A.K.; Coulson, S.J. Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic. Insects 2020, 11, 226. [Google Scholar] [CrossRef]
- Kaufmann, R.; Fuchs, M.; Gosterxeier, N. The soil fauna of an alpine glacier foreland: Colonization and succession. Arct. Antarct. Alp. Res. 2002, 34, 242–250. [Google Scholar] [CrossRef]
- Vater, A.E. Invertebrate and arachnid succession on selected glacier forelands in southern Norway. Ph.D. Thesis, University of Wales Swansea, Swansea, UK, 2006; 372p. [Google Scholar]
- Franzén, M.; Dieker, P. The influence of terrain age and altitude on the arthropod communities found on recently deglaciated terrain. Curr. Zool. 2014, 60, 203–220. [Google Scholar] [CrossRef]
- Gobbi, M.; Caccianiga, M.; Cerabolini, B.; Bernardi, F.; Luzzaro, A.; Pierce, S. Plant adaptive responses during primary succession are associated with functional adaptations in ground beetles on deglaciated terrain. Community Ecol. 2010, 11, 223–231. [Google Scholar] [CrossRef]
- Gobbi, M.; De Bernardi, F.; Pelfini, M.; Rossaro, B.; Brandmayr, P. Epigean arthropod succession along a 154-year glacier foreland chronosequence in the Forni Valley (Central Italian Alps). Arct. Antarct. Alp. Res. 2006, 38, 357–362. [Google Scholar] [CrossRef]
- Gobbi, M.; Fontaneto, D.; De Bernardi, F. Influence of climate changes on animal communities in space and time: The case of spider assemblages along an alpine glacier foreland. Glob. Chang. Biol. 2006, 12, 1985–1992. [Google Scholar] [CrossRef]
- Pelfini, M.; Gobbi, M. Enhancement of the ecological value of Forni Glacier (Central Alps) as a possible geomorphosite: New data from arthropod communities. Geografia Fisica e Dinamica Quaternaria. 2005, 28, 211–217. [Google Scholar]
- Tribsch, A.; Schönswetter, P. Patterns of endemism and comparative phylogeography confirm palaeo-environmental evidence for Pleistocene refugia in the Eastern Alps. Taxon 2003, 52, 477–497. [Google Scholar] [CrossRef]
- Porco, D.; Greenslade, P.; Janion, C.; Skarzybski, D. Challenging species delimitation in Collembola: Cryptic diversity among common springtails unveiled by DNA barcoding. Invertebr. Syst. 2012, 26, 470. [Google Scholar] [CrossRef]
- Vater, A.E.; Matthews, J.A. Succession of pitfall-trapped insects and arachnids on eight Norwegian glacier forelands along an altitudinal gradient: Patterns and models. Holocene 2015, 25, 108–129. [Google Scholar] [CrossRef]
- Hågvar, S.; Ohlson, M. Ancient carbon from a melting glacier gives high 14C age in living pioneer invertebrates. Sci. Rep. 2013, 3, 2820. [Google Scholar] [CrossRef] [PubMed]
- Füreder, L.; Schütz, C.; Wallinger, M.; Burger, R. Physico-chemistry and aquatic insects of a glacier-fed and a spring-fed alpine stream. Freshw. Biol. 2001, 46, 1673–1690. [Google Scholar] [CrossRef]
- König, T.; Kaufmann, R.; Scheu, S. The formation of terrestrial food webs in glacier foreland: Evidence for the pivotal role of decomposer prey and intraguild predation. Pedobiologia 2011, 54, 147–152. [Google Scholar] [CrossRef]
- Stubbins, A.; Hood, E.; Raymond, P.A.; Aiken, G.R.; Sleighter, R.L.; Hernes, P.J.; Butman, D.; Hatcher, P.G.; Striegl, R.G.; Schuster, P.; et al. Anthropogenic aerosols as a source of ancient dissolved organic matter in glaciers. Nat. Geosci. 2012, 5, 198–201. [Google Scholar] [CrossRef]
- Dresch, P.; Falbesoner, J.; Ennemoser, C.; Hittorf, M.; Kuhnert, R.; Peintner, U. Emerging from the ice-fungal communities are diverse and dynamic in earliest soil developmental stages of a receding glacier. Environ. Microbiol. 2019, 21, 1864–1880. [Google Scholar] [CrossRef]
- Vater, A.E. Insect and arachnid colonization on the Storbreen glacier foreland, Jotunheimen, Norway: Persistence of taxa suggests an alternative model of succession. Holocene 2012, 22, 1123–1133. [Google Scholar] [CrossRef]
- Ingimarsdóttir, M.; Michelsen, A.; Ripa, J.; Hedlund, K. Food sources of early colonising arthropods: The importance of allochthonous input. Pedobiologia 2014, 57, 21–26. [Google Scholar] [CrossRef]
- Pizzolotto, R.; Gobbi, M.; Brandmayr, P. Changes in ground beetle assemblages above and below the treeline of the Dolomites after almost 30 years (1980/2009). Ecol. Evol. 2014, 4, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, M.; Gobbi, M. A century of chasing the ice: Delayed colonisation of ice-free sites by ground beetles along glacier forelands in the Alps. Ecography 2013, 37, 33–42. [Google Scholar] [CrossRef]
- Pelfini, M.; Diolaiuti, G.A.; Leonelli, G.; Bozzoni, M.; Bressan, N.; Brioschi, D.; Riccardi, A. The influence of glacier surface processes on the short-term evolution of supraglacial tree vegetation: A case study of the Miage Glacier, Italian Alps. Holocene 2012, 22, 847–856. [Google Scholar] [CrossRef]
- Cauvy-Fraunié, S.; Dangles, O. A global synthesis of biodiversity responses to glacier retreat. Nat. Ecol. Evol. 2019, 3, 1–11. [Google Scholar] [CrossRef]
- Darcy, J.L.; Schmidt, S.K.; Knelman, J.E.; Cleveland, C.C.; Castle, S.C.; Nemergut, D.R. Phosphorus, not nitrogen, limits plants and microbial primary producers following glacial retreat. Sci. Adv. 2018, 4, 1–7. [Google Scholar] [CrossRef]
- Carlson, M.L.; Flagstad, L.A.; Gillet, F.; Mitchell, E.A.D. Community development along a proglacial chronosequence: Are above-ground and below-ground community structure controlled more by biotic than abiotic factors? J. Ecol. 2010, 98, 1084–1095. [Google Scholar] [CrossRef]
- Azzoni, R.S.; Franzetti, A.; Fontaneto, D.; Zullini, A.; Ambrosini, R. Nematodes and rotifers on two Alpine debris-covered glaciers. Ital. J. Zool. 2015, 82, 616–623. [Google Scholar] [CrossRef]
- Matthews, J.A.; Vater, A.E. Pioneer zone geo-ecological change: Observations from a chronosequence on the Storbreen glacier foreland, Jotunheimen, southern Norway. Catena 2015, 135, 219–230. [Google Scholar] [CrossRef]










| Species | Group | Bio-Film | Fungal Hyphae | Bryo-Phytes | Vascular Plants | Arthro-Pods | Ancient Carbon via Chironomidae |
|---|---|---|---|---|---|---|---|
| Agrenia bidenticulata | Collembola | x | |||||
| Desoria olivacea | Collembola | x | |||||
| Isotoma viridis | Collembola | x | x | ||||
| Lepidocyrtus lignorum | Collembola | x | |||||
| Bourletiella hortensis | Collembola | x | x | ||||
| Simplocaria metallica | Coleoptera | x | |||||
| Amara alpina | Coleoptera | x | x | x | |||
| Amara quenseli | Coleoptera | x | x | ||||
| Nebria nivalis | Coleoptera | x | x | ||||
| Bembidion hastii | Coleoptera | x | x | ||||
| Mitopus morio | Opiliones | x | x |
| Country and Locality | Iceland, Nunataks | Southern Norway, Hardangerjøkulen | Svalbard | ||||
|---|---|---|---|---|---|---|---|
| Altitude (m) | 460–728 | 1400 | 50 | ||||
| Reference | [19] | [23,30,45] | [14] | [46] | |||
| Age of ground (years) | <10 | 0 | 3 | 32–34 | 2 | 16 | 0 |
| COLLEMBOLA | |||||||
| SYMPHYPLEONA | |||||||
| Bourletiella hortensis | X | X | |||||
| Heterosminthurus claviger | X | ||||||
| Sminthurides inequalis | X | X | |||||
| Sminthurides malmgreni | X | ||||||
| ENTOMOBRYOMORPHA | |||||||
| Agrenia bidenticulata | X | X | X | X | |||
| Desoria infuscata | X | X | X | ||||
| Desoria olivacea | X | X | X | ||||
| Desoria tolya | X | X | X | ||||
| Folsomia brevicauda | X | ||||||
| Folsomia quadrioculata | X | X | X | ||||
| Isotoma anglicana | X | X | X | ||||
| Isotoma viridis | X | X | |||||
| Lepidocyrtus lignorum | X | X | X | ||||
| Pseudisotoma sensibilis | X | ||||||
| Tetracanthella arctica | X | ||||||
| Tetracanthella wahlgreni | X | ||||||
| PODUROMORPHA | |||||||
| Ceratophysella sp. | X | ||||||
| Ceratophysella scotica | X | X | |||||
| Hypogastrura tullbergi | X | X | |||||
| Hypogastrura concolor | X | ||||||
| Mesaphorura sp. | X | ||||||
| Micranurida pygmaea | X | ||||||
| Oligophorura groenlandica | X | ||||||
| Oligophorura schoetti | X | ||||||
| Protaphorura sp. | X | ||||||
| ACARI | |||||||
| ORIBATIDA | |||||||
| Camisia anomia | X | X | |||||
| Liochthonius cf. sellnicki | X | ||||||
| Liochthonius clavatus | X | ||||||
| Liochthonius strenzkei | X | ||||||
| Pantelozetes paolii | X | ||||||
| Tectocepheus velatus | X | X | X | ||||
| PROSTIGMATA | X | X | |||||
| GAMASIDA | X | X | X | ||||
| Country and Locality | W-Sorapiss, Italy | C-Sorapiss, Italy | Agola Glacier, Italy | Amola Glacier, Italy | Rotmoos Glacier, Austria | Hintereis Glacier, Austria | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Altitude (m) | 2300–2400 | 2200–2300 | 2500–2600 | 2500–2680 | 2450 | 2400 | ||||||||
| Reference | Valle et al. (unpublished data) | Valle et al. (unpublished data) | Valle et al. (unpublished data) | [47] | [25] | [42] | ||||||||
| Age of ground (years) | On ice | <50 | 50 | On ice | 40 | On ice | <25 | <50 | On ice | <18 | <40 | 0–3 | 0–1 | 2–4 |
| SYMPHYPLEONA | ||||||||||||||
| Symphypleona indet. | X | X | X | X | X | X | ||||||||
| Heterosminthurus diffusus | X | X | X | X | ||||||||||
| Sminthurinus trinotatus | X | |||||||||||||
| Bourletiella repanda | X | X | ||||||||||||
| ENTOMOBRYO-MORPHA | ||||||||||||||
| Orchesella cf. alticola | X | X | X | X | X | X | X | X | X | X | X | |||
| Orchesella bifasciata | X | X | ||||||||||||
| Orchesella sp. | X | |||||||||||||
| Entomobryanivalis | X | |||||||||||||
| Isotomidae indet. | X | X | X | X | X | X | ||||||||
| Folsomia manolachei | X | |||||||||||||
| Isotoma viridis | X | |||||||||||||
| Desoria saltans | X | |||||||||||||
| Parisotoma notabilis | X | |||||||||||||
| Proisotoma schoetti | X | X | ||||||||||||
| Proisotoma crassicauda | X | X | ||||||||||||
| Tetracanthella specifica | X | |||||||||||||
| Desoria nivalis | X | |||||||||||||
| Pachyotoma crassicauda | X | X | ||||||||||||
| Pachyotoma pseudorecta | X | |||||||||||||
| Isotomurus pseudopalustris | X | |||||||||||||
| Isotomurus maculatus | X | |||||||||||||
| Isotomurus palliceps | X | X | ||||||||||||
| Isotomurus alticolus | X | |||||||||||||
| Lepidocyrtus curvicollis gr. | X | |||||||||||||
| Lepidocyrtus sp. | X | X | X | X | X | |||||||||
| Tomocerus cf. minor | X | X | X | |||||||||||
| PODUROMORPHA | ||||||||||||||
| Pseudachorudina alpina | X | X | ||||||||||||
| Ceratophysella tuberculata | X | |||||||||||||
| Micranurida pygmaea | X | |||||||||||||
| Mesaphorura critica | X | |||||||||||||
| Hypogastrura cf. socialis | X | |||||||||||||
| Hypogastrura parva | X | |||||||||||||
| Hypogastruridae indet. | X | |||||||||||||
| Country and Locality | Jostedalen, Norway | Jotunheimen, Norway | Hardanger-Vidda, Norway | North-Ern Swe-Den | Ice-Land | Sval-Bard | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glacier name | Aus-ter-dals-breen | Berg-set-breen | Fåberg-støls-breen | Bø-dals-breen | Stygge-dals-breen | Bøver-breen | Stor-breen | Vesle-juv-breen | Hardanger-jøkulen | Ålma-jallo-jeknaglacier | Nu-na-taks | MidtreLovén-bre | ||
| Altitude (m) | 320–390 | 400–500 | 480–620 | 560–600 | 1270 | 1400 | 1400 | 1860 | 1400 | 1180–1344 | 460–728 | 50 | ||
| Reference [numbers] | [48] | [48] | [48] | [48] | [48] | [48] | [48] | [48] | [36] | [49] | [19] | [14] | ||
| Climatic zone | Sub-alpine | Sub-alpine | Sub-alpine | Sub-alpine | Low alpine | Low/mid-alpine | Low/mid-alpine | High alpine | Mid-alpine | Low/mid-alpine | Arctic | High arctic | ||
| Age of ground (years) | <20 | <20 | <20 | <20 | <20 | <20 | <20 | <20 | 3 | 40 | <40 | <10 | 2 | 16 |
| COLEOPTERA, CARABIDAE | ||||||||||||||
| Amara alpina | x | x | x | x | x | x | x | |||||||
| Amara quenseli | x | x | x | x | x | |||||||||
| Nebria sp. | sp. | sp. | sp. | N. nivalis | sp. | sp. | N. nivalis | N. nivalis N. rufescens | N. nivalis N. rufescens | |||||
| Bembidion sp. | B. fellmanni | B. fellmanni | B. fellmanni | B. hastii | B. hastii | B. hastii | ||||||||
| Patrobus septentrionis | x | |||||||||||||
| Notiophilus aquaticus | x | x | ||||||||||||
| Miscodera arctica | x | x | x | |||||||||||
| COLEOPTERA, BYRRHIDAE | ||||||||||||||
| Simplocaria metallica | x | x | ||||||||||||
| Byrrhus arietinus | x | x | ||||||||||||
| COLEOPTERA, CURCULIONIDAE | ||||||||||||||
| Otiorhynchus nodosus | x | |||||||||||||
| Otiorhynchus arcticus | x | |||||||||||||
| COLEOPTERA, STAPHYLINIDAE | ||||||||||||||
| Oxypoda annularis | x | |||||||||||||
| Boreaphilus henningianus | x | |||||||||||||
| Geodromicus longipes | x | x | ||||||||||||
| Acidota crenata | x | |||||||||||||
| Anthophagus alpinus | x | |||||||||||||
| Arpedium quadrum | x | x | ||||||||||||
| Coryphiomorphus hyperboreus | x | |||||||||||||
| Olophrum boreale | x | |||||||||||||
| Tachinus elongatus | x | |||||||||||||
| Quedius sp. | x | x | ||||||||||||
| COLEOPTERA, HYDROPHILIDAE | ||||||||||||||
| Helophorus glacialis | x | |||||||||||||
| COLEOPTERA, DYTISCIDAE | ||||||||||||||
| Agabus bipustulatus | x | |||||||||||||
| Agabus thomsoni/lapponicus | x | |||||||||||||
| COLEOPTERA, CHRYSOMELIDAE | ||||||||||||||
| Phratora polaris | x | |||||||||||||
| COLEOPTERA, NITIDULIDAE | ||||||||||||||
| Meligethes aeneus | x | |||||||||||||
| COLEOPTERA, SILPHIDAE | ||||||||||||||
| Thanatophilus lapponicus | x | |||||||||||||
| COLEOPTERA, LEIODIDAE | ||||||||||||||
| Catops tristis | x | x | ||||||||||||
| OPILIONES, PHALANGIIDAE | ||||||||||||||
| Mitopus morio | x | x | x | x | x | x | x | x | x | x | x | |||
| ARANEAE, LYCOSIDAE | ||||||||||||||
| Pardosa trailli | x | x | x | x | ||||||||||
| Pardosa palustris | x | x | ||||||||||||
| Pardosa pullata | x | |||||||||||||
| ARANEAE, GNAPHOSIDAE | ||||||||||||||
| Zelotes subterraneus | x | |||||||||||||
| Gnaphosa leporina | x | |||||||||||||
| Micaria alpina | x | |||||||||||||
| ARANEAE, LINYPHIIDAE | ||||||||||||||
| Gonatium rubellum | x | |||||||||||||
| Oedothorax retusus | x | |||||||||||||
| Erigone longipalpis | x | x | x | x | ||||||||||
| Erigone tirolensis | x | x | ||||||||||||
| Erigone psychrophila | x | x | ||||||||||||
| Erigone arctica | x | x | x | |||||||||||
| Meioneta nigripes | x | x | x | x | ||||||||||
| Collinsia spetsbergensis | x | x | x | |||||||||||
| Collinsia holmgreni | x | x | x | |||||||||||
| Islandiana princeps | x | |||||||||||||
| Improphantes complicatus | x | |||||||||||||
| Country and Glacier Name | Rotmoos-tal, Austria | Cedec Glacier, Italy | Forni Valley, Italy | Forni Valley, Italy | Trobio Glacier, Italy | W-Sorapiss Glacier, Italy | C-Sorapiss Glacier, Italy | D’Agola Glacier, Italy | Amola Glacier, Italy | Hintereis, Austria | Hornkees, Austria | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Altitude (m) | 2500 | 2700 | 2500 | 2500 | 2500 | 2300–2400 | 2200–2300 | 2500–2600 | 2500–2680 | 2400 | 2200 | ||||||||||
| Reference | [31] and Kauf-mann, unpubl. | [50] | [38] | [51,52,53] | [10] | [43] | [43] | Gobbi et al. unpubl. | [39] | [42] | [41] | ||||||||||
| Age of ground (years) | <10 | <20 | <5 | On ice | 24 | <30 | On ice | <50 | 50 | On ice | 40 | On ice | <20 | <50 | 50 | On ice | <18 | 0–1 | 2–4 | 2 | 6–10 |
| COLEOPTERA, CARABIDAE | |||||||||||||||||||||
| Nebria germari | X | X | X | X | X | X | X | X | X | X | X | X | X | X | |||||||
| Nebria jockischii | X | X | X | X | X | X | |||||||||||||||
| Nebria hellwigi | X | ||||||||||||||||||||
| Nebria rufescens | X | X | |||||||||||||||||||
| Oreonebria castanea | X | X | X | X | |||||||||||||||||
| Oreonebria diaphana | X | X | X | X | X | ||||||||||||||||
| Oreonebria soror | X | X | |||||||||||||||||||
| Amara quenseli | X | X | X | X | |||||||||||||||||
| Amara erratica | X | ||||||||||||||||||||
| Pterostichus jurinei | X | ||||||||||||||||||||
| Carabus sylvestris | X | X | |||||||||||||||||||
| Carabus adamellicola | X | X | X | ||||||||||||||||||
| Trechus dolomitanus | X | X | |||||||||||||||||||
| Sinechostichus doderoi | X | ||||||||||||||||||||
| Princidium bipunctatum | X | ||||||||||||||||||||
| Bembidion geniculatum | X | ||||||||||||||||||||
| COLEOPTERA, BYRRHIDAE | |||||||||||||||||||||
| Simplocaria semistriata | X | ||||||||||||||||||||
| COLEOPTERA, ELATERIDAE | |||||||||||||||||||||
| Fleutiauxellus maritimus | X | X | X | X | |||||||||||||||||
| COLEOPTERA, HYDROPHILIDAE | |||||||||||||||||||||
| Helophorus glacialis | X | ||||||||||||||||||||
| COLEOPTERA, STAPHYLINIDAE | |||||||||||||||||||||
| Eusphalerum anale | X | ||||||||||||||||||||
| OPILIONES, PHALANGIIDAE | |||||||||||||||||||||
| Dicranopalpus gasteinensis | X | ||||||||||||||||||||
| Mitopus glacialis | X | X | |||||||||||||||||||
| Gyas annulatus | X | ||||||||||||||||||||
| ARANEAE, LYCOSIDAE | |||||||||||||||||||||
| Pardosa nigra | X | X | X | X | X | ||||||||||||||||
| Acantholycosa pedestris | X | X | X | X | X | X | |||||||||||||||
| Pardosa saturatior ARANEAE, | X | X | |||||||||||||||||||
| GNAPHOSIDAE | |||||||||||||||||||||
| Drassodex heeri | X | X | X | ||||||||||||||||||
| ARANEAE, ERIGONIDAE | |||||||||||||||||||||
| Erigone atra | X | ||||||||||||||||||||
| Erigone tirolensis | X | ||||||||||||||||||||
| Mecynargus paetulus | X | ||||||||||||||||||||
| Entelecara media | X | X | |||||||||||||||||||
| Walckenaeria vigilax | X | ||||||||||||||||||||
| ARANEAE, THERIDIIDAE | |||||||||||||||||||||
| Robertus arundineti | X | ||||||||||||||||||||
| ARANEAE, AGELENIDAE | |||||||||||||||||||||
| Thanatus formicinus | X | ||||||||||||||||||||
| Coelotes mediocris | X | ||||||||||||||||||||
| Coelotes pickardiitirolensis | X | X | |||||||||||||||||||
| ARANEAE, THOMISIDAE | |||||||||||||||||||||
| Xysticus audax | X | ||||||||||||||||||||
| Xysticus alpinus | X | X | X | ||||||||||||||||||
| ARANEAE, LINYPHIIDAE | |||||||||||||||||||||
| Meioneta rurestris | X | ||||||||||||||||||||
| Meioneta gulosa | X | ||||||||||||||||||||
| Meioneta nigripes | X | ||||||||||||||||||||
| Lepthyphantes variabilis | X | ||||||||||||||||||||
| Agyneta rurestris | X | X | X | X | X | ||||||||||||||||
| Mughiphantes brunnerii | X | X | |||||||||||||||||||
| Mughiphantes handschini | X | X | X | ||||||||||||||||||
| Mughiphantes cf. merretti | X | X | |||||||||||||||||||
| Mughiphantes pulcher | X | ||||||||||||||||||||
| Tenuiphanthes jacksonoides | X | ||||||||||||||||||||
| Diplocephalus helleri | X | X | X | X | X | X | |||||||||||||||
| Troglohyphantes fagei | X | X | |||||||||||||||||||
| Erigone dentipalpis | X | ||||||||||||||||||||
| Oreonetides glacialis | X | ||||||||||||||||||||
| Janetschekia lesserti | X | ||||||||||||||||||||
| Country and Reference | Method | Collembola | Chironomid Midges | Other Predators | Foreign Prey? | Locally Produced Prey? |
|---|---|---|---|---|---|---|
| Norway [57] | Microscope study of gut content | Rare | Common | Rare | Yes, but rare? | Yes, locally produced chironomid midges |
| Austria [17] | DNA study of gut content | Common | Rare | Common | Yes, Mostly Dipterans and aphids | Yes, locally produced Collembola, and maybe other predators |
| Family | Genus/Species | 0–3 Years | 9–14 Years | 18–25 Years | On Glacier | Total Numbers |
|---|---|---|---|---|---|---|
| Bourletiellidae | Heterosminthurus diffusus | 1927 | 320 | 32 | 5 | 2284 |
| Entomobryidae | Orchesella sp. | 375 | 61 | 9 | 2 | 447 |
| Lepidocyrtus cf. curvicollis | 10 | 104 | 114 | 2 | 230 | |
| Isotomidae | Pachyotoma pseudorecta | 33 | 1 | 3 | 0 | 37 |
| Isotomurus cf. alticolus | 57 | 1 | 41 | 1 | 100 | |
| Isotomurus palliceps | 1 | 24 | 11 | 0 | 36 | |
| Agrenia bidenticulata | 0 | 0 | 1 | 1 | 2 | |
| Desoria sp. | 0 | 0 | 0 | 2 | 2 | |
| Isotomidae indet. | 0 | 0 | 9 | 0 | 9 | |
| Hypogastruridae | Hypogastruridae sp. 1 | 6 | 4 | 0 | 0 | 10 |
| Hypogastruridae sp. 2 | 0 | 0 | 0 | 3 | 3 | |
| Hypogastruridae sp. 3 | 0 | 0 | 1 | 0 | 1 | |
| Hypogastrura sp. | 0 | 0 | 0 | 1 | 1 |
| Age (Year) | 0 | 3 | 32–36 | 41–47 |
|---|---|---|---|---|
| Sampling method | Flotation | Pitfall | Pitfall | Pitfall |
| Agrenia bidenticulata | 84.6 | 24.7 | ||
| Desoria infuscata | 15.4 | 1.5 | 6.5 | |
| Bourletiella hortensis | 59.9 | 1.1 | ||
| Isotoma viridis | 5.2 | 28.3 | 21.3 | |
| Lepidocyrtus lignorum | 0.4 | 50.0 | 65.3 | |
| Desoria olivacea | 8.0 | 8.7 | 4.5 | |
| Desoria tolya | 0.2 | 4.3 | 3.0 | |
| Ceratophysella scotica | 0.1 | 1.1 | 6.1 | |
| Number of animals sampled | 26 | 1465 | 92 | 66 |
| Species | Gut Content | 3 Years | 30–40 Years | 63 Years |
|---|---|---|---|---|
| Isotoma viridis | Mineral particles | ++++ | ++++ | + |
| Diatom algae | + | - | - | |
| Fungal hyphae | + | ++ | ++++ | |
| Fungal spores | + | ++ | - | |
| Lepidocyrtus lignorum | Mineral particles | ++++ | ++++ | + |
| Diatom algae | - | - | - | |
| Fungal hyphae | + | ++ | +++ | |
| Fungal spores | - | ++ | +++ |
| Species | Group | Climate | Habitat | Food | ||||
|---|---|---|---|---|---|---|---|---|
| TERRESTRIAL | Specialist | Generalist | Specialist | Generalist | Specialist | Generalist | ||
 | Diatom algaein biofilm | Algae | Open | |||||
 | Agrenia bidenticulata | Collem-bola | Cold adapted | (Open) | Biofilm | |||
 | Nebria nivalis | Cara-bidae | Cold adapted | (Open) | Generalist predator | |||
 | Bembidion hastii | Cara-bidae | Open | Generalist predator | ||||
 | Pardosa trailli | Lyco-sidae | Open | Generalist predator | ||||
 | Mitopus morio | Opili-ones | Generalist | Generalist | Generalist predator | |||
 | Pioneer mosses | Moss | Open | |||||
 | Bourletiella hortensis | Collem-bola | Open | Moss | ||||
 | Simplocaria metallica | Byrr-hidae | (Open) | Moss | ||||
 | Amara alpina | Cara-bidae | (Alpine) | (Moss) | Omnivore | |||
 | Amara quenseli | Cara-bidae | (Moss) | Omnivore | ||||
 | Isotoma viridis | Collem-bola | Biofilm and fungal hyphae | |||||
| AQUATIC | ||||||||
 | Chironomidae larvae | Diptera | Young ponds | Bio-available ancient carbon | ||||
 | Tipulidae larvae | Diptera | Young ponds | Bio-available ancient carbon | ||||
 | Agabus bipustulatus | Dytis-cidae | Young ponds | Predator on Chirono-mid larvae | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hågvar, S.; Gobbi, M.; Kaufmann, R.; Ingimarsdóttir, M.; Caccianiga, M.; Valle, B.; Pantini, P.; Fanciulli, P.P.; Vater, A. Ecosystem Birth near Melting Glaciers: A Review on the Pioneer Role of Ground-Dwelling Arthropods. Insects 2020, 11, 644. https://doi.org/10.3390/insects11090644
Hågvar S, Gobbi M, Kaufmann R, Ingimarsdóttir M, Caccianiga M, Valle B, Pantini P, Fanciulli PP, Vater A. Ecosystem Birth near Melting Glaciers: A Review on the Pioneer Role of Ground-Dwelling Arthropods. Insects. 2020; 11(9):644. https://doi.org/10.3390/insects11090644
Chicago/Turabian StyleHågvar, Sigmund, Mauro Gobbi, Rüdiger Kaufmann, María Ingimarsdóttir, Marco Caccianiga, Barbara Valle, Paolo Pantini, Pietro Paolo Fanciulli, and Amber Vater. 2020. "Ecosystem Birth near Melting Glaciers: A Review on the Pioneer Role of Ground-Dwelling Arthropods" Insects 11, no. 9: 644. https://doi.org/10.3390/insects11090644
APA StyleHågvar, S., Gobbi, M., Kaufmann, R., Ingimarsdóttir, M., Caccianiga, M., Valle, B., Pantini, P., Fanciulli, P. P., & Vater, A. (2020). Ecosystem Birth near Melting Glaciers: A Review on the Pioneer Role of Ground-Dwelling Arthropods. Insects, 11(9), 644. https://doi.org/10.3390/insects11090644