Strong Gene Flow Undermines Local Adaptations in a Host Parasite System
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
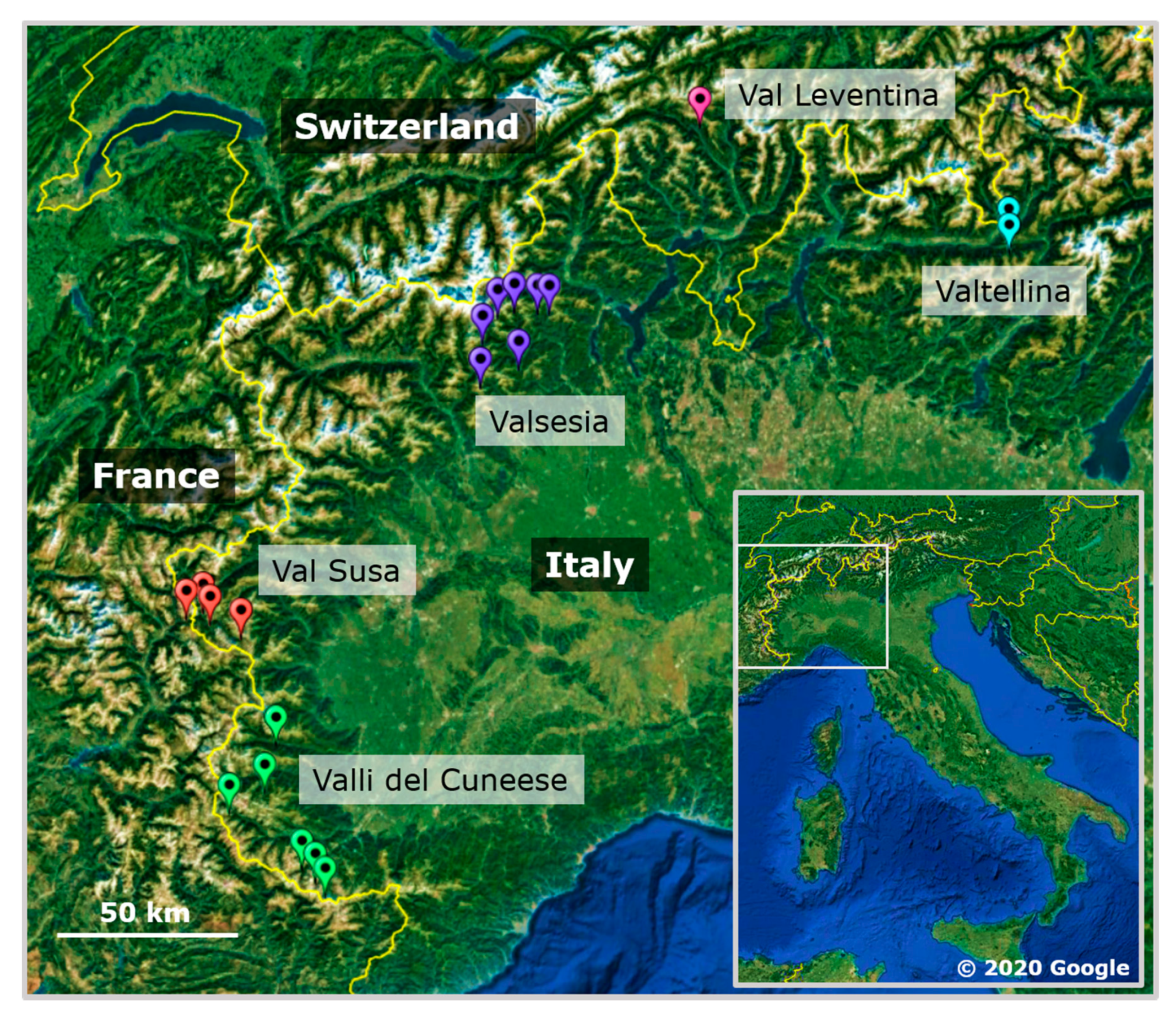
2.1. Sampling
2.2. Molecular Analyses
2.3. Population Genetic Analyses
2.4. Phylogenetic Analyses
3. Results
3.1. Genetic Variation in the Host and the Parasite
3.1.1. Nuclear Markers
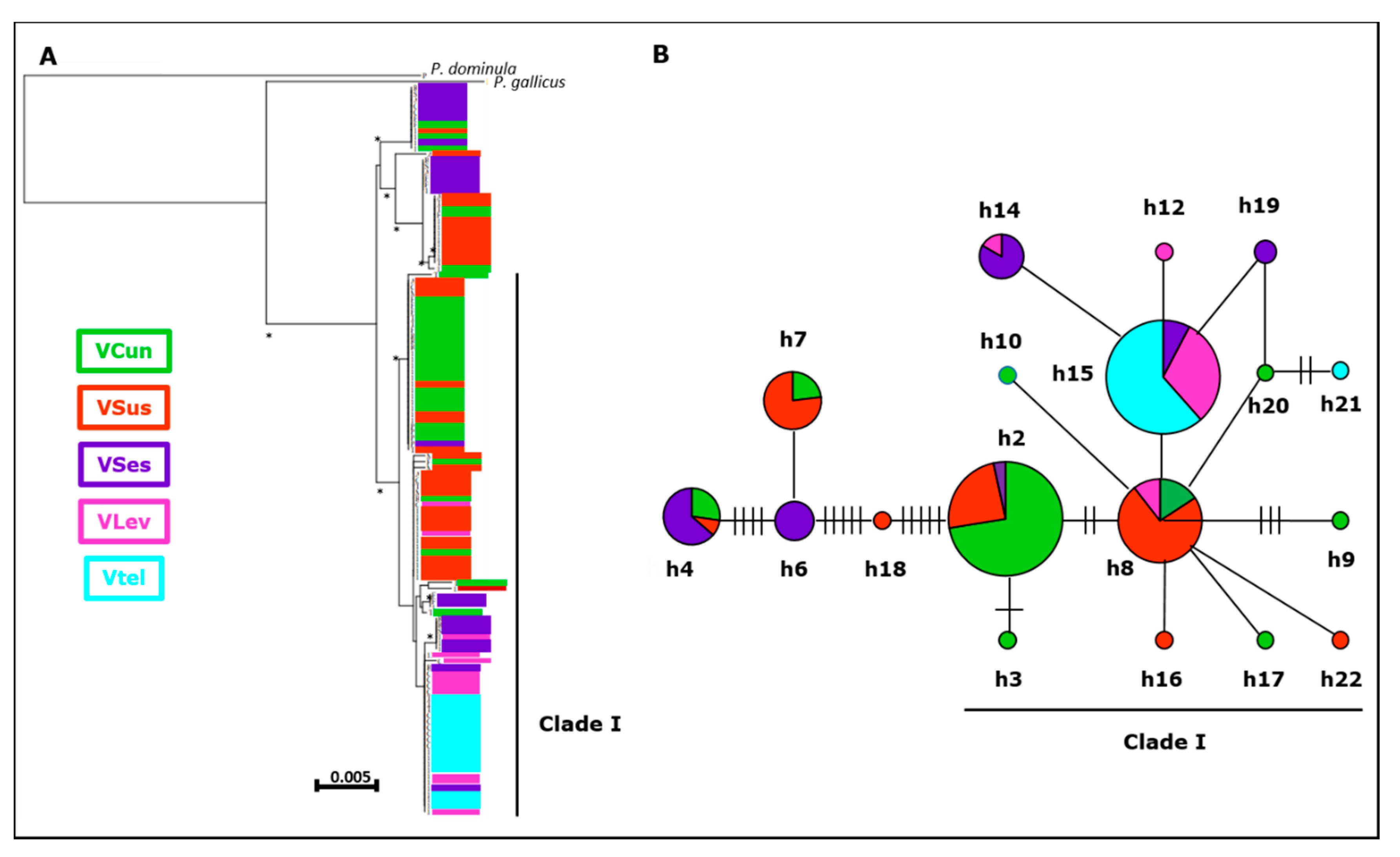
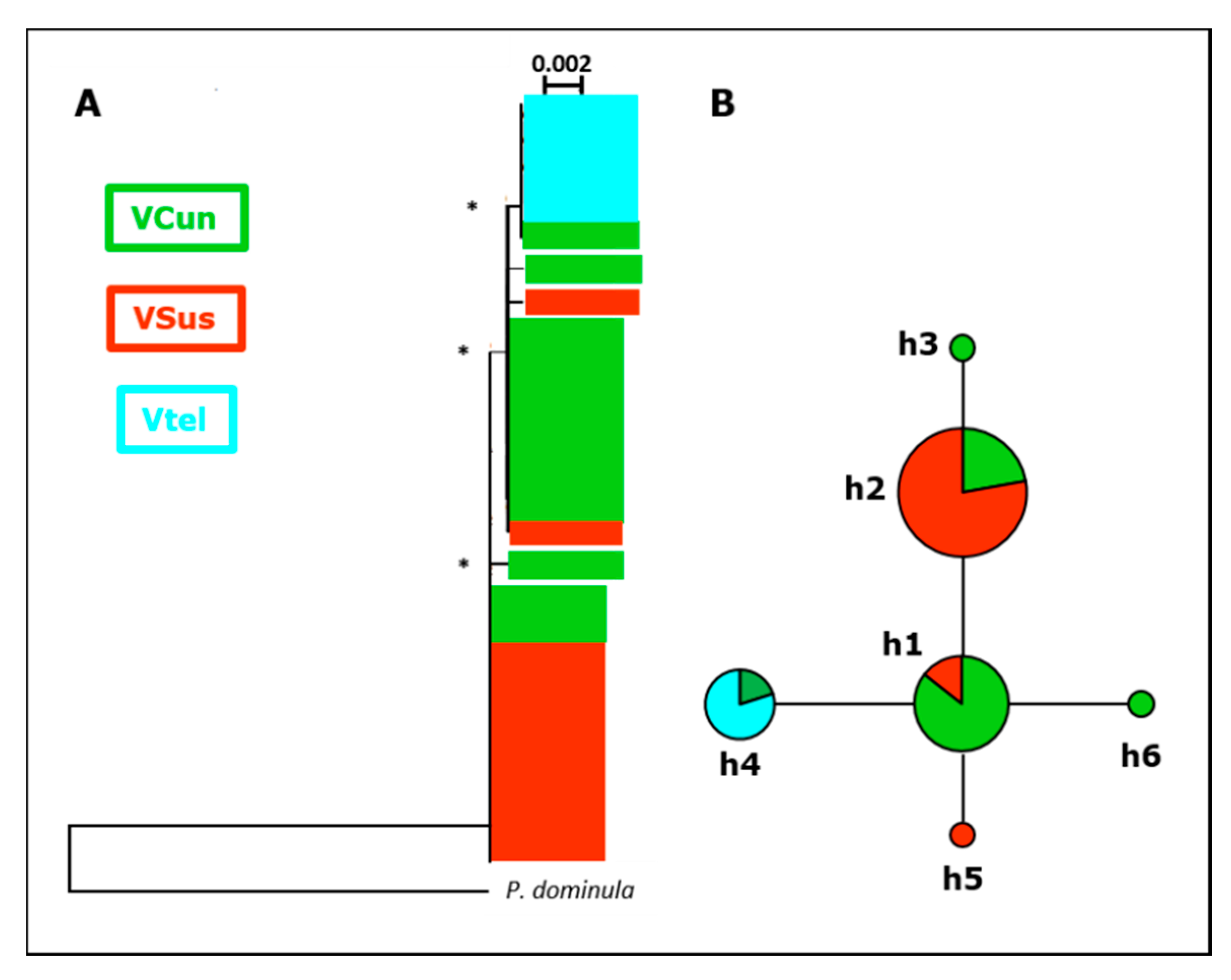
3.1.2. Mitochondrial Sequences
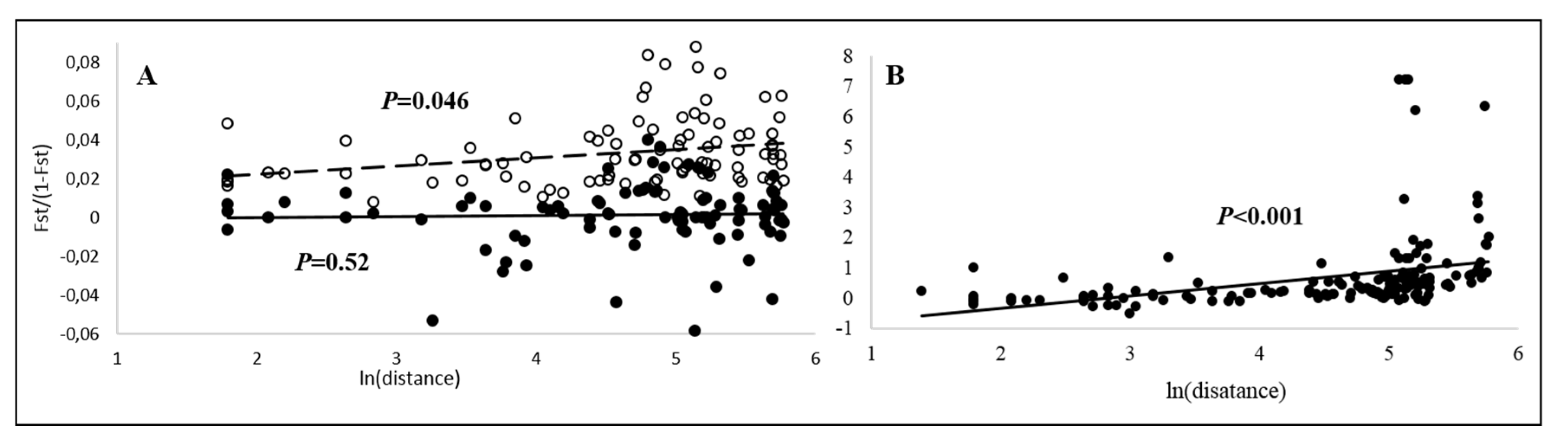
3.2. Spatial Population Structure
3.3. Phylogenetic Analyses
4. Discussion
4.1. Spatial Genetic Structure
4.1.1. Gene Flow and Dispersal
4.1.2. Selection of the Host Nest by the Parasite
4.2. Genetic Variation in the Host and the Parasite
4.3. Signatures of Historical Gene Flow
4.4. Assumptions Revisited
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ehrich, P.R.; Raven, P.H. Butterflies and plants: A study in coevolution. Evolution 1964, 18, 586–608. [Google Scholar] [CrossRef]
- Janzen, D.H. When is it Coevolution? Evolution 1980, 34, 611–612. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.N. The Coevolutionary Process; University of Chicago Press: Chicago, IL, USA, 1994; 383p. [Google Scholar]
- Dawkins, R.; Krebs, J.R. Arms races between and within species. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1979, 202, 489–511. [Google Scholar]
- Davies, N.B.; Bourke, A.F.G.; Brooke, M.D.L. Cuckoos and parasitic ants: Interspecific brood parasitism as an evolutionary arms race. Trends Ecol. Evol. 1989, 4, 274–278. [Google Scholar] [CrossRef]
- Dybdahl, M.F.; Storfer, A. Parasite local adaptation: Red queen versus suicide king. Trends Ecol. Evol. 2003, 18, 523–530. [Google Scholar] [CrossRef]
- Gandon, S.; Michalakis, Y. Local adaptation, evolutionary potential and host-parasite coevolution: Interactions between migration, mutation, population size and generation time. J. Evol. Biol. 2002, 15, 451–462. [Google Scholar] [CrossRef]
- Kaltz, O.; Shykoff, J.A. Local adaptation in host-parasite systems. Heredity 1998, 81, 361–370. [Google Scholar] [CrossRef]
- Gandon, S.; Capowiez, Y.; Dubois, Y.; Michalakis, Y.; Olivieri, I. Local adaptation and gene-for-gene coevolution in a metapopulation model. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1996, 263, 1003–1009. [Google Scholar]
- Haldane, J.B.S. Theoretical genetics of autopolyploids. J. Genet. 1930, 22, 359–372. [Google Scholar] [CrossRef]
- Wright, S. Evolution and the Genetics of Populations. Vol2 The Theory of Gene Frequencies; The Chicago University Press: Chicago, IL, USA, 1969; 520p. [Google Scholar]
- Swindell, W.R.; Bouzat, J. Gene flow and adaptive potential in Drosophila melanogaster. Conserv. Genet. 2006, 7, 79–89. [Google Scholar] [CrossRef]
- Tigano, A.; Friesen, V.L. Genomics of local adaptation with gene flow. Mol. Ecol. 2016, 25, 2144–2164. [Google Scholar] [CrossRef] [PubMed]
- Hämälä, T.; Savolainen, O. Genomic Patterns of Local Adaptation under Gene Flow in Arabidopsis lyrata. Mol. Biol. Evol. 2019, 36, 2557–2571. [Google Scholar] [CrossRef]
- Thompson, J.N. The Geographic Mosaic of Coevolution; University of Chicago Press: Chicago, IL, USA, 2005; 400p. [Google Scholar]
- Turillazzi, S.; West Eberhard, M.J. (Eds.) Natural History and the Evolution of Paper Wasps; Oxford University Press: Oxford, UK, 1996; 414p. [Google Scholar]
- Cervo, R. Polistes wasps and their social parasites, an overview. Ann. Zool. Fenn. 2006, 43, 531–549. [Google Scholar]
- Brandt, M.; Foitzik, S.; Blass, B.; Heinze, J.; Foitzik, S. The coevolutionary dynamics of obligate ant social parasite systems—Between prudence and antagonism. Biol. Rev. 2005, 80, 251–267. [Google Scholar] [CrossRef] [PubMed]
- Brandt, M.; Fischer-Blass, B.; Heinze, J.; Foitzik, S. Population structure and the co-evolution between social parasites and their hosts. Mol. Ecol. 2007, 16, 2063–2078. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, M.C.; Cervo, R.; Turillazzi, S. Effects of social parasitism of Polistes atrimandibularis on the colony cycle and brood production of Polistes biglumis bimaculatus (Hymenoptera, Vespidae). Ethology 1992, 59, 267–271. [Google Scholar]
- Foitzik, S.; Herbers, J.M. Colony structure of a slavemaking ant. II. Frequency of slave raids and impact on the host population. Evolution 2001, 55, 316–323. [Google Scholar] [CrossRef]
- Foitzik, S.; DeHeer, C.J.; Hunjan, D.N.; Herbers, J.M. Coevolution in host-parasite systems: Behavioural strategies of slave-making ants and their hosts. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2001, 268, 1139–1146. [Google Scholar] [CrossRef]
- Fischer, B.; Foitzik, S. Local co-adaptation leading to a geographical mosaic of coevolution in a social parasite system. J. Evol. Biol. 2004, 17, 1026–1034. [Google Scholar] [CrossRef]
- Lorenzi, M.C.; Thompson, J.N. The geographic structure of selection on a coevolving interaction between social parasitic wasps and their hosts hampers social evolution. Evolution 2011, 65, 3527–3542. [Google Scholar] [CrossRef]
- Pekkarinen, A.; Gustafsson, B. The Polistes species in northern Europe (Hymenoptera: Vespidae). Entomol. Fennica 1999, 10, 191–194. [Google Scholar] [CrossRef]
- Guiglia, D. Les guêpes sociales (Hymenoptera Vespidae) d’Europe Occidentale et Septentrionale. In Faune de l’Europe et du Bassin Mediterraneen; Masson: Paris, France, 1972; Volume 6, pp. 1–181. [Google Scholar]
- Neumeyer, R.; Baur, H.; Guex, G.-D.; Praz, C. A new species of the paper wasp genus Polistes (Hymenoptera, Vespidae, Polistinae) in Europe revealed by morphometrics and molecular analyses. ZooKeys 2014, 400, 67–118. [Google Scholar] [CrossRef]
- Fanelli, D.; Cervo, R.; Turillazzi, S. Three new host species of the social wasp parasite, Polistes atrimandibularis (Hymenoptera, Vespidae). Insect. Soc. 2001, 48, 352–354. [Google Scholar] [CrossRef]
- Bagnères, A.-G.; Lorenzi, M.C.; Clément, J.L.; Dusticier, G.; Turillazzi, S. Chemical usurpation of a nest by paper wasp parasites. Science 1996, 272, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, M.C.; Filippone, F. Opportunistic discrimination of alien eggs by social wasps (Polistes biglumis, Hymenoptera Vespidae): A defense against social parasitism? Behav. Ecol. Sociobiol. 2000, 48, 402–406. [Google Scholar] [CrossRef]
- Fucini, S.; Di Bona, V.; Mola, F.; Piccaluga, C.; Lorenzi, M.C. Social wasps without workers: Geographic variation of caste expression in the paper wasp Polistes biglumis. Insect. Soc. 2009, 56, 347–358. [Google Scholar] [CrossRef]
- Ortolani, I.; Cervo, R. Intra-specific body size variation in Polistes paper wasps as a response to social parasite pressure. Ecol. Entomol. 2010, 35, 352–359. [Google Scholar] [CrossRef]
- Uboni, A.; Lorenzi, M.C. Poor odors, strength, and persistence give their rewards to Mutilla europaea visiting dangerous wasp nests. J. Insect Behav. 2013, 26, 246–252. [Google Scholar] [CrossRef]
- Lorenzi, M.C. The result of an arms race: The chemical strategies of Polistes social parasites. Ann. Zool. Fenn. 2006, 43, 550–563. [Google Scholar]
- Lorenzi, M.C.; Azzani, L.; Bagnères, A.-G. Evolutionary consequences of deception: Complexity and informational content of colony signature are favored by social parasitism. Curr. Zool. 2014, 60, 137–149. [Google Scholar] [CrossRef]
- Cervo, R.; Lorenzi, M.C.; Turillazzi, S. Nonaggressive usurpation of the nest of Polistes biglumis bimaculatus by the social parasite Sulcopolistes atrimandibularis (Hymenoptera Vespidae). Insect. Soc. 1990, 37, 333–347. [Google Scholar] [CrossRef]
- Lorenzi, M.C.; Bagnères, A.-G. Concealing identity and mimicking hosts: A dual chemical strategy for a single social parasite? (Polistes atrimandibularis, Hymenoptera: Vespidae). Parasitology 2002, 125, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Blancato, G.; Picchi, L.; Lucas, C.; Bagnères, A.-G.; Lorenzi, M.C. Dynamics of the nest signature in a social wasp: Changes are associated with colony cycle and social parasites. PLoS ONE 2017, 12, e0190018. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Khalil, A.; Bagnères, A.-G.; Lorenzi, M.C. Appeasing their hosts: A novel strategy for parasite brood. Anim. Behav. 2018, 146, 123–134. [Google Scholar] [CrossRef]
- Bonelli, M.; Lorenzi, M.C.; Christidès, J.-P.; Dupont, S.; Bagnères, A.-G. Population diversity in cuticular hydrocarbons and mtDNA in a mountain social wasp. J. Chem. Ecol. 2015, 41, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Mignini, M.; Lorenzi, M.C. Vibratory signals predict rank and offspring caste ratio in a social insect. Behav. Ecol. Sociobiol. 2015, 69, 1739–1748. [Google Scholar] [CrossRef]
- Seppä, P.; Fogelqvist, J.; Gyllenstrand, N.; Lorenzi, M.C. Colony kin structure and breeding patterns in the social wasp, Polistes biglumis. Insect. Soc. 2011, 58, 345–355. [Google Scholar] [CrossRef]
- Lorenzi, M.C.; Turillazzi, S. Behavioural and ecological adaptations to the high mountain environment of Polistes biglumis bimaculatus. Ecol. Entomol. 1986, 11, 199–204. [Google Scholar] [CrossRef]
- Lorenzi, M.C. (University of Sorbonne Paris Nord, Villetaneuse, France). Personal communication, 2020.
- Reeve, H.K. Polistes. In The Social Biology of Wasps; Ross, K.G., Matthews, R.W., Eds.; Cornell University Press: Ithaca, NY, USA, 1991; pp. 99–148. [Google Scholar]
- Peters, J.M.; Queller, D.C.; Strassmann, J.E.; Solıs, C.R. Maternity assignment and queen replacement in a social wasp. Proc. R. Soc. Lond. Ser. B Biol Sci 1995, 260, 7–12. [Google Scholar]
- Strassmann, J.E. Selective altruism towards closer over more distant relatives in colonies of the primitively eusocial wasp, Polistes. In Natural History and Evolution of Paper Wasps; Turillazzi, S., West-Eberhard, M.J., Eds.; Oxford University Press: Oxford, UK, 1996; pp. 190–201. [Google Scholar]
- Field, J.; Solıs, C.R.; Queller, D.C.; Strassmann, J.E. Social and genetic structure of paper-wasp co-foundress associations: Tests of reproductive skew models. Am. Nat. 1998, 151, 545–563. [Google Scholar] [CrossRef]
- Seppä, P.; Queller, D.C.; Strassmann, J.E. Reproduction in foundress associations of the social wasp, Polistes carolina: Conventions, competition, and skew. Behav. Ecol. 2002, 13, 531–542. [Google Scholar] [CrossRef]
- Tsuchida, K.; Yukikudo, K.; Ishiguro, N. Genetic structure of an introduced paper wasp, Polistes chinensis antennalis (Hymenoptera, Vespidae) in New Zealand. Mol. Ecol. 2014, 23, 4018–4034. [Google Scholar] [CrossRef]
- Cervo, R.; Dani, F.R. Social parasitism and its evolution in Polistes. In Natural History and Evolution of Paper Wasps; Turillazzi, S., West-Eberhard, M.J., Eds.; Oxford University Press: Oxford, UK, 1996; pp. 98–112. [Google Scholar]
- Henshaw, M.T. Microsatellite loci for the social wasp Polistes dominulus and their application in other polistine wasps. Mol. Ecol. 2000, 9, 2155–2157. [Google Scholar] [CrossRef] [PubMed]
- Vos, P.; Hogers, R.; Bleeker, M.; Reijans, M.; van de Lee, T.; Hornes, M.; Frijters, A.; Pot, J.; Peleman, J.; Kuiper, M. AFLP: A new technique for DNA fingerprinting. Nucleic Acids Res. 1995, 23, 4407–4414. [Google Scholar] [CrossRef]
- Whitlock, R.; Hipperson, M.; Mannarelli, R.; Butlin, R.K.; Burke, T. An objective, rapid and reproducible method for scoring AFLP peak-height data that minimizes genotyping error. Mol. Ecol. Resour. 2008, 8, 725–735. [Google Scholar] [CrossRef] [PubMed]
- El Mousadik, A.; Petit, R.J. High level of genetic differentiation for allelic richness among populations of the argan tree [Argania spinosa (L.) Skeels] endemic to Morocco. Theor. Appl. Genet. 1996, 92, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Nei, M. Molecular Evolutionary Genetics; Columbia University Press: New York, NY, USA, 1987; 512p. [Google Scholar]
- IBM Corp. IBM SPSS Statistics for Windows; Version 25.0; IBM Corp: Armonk, NY, USA, 2017. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5-6. Available online: https://CRAN.R-project.org/package=vegan (accessed on 9 August 2020).
- Excoffier, L.; Smouse, P.E.; Quattro, J.M. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitocondrial DNA restriction sites. Genetics 1992, 131, 479–491. [Google Scholar]
- Rousset, F. Genetic differentiation and estimation of gene flow from F-statistics under isolation by distance. Genetics 1997, 145, 1219–1228. [Google Scholar]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Mantel, N. The detection of disease clustering and a generalized regression approach. Cancer Res. 1967, 27, 209–220. [Google Scholar]
- Goudet, J. FSTAT, a Program to Estimate and Test Gene Diversities and Fixation Indices (version 2.9.3). Updated from Goudet (1995). 2001. Available online: http://www.unil.ch/izea/softwares/fstat.html (accessed on 19 August 2016).
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Raymond, M.; Rousset, F. GENEPOP (version 1.2): Population genetics software for exact tests and ecumenicism. J. Hered. 1995, 86, 248–249. [Google Scholar] [CrossRef]
- Corander, J.; Sirén, J.; Arjas, E. Bayesian Spatial Modelling of Genetic Population Structure. Comput. Stat. 2008, 23, 111–129. [Google Scholar] [CrossRef]
- Cheng, L.; Connor, T.R.; Sirén, J.; Aanensen, D.M.; Corander, J. Hierarchical and spatially explicit clustering of DNA sequences with BAPS software. Mol. Biol. Evol. 2013, 30, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. Clustal-w—Improving the sensitivity of progressive multiple sequence alignment through sequence weigthing, position-specific, gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef]
- Galtier, N.; Gouy, M.; Gautier, C. SEAVIEW and PHYLO_WIN: Two graphic tools for sequence alignment and molecular phylogeny. Comput. Appl. Biosci. 1996, 12, 543–548. [Google Scholar] [CrossRef]
- Guindon, S.; Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES, Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef]
- Nylander, J.A.A. 2004 MrAIC.pl; Program Distributed by the Author; Evolutionary Biology Centre, Uppsala University: Uppsala, Sweden, 2004. [Google Scholar]
- Clement, M.; Posada, D.; Crandall, K.A. TCS: A computer program to estimate gene genealogies. Mol. Ecol. 2000, 9, 1657–1660. [Google Scholar] [CrossRef]
- Seppä, P.; Gyllenstrand, N.; Corander, J.; Pamilo, P. Coexistence of the social types: Genetic population structure in the ant, Formica exsecta. Evolution 2004, 58, 2462–2471. [Google Scholar]
- Uddin, M.M.; Tsuchida, K. Genetic population structure of the paper wasp Polistes olivaceus (Hymenoptera: Vespidae) in Bangladesh. Popul. Ecol. 2012, 54, 103–114. [Google Scholar] [CrossRef]
- Davis, S.K.; Strassmann, J.E.; Hughes, C.; Pletscher, L.S.; Templeton, A.R. Population structure and kinship I Polistes (Hymenoptera, Vespidae): An analysis using ribosomal DNA and protein electrophoresis. Evolution 1990, 44, 1242–1253. [Google Scholar] [CrossRef]
- Queller, D.C.; Strassmann, J.E.; Hughes, C.R. Genetic relatedness and population structure in primitively eusocial wasps in the genus Mischocyttarus (Hymenoptera: Vespidae). J. Hymenopt. Res. 1992, 1, 81–89. [Google Scholar]
- Strassmann, J.E.; Queller, D.C.; Solís, C.R. Genetic relatedness and population structure in the social wasp, Mischocyttarus mexicanus (Hymenoptera: Vespidae). Insect. Soc. 1995, 42, 379–383. [Google Scholar] [CrossRef]
- Lengronne, T.; Leadbeater, E.; Patalano, S.; Dreier, S.; Field, J.; Sumner, S.; Keller, L. Little effect of seasonal constraints on population genetic structure in eusocial paper wasps. Ecol. Evol. 2012, 2, 2610–2619. [Google Scholar] [CrossRef] [PubMed]
- Kozyra, K.B.; Melosik, I.; Baraniak, E. Genetic diversity and population structure of Polistes nimpha based on DNA microsatellite markers. Insect. Soc. 2015, 62, 423–432. [Google Scholar] [CrossRef]
- Parsons, P.J.; Grinsted, L.; Field, J. Partner choice correlates with fine scale kin structuring in the paper wasp Polistes dominula. PLoS ONE 2019, 14, e0221701. [Google Scholar] [CrossRef]
- Fanelli, D.; Henshaw, M.; Cervo, R.; Turillazzi, S.; Queller, D.C.; Strassmann, J.E. The social parasite wasp Polistes atrimandibularis does not form host races. J. Evol. Biol. 2005, 18, 1362–1367. [Google Scholar] [CrossRef]
- Hoffman, E.A.; Kovacs, J.L.; Goodisman, M.A.D. Genetic structure and breeding system in a social wasp and its social parasite. BMC Evol. Biol. 2008, 8, 239. [Google Scholar] [CrossRef]
- Gandon, S.; Michalakis, Y.; Ebert, D. Temporal variability and local adaptation. Trends Ecol. Evol. 1996, 11, 431. [Google Scholar] [CrossRef]
- van Zweden, J.S.; d’Ettorre, P. Nestmate recognition in social insects and the role of hydrocarbons. In Insect Hydrocarbons: Biology, Biochemistry and Chemical Ecology; Blomquist, G.J., Bagnères, A.-G., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 222–243. [Google Scholar]
- Boomsma, J.J.; Nielsen, J.; Sundström, L.; Oldham, N.J.; Tentschert, J.; Petersen, H.C.; Morgan, E.D. Informational constraints on optimal sex allocation in ants. Proc. Natl. Acad. Sci. USA 2003, 100, 8799–8804. [Google Scholar] [CrossRef] [PubMed]
- Dani, F.R.; Foster, K.R.; Zacchi, F.; Seppä, P.; Massolo, A.; Carelli, A.; Arévalo, E.; Strassmann, J.E.; Turillazzi, S. Can cuticular lipids provide sufficient information for within-colony nepotism in wasps? Proc. R. Soc. Lond. Ser. B Biol. Sci. 2004, 271, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Dronnet, S.; Lohou, C.; Christidès, J.-P.; Bagnères, A.-G. Cuticular hydrocarbon composition reflects genetic relationship among colonies of the introduced termite Reticulitermes santonensis Feytaud. J. Chem. Ecol. 2006, 32, 1027–1042. [Google Scholar] [CrossRef]
- Avise, J.C.; Arnold, J.; Ball, R.M.; Bermingham, E.; Lamb, T.; Neigel, J.E.; Reeb, C.A.; Saunders, N.C. Intraspecific phylogeography: The mitochondrial DNA bridge between population genetics and systematics. Annu. Rev. Ecol. Syst. 1987, 18, 489–522. [Google Scholar] [CrossRef]
- Hardie, D.C.; Hutchings, J.A. Evolutionary ecology at the extremes of species’ ranges. Environ. Rev. 2010, 18, 1–20. [Google Scholar] [CrossRef]
- Fitzpatrick, S.W.; Gerberich, J.C.; Kronenberger, J.A.; Angeloni, L.M.; Funk, W.C. Locally adapted traits maintained in the face of high gene flow. Ecol. Lett. 2015, 18, 37–47. [Google Scholar] [CrossRef]
- Fitzpatrick, S.W.; Handelsman, C.A.; Torres-Dowdall, J.; Ruell, E.W.; Broder, E.D.; Kronenberger, J.A.; Reznick, D.N.; Ghalambor, C.K.; Angeloni, L.M.; Funk, W.C. Gene Flow Constrains and Facilitates Genetically Based Divergence in Quantitative Traits. Copeia 2017, 105, 462–474. [Google Scholar] [CrossRef]
- Jacob, L.D.; Chaine, A.S.; Bonte, D.; Schtickzelle, N.; Huet, M.; Clobert, J. Gene flow favours local adaptation under habitat choice in ciliate microcosms. Nat. Ecol. Evol. 2017, 1, 1407–1410. [Google Scholar] [CrossRef]
| Population | DNA Microsatellites | AFLP | mtDNA | |||
|---|---|---|---|---|---|---|
| n | HE [SE] | hAFLP [SE] | Haplotypes Found | hmt | π [SD] | |
| P. biglumis | ||||||
| Valli del Cuneese | ||||||
| Ferrere * | 0.65 [0.07] | 0.23 [0.02] | 10*h2, h4, h7, h8, h9, h10 | 0.57 | 0.003 [0.002] | |
| Terme di Valdieri | 9 | 0.64 [0.09] | 0.30 [0.03] | 4*h2, h3, h4, h7, h20 | 0.80 | 0.004 [0.003] |
| Lago della Rovina | 4 | 0.61 [0.08] | 0.11 [0.03] | 4*h2, h8 | 0.50 | 0.001 [0.001] |
| Fondovet | 7 | 0.59 [0.11] | 0.20 [0.02] | 4*h2, h4, h7 | 0.60 | 0.004 [0.003] |
| Colle di Sampeyre | 1 | - | - | h17 | - | - |
| Valle Pesio | 1 | - | - | h8 | - | - |
| Average | 0.62 | 0.21 | 0.61 | 0.003 | ||
| Val Susa | ||||||
| Montgenèvre * | 48 | 0.69 [0.06] | 0.17 [0.01] | 3*h2, h4, 4*h7, 6*h8, h16, h22 | 0.80 | 0.004 [0.002] |
| Alpe Plane | 16 | 0.68 [0.08] | 0.12 [0.02] | 2*h2, 2*h7, 5*h8, h18 | 0.73 | 0.004 [0.003] |
| Val di Thuras * | 8 | 0.58 [0.12] | 0.16 [0.02] | h2, 2*h7, h8 | 0.83 | 0.006 [0.004] |
| Cesana | 9 | 0.62 [0.09] | 0.15 [0.02] | h2, 2*h7, 2*h8 | 0.80 | 0.005 [0.003] |
| Average | 0.64 | 0.15 | 0.79 | 0.005 | ||
| Valsesia | ||||||
| Frazione Dorf | 5 | 0.70 [0.03] | 0.15 [0.03] | h6, 2*h19 | 0.67 | 0.006 [0.005] |
| Rimella | 11 | 0.64 [0.05] | 0.25 [0.02] | h4, 4*h14, 2*h15 | 0.67 | 0.002 [0.002] |
| Fobello | 2 | - | - | h4, h6 | 1 | 0.005 [0.005] |
| Rima | 2 | - | - | h2, h6 | 1 | 0.007 [0.007] |
| Carcoforo | 2 | - | - | 2*h4 | 0 | 0 [0] |
| Alpe di Mera | 2 | - | - | h4, h6 | 1 | 0.005 [0.005] |
| Sant’Antonio | 9 | 0.70 [0.06] | 0.20 [0.02] | 2*h4, 2*h6, h14, h19 | 0.87 | 0.005 [0.003] |
| Average | 0.68 | 0.20 | 0.80 | 0.005 | ||
| Val Leventina | 21 | |||||
| Cari | 0.69 [0.02] | 0.19 [0.02] | 2*h8, h12, h14, 8*h15 | 0.79 | 0.001 [0.001] | |
| Valtellina | ||||||
| Trivigno * | 11 | 0.64 [0.07] | 0.13 [0.02] | 6*h16, h21 | 0.29 | 0.001 [0.001] |
| Campovecchio * | 12 | 0.68 [0.06] | 0.23 [0.02] | 10*h16 | 0 | 0 [0] |
| Average | 0.66 | 0.18 | 0.14 | 0.001 | ||
| Average across all populations | 0.65 | 0.19 | 0.66 | 0.002 | ||
| P. atrimandibularis | ||||||
| Valli del Cuneese | 11 | 0.68 [0.04] | 0.21 [0.02] | 6*h1, 2*h2, h3, h4, h6 | 0.71 | 0.001 [0.001] |
| Val Susa | 18 | 0.72 [0.06] | 0.22 [0.02] | h1, 7*h2, h5 | 0.42 | 0.001 [0.001] |
| Valtellina | 4 | 0.70 [0.04] | 0.19 [0.03] | 4*h4 | 0 | 0 [0] |
| Average across all populations | 0.70 | 0.21 | 0.56 | 0.001 | ||
| Source of Variation | DNA Microsatellites | AFLP | mtDNA | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| % | F | p | % | Φ | p | % | Φ | p | Φmt/F | |
| P. biglumis/populations divided to regions | ||||||||||
| Among regions, FCT, ΦCT | 2 | 0.017 | 0.001 | 12 | 0.118 | 0.001 | 28 | 0.280 | 0.001 | 16.5 |
| Among pops within regions, FSC, ΦSC | 0 | −0.002 | 0.611 | 1 | 0.016 | 0.164 | 0 | −0.020 | 0.693 | |
| Among individuals within pops, FIS | 10 | 0.104 | 0.001 | |||||||
| Within individuals, FIT | 88 | 0.118 | 0.001 | |||||||
| Within populations | 87 | 72 | ||||||||
| P. biglumis/three regions shared by P. biglumis and P. atrimandibularis, no population level | ||||||||||
| Among regions, FCT, ΦCT | 1 | 0.005 | 0.015 | 7 | 0.065 | 0.002 | 28 | 0.280 | 0.001 | 46.7 |
| Among individuals within pops, FIS | 9 | 0.093 | 0.001 | |||||||
| Within individuals, FIT | 90 | 0.098 | 0.001 | |||||||
| Within populations | 93 | 72 | ||||||||
| P. atrimandibularis/three regions, no population level | ||||||||||
| Among regions, FCT, ΦCT | 7 | 0.073 | 0.001 | 9 | 0.085 | 0.063 | 47 | 0.469 | 0.001 | 6.42 |
| Among individuals within pops, FIS | 10 | 0.113 | 0.002 | |||||||
| Within individuals, FIT | 82 | 0,178 | 0.001 | |||||||
| Within regions | 91 | 53 | ||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seppä, P.; Bonelli, M.; Dupont, S.; Hakala, S.M.; Bagnères, A.-G.; Lorenzi, M.C. Strong Gene Flow Undermines Local Adaptations in a Host Parasite System. Insects 2020, 11, 585. https://doi.org/10.3390/insects11090585
Seppä P, Bonelli M, Dupont S, Hakala SM, Bagnères A-G, Lorenzi MC. Strong Gene Flow Undermines Local Adaptations in a Host Parasite System. Insects. 2020; 11(9):585. https://doi.org/10.3390/insects11090585
Chicago/Turabian StyleSeppä, Perttu, Mariaelena Bonelli, Simon Dupont, Sanja Maria Hakala, Anne-Geneviève Bagnères, and Maria Cristina Lorenzi. 2020. "Strong Gene Flow Undermines Local Adaptations in a Host Parasite System" Insects 11, no. 9: 585. https://doi.org/10.3390/insects11090585
APA StyleSeppä, P., Bonelli, M., Dupont, S., Hakala, S. M., Bagnères, A.-G., & Lorenzi, M. C. (2020). Strong Gene Flow Undermines Local Adaptations in a Host Parasite System. Insects, 11(9), 585. https://doi.org/10.3390/insects11090585






