The Midgut Microbiota of Colombian Aedes aegypti Populations with Different Levels of Resistance to the Insecticide Lambda-cyhalothrin
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Mosquito Collections
2.2. Determination of Lambda-Cyhalothrin Resistance Profile of Larvae
2.3. Allele-Specific PCR (AS-PCR) for the kdr Mutation V1016I
2.4. Generation of Axenic Adult Mosquitoes
2.5. Preparation of Genomic DNA, and Library for Metagenome 16S Sequencing
2.6. Statistical Analyses
2.7. Bacterial Diversity
2.8. Taxonomic Annotation and Relative Abundance of Taxa
2.9. Data Accessibility
3. Results
3.1. Mosquito Populations from Colombia Show Different Susceptibility Profiles to Lambda-Cyhalothrin
3.2. Genotyping of kdr Mutation V1016I
3.3. Axenic Adult Mosquitoes are More Susceptible to Lambda-Cyhalothrin
3.4. Metagenome Analyses
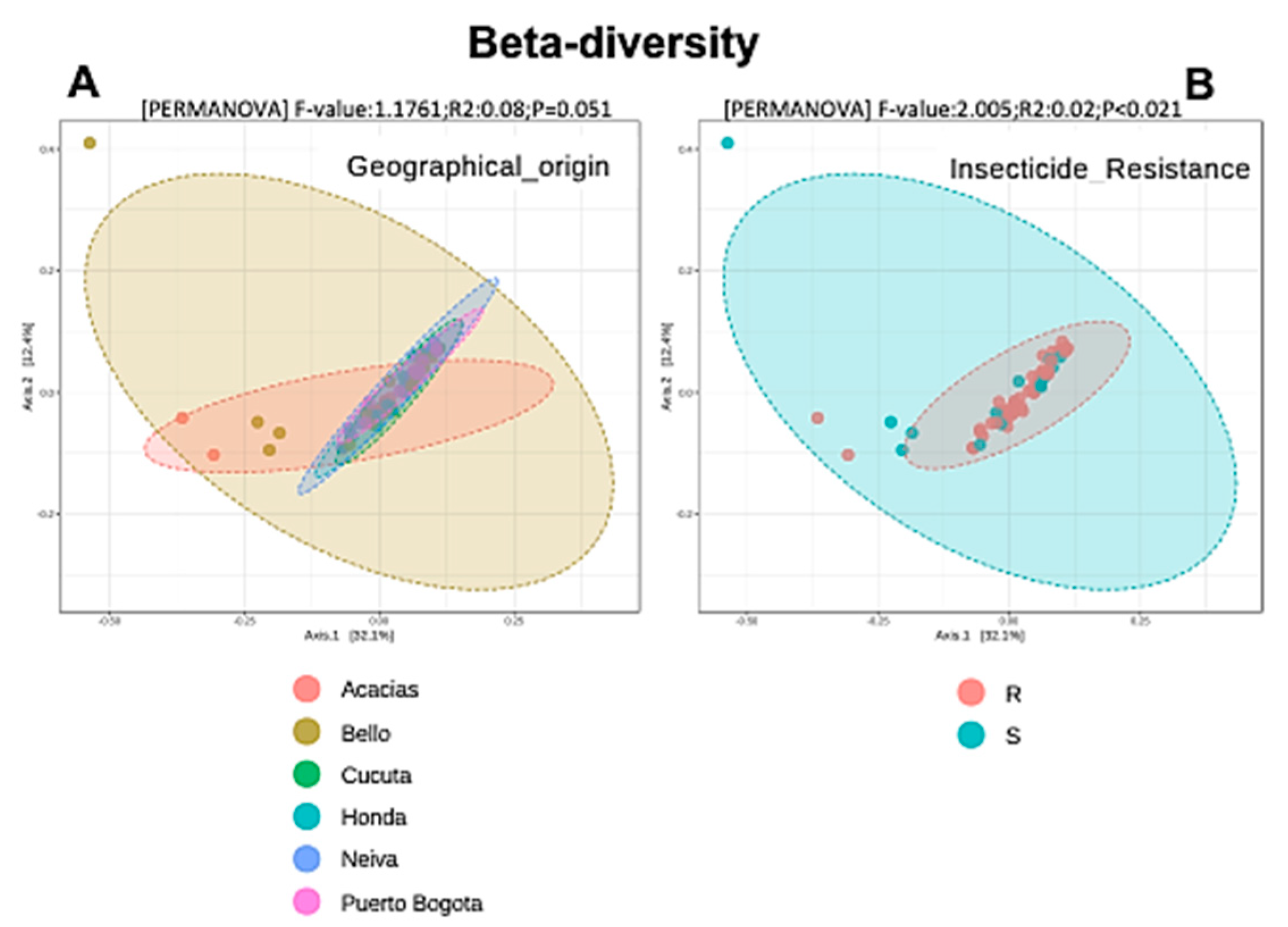
3.5. Alpha and Beta Diversity Analyses
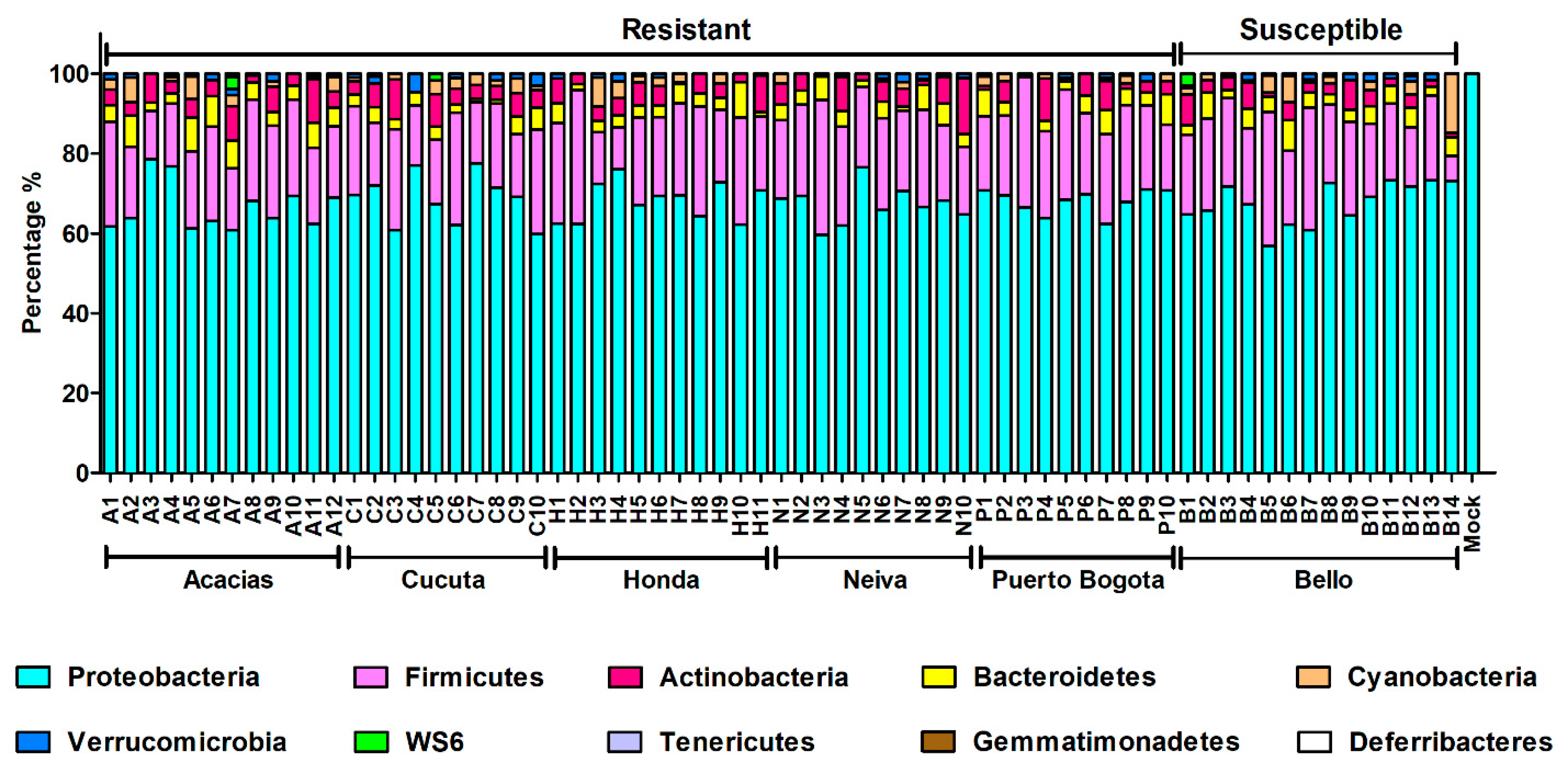
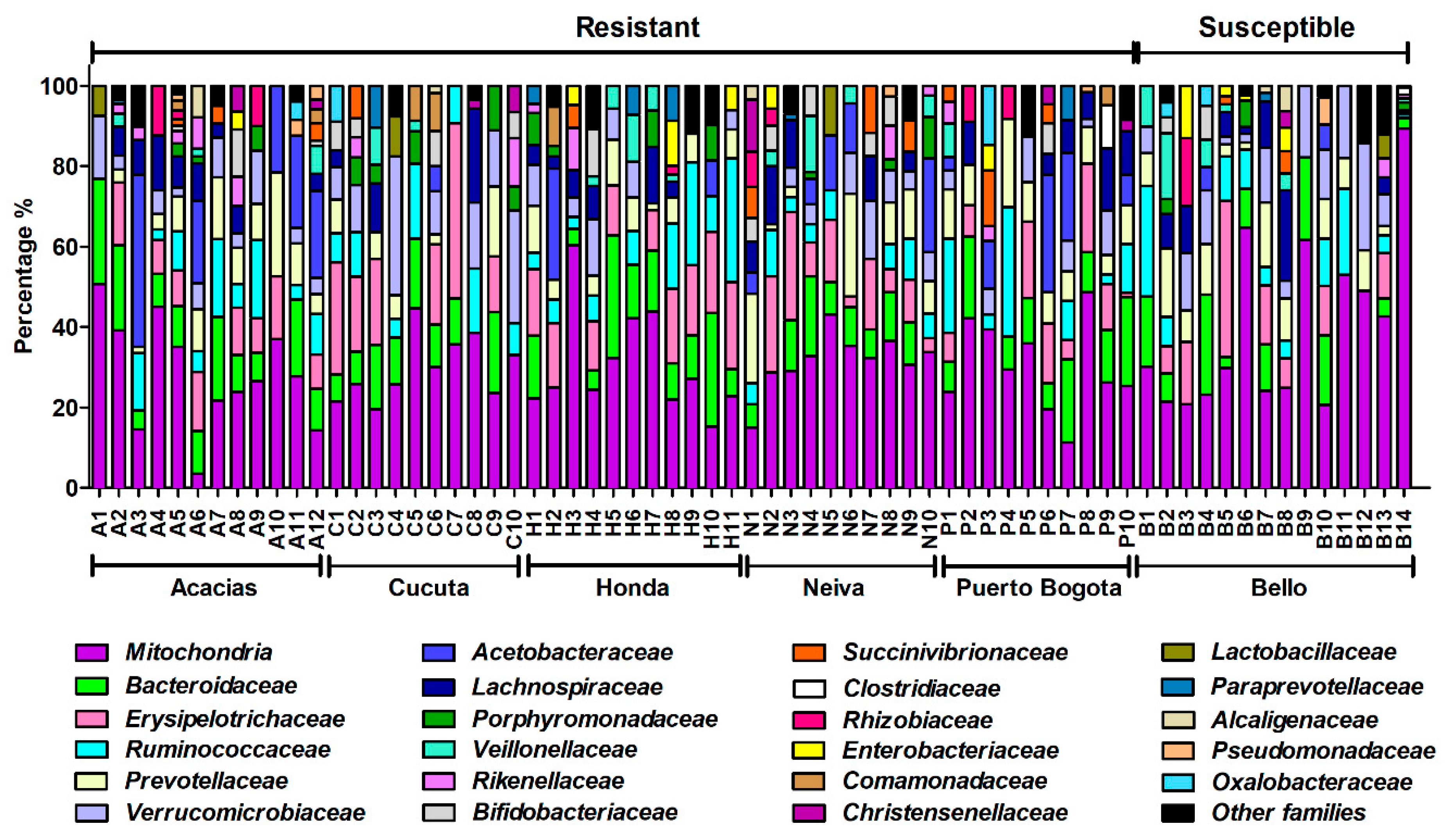
3.6. Taxonomic Composition and Abundance Analyses
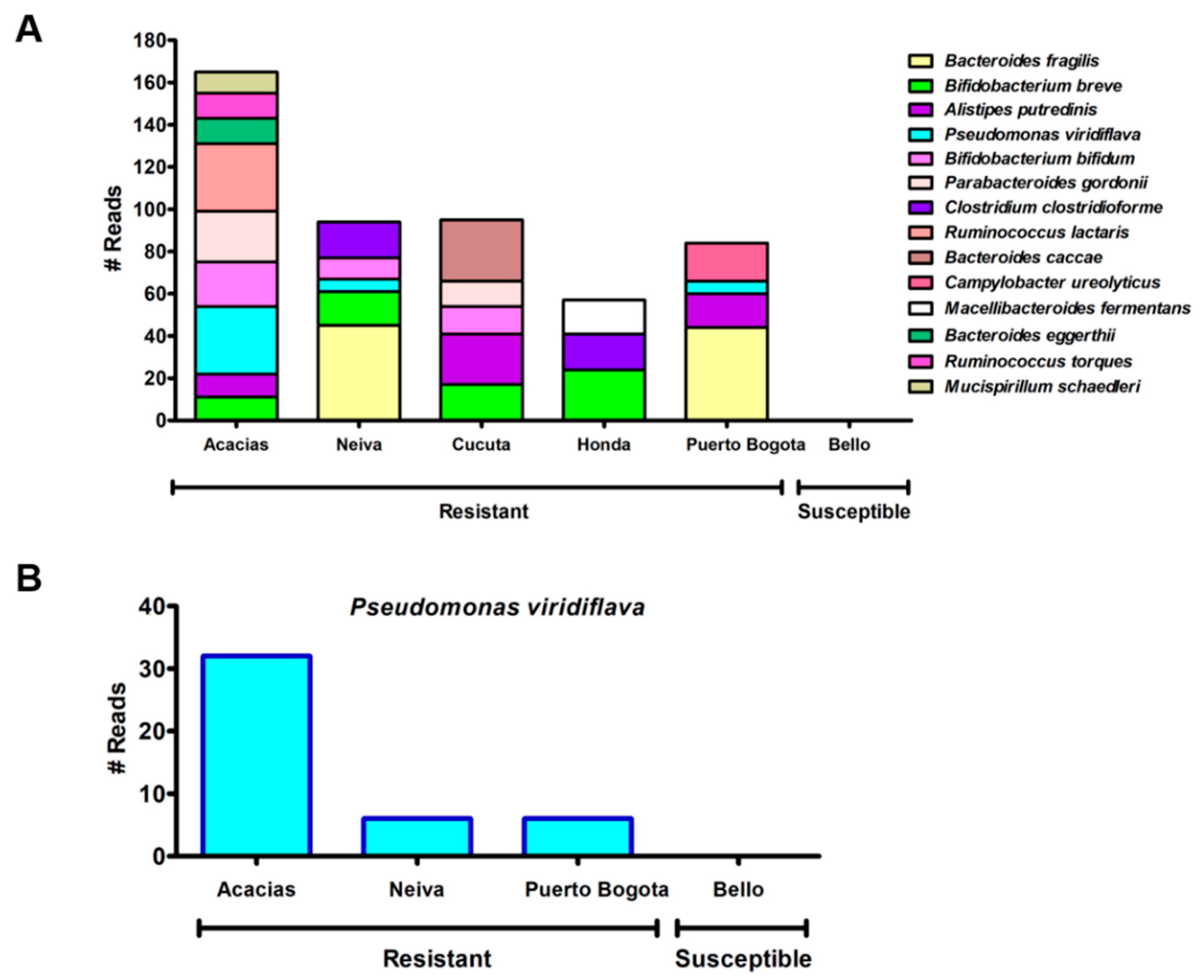
3.7. The Abundance Analyses Revealed Significant Differences Between Bacteria from Lambda-Cyhalothrin-Resistant and -Susceptible Mosquitoes
4. Discussion
4.1. Evidence of High Resistance Ratio (RR) to Lambda-Cyhalothrin in Colombian Populations of Ae. aegypti
4.2. Lambda-Cyhalothrin Resistance is Partially Explained by the 1016I Mutation
4.3. Midgut-Associated Bacteria Participate in the Resistance to Lambda-Cyhalothrin in Ae. aegypti
4.4. Aedes aegypti Midgut from Colombia Presented a Core Microbiota, Inter-Individual Variation, and Low Bacterial Diversity
4.5. Bacterial Taxa Associated with Lambda-Cyhalothrin-Resistant and -Susceptible Ae. aegypti Populations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Boeuf, P.; Drummer, H.E.; Richards, J.S.; Scoullar, M.J.L.; Beeson, J.G. The global threat of Zika virus to pregnancy: Epidemiology, clinical perspectives, mechanisms, and impact. BMC Med. 2016, 14, 1–9. [Google Scholar] [CrossRef]
- Messina, J.; Kraemer, M.U.G.; Brady, O.J.; Pigott, D.M.; Shearer, F.M.; Weiss, D.J.; Golding, N.; Ruktanonchai, C.W.; Gething, P.W.; Cohn, E.; et al. Mapping global environmental suitability for Zika virus. eLife 2016, 5. [Google Scholar] [CrossRef]
- Wahid, B.; Ali, A.; Rafique, S.; Idrees, M. Global expansion of chikungunya virus: Mapping the 64-year history. Int. J. Infect Dis. 2017, 58, 69–76. [Google Scholar] [CrossRef]
- Ocampo, C.B.; Salazar-Terreros, M.J.; Mina, N.J.; McAllister, J.; Brogdon, W. Insecticide resistance status of Aedes aegypti in 10 localities in Colombia. Acta Trop. 2011, 118, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Santacoloma Varón, L.; Chaves Córdoba, B.; Brochero, H.L. Susceptibilidad de Aedes aegypti a DDT, deltametrina y lambdacialotrina en Colombia. Rev. Panam Salud Pública 2010, 27, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Ardila-Roldán, S.; Santacoloma, L.; Brochero, H. Estado de la susceptibilidad a insecticidas de uso en salud pública en poblaciones naturales de Aedes aegypti (Diptera: Culicidae) del departamento de Casanare, Colombia. Biomédica 2013, 33, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Granada, Y.; Mejía-Jaramillo, A.; Strode, C.; Triana-Chavez, O. A Point Mutation V419L in the Sodium Channel Gene from Natural Populations of Aedes aegypti is Involved in Resistance to λ-Cyhalothrin in Colombia. Insects 2018, 9, 23. [Google Scholar] [CrossRef]
- Maestre-Serrano, R.; Gomez-Camargo, D.; Ponce-Garcia, G.; Flores, A.E. Susceptibility to insecticides and resistance mechanisms in Aedes aegypti from the Colombian Caribbean Region. Pestic Biochem. Physiol. 2014, 116, 63–73. [Google Scholar] [CrossRef]
- Aponte, A.; Penilla, R.P.; Rodríguez, A.D.; Ocampo, C.B. Mechanisms of pyrethroid resistance in Aedes (Stegomyia) aegypti from Colombia. Acta Trop. 2019, 191, 146–154. [Google Scholar] [CrossRef]
- De Almeida, L.G.; De Moraes, L.A.B.; Trigo, J.R.; Omoto, C.; Cônsoli, F.L. The gut microbiota of insecticide-resistant insects houses insecticide-degrading bacteria: A potential source for biotechnological exploitation. PLoS ONE 2017, 12, e0174754. [Google Scholar] [CrossRef] [PubMed]
- Dada, N.; Sheth, M.; Liebman, K.; Pinto, J.; Lenhart, A. Whole metagenome sequencing reveals links between mosquito microbiota and insecticide resistance in malaria vectors. Sci. Rep. 2018, 8, 2084. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, Y.; Hayatsu, M.; Hosokawa, T.; Nagayama, A.; Tago, K.; Fukatsu, T. Symbiont-mediated insecticide resistance. Proc. Natl. Acad. Sci. USA 2012, 109, 8618–8622. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, K.; Kikuchi, Y. Riptortus pedestris and Burkholderia symbiont: An ideal model system for insect–microbe symbiotic associations. Res. Microbiol. 2017, 168, 175–187. [Google Scholar] [CrossRef]
- Xia, X.; Zheng, D.; Zhong, H.; Qin, B.; Gurr, G.M.; Vasseur, L.; Lin, H.; Bai, J.; He, W.; You, M. DNA Sequencing Reveals the Midgut Microbiota of Diamondback Moth, Plutella xylostella (L.) and a Possible Relationship with Insecticide Resistance. PLoS ONE 2013, 8, e68852. [Google Scholar] [CrossRef]
- Sharma, A.; Bhatt, P.; Khati, P.; Gangola, S.; Kumar, G. Microbial Degradation of Pesticides for Environmental Cleanup. In Bioremediation of Industrial Pollutants; Write and Print Publications: New Dheli, India, 2016; pp. 178–205. [Google Scholar]
- Li, W.; Jin, D.; Shi, C.; Li, F. Midgut bacteria in deltamethrin-resistant, deltamethrin-susceptible, and field-caught populations of Plutella xylostella, and phenomics of the predominant midgut bacterium Enterococcus mundtii. Sci. Rep. 2017, 7, 1947. [Google Scholar] [CrossRef]
- Pietri, J.E.; Liang, D. The Links between Insect Symbionts and Insecticide Resistance: Causal Relationships and Physiological Tradeoffs. Ann. Entomol. Soc. Am. 2018, 111, 92–97. [Google Scholar] [CrossRef]
- Itoh, H.; Hori, T.; Sato, Y.; Nagayama, A.; Tago, K.; Hayatsu, M.; Kikuchi, Y. Infection dynamics of insecticide-degrading symbionts from soil to insects in response to insecticide spraying. ISME J. 2018, 12, 909–920. [Google Scholar] [CrossRef]
- Maestre, R.; Rey, G.; De Las Salas, J.; Vergara, C.; Santacoloma, L.; Goenaga, S. Susceptibility status of Aedes aegypti to insecticides in Atlántico (Colombia). Revista Colombiana de Entomología 2010, 36, 242–248. [Google Scholar]
- Aguirre-Obando, O.A.; Bona, A.C.D.; Duque, L.J.E.; Navarro-Silva, M.A. Insecticide resistance and genetic variability in natural populations of Aedes (Stegomyia) aegypti (Diptera: Culicidae) from Colombia. Zool Curitiba 2015, 32, 14–22. [Google Scholar] [CrossRef]
- Pinto, J.; Palomino, M.; Mendoza-Uribe, L.; Sinti, C.; Liebman, K.A.; Lenhart, A. Susceptibility to insecticides and resistance mechanisms in three populations of Aedes aegypti from Peru. Parasit Vectors 2019, 12, 494. [Google Scholar] [CrossRef] [PubMed]
- Maestre-Serrano, R.; Pareja-Loaiza, P.; Gomez Camargo, D.; Ponce-García, G.; Flores, A.E. Co-occurrence of V1016I and F1534C mutations in the voltage-gated sodium channel and resistance to pyrethroids in Aedes aegypti (L.) from the Colombian Caribbean region. Pest. Manag. Sci. 2019, 75, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, L.C.; Ponce, G.; Saavedra-Rodriguez, K.; Lopez, B.; Flores, A.E. Frequency of V1016I and F1534C mutations in the voltage-gated sodium channel gene in Aedes aegypti in Venezuela. Pest. Manag. Sci. 2015, 71, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Linss, J.G.B.; Brito, L.P.; Garcia, G.A.; Araki, A.S.; Bruno, R.V.; Lima, J.B.P.; Valle, D.; Martins, A.J. Distribution and dissemination of the Val1016Ile and Phe1534Cys Kdr mutations in Aedes aegypti Brazilian natural populations. Parasit Vectors 2014, 7, 25. [Google Scholar]
- Bingham, G.; Strode, C.; Tran, L.; Khoa, P.T.; Jamet, H.P. Can piperonyl butoxide enhance the efficacy of pyrethroids against pyrethroid-resistant Aedes aegypti? Trop. Med. Int. Health 2011, 16, 492–500. [Google Scholar] [CrossRef]
- Kawada, H.; Higa, Y.; Futami, K.; Muranami, Y.; Kawashima, E.; Osei, J.H.N.; Sakyi, K.Y.; Dadzie, S.; De Souza, D.K.; Appawu, M.; et al. Discovery of Point Mutations in the Voltage-Gated Sodium Channel from African Aedes aegypti Populations: Potential Phylogenetic Reasons for Gene Introgression. McCall PJ, editor. PLoS Negl. Trop. Dis. 2016, 10, e0004780. [Google Scholar] [CrossRef]
- Sombié, A.; Saiki, E.; Yaméogo, F.; Sakurai, T.; Shirozu, T.; Fukumoto, S.; Sanon, A.; Weetman, D.; McCall, P.J.; Kanuka, H.; et al. High frequencies of F1534C and V1016I kdr mutations and association with pyrethroid resistance in Aedes aegypti from Somgandé (Ouagadougou), Burkina Faso. Trop. Med. Health 2019, 47, 2. [Google Scholar] [CrossRef]
- Contreras-Perera, Y.; Ponce-Garcia, G.; Villanueva-Segura, K.; Lopez-Monroy, B.; Rodríguez-Sanchez, I.P.; Lenhart, A.; Manrique-Saide, P.; Flores, A.E. Impact of deltamethrin selection on kdr mutations and insecticide detoxifying enzymes in Aedes aegypti from Mexico. Parasit. Vectors 2020, 13, 224. [Google Scholar] [CrossRef]
- WHO. Guidelines for Laboratory and Field Testing of Mosquito Larvicides [Internet]; World Health Organization: Geneva, Switzerland, 2005; Available online: https://apps.who.int/iris/bitstream/handle/10665/69101/WHO_CDS_WHOPES_GCDPP_2005.13.pdf?sequence=1&isAllowed=y (accessed on 25 August 2020).
- Collins, F.H.; Mendez, M.A.; Rasmussen, M.O.; Mehaffey, P.C.; Besansky, N.J.; Finnerty, V. A Ribosomal RNA Gene Probe Differentiates Member Species of the Anopheles gambiae Complex. Am. J. Trop. Med. Hyg. 1987, 37, 37–41. [Google Scholar] [CrossRef]
- Li, C.-X.; Kaufman, P.; Xue, R.-D.; Zhao, M.; Wang, G.; Yan, T.; Guo, X.; Zhang, Y.; Dong, Y.; Xing, D.; et al. Relationship between insecticide resistance and kdr mutations in the dengue vector Aedes aegypti in Southern China. Parasit. Vectors 2015, 8, 325. [Google Scholar] [CrossRef]
- Ramirez, J.L.; Souza-Neto, J.A.; Cosme, R.T.; Rovira, J.; Ortiz, A.; Pascale, J.M.; Dimopoulos, G. Reciprocal Tripartite Interactions between the Aedes aegypti Midgut Microbiota, Innate Immune System and Dengue Virus Influences Vector Competence. PLoS Negl. Trop. Dis. 2012, 6, e1561. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Dhariwal, A.; Chong, J.; Habib, S.; King, I.L.; Agellon, L.B.; Xia, J. MicrobiomeAnalyst: A web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res. 2017, 45, W180–W188. [Google Scholar] [CrossRef]
- King, G.M. Microbiomes of the Enteropneust, Saccoglossus bromophenolosus, and Associated Marine Intertidal Sediments of Cod Cove, Maine. Front Microbiol. 2018, 9, 3066. [Google Scholar] [CrossRef]
- Selvam, A.D.G.; Thatheyus, A.; Vidhya, R. Biodegradation of the Synthetic Pyrethroid, Fenvalerate by Pseudomonas viridiflava. Am. J. Microbiol. Res. 2013, 1, 32–38. [Google Scholar] [CrossRef]
- Chaverra-Rodríguez, D.; Jaramillo-Ocampo, N.; Fonseca-González, I. Selección artificial de resistencia a lambda-cialotrina en Aedes aegypti y resistencia cruzada a otros insecticidas. Revista Colombiana de Entomología 2012, 38, 100–107. [Google Scholar]
- Rodríguez, M.M.; Bisset, J.A.; Fernández, D. Levels of Insecticide Resistance and Resistance Mechanisms in Aedes aegypti From Some Latin American Countries. J. Am. Mosq. Control Assoc. 2007, 23, 420–429. [Google Scholar] [CrossRef]
- Vontas, J.; Kioulos, E.; Pavlidi, N.; Morou, E.; Delta Torre, A.; Ranson, H. Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic. Biochem. Physiol. 2012, 104, 126–131. [Google Scholar] [CrossRef]
- Liu, N. Insecticide Resistance in Mosquitoes: Impact, Mechanisms, and Research Directions. Ann. Rev. Entomol. 2015, 60, 537–559. [Google Scholar] [CrossRef]
- Faucon, F.; Dusfour, I.; Gaude, T.; Navratil, V.; Boyer, F.; Chandre, F.; Sirisopa, P.; Thanispong, K.; Juntarajumnong, W.; Poupardin, R.; et al. Identifying genomic changes associated with insecticide resistance in the dengue mosquito Aedes aegypti by deep targeted sequencing. Genome Res. 2015, 25, 1347–1359. [Google Scholar] [CrossRef] [PubMed]
- Weetman, D.; Djogbenou, L.S.; Lucas, E.R. Copy number variation (CNV) and insecticide resistance in mosquitoes: Evolving knowledge or an evolving problem? Curr. Opin. Insect Sci. 2018, 27, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.J.; Brito, L.P.; Linss, J.G.B.; Rivas, G.B.S.; Machado, R.; Bruno, R.V.; Lima, J.B.P.; Valle, D.; Peixoto, A.A. Evidence for gene duplication in the voltage-gated sodium channel gene of Aedes aegypti. Evol. Med. Public Health 2013, 2013, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Su, X.; Bonizzoni, M.; Zhong, D.; Li, Y.; Zhou, G.; Nguyen, H.; Tong, S.; Yan, G.; Chen, X. Comparative transcriptome analysis and RNA interference reveal CYP6A8 and SNPs related to pyrethroid resistance in Aedes albopictus. PLoS Negl. Trop. Dis. 2018, 12, e0006828. [Google Scholar] [CrossRef] [PubMed]
- Oppold, A.; Kreß, A.; Bussche, J.V.; Diogo, J.; Kuch, U.; Oehlmann, J.; Vandegehuchte, M.; Müller, R. Epigenetic alterations and decreasing insecticide sensitivity of the Asian tiger mosquito Aedes albopictus. Ecotoxicol. Environ. Saf. 2015, 122, 45–53. [Google Scholar] [CrossRef]
- Scates, S.S.; O’Neal, S.T.; Anderson, T.D. Bacteria-mediated modification of insecticide toxicity in the yellow fever mosquito, Aedes aegypti. Pestic. Biochem. Physiol. 2019, 161, 77–85. [Google Scholar] [CrossRef]
- Scolari, F.; Casiraghi, M.; Bonizzoni, M. Aedes spp. and Their Microbiota: A Review. Front. Microbiol. 2019, 10, 2036. [Google Scholar] [CrossRef]
- Guégan, M.; Zouache, K.; Démichel, C.; Minard, G.; Van, V.T.; Potier, P.; Mavingui, P.; Moro, C.V. The mosquito holobiont: Fresh insight into mosquito-microbiota interactions. Microbiome 2018, 6, 49. [Google Scholar] [CrossRef]
- Strand, M.R. The Gut Microbiota of Mosquitoes. In Arthropod Vector: Controller of Disease Transmission; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 185–199. Available online: https://linkinghub.elsevier.com/retrieve/pii/B9780128053508000118 (accessed on 2 July 2020).
- Dada, N.; Jumas-Bilak, E.; Manguin, S.; Seidu, R.; Stenström, T.-A.; Overgaard, H.J. Comparative assessment of the bacterial communities associated with Aedes aegypti larvae and water from domestic water storage containers. Parasit. Vectors 2014, 7, 391. [Google Scholar] [CrossRef]
- Bennett, K.L.; Gómez-Martínez, C.; Chin, Y.; Saltonstall, K.; McMillan, W.O.; Rovira, J.R.; Loaiza, J.R. Dynamics and diversity of bacteria associated with the disease vectors Aedes aegypti and Aedes albopictus. Sci. Rep. 2019, 9, 12160. [Google Scholar] [CrossRef]
- Coatsworth, H.; Caicedo, P.A.; Van Rossum, T.; Ocampo, C.B.; Lowenberger, C. The Composition of Midgut Bacteria in Aedes aegypti (Diptera: Culicidae) That Are Naturally Susceptible or Refractory to Dengue Viruses. J. Insect Sci. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Muturi, E.J.; Dunlap, C.; Ramirez, J.L.; Rooney, A.P.; Kim, C.-H. Host blood meal source has a strong impact on gut microbiota of Aedes aegypti. FEMS Microbiol. Ecol. 2018, 95, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yadav, K.K.; Bora, A.; Datta, S.; Chandel, K.; Gogoi, H.K.; Prasad, G.B.K.S.; Veer, V. Molecular characterization of midgut microbiota of Aedes albopictus and Aedes aegypti from Arunachal Pradesh, India. Parasit. Vectors 2015, 8, 641. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.V.; Damiani, C.; Accoti, A.; Tallarita, M.; Nunzi, E.; Cappelli, A.; Bozić, J.; Catanzani, R.; Rossi, P.; Valzano, M.; et al. Estimating bacteria diversity in different organs of nine species of mosquito by next generation sequencing. BMC Microbiol. 2018, 18, 126. [Google Scholar] [CrossRef] [PubMed]
- Dickson, L.B.; Ghozlane, A.; Volant, S.; Bouchier, C.; Ma, L.; Vega-Rúa, A.; Dusfour, I.; Jiolle, D.; Paupy, C.; Mayanja, M.; et al. Diverse laboratory colonies of Aedes aegypti harbor the same adult midgut bacterial microbiome. Parasit. Vectors 2018, 11, 207. [Google Scholar] [CrossRef]
- Short, S.M.; Mongodin, E.F.; MacLeod, H.J.; Talyuli, O.A.C.; Dimopoulos, G.; Olson, K.E. Amino acid metabolic signaling influences Aedes aegypti midgut microbiome variability. PLoS Negl. Trop. Dis. 2017, 11, e0005677. [Google Scholar] [CrossRef]
- Gimonneau, G.; Tchioffo, M.T.; Abate, L.; Boissiere, A.; Awono-Ambene, P.; Nsango, S.E.; Christen, R.; Morlais, I. Composition of Anopheles coluzzii and Anopheles gambiae microbiota from larval to adult stages. Infect. Genet. Evol. 2014, 28, 715–724. [Google Scholar] [CrossRef]
- Coon, K.L.; Vogel, K.J.; Brown, M.R.; Strand, M.R. Mosquitoes rely on their gut microbiota for development. Mol. Ecol. 2014, 23, 2727–2739. [Google Scholar] [CrossRef]
- Boissière, A.; Tchioffo, M.T.; Bachar, D.; Abate, L.; Marie, A.; Nsango, S.E.; Shahbazkia, H.R.; Awono-Ambene, P.H.; Levashina, E.A.; Christen, R.; et al. Midgut Microbiota of the Malaria Mosquito Vector Anopheles gambiae and Interactions with Plasmodium falciparum Infection. PLoS Pathog. 2012, 8, e1002742. [Google Scholar] [CrossRef]
- Hegde, S.; Khanipov, K.; Albayrak, L.; Golovko, G.; Pimenova, M.; Saldaña, M.A.; Rojas, M.M.; Hornett, E.A.; Motl, G.C.; Fredregill, C.L.; et al. Microbiome Interaction Networks and Community Structure From Laboratory-Reared and Field-Collected Aedes aegypti, Aedes albopictus, and Culex quinquefasciatus Mosquito Vectors. Front. Microbiol. 2018, 9, 2160. [Google Scholar] [CrossRef]
- Osei-Poku, J.; Mbogo, C.M.; Palmer, W.J.; Jiggins, F.M. Deep sequencing reveals extensive variation in the gut microbiota of wild mosquitoes from Kenya. Mol. Ecol. 2012, 21, 5138–5150. [Google Scholar] [CrossRef] [PubMed]
- Lazaro, J.E.H.; Nitcheu, J.; Predicala, R.Z.; Mangalindan, G.C.; Nesslany, F.; Marzin, D.; Concepcion, G.P.; Diquet, B. Heptyl prodigiosin, a bacterial metabolite, is antimalarial in vivo and non-mutagenic in vitro. J. Nat. Toxins 2002, 11, 367–377. [Google Scholar] [PubMed]
- Itoh, H.; Tago, K.; Hayatsu, M.; Kikuchi, Y. Detoxifying symbiosis: Microbe-mediated detoxification of phytotoxins and pesticides in insects. Nat. Prod. Rep. 2018, 35, 434–454. [Google Scholar] [CrossRef] [PubMed]
- Strand, M.R. Composition and functional roles of the gut microbiota in mosquitoes. Curr. Opin. Insect Sci. 2018, 28, 59–65. [Google Scholar] [CrossRef]
- Zhang, S.; Yin, L.; Liu, Y.; Zhang, D.; Luo, X.; Cheng, J.; Cheng, F.; Dai, J. Cometabolic biotransformation of fenpropathrin by Clostridium species strain ZP3. Biodegradation 2011, 22, 869–875. [Google Scholar] [CrossRef]
- Cycoń, M.; Piotrowska-Seget, Z. Pyrethroid-Degrading Microorganisms and Their Potential for the Bioremediation of Contaminated Soils: A Review. Front. Microbiol. 2016, 7, 1–26. [Google Scholar] [CrossRef]
- Bhatt, P.; Huang, Y.; Zhan, H.; Chen, S. Insight into Microbial Applications for the Biodegradation of Pyrethroid Insecticides. Front. Microbiol. 2019, 10, 1778. [Google Scholar] [CrossRef]
- Xiong, J.; Zhang, X.; Huang, J.; Chen, C.; Chen, Z.; Liu, L.; Zhang, G.; Yang, J.; Zhang, Z.; Zhang, Z.; et al. Fenpropathrin, a Widely Used Pesticide, Causes Dopaminergic Degeneration. Mol. Neurobiol. 2016, 53, 995–1008. [Google Scholar] [CrossRef]
- Kanade, S.N.; Shaikh, S.M.; Ade, A.B.; Khilare, V.C. Degradation of Malathion by Rhizobium isolated from fenugreek (Trigonella foenum graecum). J. Biotechnol. Bioinform. 2010, 1, 240–242. [Google Scholar]
- Sabourmoghaddam, N.; Zakaria, M.P.; Omar, D. Evidence for the microbial degradation of imidacloprid in soils of Cameron Highlands. J. Saudi Soc. Agric. Sci. 2015, 14, 182–188. [Google Scholar] [CrossRef]
- Dada, N.; Vannavong, N.; Seidu, R.; Lenhart, A.; Stenström, T.A.; Chareonviriyaphap, T.; Overgaard, H.J. Relationship between Aedes aegypti production and occurrence of Escherichia coli in domestic water storage containers in rural and sub-urban villages in Thailand and Laos. Acta Trop. 2013, 126, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Minard, G.; Tran, F.-H.; Dubost, A.; Tran-Van, V.; Mavingui, P.; Moro, C.V. Pyrosequencing 16S rRNA genes of bacteria associated with wild tiger mosquito Aedes albopictus: A pilot study. Front. Cell Infect. Microbiol. 2014, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Ozato, N.; Saito, S.; Yamaguchi, T.; Katashima, M.; Tokuda, I.; Sawada, K. Blautia genus associated with visceral fat accumulation in adults 20–76 years of age. NPJ Biofilms Microbiomes. 2019, 5, 28. [Google Scholar] [CrossRef]
- Allison, M.J.; Dawson, K.A.; Mayberry, W.R.; Foss, J.G. Oxalobacter formigenes gen. nov., sp. nov.: Oxalate-degrading anaerobes that inhabit the gastrointestinal tract. Arch. Microbiol. 1985, 141, 1–7. [Google Scholar] [CrossRef]
- PeBenito, A.; Nazzal, L.; Wang, C.; Li, H.; Jay, M.; Noya-Alarcon, O.; Contreras, M.; Lander, O.; Leach, J.; Dominguez-Bello, M.G.; et al. Comparative prevalence of Oxalobacter formigenes in three human populations. Sci. Rep. 2019, 9, 574. [Google Scholar] [CrossRef]
- Tallur, P.N.; Megadi, V.B.; Ninnekar, H.Z. Biodegradation of Cypermethrin by Micrococcus sp. strain CPN 1. Biodegradation 2008, 19, 77–82. [Google Scholar] [CrossRef]
- Chen, S.; Geng, P.; Xiao, Y.; Hu, M. Bioremediation of β-cypermethrin and 3-phenoxybenzaldehyde contaminated soils using Streptomyces aureus HP-S-01. Appl. Microbiol. Biotechnol. 2012, 94, 505–515. [Google Scholar] [CrossRef]
- Gangola, S.; Sharma, A.; Bhatt, P.; Khati, P.; Chaudhary, P. Presence of esterase and laccase in Bacillus subtilis facilitates biodegradation and detoxification of cypermethrin. Sci. Rep. 2018, 8, 12755. [Google Scholar] [CrossRef]
- Vijayakumar, M.M.; More, R.P.; Rangasamy, A.; Gandhi, G.R.; Muthugounder, M.; Thiruvengadam, V.; Samaddar, S.; Jalali, S.K.; Sa, T. Gut Bacterial Diversity of Insecticide-Susceptible and -Resistant Nymphs of the Brown Planthopper Nilaparvata lugens Stå l (Hemiptera: Delphacidae) and Elucidation of Their Putative Functional Roles. J. Microbiol. Biotechnol. 2018, 28, 976–986. [Google Scholar] [CrossRef]
- Echaubard, P.; Duron, O.; Agnew, P.; Sidobre, C.; Noel, V.; Weill, M.; Michalakis, Y.; No, V. Rapid evolution of Wolbachia density in insecticide resistant Culex pipiens. Heredity 2010, 104, 15–19. [Google Scholar] [CrossRef] [PubMed]
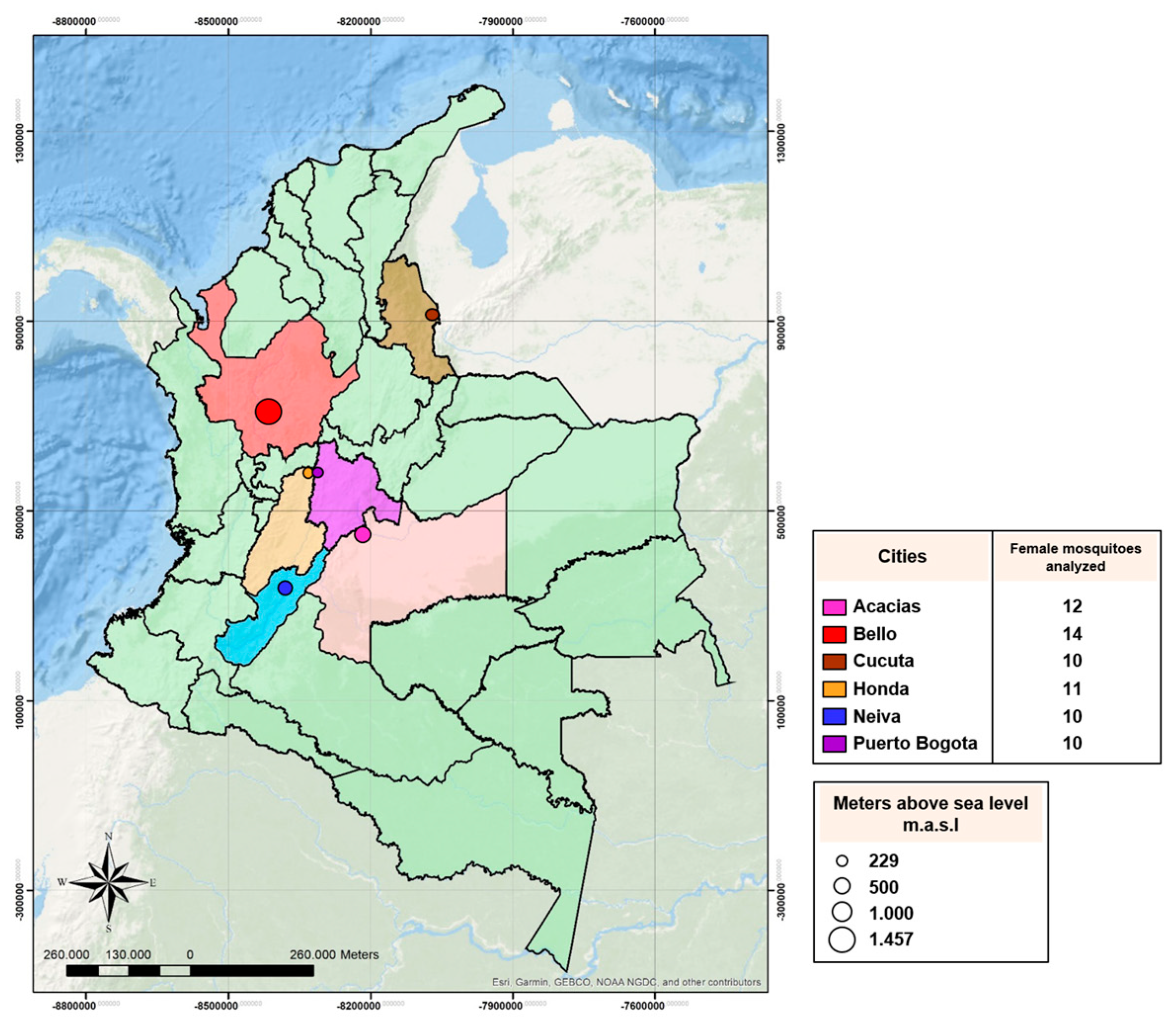
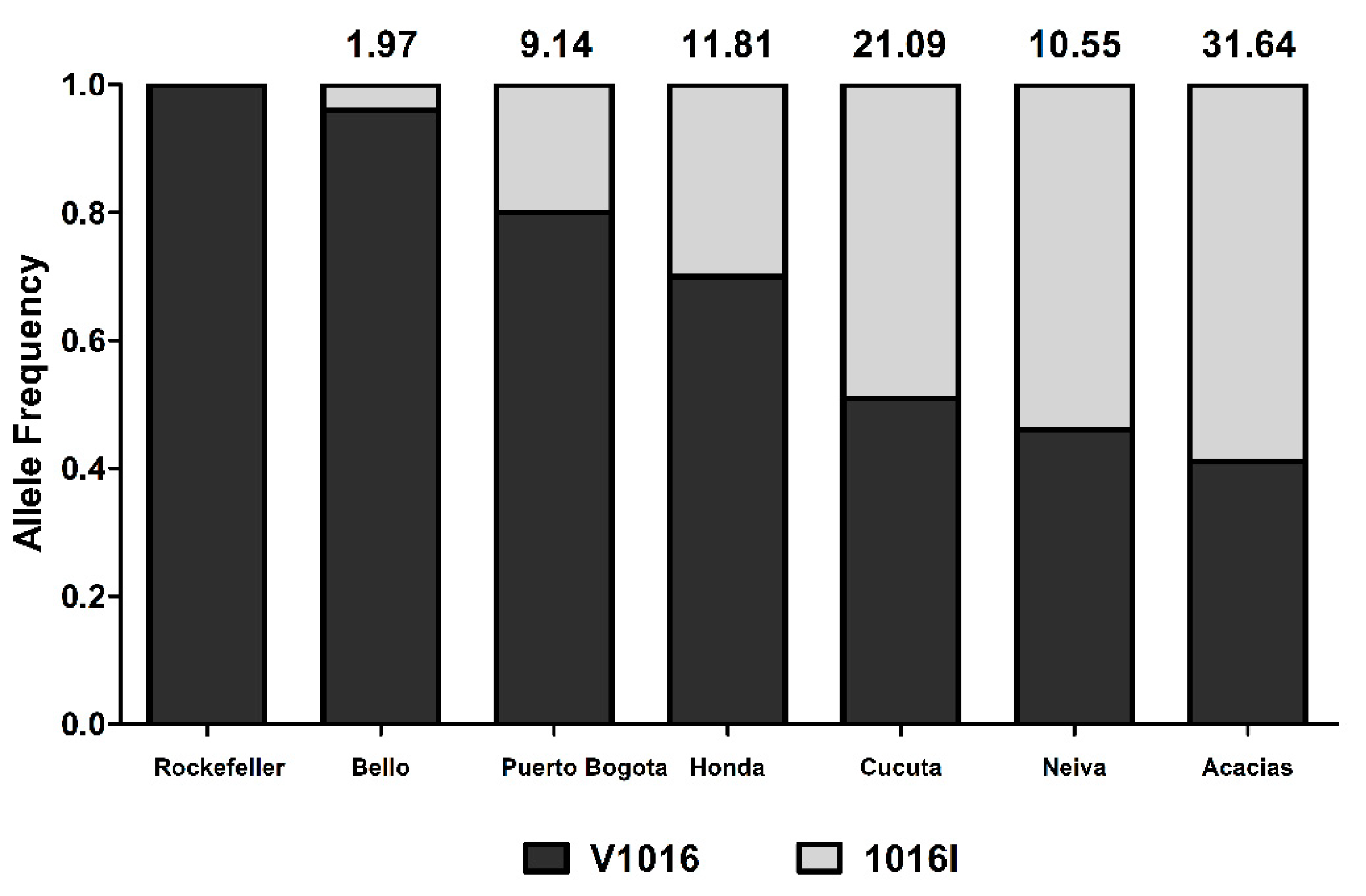
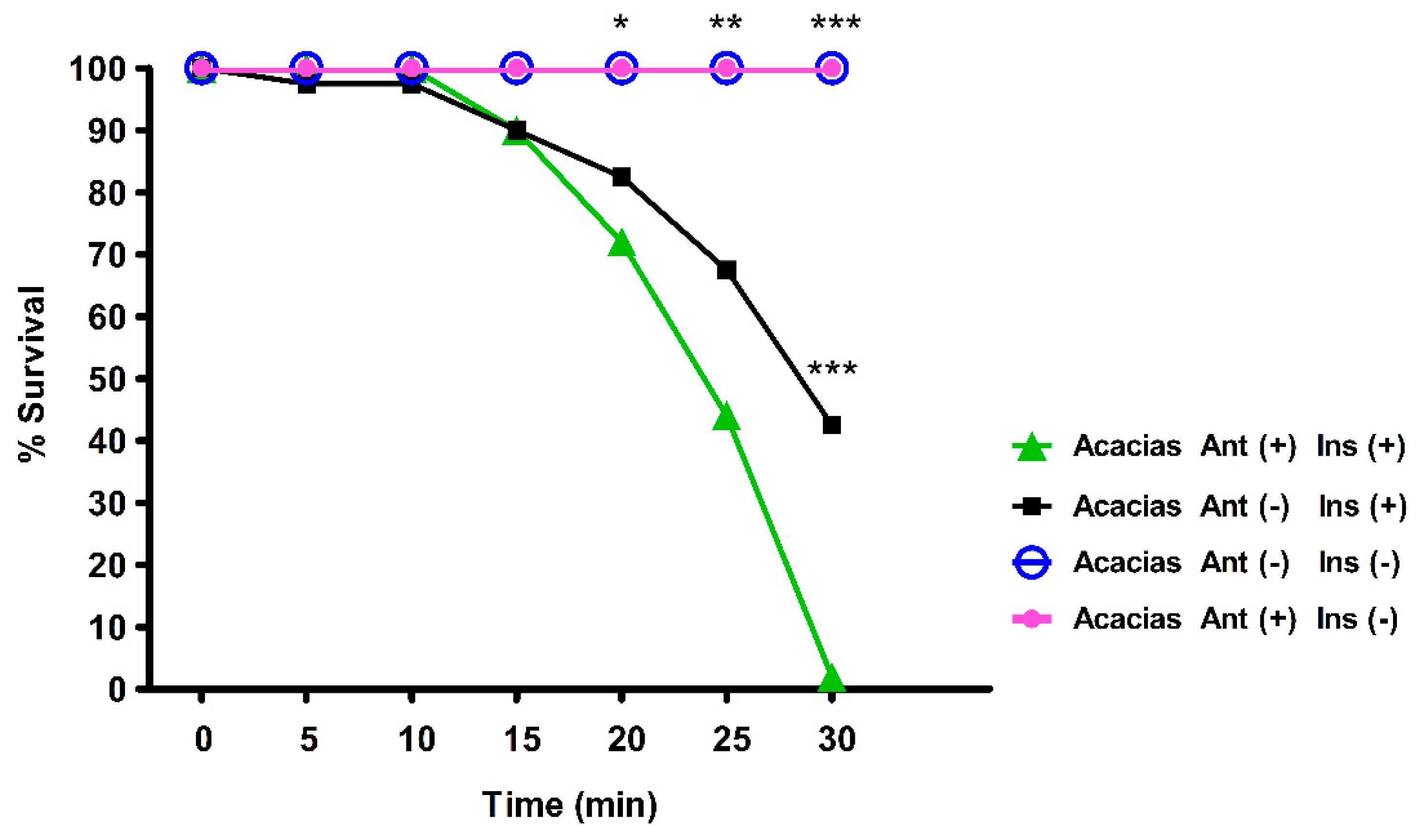
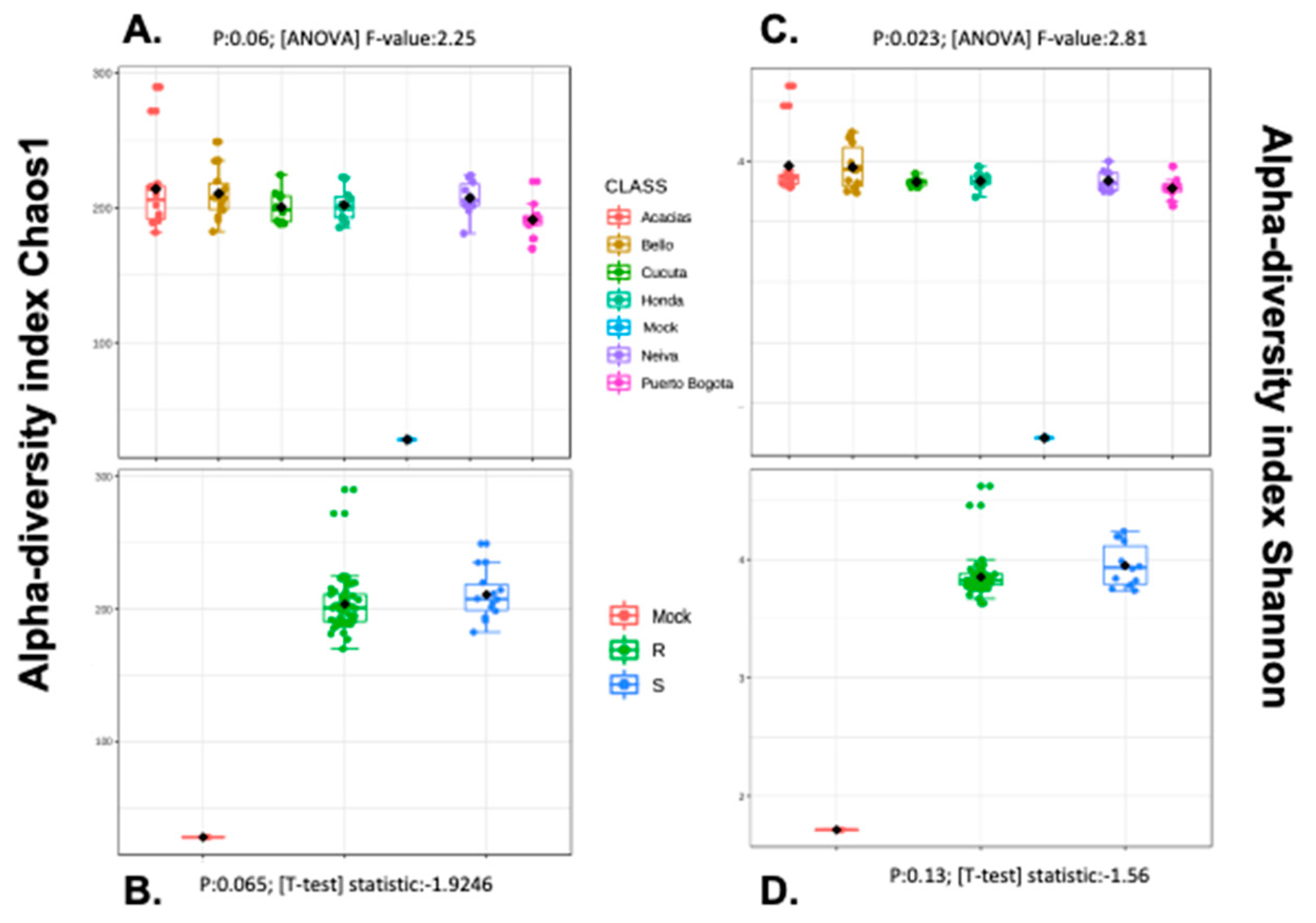
| Cities | RR50 | Phenotype |
|---|---|---|
| Bello (B) | 1.97 | Susceptible |
| Puerto Bogota (P) | 9.14 | Moderate Resistant |
| Neiva (N) | 10.55 | High Resistant |
| Honda (H) | 11.81 | High Resistant |
| Cucuta (C) | 21.09 | High Resistant |
| Acacias (A) | 31.64 | High Resistant |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arévalo-Cortés, A.; Mejia-Jaramillo, A.M.; Granada, Y.; Coatsworth, H.; Lowenberger, C.; Triana-Chavez, O. The Midgut Microbiota of Colombian Aedes aegypti Populations with Different Levels of Resistance to the Insecticide Lambda-cyhalothrin. Insects 2020, 11, 584. https://doi.org/10.3390/insects11090584
Arévalo-Cortés A, Mejia-Jaramillo AM, Granada Y, Coatsworth H, Lowenberger C, Triana-Chavez O. The Midgut Microbiota of Colombian Aedes aegypti Populations with Different Levels of Resistance to the Insecticide Lambda-cyhalothrin. Insects. 2020; 11(9):584. https://doi.org/10.3390/insects11090584
Chicago/Turabian StyleArévalo-Cortés, Andrea, Ana M. Mejia-Jaramillo, Yurany Granada, Heather Coatsworth, Carl Lowenberger, and Omar Triana-Chavez. 2020. "The Midgut Microbiota of Colombian Aedes aegypti Populations with Different Levels of Resistance to the Insecticide Lambda-cyhalothrin" Insects 11, no. 9: 584. https://doi.org/10.3390/insects11090584
APA StyleArévalo-Cortés, A., Mejia-Jaramillo, A. M., Granada, Y., Coatsworth, H., Lowenberger, C., & Triana-Chavez, O. (2020). The Midgut Microbiota of Colombian Aedes aegypti Populations with Different Levels of Resistance to the Insecticide Lambda-cyhalothrin. Insects, 11(9), 584. https://doi.org/10.3390/insects11090584






