Control of Pest Grasshoppers in North America †
Simple Summary
Abstract
1. Introduction
2. Rangeland Grasshoppers (Orthoptera: Acrididae) and Their Economic Importance
3. Strategies for Managing Grasshoppers
3.1. Chemical Control
3.1.1. Carbaryl
3.1.2. Malathion
3.1.3. Diflubenzuron
3.2. Biological Control
3.2.1. Viruses
3.2.2. Bacteria
3.2.3. Nematodes
3.2.4. Microsporidia
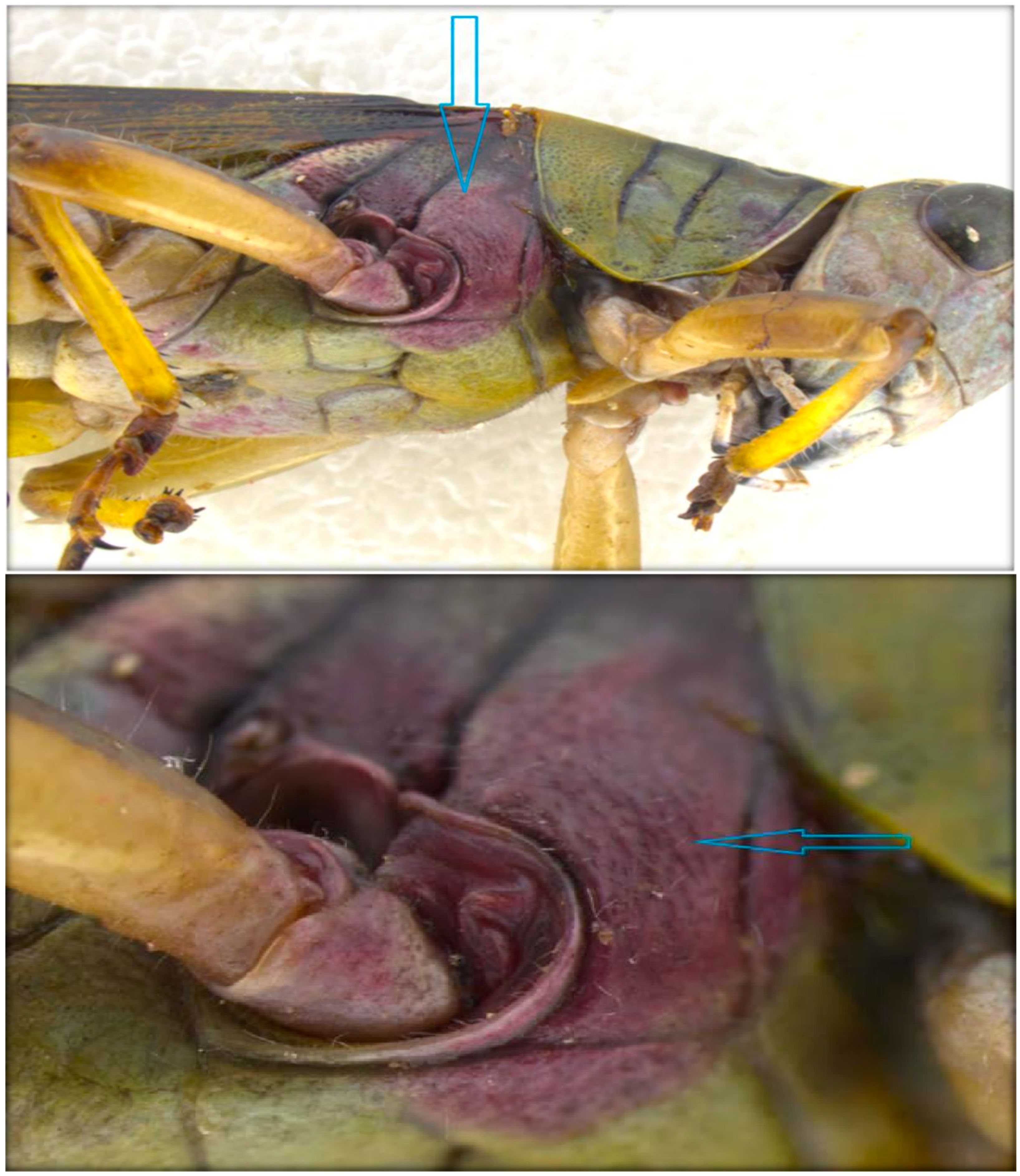
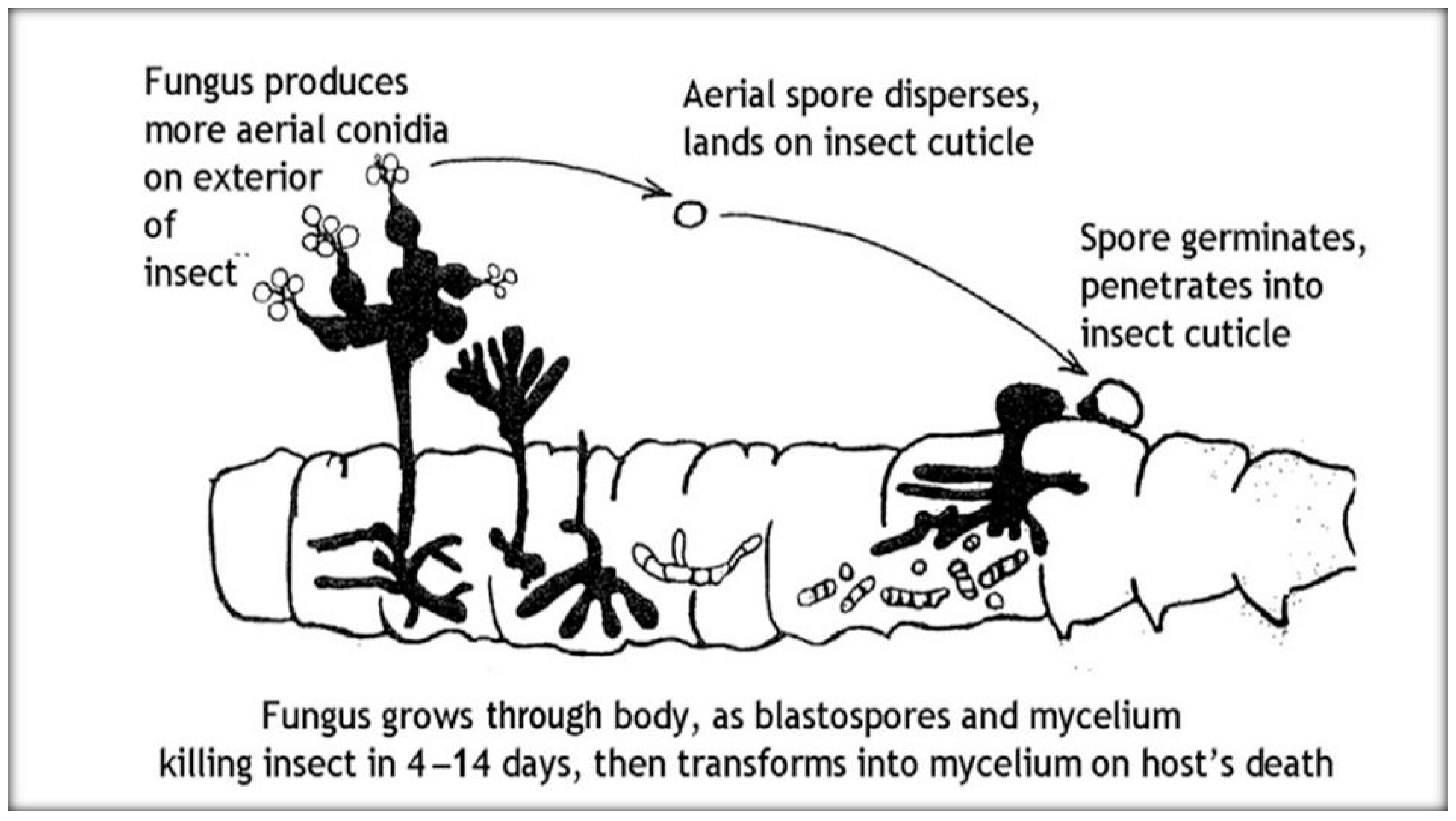
3.2.5. Fungi
3.3. Bait Formulation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Branson, D.H.; Sword, G.A. An experimental analysis of grasshopper community responses to fire and livestock grazing in a northern mixed-grass prairie. Environ. Entomol. 2010, 39, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Pfadt, R.E. Field Guide to Common Western Grasshoppers; Wyoming Agricultural Experiment Station: Laramie, WY, USA, 1994; Volume 912. [Google Scholar]
- Dakhel, W.H.; Latchininsky, A.V.; Jaronski, S.T. Efficacy of two entomopathogenic fungi, Metarhizium brunneum, strain F52 alone and combined with Paranosema locustae against the migratory grasshopper, Melanoplus sanguinipes, under laboratory and greenhouse conditions. Insects 2019, 10, 94. [Google Scholar] [CrossRef] [PubMed]
- DeBrey, L.; Brewer, M.J.; Lockwood, J.A. Rangeland Grasshopper Management; Extension Bulletin B-980; College of Agriculture, University of Wyoming: Laramie, WY, USA, 1993. [Google Scholar]
- Latchininsky, A.; Sword, G.; Sergeev, M.; Cigliano, M.M.; Lecoq, M. Locusts and grasshoppers: Behavior, ecology and biogeography. Open Access 2011. [Google Scholar] [CrossRef]
- Hostetter, D.L. Natural enemies attacking grasshopper nymphs and adults. In Grasshopper Integrated Pest Management User Handbook; Cunningham, G.L., Sampson, M.W., Eds.; U.S. Department of Agriculture: Washington, DC, USA, 2000; pp. I.8-1–I.8-7. [Google Scholar]
- Pfadt, R.E. A Field Guide to Common Western Grasshoppers, 3rd ed.; Wyoming Agricultural Experiment Station Bulletin 912; Wyoming Agricultural Experiment Station: Laramie, WY, USA, 2002; p. 228. [Google Scholar]
- Murray, D.W. The Biology, Ecology, and Management of the Migratory Grasshopper, Melanoplus sanguinipes (Fab.); Distance Master of science in Entomology Project 13; University of Nebraska: Lincoln, NE, USA, 2016. [Google Scholar]
- Zhang, L.; Lecoq, M.; Latchininsky, A.; Hunter, D. Locust and grasshopper management. Annu. Rev. Entomol. 2019, 64, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Bernays, E.A. Food tastes and toxic effects: Associative learning by the polyphagous grasshopper Schistocerca americana (Drury) (Orthoptera: Acrididae). Anim. Behav. 1990, 39, 163–173. [Google Scholar] [CrossRef]
- Branson, D.H.; Sword, G.A. Grasshopper herbivory affects native plant diversity and abundance in a grassland dominated by the exotic grass Agropyron cristatum. Restor. Ecol. 2009, 17, 89–96. [Google Scholar] [CrossRef]
- Hastings, J.D.; Latchininsky, A.V.; Schell, S.P. Sustainability of grasshopper management and support through CARMA. In Proceedings of the 2009 42nd Hawaii International Conference on System Sciences, Waikoloa, Hawaii, 26 June 2009; pp. 1–10. [Google Scholar]
- Lockwood, J.R. The fate of the Rocky Mountain locust, Melanoplus spretus Walsh: Implications for conservation biology. Terr. Arthropod Rev. 2010, 3, 129–160. [Google Scholar] [CrossRef]
- Skinner, K.M. The past, present, and future of rangeland grasshopper management. Rangel. Arch. 2000, 22, 24–28. [Google Scholar] [CrossRef]
- Davis, R.M.; Skold, M.D.; Berry, J.S.; Kemp, W.P. The economic threshold for grasshopper control on public rangelands. J. Agri. Resour. Econ. 1992, 17, 56–65. [Google Scholar]
- Cunningham, G.L.; Sampson, M.W. Grasshopper Integrated Pest Management User Handbook; US Department of Agriculture, Animal and Plant Health Inspection Service: Washington, DC, USA, 2000.
- Hastings, J.D.; Latchininsky, A.V.; Adelung, T.J.; Schell, S.P. Early Assessment of an Approach to Determining the Predictive Coverage of Case-Based Reasoning with Adaptation through CARMA. In Proceedings of the 2014 47th Hawaii International Conference on System Sciences, Waikoloa, HI, USA, 6–9 January 2014; pp. 857–864, IEEE. [Google Scholar]
- Faust, R.M. General introduction to areawide pest management. In Areawide Pest Management Theory Implementation; Koul, O., Cuperus, G., Elliot, N., Eds.; CABI: Wallingford, UK, 2008; Chapter 1; pp. 1–14. [Google Scholar]
- Headley, J.C. Defining the economic threshold. Pest Control Strateg. Future 2013, 1972, 100–108. [Google Scholar]
- United States Department of Agriculture. Rangeland Grasshopper and Mormon Cricket Suppression Program, Final Environmental Impact Statement—2002; U.S. Department of Agriculture: Washington, DC, USA, 2002.
- Latchininsky, A.V.; VanDyke, K.A. Grasshopper and locust control with poisoned baits: A renaissance of the old strategy? Outlooks Pest Manag. 2006, 17, 105–111. [Google Scholar] [CrossRef]
- Quinn, M.A.; Kepner, R.L.; Walgenbach, D.D.; Foster, R.N.; Bohls, R.A.; Pooler, P.D.; Reuter, K.C.; Swain, J.L. Effect of habitat characteristics and perturbation from insecticides on the community dynamics of ground beetles (Coleoptera: Carabidae) on mixed-grass rangeland. Environ. Entomol. 1991, 20, 1285–1294. [Google Scholar] [CrossRef]
- Smith, D.I.; Lockwood, J.A.; Latchininsky, A.V.; Legg, D.E. Changes in non-target arthropod populations following application of liquid bait formulations of insecticides for control of rangeland grasshoppers. J. Int. Pest Manag. 2006, 52, 125–139. [Google Scholar] [CrossRef]
- Lockwood, J.A.; Schell, S.P.; Foster, R.N.; Reuter, C.; Rachadi, T. Reduced agent-area treatments (RAAT) for management of rangeland grasshoppers: Efficacy and economics under operational conditions. J. Int. Pest Manag. 2000, 46, 29–42. [Google Scholar] [CrossRef]
- McNary, T.J.; Shambaugh, B.A.; Elliston, R.J.; Brown, C.L. Cooperative rangeland grasshopper suppression in Wyoming (USA) in 2010. Metaleptea 2011, 31, 10–12. [Google Scholar]
- Foster, R.N.; Onsager, J.A. A review of chemical sprays in cooperative rangeland control programs. In Grasshopper Integrated Pest Management User Handbook; Tech. Bul. No. 1809, Sec. II.4; U.S. Department of Agriculture, Animal and Plant Health Inspection Service: Washington, DC, USA, 1996. [Google Scholar]
- Pfadt, R.E. Effect of ULV Malathion Grasshopper Spray on Nontarget Arthropods; Agricultural Experiment Station: Laramie, WY, USA, 1985. [Google Scholar]
- Foster, R.N.; Onsager, J.A. Sprays versus baits. In Grasshopper Integrated Pest Management User Handbook; Tech. Bul. No. 1809, Sec. II.3; U.S. Department of Agriculture, Animal and Plant Health Inspection Service: Washington, DC, USA, 1996. [Google Scholar]
- Catangui, M.A.; Fuller, B.W.; Walz, A.W. Impact of Dimilin® on nontarget arthropods and its efficacy against rangeland grasshoppers. In Grasshoppers: Their Biology, Identification and Management; Branson, D.H., Redlin, B., Eds.; USDA Agricultural Research Service: Washington, DC, USA, 2001. [Google Scholar]
- FAO. Desert Locust Guidelines; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001. [Google Scholar]
- St Leger, R.J.; Wang, C.; Stock, S.; Vandenberg, J.; Glazer, I. Entomopathonic fungi and the genomics era. In Insect Pathogens: Molecular Approaches and Techniques; CABI: Wallingford, UK, 2009; pp. 365–400. [Google Scholar]
- Soper, R.S. Agricultural Research Service national biological control program: Policy, and constraints. In Regulations and Guidelines: Critical Issues in Biological Control; Charudattan, R., Browning, W.H., Eds.; Technical Bulletin No. 66; USDA: Washington, DC, USA, 1992; pp. 49–52. [Google Scholar]
- Fiedler, A.; Tuell, J.; Isaacs, R.; Landis, D. Attracting Beneficial Insects with Native Flowering Plants; Michigan State University, Extension Service: East Lansing, MI, USA, 2007. [Google Scholar]
- Baker, G.L.; Dysart, R.J.; Pigott, R.G. Parasitism of grasshopper and locust eggs (Orthoptera: Acrididae) by Scelio species (Hymenoptera: Scelionidae) in southern Australia. Aust. J. Zool. 1996, 44, 427–443. [Google Scholar] [CrossRef]
- Catangui, M.A. Economic Thresholds in Soybeans: Grasshopper and Bean Leaf Beetle; SDSU CES FS905; South Dakota State University: Brookings, SD, USA, 1999. [Google Scholar]
- Bidochka, M.J.; Walsh, S.R.; Ramos, M.E.; St Leger, R.J.; Silver, J.C.; Roberts, D.W. Fate of biological control introductions: Monitoring an Australian fungal pathogen of grasshoppers in North America. Proc. Natl. Acad. Sci. USA 1996, 93, 918–921. [Google Scholar] [CrossRef]
- Streett, D.A.; Woods, S.A.; Erlandson, M.A. Entomopoxviruses of grasshoppers and locusts: Biology and biological control potential. Mem. Entomol. Soc. Can. 1997, 129, 115–130. [Google Scholar] [CrossRef]
- Sandhu, S.S.; Sharma, A.K.; Beniwal, V.; Goel, G.; Batra, P.; Kumar, A.; Jaglan, S.; Sharma, A.K.; Malhotra, S. Myco-biocontrol of insect pests: Factors involved, mechanism, and regulation. J. Pathog. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Clem, R.J.; Passarelli, A.L. Baculoviruses: Sophisticated pathogens of insects. PLoS Pathog. 2013, 9, e1003729. [Google Scholar] [CrossRef]
- Hernandez-Crespo, P.; Veyrunes, J.C.; Cousserans, F.; Bergoin, M. The spheroidin of an entomopoxvirus isolated from the grasshopper Anacridium aegyptium (AaEPV) shares low homology with spheroidins from lepidopteran or coleopteran EPVs. Virus Res. 2000, 67, 203–213. [Google Scholar] [CrossRef]
- Streett, D.A.; McGuire, M.R. Pathogenic diseases of grasshoppers. In Biology of Grasshoppers; Streett, D.A., McGuire, M.R., Chapman, R.F., Joern, A., Eds.; Wiley-Interscience: New York, NY, USA, 1990; pp. 483–516. [Google Scholar]
- Waterfield, N.R.; Daborn, P.J. Genomic islands in Photorhabdus. Trends. Microbiol. 2002, 10, 541–545. [Google Scholar] [CrossRef]
- Lord, J.C. From Metchnikoff to Monsanto and beyond: The path of microbial control. J. Invertebr. Pathol. 2005, 89, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Tao, K.; Long, Z.; Liu, K.; Tao, Y.; Liu, S. Purification and properties of a novel insecticidal protein from the locust pathogen Serratia marcescens HR-3. Curr. Microbial. 2006, 52, 45–49. [Google Scholar] [CrossRef]
- Schünemann, R.; Knaak, N.; Fiuza, L.M. Mode of action and specificity of Bacillus thuringiensis toxins in the control of caterpillars and stink bugs in soybean culture. Int. Sch. Res. Not. 2014, 135675. [Google Scholar] [CrossRef]
- Song, L.; Gao, M.; Dai, S.; Wu, Y.; Yi, D.; Li, R. Specific activity of a Bacillus thuringiensis strain against Locusta migratoria manilensis. J. Invertebr. Pathol. 2008, 98, 169–176. [Google Scholar] [CrossRef]
- Wu, Y.; Lei, C.F.; Yi, D.; Liu, P.M.; Gao, M.Y. Novel Bacillus thuringiensis δ-endotoxin active against Locusta migratoria manilensis. Appl. Environ. Microbiol. 2011, 77, 3227–3233. [Google Scholar] [CrossRef]
- Baker, G.L.; Capinera, J.L. Nematodes and nematomorphs as control agents of grasshoppers and locusts. Mem. Entomol. Soc. Can. 1997, 129, 157–211. [Google Scholar] [CrossRef]
- Lewis, E.E.; Clarke, D.J. Nematode parasites and entomopathogens. In Insect Pathology; Academic Press: Cambridge, MA, USA, 2012; pp. 395–424. [Google Scholar]
- Lacey, L.A.; Georgis, R. Entomopathogenic nematodes for control of insect pests above and below ground with comments on commercial production. J. Nematol. 2012, 44, 218. [Google Scholar]
- Ferreira, T.; Malan, A.P. Xenorhabdus and Photorhabdus, bacterial symbionts of the entomopathogenic nematodes Steinernema and Heterorhabditis and their in vitro liquid mass culture: A review. Afr. Entomol. 2014, 22, 1–14. [Google Scholar] [CrossRef]
- Nicolas, B.; Epsky, N.D.; Capinera, J.L. Susceptibility of Melanoplus sanguinipes (Orthoptera: Acrididae) nymphs to Steinernema carpocapsae and S. scapterisci (Nematoda: Steinernematidae). Environ. Entomol. 1995, 24, 762–769. [Google Scholar] [CrossRef]
- Keeling, P. Five questions about microsporidia. PLoS Pathog. 2009, 5, e1000489. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Weiss, L.M. Microsporidia: Obligate intracellular pathogens within the fungal kingdom. In The Fungal Kingdom; Wiley Online Library: New York, NY, USA, 2017; pp. 97–113. [Google Scholar] [CrossRef]
- Mathis, A.; Weber, R.; Deplazes, P. Zoonotic potential of the microsporidia. Clin. Microbiol. Rev. 2005, 18, 423–445. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Guo, Y.; Xu, C.; Tan, S.; Miao, J.; Feng, Y.; Zhao, H.; Leger, R.J.S.; Fang, W. Unveiling the mechanism by which microsporidian parasites prevent locust swarm behavior. Proc. Natl. Acad. Sci. USA 2014, 111, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.J.; Hunter, D.M.; Shi, W.P. Effect of Paranosema (Nosema) locustae (Microsporidia) on morphological phase transformation of Locusta migratoria manilensis (Orthoptera: Acrididae). Biocontrol Sci. Technol. 2010, 20, 683–693. [Google Scholar] [CrossRef]
- United States Environmental Protection Administration (USEPA). Nosema Locustae (117001) Fact Sheet; USEPA: Washington, DC, USA, 2000.
- Solter, L.F.; Becnel, J.J.; Vávra, J. Research methods for entomopathogenic microsporidia and other protists. In Manual of Techniques in Invertebrate Pathology; Lacey, L.A., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 12, pp. 329–371. [Google Scholar]
- Tounou, A.K.; Kooyman, C.; Douro-Kpindou, O.K.; Poehling, H.M. Interaction between Paranosema locustae and Metarhizium anisopliae var. acridum, two pathogens of the desert locust, Schistocerca gregaria under laboratory conditions. J. Invertebr. Pathol. 2008, 97, 203–210. [Google Scholar] [CrossRef]
- Tounou, A.K.; Kooyman, C.; Douro-Kpindou, O.K.; Poehling, H.M. Combined field efficacy of Paranosema locustae and Metarhizium anisopliae var. acridum for the control of sahelian grasshoppers. Biocontrol 2008, 53, 813–828. [Google Scholar] [CrossRef]
- Johnson, D.L.; Henry, J.E. Low rates of insecticides and Nosema locustae (Microsporidia: Nosematidae) on baits applied to roadsides for grasshopper (Orthoptera: Acrididae) control. J. Econ. Entomol. 1987, 80, 685–689. [Google Scholar] [CrossRef]
- Vega, F.E.; Kaya, H.K. Insect Pathology; Academic Press: Cambridge, MA, USA, 2012. [Google Scholar]
- Streett, D.A. Nosema locustae. In Grasshoppers: Their Biology, Identification and Management; Branson, D.H., Redlin, B., Eds.; USDA Agricultural Research Service: Washington, DC, USA, 2001. [Google Scholar]
- Köhler, J.R.; Casadevall, A.; Perfect, J. The spectrum of fungi that infects humans. Cold Spring Harb. Perspect. Med. 2015, 5, 019273. [Google Scholar] [CrossRef]
- Jaronski, S.T. Mass production of entomopathogenic fungi: State of the art. In Mass Production of Beneficial Organisms; Academic Press: London, UK, 2014; pp. 357–413. [Google Scholar]
- De Faria, M.R.; Wraight, S.P. Mycoinsecticides and mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control. 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Sinha, K.K.; Choudhary, A.K.; Kumari, P. Entomopathogenic fungi. In Ecofriendly Pest Management for Food Security; Academic Press: Cambridge, MA, USA, 2016; pp. 475–505. [Google Scholar]
- Jaronski, S.T.; Goettel, M.S. Development of Beauveria bassiana for control of grasshoppers and locusts. Mem. Entomol. Soc. Can. 1997, 129, 225–237. [Google Scholar] [CrossRef]
- Zimmermann, G. Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii. Biocontrol Sci. Technol. 2007, 17, 553–596. [Google Scholar] [CrossRef]
- Kim, J.J.; Goettel, M.S.; Gillespie, D.R. Evaluation of Lecanicillium longisporum, Vertalec® for simultaneous suppression of cotton aphid, Aphis gossypii, and cucumber powdery mildew, Sphaerotheca fuliginea, on potted cucumbers. Biol. Control. 2008, 45, 404–409. [Google Scholar] [CrossRef]
- Dakhel, W.H. Biological Control of Pest Grasshoppers with Fungal Entomopathogens. Ph.D. Thesis, University of Wyoming, Laramie, WY, USA, 2016. [Google Scholar]
- Zimmermann, G. Review on safety of the entomopathogenic fungus Metarhizium anisopliae. Biocontrol Sci.Technol. 2007, 17, 879–920. [Google Scholar] [CrossRef]
- Aw, K.M.S.; Hue, S.M. Mode of infection of Metarhizium spp. fungus and their potential as biological control agents. J. Fungi 2017, 3, 30. [Google Scholar] [CrossRef]
- Foster, R.N.; Jaronski, S.; Reuter, K.C.; Black, L.R.; Schlothauer, R.; Harper, J.; Jech, L.E. Simulated aerial sprays for field cage evaluation of Beauveria bassiana and Metarhizium brunneum (Ascomycetes: Hypocreales) against Anabrus simplex (Orthoptera: Tettigoniidae) in Montana. Biocontrol Sci. Technol. 2011, 21, 1331–1350. [Google Scholar] [CrossRef]
- Athanassiou, C.G.; Kavallieratos, N.G.; Rumbos, C.I.; Kontodimas, D.C. Influence of temperature and relative humidity on the insecticidal efficacy of Metarhizium anisopliae against larvae of Ephestia kuehniella (Lepidoptera: Pyralidae) on wheat. J. Insect Sci. 2017, 17, 1–7. [Google Scholar] [CrossRef]
- Bateman, R.P.; Carey, M.; Moore, D.E.; Prior, C. The enhanced infectivity of Metarhizium flavoviride in oil formulations to desert locusts at low humidities. Ann. Appl. Biol. 1993, 122, 145–152. [Google Scholar] [CrossRef]
- Rangel, D.E.; Alston, D.G.; Roberts, D.W. Effects of physical and nutritional stress conditions during mycelial growth on conidial germination speed, adhesion to host cuticle, and virulence of Metarhizium anisopliae, an entomopathogenic fungus. Mycol. Res. 2008, 112, 1355–1361. [Google Scholar] [CrossRef]
- Fuller, B.W.; Catangui, M.A.; Boetel, M.A.; Foster, R.N.; Wang, T.; Walgenbach, D.D.; Walz, A.W. Bran bait or liquid insecticide treatments for managing grasshoppers on croplands adjacent to rangeland or conservation reserve program acreages. In Grasshopper Integrated Pest Management User Handbook; Technology Bull. 1809; U.S. Department of Agriculture, Animal and Plant Health Inspection Service: Washington, DC, USA, 1996. [Google Scholar]
- United States Entomological Commission. Report of the United States Entomological Commission; US Government Printing Office: Washington, DC, USA, 1878.
- Foster, R.N.; Quinn, M.A.; Reuter, K.C.; Colletto, D.; Houston, R.; Puclik, M.J.; Scott, A.; Radsick, B. Comparison of single and multiple applications of an insecticidal bait for controlling grasshoppers (Orthoptera: Acrididae) on mixed-grass prairie. J. Kans. Entomol. Soc. 1999, 1, 181–189. [Google Scholar]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dakhel, W.H.; Jaronski, S.T.; Schell, S. Control of Pest Grasshoppers in North America. Insects 2020, 11, 566. https://doi.org/10.3390/insects11090566
Dakhel WH, Jaronski ST, Schell S. Control of Pest Grasshoppers in North America. Insects. 2020; 11(9):566. https://doi.org/10.3390/insects11090566
Chicago/Turabian StyleDakhel, Wahid H., Stefan T. Jaronski, and Scott Schell. 2020. "Control of Pest Grasshoppers in North America" Insects 11, no. 9: 566. https://doi.org/10.3390/insects11090566
APA StyleDakhel, W. H., Jaronski, S. T., & Schell, S. (2020). Control of Pest Grasshoppers in North America. Insects, 11(9), 566. https://doi.org/10.3390/insects11090566





