Foliar Spraying of Tomato Plants with Systemic Insecticides: Effects on Feeding Behavior, Mortality and Oviposition of Bemisia tabaci (Hemiptera: Aleyrodidae) and Inoculation Efficiency of Tomato Chlorosis Virus
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Whitefly Colony and Test Plants
2.2. Stylet Activity of Bemisia tabaci MEAM1 in Plants Treated with Insecticides
2.3. Effect of Insecticides on Mortality, Oviposition and Transmission of ToCV by Bemisia tabaci MEAM1 in Tomato Plants
2.4. Data Analysis
3. Results
3.1. Stylet Activity of B. tabaci on Plants Treated with Insecticides
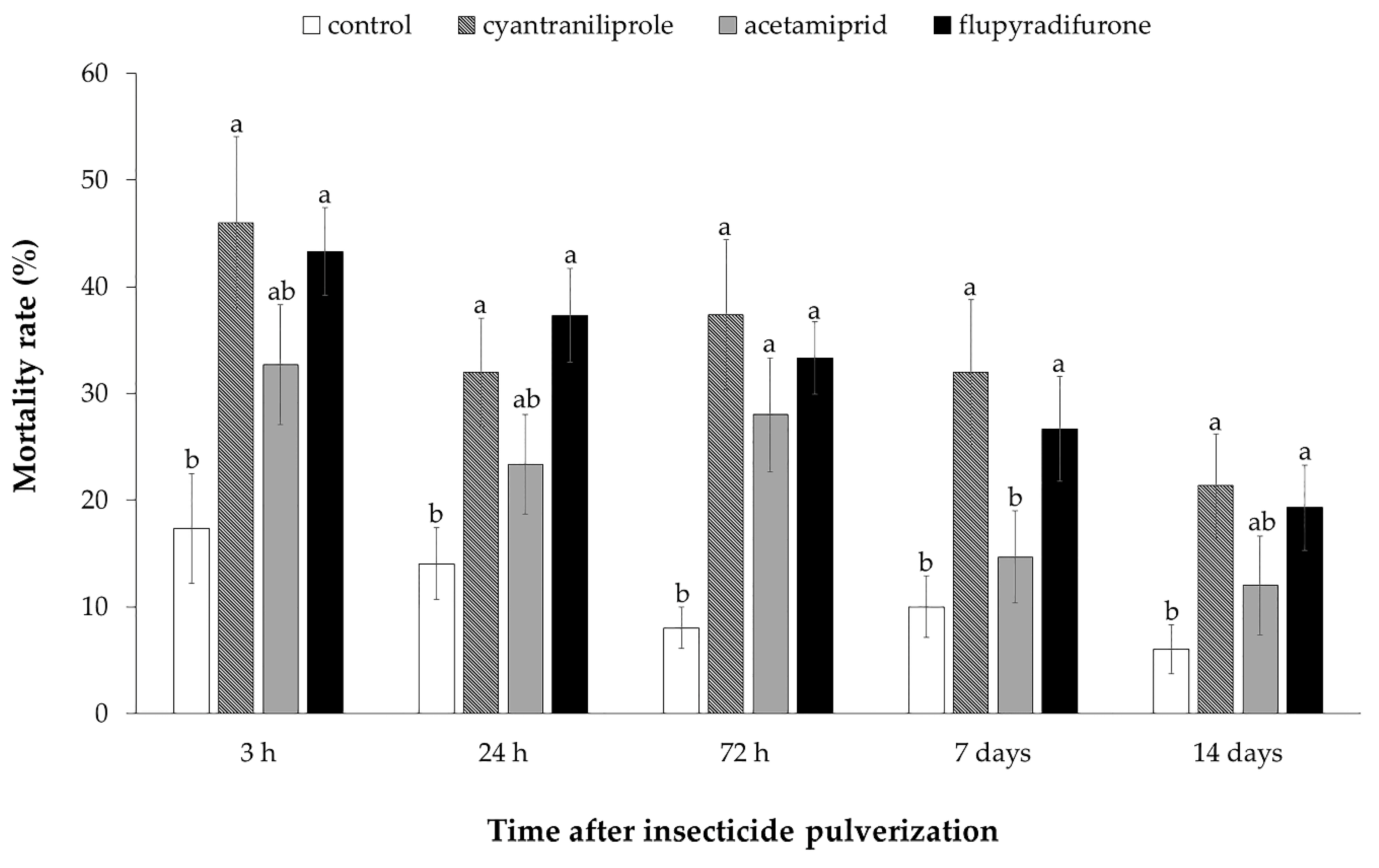
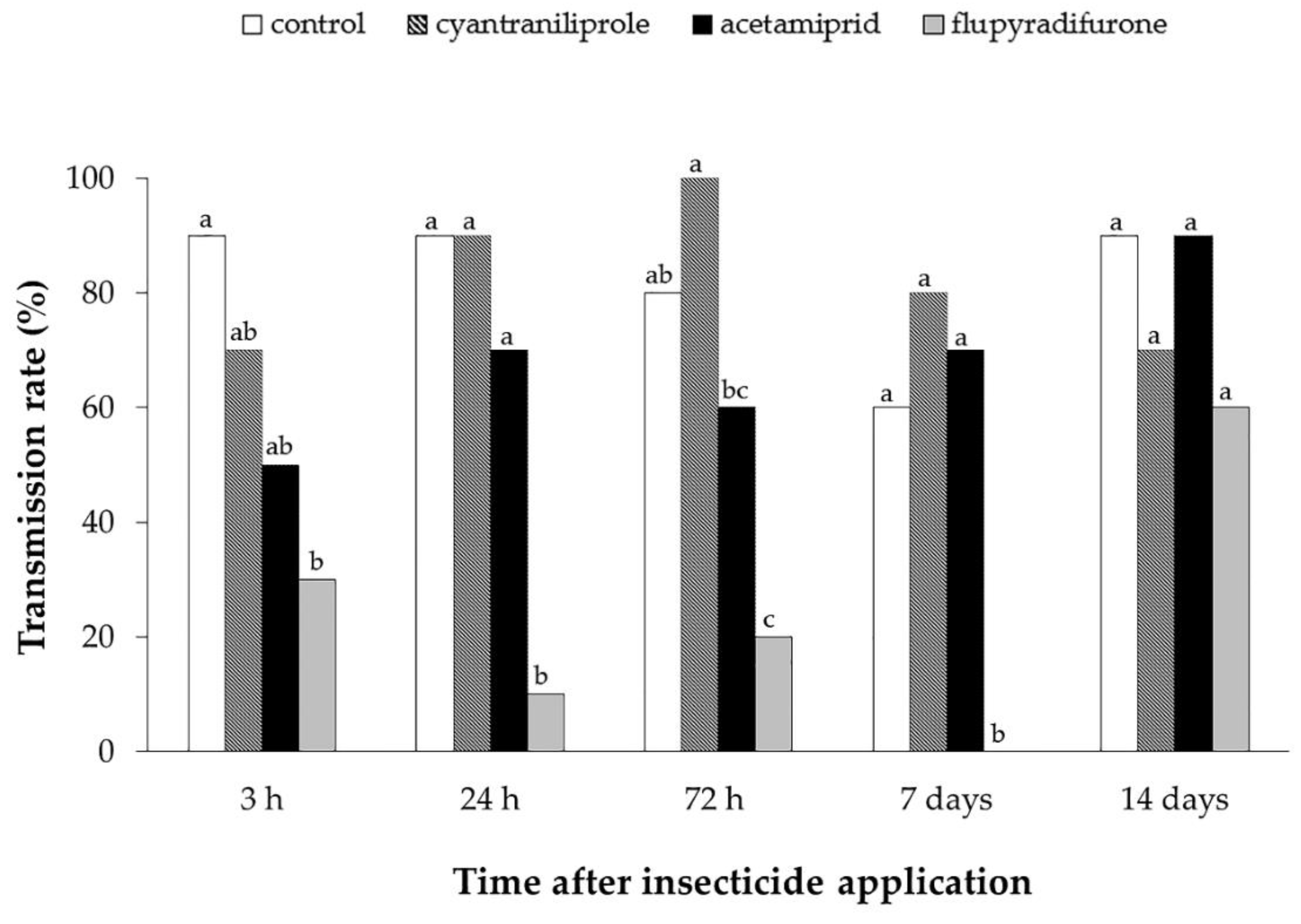
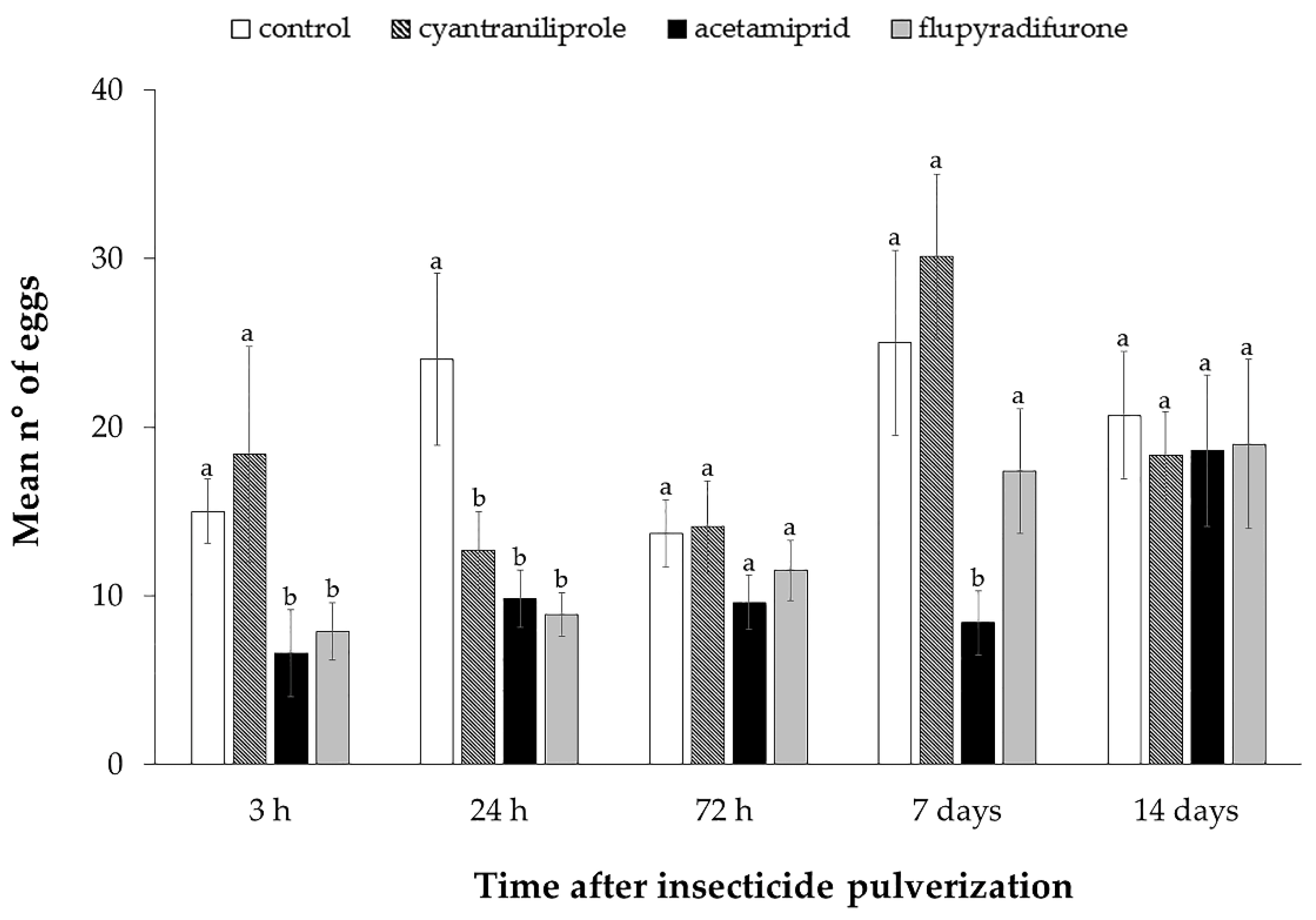
3.2. Effect of Insecticides on Mortality, Oviposition and ToCV Transmission by Bemisia tabaci MEAM1 in Tomato Plants
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Castle, S.; Palumbo, J.; Prabhaker, N. Newer insecticides for plant virus disease management. Virus Res. 2009, 141, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.K. Phylogenetic biology of the Bemisia tabaci sibling species group. In Bemisia: Bionomics and Management of a Global Pest; Stansly, P.A., Naranjo, S.E., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 31–67. [Google Scholar]
- Dinsdale, A.; Cook, L.; Riginos, C.; Buckley, Y.; De Barro, P.J. Refined global analysis of Bemisia tabaci (Hemiptera: Sternorrhyncha: Aleyrodoidea: Aleyrodidae) mitochondrial cytochrome oxidase 1 to identify species level genetic boundaries. Ann. Entomol. So. Am. 2010, 103, 196–208. [Google Scholar] [CrossRef]
- De Barro, P.J.; Liu, S.S.; Boykin, L.M.; Dinsdale, A.B. Bemisia tabaci: A statement of species status. Annu. Rev. Entomol. 2011, 56, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Martinez, S.S.; Carvalho, A.O.R.; Vieira, L.G.; Nunes, L.M.; Bianchini, A. Identification, geographical distribution and host plants of Bemisia tabaci (Genn.) biotypes (Homoptera: Aleyrodidae) in the State of Paraná, Brazil. An. Soc. Entomol. Bras. 2000, 29, 597–603. [Google Scholar] [CrossRef]
- Li, S.J.; Ahmed, M.Z.; Ren, S.X.; Du, Y.Z.; Wu, J.H.; Cuthbertson, A.G.S.; Qiu, B.-L. Host plants and natural enemies of Bemisia tabaci (Hemiptera: Aleyrodidae) in China. Insect Sci. 2011, 18, 101–120. [Google Scholar] [CrossRef]
- Polston, J.E.; De Barro, P.; Boykin, L.M. Transmission specificities of plant viruses with the newly identified species of the Bemisia tabaci species complex. Pest. Manag. Sci. 2014, 70, 1547–1552. [Google Scholar] [CrossRef]
- Lourenção, A.L.; Krause-Sakate, R.; Valle, G.E. Mosca-branca Bemisia tabaci (Gennadius) biótipo B. In Pragas Introduzidas no Brasil: Insetos e Ácaros; Vilela, E.F., Zucchi, R.A., Eds.; Fealq: Piracicaba, Brazil, 2015; pp. 682–707. [Google Scholar]
- Rojas, M.R.; Macedo, M.A.; Maliano, M.R.; Soto-Aguilar, M.; Souza, J.O.; Briddon, R.W.; Kenyon, L.; Bustamante, R.F.R.; Zerbini, F.M.; Adkins, S.; et al. World Management of Geminiviruses. Annu. Revi. Phytopathol. 2018, 56, 637–677. [Google Scholar] [CrossRef]
- Navas-Castillo, J.; Fiallo-Olivé, E.; Sánchez-Campos, S. Emerging virus diseases transmitted by whiteflies. Annu. Rev. Phytopathol. 2011, 49, 219–248. [Google Scholar] [CrossRef]
- Tzanetakis, I.E.; Martin, R.R.; Wintermantel, W.M. Epidemiology of criniviruses: An emerging problem in world agriculture. Front. Microbiol. 2013, 4, 119. [Google Scholar] [CrossRef]
- Fiallo-Olivé, E.; Navas-Castillo, J. Tomato chlorosis virus, an emergent plant virus still expanding its geographical and host ranges. Mol. Plant. Pathol. 2019, 20, 1307–1320. [Google Scholar] [CrossRef]
- Wisler, G.C.; Li, R.H.; Liu, H.Y.; Lowry, D.S.; Duffus, J.E. Tomato chlorosis virus: A new whitefly-transmitted, phloem limited, bipartite closterovirus of tomato. Phytopathology 1998, 88, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Wisler, G.C.; Duffus, J.E.; Liu, H.Y.; Li, R.H. Ecology and epidemiology of whitefly-transmitted closteroviruses. Plant. Dis. 1998, 82, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Navas-Castillo, J.; Camero, R.; Bueno, M.; Moriones, E. Severe yellowing outbreaks in tomato in Spain associated with infections of Tomato chlorosis virus. Plant. Dis. 2000, 84, 835–837. [Google Scholar] [CrossRef] [PubMed]
- Wintermantel, W.M.; Wisler, G.C. Vector specificity, host range, and genetic diversity of Tomato chlorosis virus. Plant. Dis. 2006, 90, 814–819. [Google Scholar] [CrossRef]
- Gilbertson, R.L.; Rojas, M.; Natwick, E. Development of Integrated Pest Management (IPM) strategies for whitefly (Bemisia tabaci)-transmissible geminiviruses. In The Whitefly, Bemisia tabaci (Homoptera: Aleyrodidae) Interaction with Geminivirus-Infected Host Plants; Thompson, W.M.O., Ed.; Springer: Dordrecht, The Netherlands, 2011; pp. 323–356. [Google Scholar] [CrossRef]
- Prado-Maluta, N.K.; Garzo, E.; Moreno, A.; Navas-Castillo, J.; Fiallo-Olivé, E.; Lopes, J.R.S.; Fereres, A. Stylet penetration activities of the whitefly Bemisia tabaci associated with inoculation of the crinivirus Tomato chlorosis virus. J. Gen. Virol. 2017, 6, 1515–1520. [Google Scholar] [CrossRef]
- McLean, D.L.; Kinsey, M.G. A technique for electronically recording aphid feeding and salivation. Nature 1964, 202, 1358–1359. [Google Scholar] [CrossRef]
- Tjallingii, W.F. Electrical nature of recorded signals during stylet penetration by aphids. Entomol. Exp. Appl. 1985, 38, 177–186. [Google Scholar] [CrossRef]
- Mustafa, T.; Alvarez, J.M.; Munyaneza, J.E. Effect of cyantraniliprole on probing behavior of the potato psyllid (Hemiptera: Triozidae) as measured by the electrical penetration graph technique. J. Econ. Entomol. 2015, 108, 2529–2535. [Google Scholar] [CrossRef]
- Carmo-Sousa, M.; Garcia, R.B.; Wulff, N.A.; Fereres, A.; Miranda, M.P. Drench application of systemic insecticides disrupts probing behavior of Diaphorina citri (Hemiptera: Liviidae) and inoculation of Candidatus Liberibacter asiaticus. Insects 2020, 11, 314. [Google Scholar] [CrossRef]
- Dovas, C.I.; Katis, N.I.; Avgelis, A.V. Multiplex detection of criniviruses associated with epidemics of a yellowing disease of tomato in Greece. Plant. Dis. 2002, 86, 1345–1349. [Google Scholar] [CrossRef]
- Fortes, I.M.; Navas-Castillo, J. Potato, an experimental and natural host of the crinivirus Tomato chlorosis virus. Eur. J. Plant. Pathol. 2012, 134, 81–86. [Google Scholar] [CrossRef]
- Jassen, J.A.M.; Tjallingii, W.F.; Lenteren, J.C. Electrical recording and ultrastructure of stylet penetration by the greenhouse whitefly. Entomol. Exp. Appl. 1989, 52, 69–81. [Google Scholar] [CrossRef]
- Johnson, D.D.; Walker, G.P. Intracellular punctures by the adult whitefly Bemisia argentifolii on DC and AC electronic feeding monitors. Entomol. Exp. Appl. 1999, 92, 257–270. [Google Scholar] [CrossRef]
- Jiang, Y.X.; Lei, H.; Collar, J.L.; Martin, B.; Muniz, M.; Fereres, A. Probing and feeding behavior of two distinct biotypes of Bemisia tabaci (Homoptera: Aleyrodidae) on tomato plants. J. Econ. Entomol. 1999, 92, 357–366. [Google Scholar] [CrossRef]
- Sarria, E.; Cid, M.; Garzo, E.; Fereres, A. Excel Workbook for automatic parameter calculation of EPG data. Comput. Electron. Agric. 2009, 67, 35–42. [Google Scholar] [CrossRef]
- Backus, E.A.; Cline, A.R.; Ellerseick, M.R.; Serrano, M.S. Lygus hesperus (Hemiptera: Miridae) feeding on cotton: New methods and parameters for analysis of non-sequential electrical penetration graph data. Ann. Entomol. Soc. Am. 2007, 100, 296–310. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, Version 22.0; IBM Corp: Armonk, NY, USA, 2013. [Google Scholar]
- Moreno-Delafuente, A.; Garzo, E.; Moreno, A.; Fereres, A. A plant virus manipulates the behaviour of its whitefly vector to enhance its transmission efficiency and spread. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Carmo-Sousa, M.; Moreno, A.; Garzo, E.; Fereres, A. A non-persistently transmitted-virus induces a pull–push strategy in its aphid vector to optimize transmission and spread. Virus Res. 2014, 186, 38–46. [Google Scholar] [CrossRef]
- Lei, W.; Li, P.; Han, Y.; Gong, S.; Yang, L.; Hou, M. EPG recordings reveal differential feeding behaviors in Sogatella furcifera in response to plant virus infection and transmission success. Sci. Rep. 2016, 6, 30240. [Google Scholar] [CrossRef]
- Butler, C.D.; Byrne, F.J.; Keremane, M.L.; Lee, R.F.; Trumble, J.T. Effects of insecticides on behavior of adult Bactericera cockerelli (Hemiptera: Triozidae) and transmission of Candidatus Liberibacter psyllaurous. J. Econ. Entomol. 2011, 104, 586–594. [Google Scholar] [CrossRef]
- Castle, S.; Palumbo, J.; Merten, P.; Cowden, C.; Prabhaker, N. Effects of foliar and systemic insecticides on whitefly transmission and incidence of Cucurbit yellow stunting disorder virus. Pest. Manag. Sci. 2017, 73, 1462–1471. [Google Scholar] [CrossRef] [PubMed]
- Gouvêa, M.M.; Freitas, D.M.S.; Rezende, J.A.M.; Watanabe, L.F.M.; Lourenção, A.L. Bioassay of insecticides on mortality of Bemisia tabaci biotype B and transmission of Tomato severe rugose virus (ToSRV) on tomatoes. Phytoparasitica 2017, 45, 1–7. [Google Scholar] [CrossRef]
- Garzo, E.; Moreno, A.; Plaza, M.; Fereres, A. Feeding behavior and virus-transmission ability of insect vectors exposed to systemic insecticides. Plants 2020, 9, 895. [Google Scholar] [CrossRef] [PubMed]
- Buttler, C.D.; Walker, G.P.; Trumble, J.T. Feeding disruption of potato psyllid, Bactericera cockerelli, by imidacloprid as measured by electrical penetration graphs. Entomol. Exp. Appl. 2012, 142, 247–257. [Google Scholar] [CrossRef]
- Roditakis, E.; Stavrakaki, M.; Grispou, M.; Achumastou, A.; Waetermeulen, X.V.; Nauen, R.; Tsagkarakou, A. Flupyradifurone effectively manages whitefly Bemisia tabaci MED (Hemiptera: Aleyrodidae) and Tomato yellow leaf curl virus (TYLCV) in tomato. Pest. Manag. Sci. 2017, 8, 1574–1584. [Google Scholar] [CrossRef]
- Smith, H.A.; Giurcanu, M.C. New insecticides for management of Tomato yellow leaf curl, a virus vectored by the silverleaf whitefly, Bemisia tabaci. J. Insect. Sci. 2014, 14, 183. [Google Scholar] [CrossRef]
- Civolani, S.; Cassanelli, S.; Chicca, M.; Rison, J.L.; Bassi, A.; Alvarez, J.M.; Annan, I.B.; Parrella, G.; Giorgini, M.; Fano, E.A. An EPG study of the probing behavior of adult Bemisia tabaci biotype Q (Hemiptera: Aleyrodidae) following exposure to cyantraniliprole. J. Econ. Entomol. 2014, 107, 910–919. [Google Scholar] [CrossRef]
- Maluta, N.K.P.; Garzo, E.; Moreno, A.; Lopes, J.R.S.; Fereres, A. Tomato yellow leaf curl virus benefits population growth of the Q biotype of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae). Neotrop. Entomol. 2014, 43, 385–392. [Google Scholar] [CrossRef]
- Mauck, K.E.; Bosque-Pérez, N.A.; Eigenbrode, S.D.; De Moraes, C.M.; Mescher, M.C. Transmission mechanisms shape pathogen effects on host-vector interactions: Evidence from plant viruses. Funct. Ecol. 2012, 26, 1162–1175. [Google Scholar] [CrossRef]
- Mauck, K.E.; Quentin, C.; Shapiro, L.R. Evolutionary determinants of host and vector manipulation by plant viruses. Adv. Virus Res. 2018, 101, 189–250. [Google Scholar] [CrossRef]
- Chesnais, Q.; Couty, A.; Uzest, M.; Brault, V.; Ameline, A. Plant infection by two different viruses induce contrasting changes of vectors fitness and behavior. Insect Sci. 2017, 26, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Maluta, N.K.P.; Fereres, A.; Lopes, J.R.S. Settling preferences of the whitefly vector Bemisia tabaci on infected plants varies with virus family and transmission mode. Entomol. Exp. Appl. 2017, 165, 138–147. [Google Scholar] [CrossRef]
- Bedford, I.D.; Briddon, R.W.; Brown, J.K.; Rosell, R.C.; Markham, P.G. Geminivirus transmission and biological characterization of Bemisia tabaci (Gennadius) biotypes from different geographic regions. Ann. Appl. Biol. 1994, 125, 311–325. [Google Scholar] [CrossRef]
- De Barro, P.J.; Trueman, J.W.H.; Frohlich, D.R. Bemisia argentifolii is a race of B. tabaci (Hemiptera: Aleyrodidae): The molecular genetic differentiation of B. tabaci populations around the world. Bull. Entomol. Res. 2005, 95, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Hasegawa, D.K.; Kaur, N.; Kliot, A.; Pinheiro, P.V.; Luan, J.; Stensmyr, M.C.; Zheng, Y.; Liu, W.; Sun, H.; et al. The draft genome of whitefly Bemisia tabaci MEAM1, a global crop pest, provides novel insights into virus transmission, host adaptation, and insecticide resistance. BMC Biol. 2016, 14, 110. [Google Scholar] [CrossRef] [PubMed]
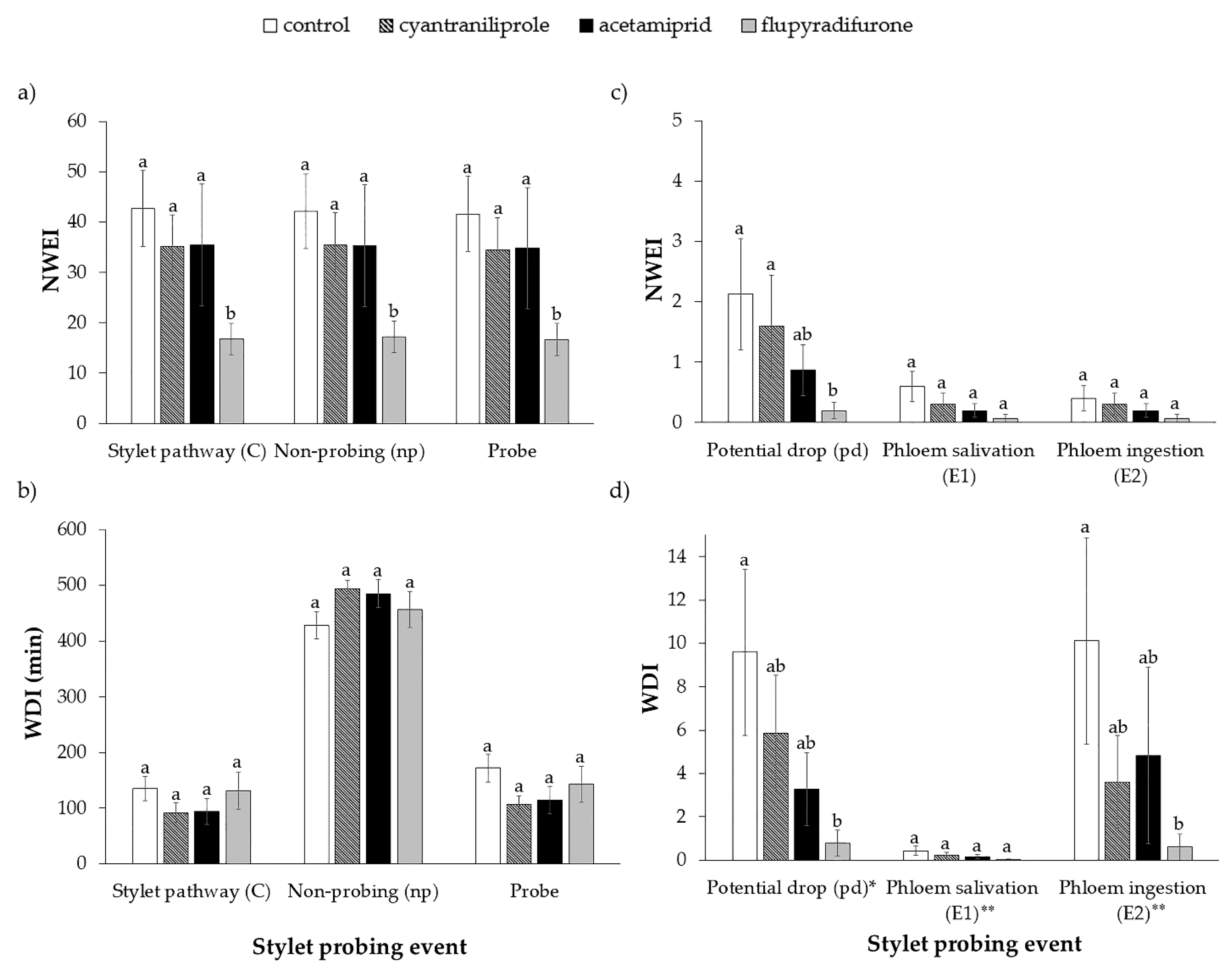
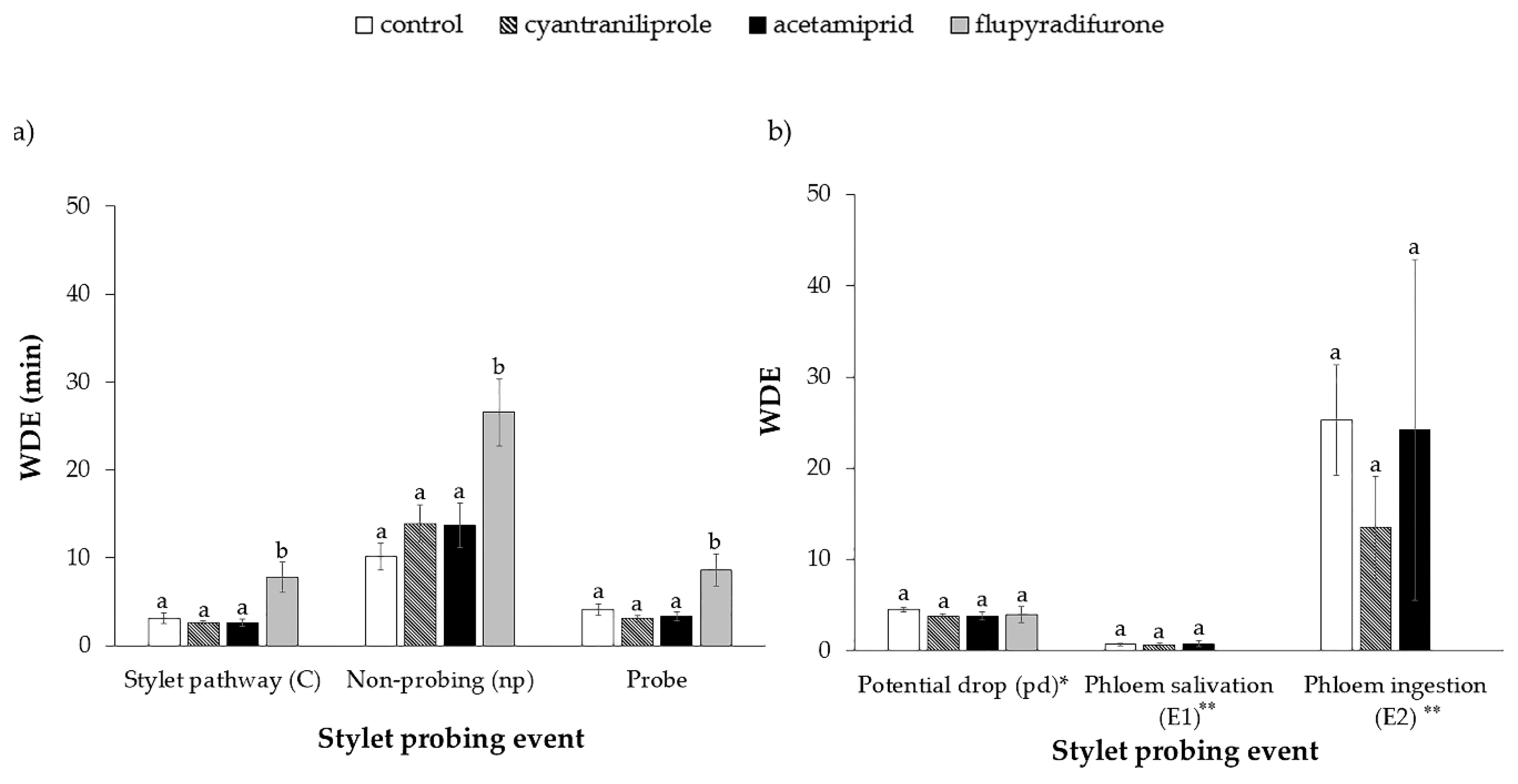
| Sequential EPG Variables a | Control n = 15 | Cyantraniliprole n = 15 | Acetamiprid n = 15 | Flupyradifurone n = 15 | H | df | p |
|---|---|---|---|---|---|---|---|
| Time to 1st probe from start of EPG | 164.19 ± 45.82 a | 199.45 ± 35.64 a | 200.33 ± 58.93 a | 125.64 ± 38.96 a | 2.74 | 3 | 0.43 |
| N° of probes to the 1st E1 | 7.0 ± 3.24 a | 1.80 ± 1.08 a | 10.53 ± 6.40 a | 0.27 ± 0.27 a | 3.77 | 3 | 0.29 |
| N° of probes after first E | 7.20 ± 3.44 a | 2.20 ± 2.13 a | 1.27 ± 1.27 a | 0.20 ± 0.20 a | 3.81 | 3 | 0.28 |
| Time from start of EPG to 1st E | 487.91 ± 48.58 a | 538.24 ± 37.43 a | 574.91 ± 17.55 a | 564.83 ± 35.17 a | 3.24 | 3 | 0.36 |
| Time from 1st probe to 1st E | 323.72 ± 48.39 a | 338.78 ± 46.86 a | 374.58 ± 59.97 a | 439.18 ± 47.91 a | 3.96 | 3 | 0.26 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maluta, N.K.P.; Lopes, J.R.S.; Fiallo-Olivé, E.; Navas-Castillo, J.; Lourenção, A.L. Foliar Spraying of Tomato Plants with Systemic Insecticides: Effects on Feeding Behavior, Mortality and Oviposition of Bemisia tabaci (Hemiptera: Aleyrodidae) and Inoculation Efficiency of Tomato Chlorosis Virus. Insects 2020, 11, 559. https://doi.org/10.3390/insects11090559
Maluta NKP, Lopes JRS, Fiallo-Olivé E, Navas-Castillo J, Lourenção AL. Foliar Spraying of Tomato Plants with Systemic Insecticides: Effects on Feeding Behavior, Mortality and Oviposition of Bemisia tabaci (Hemiptera: Aleyrodidae) and Inoculation Efficiency of Tomato Chlorosis Virus. Insects. 2020; 11(9):559. https://doi.org/10.3390/insects11090559
Chicago/Turabian StyleMaluta, Nathalie Kristine Prado, João Roberto Spotti Lopes, Elvira Fiallo-Olivé, Jesús Navas-Castillo, and André Luiz Lourenção. 2020. "Foliar Spraying of Tomato Plants with Systemic Insecticides: Effects on Feeding Behavior, Mortality and Oviposition of Bemisia tabaci (Hemiptera: Aleyrodidae) and Inoculation Efficiency of Tomato Chlorosis Virus" Insects 11, no. 9: 559. https://doi.org/10.3390/insects11090559
APA StyleMaluta, N. K. P., Lopes, J. R. S., Fiallo-Olivé, E., Navas-Castillo, J., & Lourenção, A. L. (2020). Foliar Spraying of Tomato Plants with Systemic Insecticides: Effects on Feeding Behavior, Mortality and Oviposition of Bemisia tabaci (Hemiptera: Aleyrodidae) and Inoculation Efficiency of Tomato Chlorosis Virus. Insects, 11(9), 559. https://doi.org/10.3390/insects11090559









