Identification of Immune Regulatory Genes in Apis mellifera through Caffeine Treatment
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Bee Rearing
2.2. Deformed Wing Virus (DWV) Purification
2.3. DWV Infection and Caffeine Treatment
2.4. Total RNA Extraction
2.5. cDNA Synthesis
2.6. Real-Time Polymerase Chain Reaction (PCR) and Data Analysis
2.7. DWV Titer Calculation
2.8. Other Latent Infecting Viruses in Taiwan
2.9. Statistical Analysis
3. Results
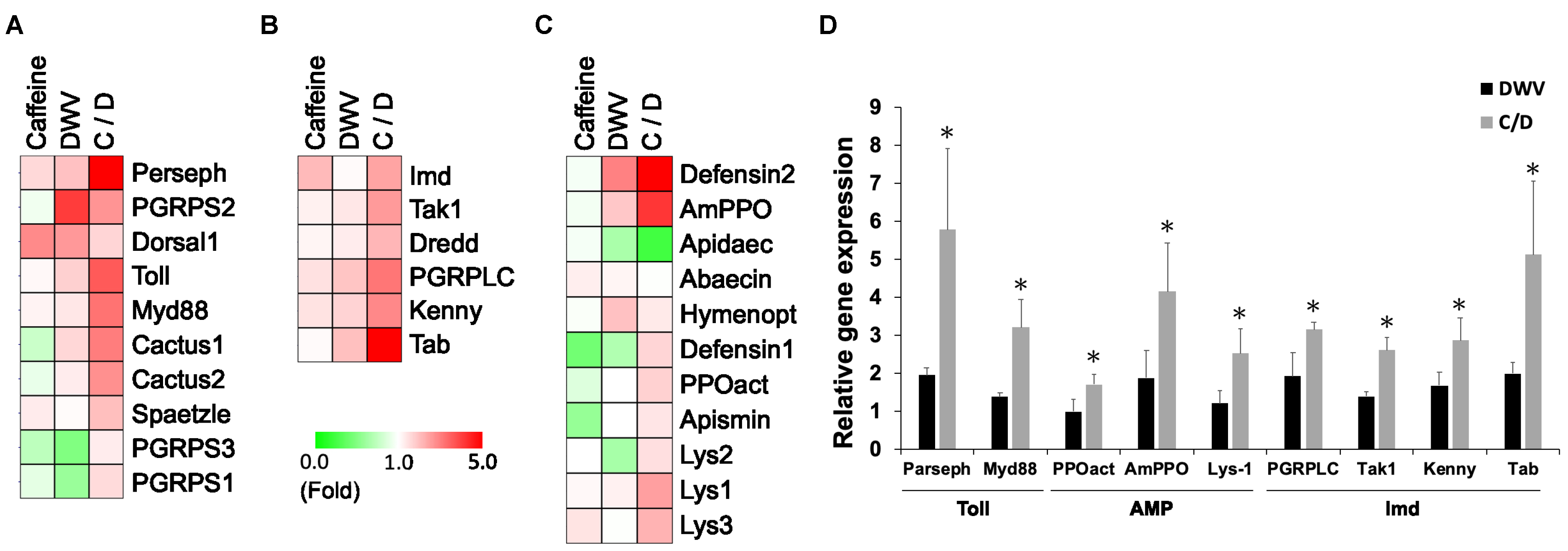
3.1. Effects of Caffeine on Immunity Gene Signaling Factors and Anti-Microbial Peptides
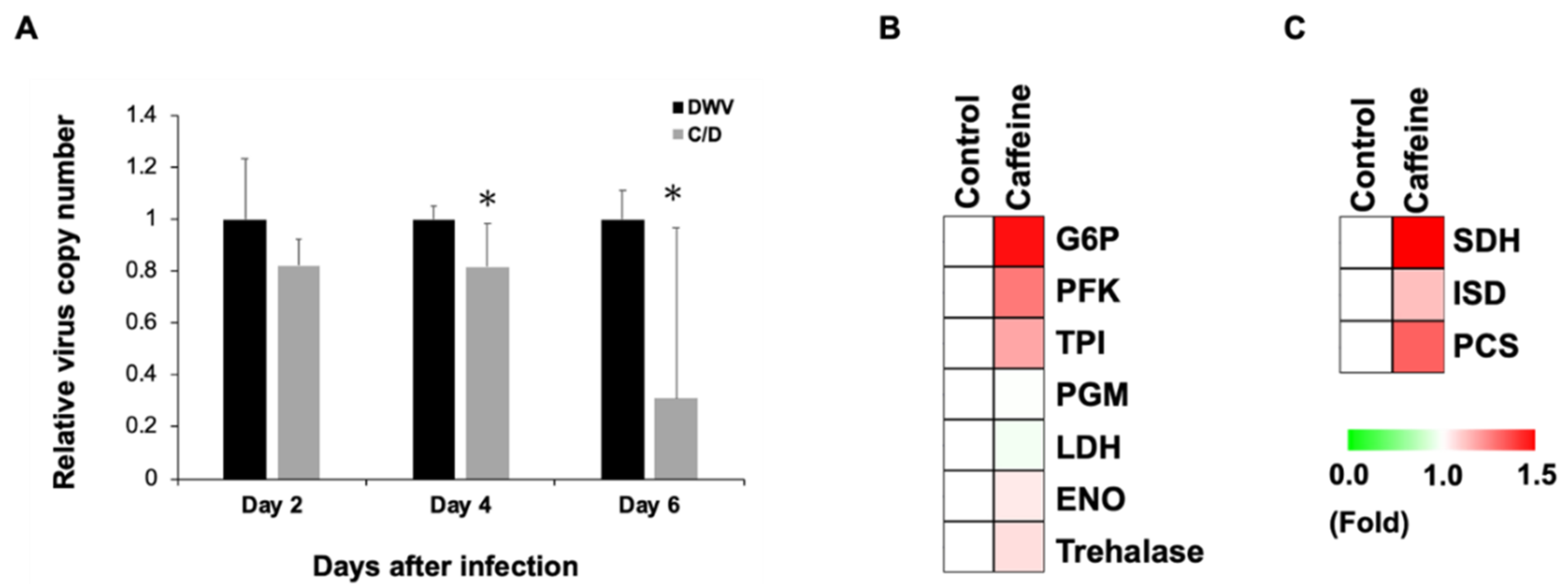
3.2. Caffeine Inhibits DWV Replication by Enhancing Carbohydrate Metabolism
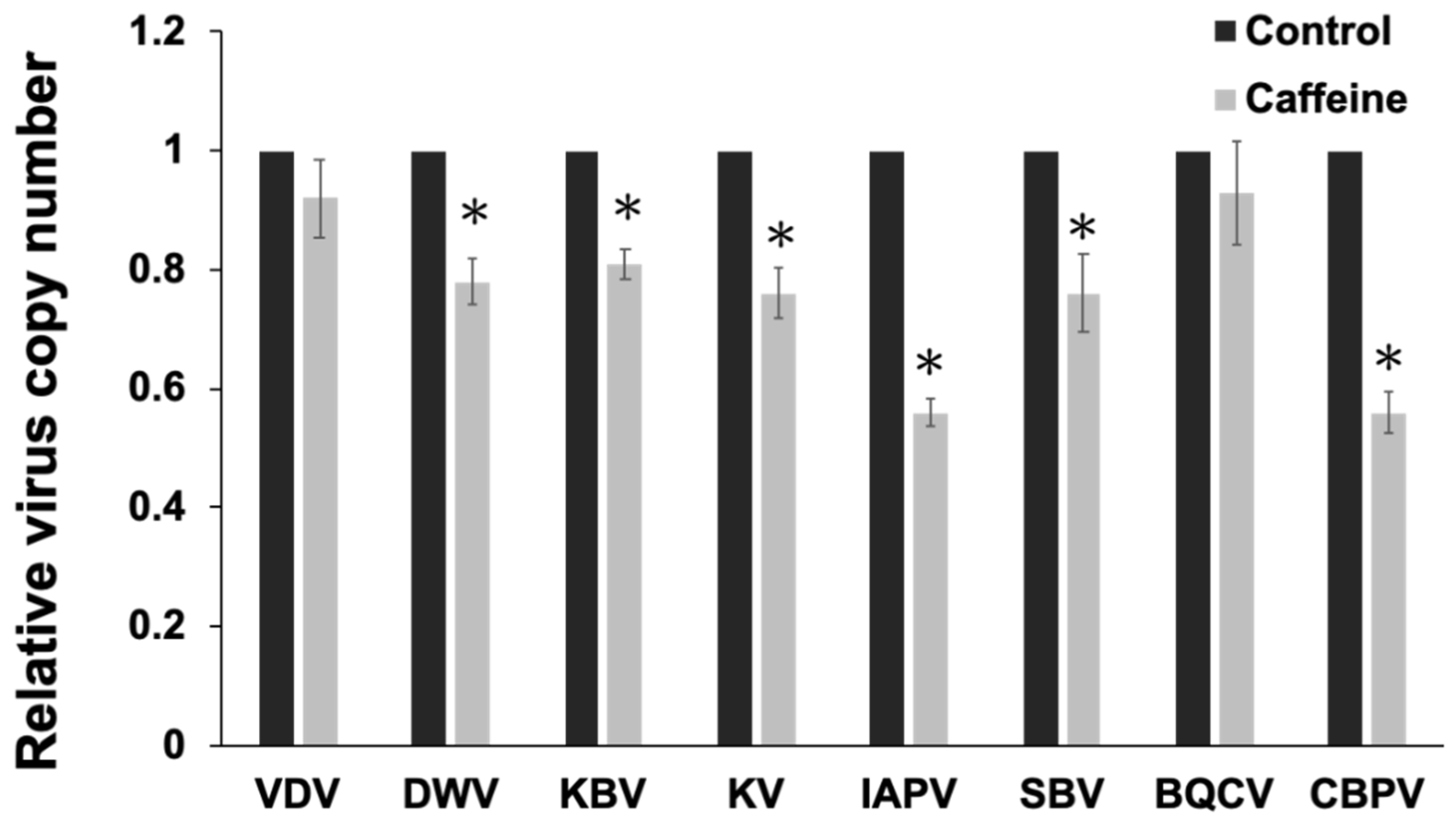
3.3. Caffeine also Inhibits the Infection of Other Prevalent Viruses in Taiwan
3.4. Caffeine Does not Affect the Expression of Immunity Related Genes in 16-Days Old Honeybee
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moritz, R.; Härtel, S.; Neumann, P. Global invasions of the western honeybee (Apis mellifera) and the consequences for biodiversity. Ecoscience 2005, 12, 289–301. [Google Scholar] [CrossRef]
- Kremen, C.; Williams, N.M.; Thorp, R.W. Crop pollination from native bees at risk from agricultural intensification. Proc. Natl. Acad. Sci. USA 2002, 99, 16812–16816. [Google Scholar] [CrossRef] [PubMed]
- Francis, R.M.; Nielsen, S.L.; Kryger, P. Varroa-virus interaction in collapsing honey bee colonies. PLoS ONE 2013, 8, e57540. [Google Scholar] [CrossRef] [PubMed]
- Vanengelsdorp, D.; Traynor, K.; Andree, M.; Lichtenberg, E.M.; Chen, Y.; Saegerman, C.; Cox-Foster, D.L. Colony Collapse Disorder (CCD) and bee age impact honey bee pathophysiology. PLoS ONE 2017, 12, e0179535. [Google Scholar] [CrossRef] [PubMed]
- Nazzi, F.; Pennacchio, F. Disentangling multiple interactions in the hive ecosystem. Trends Parasitol. 2014, 30, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Cornman, R.S.; Tarpy, D.R.; Chen, Y.; Jeffreys, L.; Lopez, D.; Pettis, J.S.; Vanengelsdorp, D.; Evans, J.D. Pathogen Webs in Collapsing Honey Bee Colonies. PLoS ONE 2012, 7, e43562. [Google Scholar] [CrossRef]
- Cox-Foster, D.L.; Conlan, S.; Holmes, E.C.; Palacios, G.F.; Evans, J.D.; Moran, N.A.; Quan, P.-L.; Briese, T.; Hornig, M.; Geiser, D.M.; et al. A Metagenomic Survey of Microbes in Honey Bee Colony Collapse Disorder. Science 2007, 318, 283–287. [Google Scholar] [CrossRef]
- Ellis, J.D.; Evans, J.D.; Pettis, J.S. Colony losses, managed colony population decline, and Colony Collapse Disorder in the United States. J. Apic. Res. 2010, 49, 134–136. [Google Scholar] [CrossRef]
- Dolezal, A.G.; Hendrix, S.D.; Scavo, N.A.; Carrillo-Tripp, J.; Harris, M.A.; Wheelock, M.J.; O’Neal, M.E.; Toth, A.L. Honey Bee Viruses in Wild Bees: Viral Prevalence, Loads, and Experimental Inoculation. PLoS ONE 2016, 11, e0166190. [Google Scholar] [CrossRef]
- Grozinger, C.M.; Flenniken, M.L. Bee Viruses: Ecology, Pathogenicity, and Impacts. Annu. Rev. Èntomol. 2019, 64, 205–226. [Google Scholar] [CrossRef]
- McMenamin, A.J.; Genersch, E. Honey bee colony losses and associated viruses. Current Opinion in Insect Science 2015, 8, 121–129. [Google Scholar] [CrossRef]
- Wilfert, L.; Long, G.; Leggett, H.C.; Schmid-Hempel, P.; Butlin, R.K.; Martin, S.J.M.; Boots, M. Deformed wing virus is a recent global epidemic in honeybees driven by Varroa mites. Science 2016, 351, 594–597. [Google Scholar] [CrossRef] [PubMed]
- Nazzi, F.; Le Conte, Y. Ecology ofVarroa destructor, the Major Ectoparasite of the Western Honey Bee, Apis mellifera. Annu. Rev. Èntomol. 2016, 61, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Berényi, O.; Bakonyi, T.; Derakhshifar, I.; Köglberger, H.; Topolska, G.; Ritter, W.; Pechhacker, H.; Nowotny, N. Phylogenetic Analysis of Deformed Wing Virus Genotypes from Diverse Geographic Origins Indicates Recent Global Distribution of the Virus. Appl. Environ. Microbiol. 2007, 73, 3605–3611. [Google Scholar] [CrossRef]
- Yang, X.; Cox-Foster, D.L. Impact of an ectoparasite on the immunity and pathology of an invertebrate: Evidence for host immunosuppression and viral amplification. Proc. Natl. Acad. Sci. USA 2005, 102, 7470–7475. [Google Scholar] [CrossRef]
- Navajas, M.; Migeon, A.; Alaux, C.; Martin-Magniette, M.; Robinson, G.E.; Evans, J.D.; Cros-Arteil, S.; Crauser, D.; Le Conte, Y. Differential gene expression of the honey bee Apis mellifera associated with Varroa destructor infection. BMC Genom. 2008, 9, 301. [Google Scholar] [CrossRef]
- Li, Q.; Verma, I.M. NF-κB regulation in the immune system. Nat. Rev. Immunol. 2002, 2, 725–734. [Google Scholar] [CrossRef]
- Gordon, M.D.; Dionne, M.S.; Schneider, D.S.; Nusse, R. WntD is a feedback inhibitor of Dorsal/NF-κB in Drosophila development and immunity. Nature 2005, 437, 746–749. [Google Scholar] [CrossRef]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti Toll Pathway Controls Dengue Virus Infection. PLoS Pathog. 2008, 4, e1000098. [Google Scholar] [CrossRef]
- Vanwalscappel, B.; Tada, T.; Landau, N.R. Toll-like receptor agonist R848 blocks Zika virus replication by inducing the antiviral protein viperin. Virology 2018, 522, 199–208. [Google Scholar] [CrossRef]
- Evans, J.D.; Aronstein, K.; Chen, Y.P.; Hetru, C.; Imler, J.; Jiang, H.; Kanost, M.; Thompson, G.J.; Zou, Z.; Hultmark, D. Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol. Biol. 2006, 15, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Brutscher, L.M.; Daughenbaugh, K.F.; Flenniken, M.L. Antiviral defense mechanisms in honey bees. Curr. Opin. Insect Sci. 2015, 10, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Nazzi, F.; Pennacchio, F. Honey Bee Antiviral Immune Barriers as Affected by Multiple Stress Factors: A Novel Paradigm to Interpret Colony Health Decline and Collapse. Viruses 2018, 10, 159. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Wightman, E.L. Herbal Extracts and Phytochemicals: Plant Secondary Metabolites and the Enhancement of Human Brain Function. Adv. Nutr. 2011, 2, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Kretschmar, J.A.; Baumann, T.W. Caffeine in Citrus flowers. Phytochemistry 1999, 52, 19–23. [Google Scholar] [CrossRef]
- Couvillon, M.J.; Al Toufailia, H.; Butterfield, T.M.; Schrell, F.; Ratnieks, F.L.W.; Schurch, R. Caffeinated forage tricks honeybees into increasing foraging and recruitment behaviors. Curr. Biol. 2015, 25, 2815–2818. [Google Scholar] [CrossRef]
- Mustard, J.; Dews, L.; Brugato, A.; Dey, K.; Wright, G.A. Consumption of an acute dose of caffeine reduces acquisition but not memory in the honey bee. Behav. Brain Res. 2012, 232, 217–224. [Google Scholar] [CrossRef]
- Si, A.; Zhang, S.-W.; Maleszka, R. Effects of caffeine on olfactory and visual learning in the honey bee (Apis mellifera). Pharmacol. Biochem. Behav. 2005, 82, 664–672. [Google Scholar] [CrossRef]
- Wright, G.A.; Baker, D.D.; Palmer, M.J.; Stabler, D.; Mustard, J.A.; Power, E.F.; Borland, A.M.; Stevenson, P.C. Caffeine in floral nectar enhances a pollinator’s memory of reward. Science 2013, 339, 1202–1204. [Google Scholar] [CrossRef]
- Ishay, J.S.; Paniry, V.A. Effects of Caffeine and Various Xanthines on Hornets and Bees. Psychopharmacology 1979, 65, 299–309. [Google Scholar] [CrossRef]
- Fernandes, F.L.; Picanço, M.C.; Fernandes, M.; Queiroz, R.B.; Xavier, V.; Martinez, H. The Effects of Nutrients and Secondary Compounds of Coffea arabica on the Behavior and Development of Coccus viridis. Environ. Èntomol. 2012, 41, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, S.; Sasaki, K.; Matsumura, K.; Lewis, Z.; Miyatake, T. Dopaminergic system as the mechanism underlying personality in a beetle. J. Insect Physiol. 2012, 58, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Nishi, Y.; Sasaki, K.; Miyatake, T. Biogenic amines, caffeine and tonic immobility in Tribolium castaneum. J. Insect Physiol. 2010, 56, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Bernklau, E.; Bjostad, L.; Hogeboom, A.; Carlisle, A.; Arathi, H.S. Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance. Insects 2019, 10, 14. [Google Scholar] [CrossRef]
- Hu, Y.-T.; Wu, T.-C.; Yang, E.-C.; Wu, P.-C.; Lin, P.-T.; Wu, Y.-L. Regulation of genes related to immune signaling and detoxification in Apis mellifera by an inhibitor of histone deacetylation. Sci. Rep. 2017, 7, 41255. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Gregorc, A.; Evans, J.D.; Scharf, M.; Ellis, J.D. Gene expression in honey bee (Apis mellifera) larvae exposed to pesticides and Varroa mites (Varroa destructor). J. Insect Physiol. 2012, 58, 1042–1049. [Google Scholar] [CrossRef]
- Biergans, S.D.; Galizia, C.G.; Reinhard, J.; Claudianos, C. Dnmts and Tet target memory-associated genes after appetitive olfactory training in honey bees. Sci. Rep. 2015, 5, 16223. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Chang, Y.; Tang, C.K.; Lin, Y.H.; Tsai, C.H.; Lu, Y.H.; Wu, Y.L. Snellenius manilae bracovirus suppresses the host immune system by regulating extracellular adenosine levels in Spodoptera litura. Sci. Rep. 2020, 10, 2096. [Google Scholar] [CrossRef]
- Mandal, A.; Poddar, M.K. Long-term caffeine consumption reverses tumor-induced suppression of the innate immune response in adult mice. Planta Med. 2008, 74, 1779–1784. [Google Scholar] [CrossRef]
- Barcelos, R.P.; Lima, F.D.; Carvalho, N.R.; Bresciani, G.; Royes, L.F. Caffeine effects on systemic metabolism, oxidative-inflammatory pathways, and exercise performance. Nutr. Res. 2020, 80, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Horrigan, L.A.; Kelly, J.P.; Connor, T.J. Caffeine suppresses TNF-alpha production via activation of the cyclic AMP/protein kinase A pathway. Int. Immunopharmacol. 2004, 4, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Torgersen, K.M.; Vang, T.; Abrahamsen, H.; Yaqub, S.; Taskén, K. Molecular mechanisms for protein kinase A-mediated modulation of immune function. Cell. Signal. 2002, 14, 1–9. [Google Scholar] [CrossRef]
- Gil Ferreira, Á.; Naylor, H.; Esteves, S.S.; Pais, I.; E Martins, N.; Teixeira, L. The Toll-Dorsal Pathway Is Required for Resistance to Viral Oral Infection in Drosophila. PLoS Pathog. 2014, 10, e1004507. [Google Scholar] [CrossRef]
- Avadhanula, V.; Weasner, B.P.; Hardy, G.G.; Kumar, J.P.; Hardy, R.W. A Novel System for the Launch of Alphavirus RNA Synthesis Reveals a Role for the Imd Pathway in Arthropod Antiviral Response. PLoS Pathog. 2009, 5, e1000582. [Google Scholar] [CrossRef]
- Galbraith, D.A.; Yang, X.Y.; Nino, E.L.; Yi, S.; Grozinger, C. Parallel Epigenomic and Transcriptomic Responses to Viral Infection in Honey Bees (Apis mellifera). PLoS Pathog. 2015, 11, e1004713. [Google Scholar] [CrossRef]
- Vannette, R.L.; Mohamed, A.; Johnson, B.R. Forager bees (Apis mellifera) highly express immune and detoxification genes in tissues associated with nectar processing. Sci. Rep. 2015, 5, 16224. [Google Scholar] [CrossRef]

| Gene Name | Forward Sequence | Reverse Sequence |
|---|---|---|
| Persephone | CCGGTGAACTTGGAAAAGAT | ATCGCAATTTGTCCCAAAAC |
| Toll | TAGAGTGGCGCATTGTCAAG | ATCGCAATTTGTCCCAAAAC |
| Spaetzle | TGCACAAATTGTTTTTCCTGA | GTCGTCCATGAAATCGATCC |
| PGRPS1 | TTTGAAAATTTCCTATGAAAGCA | TTTTTAATTGGTGGAGATGGAAA |
| PGRPS2 | TAATTCATCATTCGGCGACA | TGTTTGTCCCATCCTCTTCC |
| PGRPS3 | GAGGCTGGTACGACATTGGT | TTATAACCAGGTGCGTGTGC |
| PGRPLC | TCCGTCAGCCGTAGTTTTTC | CGTTTGTGCAAATCGAACAT |
| Myd88 | TCACATCCAGATCCAACTGC | CAGCTGACGTTTGAGATTTTTG |
| Abaecin | CAGCATTCGCATACGTACCA | GACCAGGAAACGTTGGAAAC |
| Defensin-1 | TGCGCTGCTAACTGTCTCAG | AATGGCACTTAACCGAAACG |
| Defensin-2 | GCAACTACCGCCTTTACGTC | GGGTAACGTGCGACGTTTTA |
| Cactus-1 | CACAAGATCTGGAGCAACGA | GCATTCTTGAAGGAGGAACG |
| Cactus-2 | TTAGCAGGACAAACGGCTCT | CAGAAAGTGGTTCCGGTGTT |
| Dorsal-1 | AAATGGTTCGCTCGTAGCAC | TCCATGATATGAGTGATGGAAA |
| PPOact | GTTTGGTCGACGGAAGAAAA | CCGTCGACTCGAAATCGTAT |
| AmPPO | AGATGGCATGCATTTGTTGA | CCACGCTCGTCTTCTTTAGG |
| Hymenopt | CTCTTCTGTGCCGTTGCATA | CGTCTCCTGTCATTCCATT |
| Apidaec | TAGTCGCGGTATTTGGGAAT | TTTCACGTGCTTCATATTCTTCA |
| Apisimin | TGAGCAAAATCGTTGCTGTC | AACGACATCCACGTTCGATT |
| Lys-1 | GAACACACGGTTGGTCACTG | ATTTCCAACCATCGTTTTCG |
| Lys-2 | CCAAATTAACAGCGCCAAGT | GCAATTCTTCACCCAACCAT |
| Lys-3 | ATCTGTTTGCGGACCATTTC | TCGATGAATGCGAAGAAAATC |
| Imd | TGTTAACGACCGATGCAAAA | CATCGCTCTTTTCGGATGTT |
| Tak-1 | ATGGATATGCTGCCAATGGT | TCGGATCGCATTCAACATAA |
| Dredd | GCGTCATAAAGAAAAAGGATCA | TTTCGGGTAATTGAGCAACG |
| Kenny | GCTGAACCAGAAAGCCACTT | TGCAAGTGATGATTGTTGGA |
| Tab | GCTATCATGCAGCTGTTCCA | ACACTGGGTCAGCCAATTTC |
| G6P | GGTTGGAGGACGTTATTC | CCATAAAGTGTGCTCCAC |
| PFK | ATCAGGGTATGGTAGATGGTGG | TTTTGCGACCAGCGTGAT |
| TPI | GAGGTTGTTGTTGGTGTACC | CCAAAAGCATAGCAGGAC |
| PGM | GGTACGTCATGGAGAAAGTG | TTGCGTCTCTGATTGCTT |
| LDH | GATGGAGGATAAATTAAAGGGAGAGA | TTGATTTTCGCGTTCCTCAAG |
| ENO | ACCTACAGGTGCTTCTAG | GTAGCATCAAGACCAAAC |
| Trehalase | ATGGAGCGGCACGAACA | GGGTCGAACGTGTCGTTGA |
| SDH | AGGAGGAGCGGGAAGATGTT | CAAGACCATCTCCTGTGCATGT |
| ISD | CATTGGCAGACATGCTCATG | TGCAATGCCAGGTCCTTT |
| PCS | GCAAATATTGGGAGTACGTCTATGAA | GAATCGACAGATACGGTGTTACCA |
| Gene Name | References | Caffeine | DWV | Caffeine/DWV | |||
| Toll Pathway | Up | Dn | Up | Dn | Up | Dn | |
| Perseph | Hu et al., 2017 | 60% | - | 94% | - | 478% * | - |
| Toll | Hu et al., 2017 | 12% | - | 73% | - | 258% | - |
| Spaetzle | Hu et al., 2017 | 32% | - | 8% | - | 101% | - |
| PGRPS1 | Hu et al., 2017 | - | 11% | - | 40% | 55% * | - |
| PGRPS2 | Hu et al., 2017 | - | 6% | 304% | - | 167% | - |
| PGRPS3 | Hu et al., 2017 | - | 26% | - | 51% | 28% | - |
| Myd88 | Hu et al., 2017 | 18% | - | 38% | - | 220% * | - |
| Cactus1 | Hu et al., 2017 | - | 21% | 64% | - | 203% | - |
| Cactus2 | Hu et al., 2017 | - | 9% | 31% | - | 176% | - |
| Dorsal1 | Hu et al., 2017 | 182% | - | 163% | - | 165% | - |
| Gene Name | References | Caffeine | DWV | Caffeine/DWV | |||
| Imd Pathway | Up | Dn | Up | Dn | Up | Dn | |
| PGRPLC | Hu et al., 2017 | 47% | - | 92% | - | 215% * | - |
| Imd | Hu et al., 2017 | 107% | - | 9% | - | 144% | - |
| Tak1 | Hu et al., 2017 | 23% | - | 39% | - | 160% * | - |
| Dredd | Hu et al., 2017 | 15% | - | 29% | - | 115% | - |
| Kenny | Hu et al., 2017 | 43% | - | 68% | - | 187% * | - |
| Tab | Hu et al., 2017 | 8% | - | 99% | - | 413% * | - |
| Gene Name | References | Caffeine | DWV | Caffeine/DWV | |||
| Amp (Anti-Microbial Peptide) | Up | Dn | Up | Dn | Up | Dn | |
| Abaecin | Hu et al., 2017 | 28% | - | 15% | - | - | 1% |
| Defensin-2 | Hu et al., 2017 | - | 4% | 198% | - | 2712% | - |
| Defensin-1 | Hu et al., 2017 | - | 55% | - | 31% | 67% | - |
| PPOact | Hu et al., 2017 | - | 14% | - | - | 71% * | - |
| Hymenopt | Hu et al., 2017 | - | 3% | 97% | - | 35% | - |
| AmPPO | Hu et al., 2017 | - | 5% | 87% | - | 315% * | - |
| Apidaec | Hu et al., 2017 | - | 4% | - | 33% | - | 75% |
| Apismin | Hu et al., 2017 | - | 42% | 1% | - | 40% | - |
| Lys-1 | Hu et al., 2017 | 11% | - | 21% | - | 151% * | - |
| Lys-2 | Hu et al., 2017 | - | 1% | - | 35% | 50% | - |
| Lys-3 | Hu et al., 2017 | 42% | - | - | 2% | 119% | - |
| Gene Name | References | Caffeine | |
| Glycolysis | Up | Dn | |
| G6P (Glucose-6-phosphate isomerase) | Froman et al., 1989 | 40% | - |
| PFK (6-phosphofructokinase) | Vora et al., 1985 | 26% | - |
| ENO (Enolase) | Marcaida et al., 2006 | 4% | - |
| PGM (Phosphoglycerate mutase) | Lu and Kreckner, 1994 | - | 1.4% |
| TPI (Triose-phosphate isomerase) | Daar et al., 1986 | 17% | - |
| Trehalase | Mori et al., 2009 | 6% | - |
| LDH (L-lactate dehydrogenase) | Chung et al., 1985 | - | 5% |
| Gene Name | References | Caffeine | |
| TCA Cycle | Up | Dn | |
| SDH (Succinate dehydrogenase) | Renkema et al., 2015 | 51% | - |
| ISD (isocitrate dehydrogenase) | Ceccarelli et al., 2002 | 12% | - |
| PCS (citrate synthase) | Goldenthal et al., 1998 | 31% | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Y.-H.; Wu, C.-P.; Tang, C.-K.; Lin, Y.-H.; Maaroufi, H.O.; Chuang, Y.-C.; Wu, Y.-L. Identification of Immune Regulatory Genes in Apis mellifera through Caffeine Treatment. Insects 2020, 11, 516. https://doi.org/10.3390/insects11080516
Lu Y-H, Wu C-P, Tang C-K, Lin Y-H, Maaroufi HO, Chuang Y-C, Wu Y-L. Identification of Immune Regulatory Genes in Apis mellifera through Caffeine Treatment. Insects. 2020; 11(8):516. https://doi.org/10.3390/insects11080516
Chicago/Turabian StyleLu, Yun-Heng, Carol-P Wu, Cheng-Kang Tang, Yu-Hsien Lin, Houda Ouns Maaroufi, Yi-Chi Chuang, and Yueh-Lung Wu. 2020. "Identification of Immune Regulatory Genes in Apis mellifera through Caffeine Treatment" Insects 11, no. 8: 516. https://doi.org/10.3390/insects11080516
APA StyleLu, Y.-H., Wu, C.-P., Tang, C.-K., Lin, Y.-H., Maaroufi, H. O., Chuang, Y.-C., & Wu, Y.-L. (2020). Identification of Immune Regulatory Genes in Apis mellifera through Caffeine Treatment. Insects, 11(8), 516. https://doi.org/10.3390/insects11080516





