Defense of Milkweed Bugs (Heteroptera: Lygaeinae) against Predatory Lacewing Larvae Depends on Structural Differences of Sequestered Cardenolides
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Culture
2.2. Predation Assay
2.3. Chemical Analysis
2.4. Statistical Analysis
3. Results
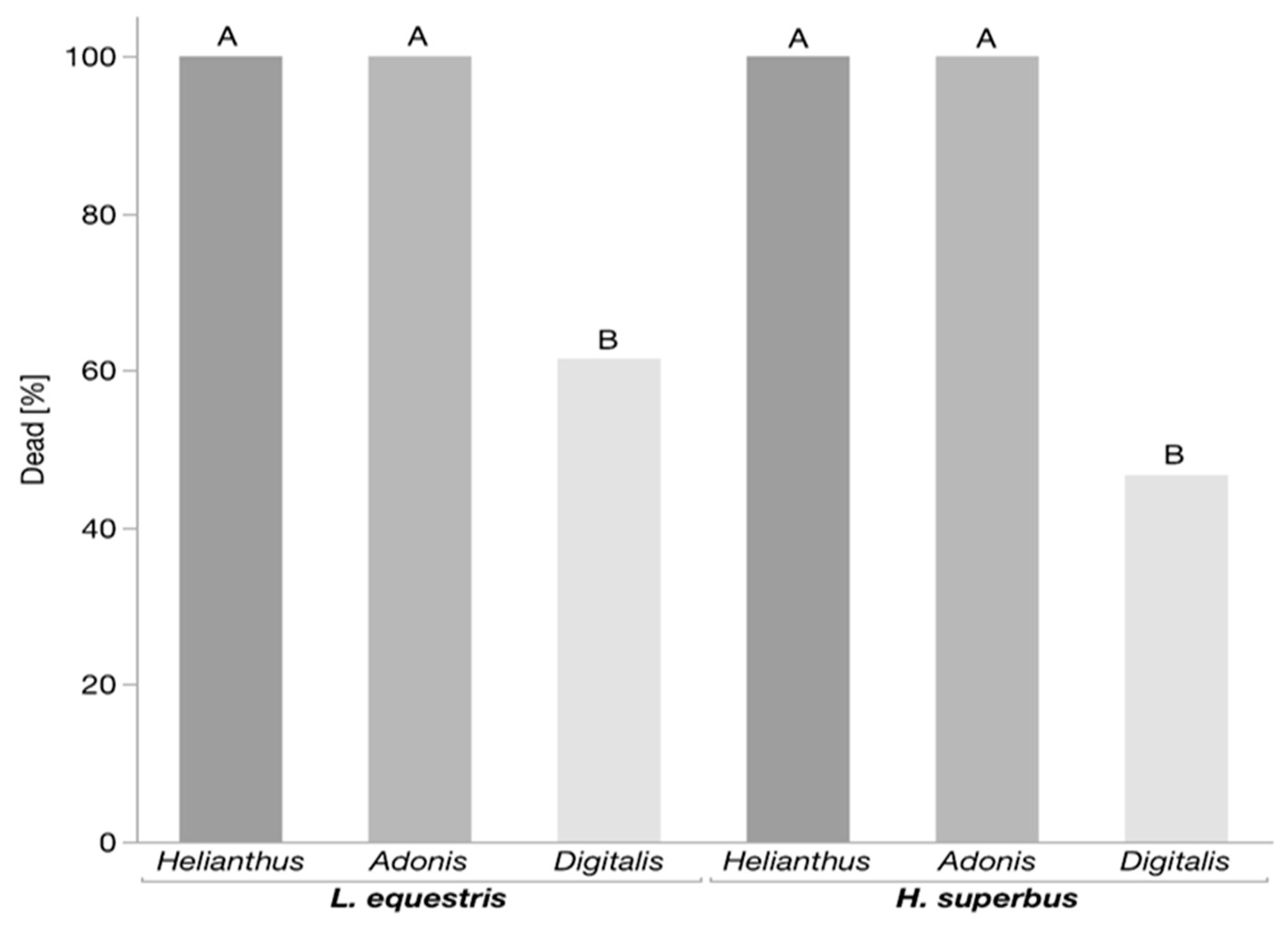
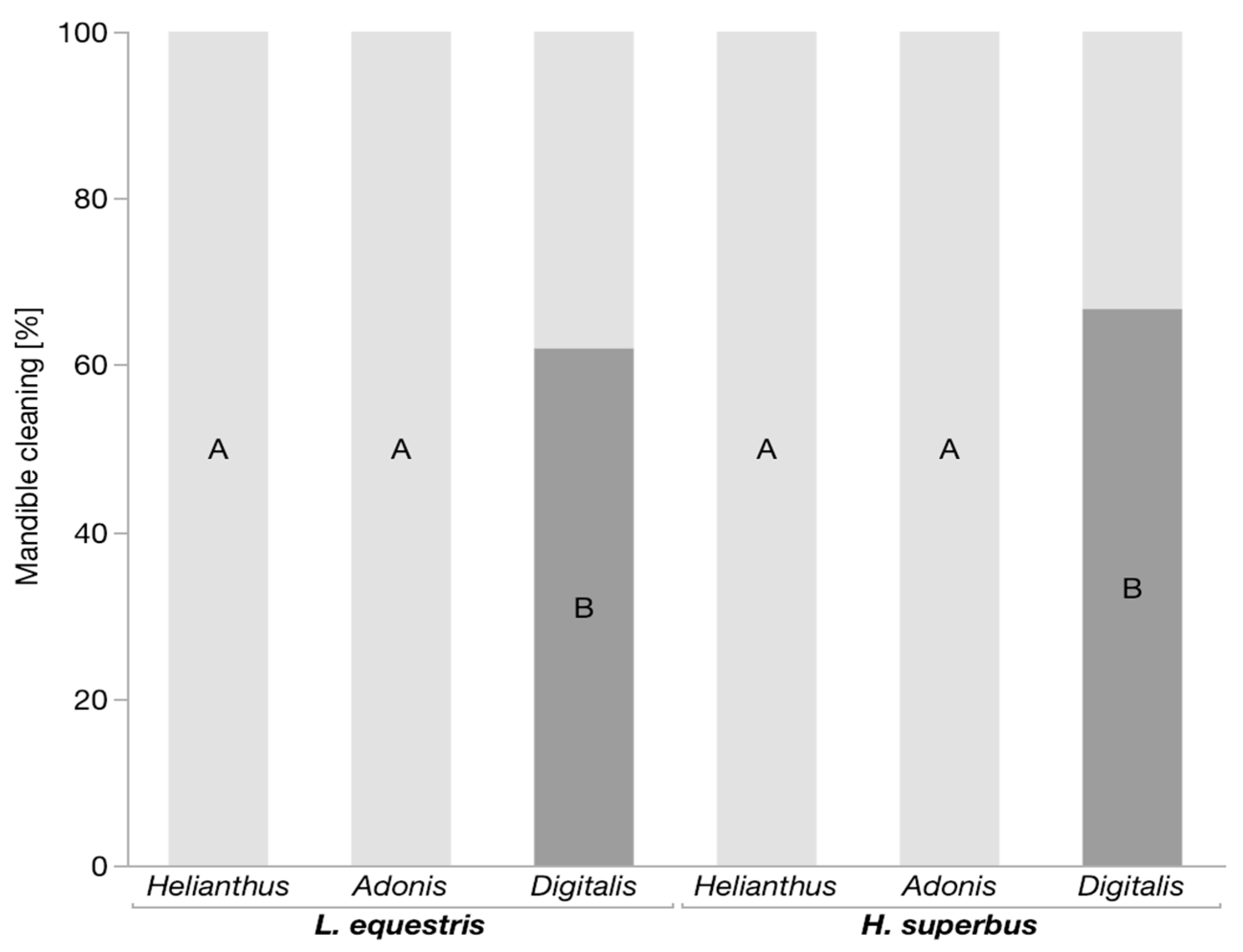
3.1. Predation Assay
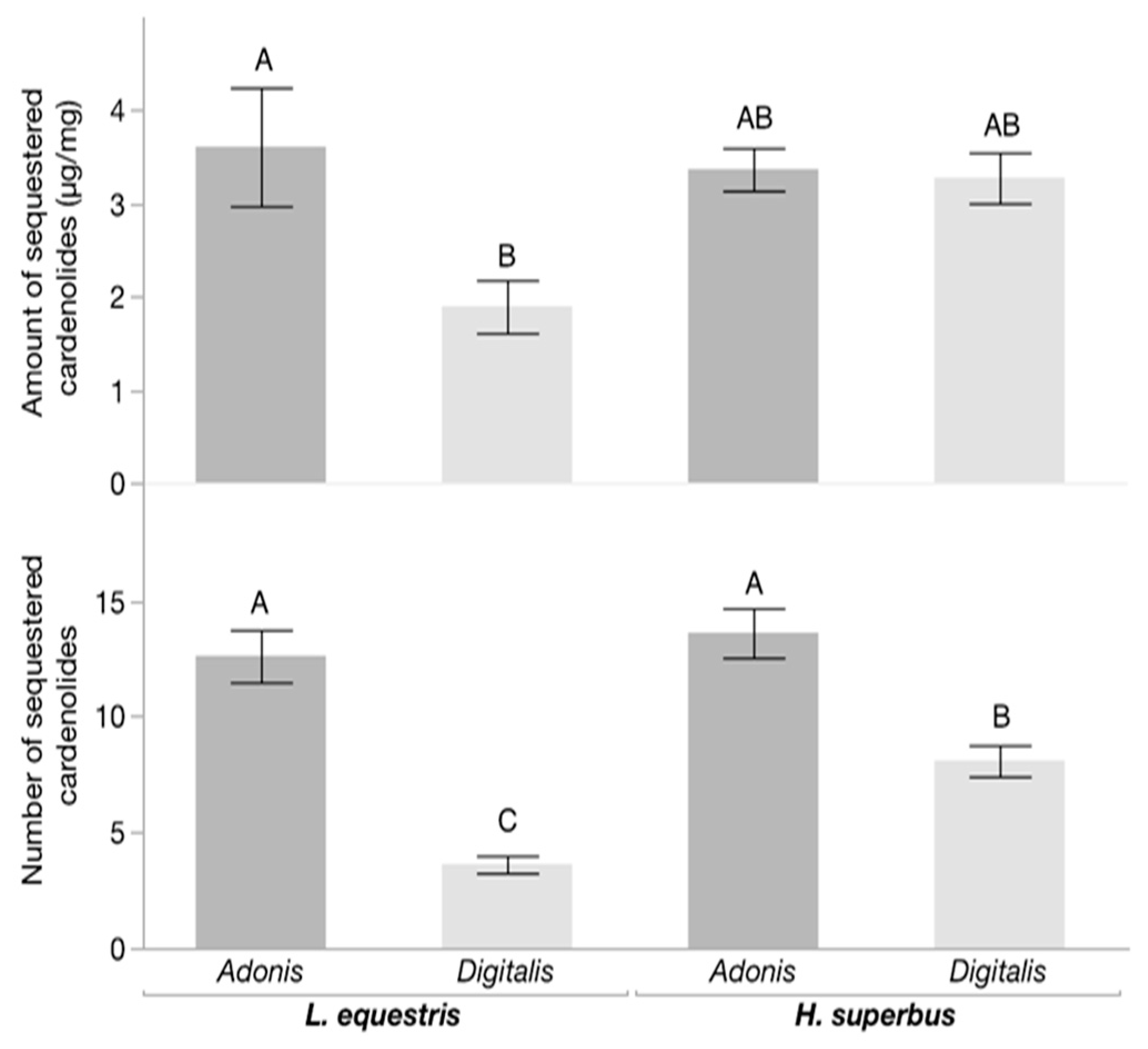
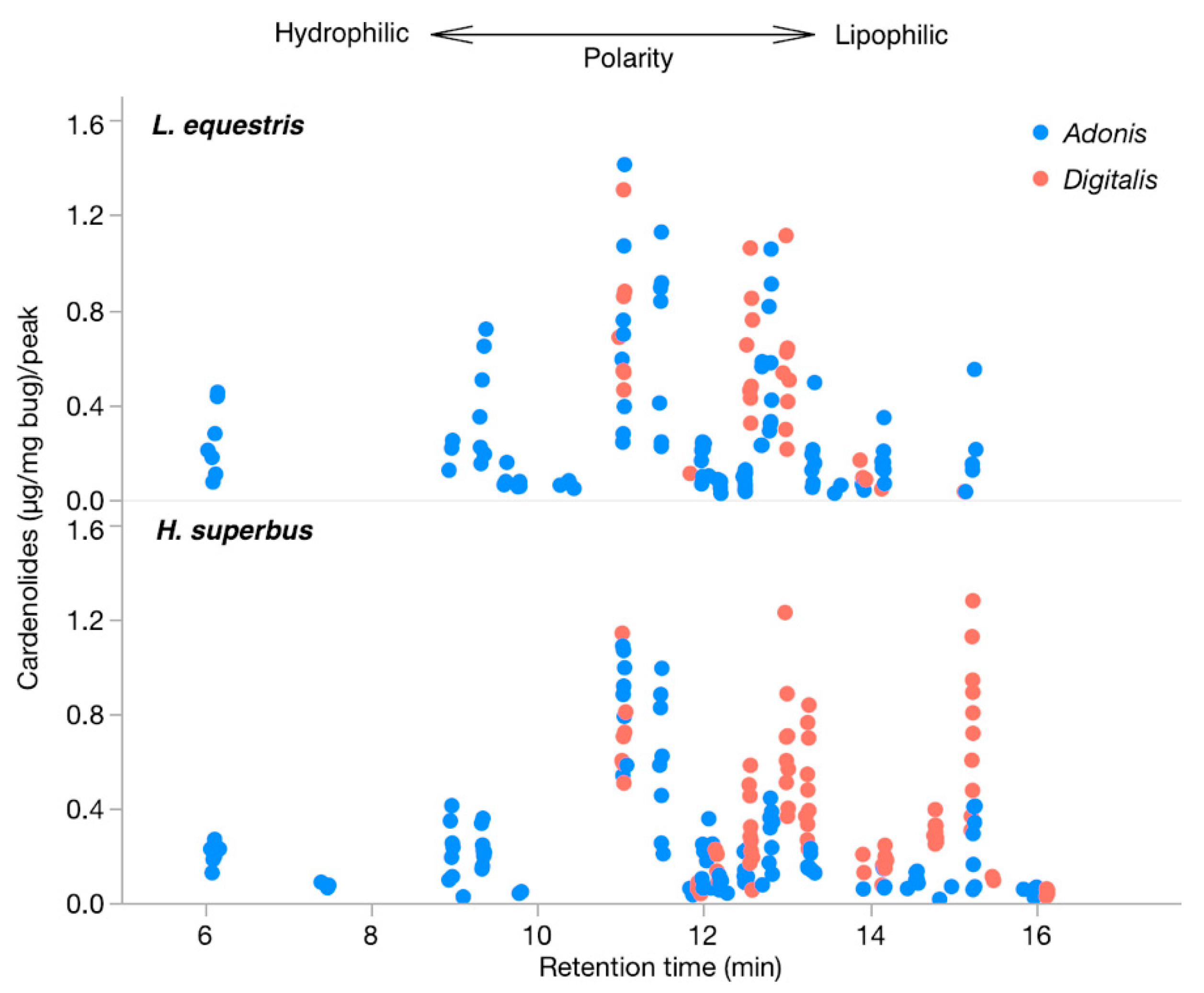
3.2. Chemical Analysis
3.3. Figures, Tables and Schemes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hairston, N.G.; Smith, F.E.; Slobodkin, L.B. Community structure, population control, and competition. Am. Nat. 1960, 94, 421–425. [Google Scholar] [CrossRef]
- Terborgh, J.; Lopez, L.; Nuñez, P.; Rao, M.; Shahabuddin, G.; Orihuela, G.; Riveros, M.; Ascanio, R.; Adler, G.H.; Lambert, T.D.; et al. Ecological meltdown in predator-free forest fragments. Science 2001, 294, 1923. [Google Scholar] [CrossRef]
- Pasteels, J.M.; Duffey, S.; Rowell-Rahier, M. Toxins in chrysomelid beetles Possible evolutionary sequence from de novo synthesis to derivation from food-plant chemicals. J. Chem. Ecol. 1990, 16, 211–222. [Google Scholar] [CrossRef]
- Opitz, S.E.W.; Müller, C. Plant chemistry and insect sequestration. Chemoecology 2009, 19, 117. [Google Scholar] [CrossRef]
- Schoonhoven, L.M.; van Loon, J.J.; Dicke, M. Insect-Plant Biology; Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Karban, R.; Baldwin, I.T. Induced Responses to Herbivory; University of Chicago Press: Chicago, IL, USA, 2007. [Google Scholar]
- Brower, L.P. Bird predation and foodplant specificity in closely related procryptic insects. Am. Nat. 1958, 92, 183–187. [Google Scholar] [CrossRef]
- Rothschild, M.; von Euw, J.; Reichstein, T. Cardiac glycosides in the oleander aphid, Aphis nerii. J. Insect Physiol. 1970, 16, 1141–1145. [Google Scholar] [CrossRef]
- Eisner, T. For love of nature: Exploration and discovery at biological field stations. BioScience 1982, 32, 321–326. [Google Scholar] [CrossRef][Green Version]
- Brown, K.S. Adult-obtained pyrrolizidine alkaloids defend ithomiine butterflies against a spider predator. Nature 1984, 309, 707–709. [Google Scholar] [CrossRef]
- Nishida, R.; Fukami, H.; Iriye, R.; Kumazawa, Z. Accumulation of highly toxic ericaceous diterpenoids by the geometrid moth, Arichanna gaschkevitchii. Agric. Biol. Chem. 1990, 54, 2347–2352. [Google Scholar] [CrossRef][Green Version]
- Nishida, R. Sequestration of defensive substances from plants by lepidoptera. Annu. Rev. Entomol. 2002, 47, 57–92. [Google Scholar] [CrossRef]
- Hartmann, T. Plant-derived secondary metabolites as defensive chemicals in herbivorous insects: A case study in chemical ecology. Planta 2004, 219, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Brower, L.P.; Ryerson, W.N.; Coppinger, L.L.; Glazier, S.C. Ecological chemistry and the palatability spectrum. Science 1968, 161, 1349. [Google Scholar] [CrossRef] [PubMed]
- Dobler, S.; Dalla, S.; Wagschal, V.; Agrawal, A.A. Community-wide convergent evolution in insect adaptation to toxic cardenolides by substitutions in the Na,K-ATPase. Proc. Natl. Acad. Sci. USA 2012, 109, 13040. [Google Scholar] [CrossRef]
- Zhen, Y.; Aardema, M.L.; Medina, E.M.; Schumer, M.; Andolfatto, P. Parallel molecular evolution in an herbivore community. Science 2012, 337, 1634. [Google Scholar] [CrossRef] [PubMed]
- Malcolm, S.B. Cardenolide-mediated interactions between plants and herbivores. Herbiv. Interact. Second. Plant Metab. Chem. Particip. 1991, 1, 251–296. [Google Scholar]
- Luckner, M.; Wichtl, M. Digitalis: Geschichte, Biologie, Biochemie, Chemie, Physiologie, Molekularbiologie, Pharmakologie, medizinische Anwendung; mit 48 Tabellen; Wiss. Verlagsges.: Shanghai, China, 2000. [Google Scholar]
- Agrawal, A.A.; Petschenka, G.; Bingham, R.A.; Weber, M.G.; Rasmann, S. Toxic cardenolides: Chemical ecology and coevolution of specialized plant–herbivore interactions. New Phytol. 2012, 194, 28–45. [Google Scholar] [CrossRef]
- Scholz, H.; Schmitz, W. Positive inotropic effects of digitoxin-and digoxin-glucuronide in human isolated ventricular heart muscle preparations. In Cardiac Glycoside Receptors and Positive Inotropy; Springer: Berlin/Heidelberg, Germany, 1984; pp. 134–139. [Google Scholar]
- Langford, S.D.; Boor, P.J. Oleander toxicity: An examination of human and animal toxic exposures. Toxicology 1996, 109, 1–13. [Google Scholar] [CrossRef]
- Lingrel, J.B. Na, K-ATPase: Isoform structure, function, and expression. J. Bioenerg. Biomembr. 1992, 24, 263–270. [Google Scholar]
- Emery, A.; Billingsley, P.; Ready, P.; Djamgoz, M. Insect Na+/K+-ATPase. J. Insect Physiol. 1998, 44, 197–210. [Google Scholar] [CrossRef]
- Al-Robai, A.A.; Khoja, S.M.; Al-Fifi, Z.I. Properties of ouabain-resistant Na+K+-transporting ATPase from the excretory system of Poekilocerus bufonius. Insect Biochem. 1990, 20, 701–707. [Google Scholar] [CrossRef]
- Holzinger, F.; Wink, M. Mediation of cardiac glycoside insensitivity in the monarch butterfly (Danaus plexippus): Role of an amino acid substitution in the ouabain binding site of Na+, K+-ATPase. J. Chem. Ecol. 1996, 22, 1921–1937. [Google Scholar] [CrossRef]
- Bramer, C.; Dobler, S.; Deckert, J.; Stemmer, M.; Petschenka, G. Na+/K+-ATPase resistance and cardenolide sequestration: Basal adaptations to host plant toxins in the milkweed bugs (Hemiptera: Lygaeidae: Lygaeinae). Proc. R. Soc. B Biol. Sci. 2015, 282, 20142346. [Google Scholar] [CrossRef] [PubMed]
- Petschenka, G.; Wagschal, V.; von Tschirnhaus, M.; Donath, A.; Dobler, S. Convergently evolved toxic secondary metabolites in plants drive the parallel molecular evolution of insect resistance. Am. Nat. 2017, 190, S29–S43. [Google Scholar] [CrossRef] [PubMed]
- Petschenka, G.; Agrawal, A.A. Milkweed butterfly resistance to plant toxins is linked to sequestration, not coping with a toxic diet. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151865. [Google Scholar] [CrossRef] [PubMed]
- Scudder, G.; Meredith, J. Morphological basis of cardiac glycoside sequestration by Oncopeltus fasciatus (Dallas)(Hemiptera: Lygaeidae). Zoomorphology 1982, 99, 87–101. [Google Scholar] [CrossRef]
- Bramer, C.; Friedrich, F.; Dobler, S. Defence by plant toxins in milkweed bugs (H eteroptera: L ygaeinae) through the evolution of a sophisticated storage compartment. Syst. Entomol. 2017, 42, 15–30. [Google Scholar] [CrossRef]
- Brower, L.P.; Van Brower, J.; Corvino, J.M. Plant poisons in a terrestrial food chain. Proc. Natl. Acad. Sci. USA 1967, 57, 893. [Google Scholar] [CrossRef]
- Glendinning, J.I.; Brower, L.P. Feeding and breeding responses of five mice species to overwintering aggregations of the monarch butterfly. J. Anim. Ecol. 1990, 1091–1112. [Google Scholar] [CrossRef]
- Glendinning, J.I.; Brower, L.P.; Montgomery, C.A. Responses of three mouse species to deterrent chemicals in the monarch butterfly. I. Taste and toxicity tests using artificial diets laced with digitoxin or monocrotaline. Chemoecology 1990, 1, 114–123. [Google Scholar] [CrossRef]
- Evans, D.L.; Castoriades, N.; Badruddine, H. Cardenolides in the defense of Caenocoris nerii (Hemiptera). Oikos 1986, 46, 325–329. [Google Scholar] [CrossRef]
- Berenbaum, M.R.; Miliczky, E. Mantids and milkweed bugs: Efficacy of aposematic coloration against invertebrate predators. Am. Midl. Nat. 1984, 111, 64–68. [Google Scholar] [CrossRef]
- Malcolm, S.B. Disruption of web structure and predatory behavior of a spider by plant-derived chemical defenses of an aposematic aphid. J. Chem. Ecol. 1989, 15, 1699–1716. [Google Scholar] [CrossRef] [PubMed]
- Bramer, C.; Schweizer, C.; Dobler, S. Cardenolide-defended milkweed bugs do not evoke learning in Nephila senegalensis spiders. Ethology 2018, 124, 504–513. [Google Scholar] [CrossRef]
- Duffey, S.; Scudder, G. Cardiac glycosides in Oncopeltus fasciatus (Dallas)(Hemiptera: Lygaeidae). I. The uptake and distribution of natural cardenolides in the body. Can. J. Zool. 1974, 52, 283–290. [Google Scholar] [CrossRef]
- Newcombe, D.; Blount, J.D.; Mitchell, C.; Moore, A.J. Chemical egg defence in the large milkweed bug, Oncopeltus fasciatus, derives from maternal but not paternal diet. Entomol. Exp. Et Appl. 2013, 149, 197–205. [Google Scholar] [CrossRef]
- Slater, J.A.; O’Donnell, J.E. A Catalogue of the Lygaeidae of the World (1960–1994); American Museum of Natural History: New York, NY, USA, 1995. [Google Scholar]
- Scudder, G.; Duffey, S. Cardiac glycosides in the Lygaeinae (Hemiptera: Lygaeidae). Can. J. Zool. 1972, 50, 35–42. [Google Scholar] [CrossRef]
- Winkler, C.; Wichtl, M. Neue cardenolide aus Adonis vernalis. Planta Med. 1986, 52, 68–70. [Google Scholar] [CrossRef]
- Wichtl, M.; Junior, P. Strophanthidin digitaloside and strophanthidin 6-deoxyguloside, two new cardenolide glycosides from Adonis vernalis L (author’s transl). Arch. Der Pharm. 1977, 310, 905–910. [Google Scholar] [CrossRef]
- Burrows, G.E.; Tyrl, R.J. Toxic Plants of North America; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Petschenka, G.; Halitschke, R.; Roth, A.; Stiehler, S.; Tenbusch, L.; Züst, T.; Hartwig, C.; Gámez, J.F.M.; Trusch, R.; Deckert, J.; et al. Predation drives specialized host plant associations in preadapted milkweed bugs (Heteroptera: Lygaeinae). bioRxiv 2020. [Google Scholar] [CrossRef]
- Kugelberg, O.; Solbreck, C. Field observations on the seasonal occurrence of lygaeus eguestris (L.)(Het., Lygaeidae) with special reference to food plant phenology. Insect Syst. Evol. 1972, 3, 189–210. [Google Scholar] [CrossRef]
- Rabitsch, W.; Deckert, J. Die Ritterwanze Lygaeus equestris (Linnaeus, 1758)(Heteroptera: Lygaeidae)–das Insekt des Jahres 2007. Beiträge Zur Entomofaunist. 2007, 8, 212–218. [Google Scholar]
- Aukema, B.; Bos, F.; Hermes, D.; Zeinstra, P. Nieuwe en interessante Nederlandse wantsen II, met een geactualiseerde naamlijst (Hemiptera: Heteroptera). Ned. Faun. Meded. 2005, 23, 37–76. [Google Scholar]
- Wachmann, E.; Melber, A.; Deckert, J. Wanzen; Goecke & Evers: Keltern, Germany, 2004; Volume 1. [Google Scholar]
- Malcolm, S.B.; Zalucki, M.P. Milkweed latex and cardenolide induction may resolve the lethal plant defence paradox. In Proceedings of the 9th International Symposium on Insect-Plant Relationships, Dordrecht, The Netherlands, 31 August 1996; pp. 193–196. [Google Scholar]
- Rasmann, S.; Johnson, M.D.; Agrawal, A.A. Induced responses to herbivory and jasmonate in three milkweed species. J. Chem. Ecol. 2009, 35, 1326. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.L.; Petschenka, G.; Flacht, L.; Agrawal, A.A. Cardenolide intake, sequestration, and excretion by the monarch butterfly along gradients of plant toxicity and larval ontogeny. J. Chem. Ecol. 2019, 45, 264–277. [Google Scholar] [CrossRef]
- Freeman, G.; Halton, J.H. Note on an exact treatment of contingency, goodness of fit and other problems of significance. Biometrika 1951, 38, 141–149. [Google Scholar] [CrossRef]
- Soper, D.S. Fisher’s Exact Test Calculator for a 2 × 3 Contingency Table [Software]. Available online: http://www.danielsoper.com/statcalc (accessed on 30 June 2020).
- Réaumur, M.D. Mém. pour. servir à l’histoire des insectes Tom. VI: 1742. Available online: https://www.biodiversitylibrary.org/bibliography/34174#/summary (accessed on 30 June 2020).
- Stiling, P.D. An Introduction to Insect Pests and Their Control; Macmillan Publishers Ltd.: Basingstoke, UK, 1985. [Google Scholar]
- Ridgway, R.; Jones, S. Field-cage releases of Chrysopa carnea for suppression of populations of the bollworm and the tobacco budworm on cotton. J. Econ. Entomol. 1968, 61, 892–898. [Google Scholar] [CrossRef]
- Ridgway, R.; Jones, S. Inundative releases of Chrysopa carnea for control of Heliothis on cotton. J. Econ. Entomol. 1969, 62, 177–180. [Google Scholar] [CrossRef]
- Shuvakhina, E. Criteria of biological effectiveness of Chrysopa carnea in the control of Colorado potato beetle on potato crops. Biulletin 1977, 41, 3–6. [Google Scholar]
- Shuvakhina, E. Experience of using Chrysopa carnea for control of Colorado potato beetle in the Voronezh Region. Biulletin’vsesoiuzngo Nauchno-Issledovatel’skogo Inst. Zashchity Rastenii 1978, 42, 3–9. [Google Scholar]
- Miszczak, M.; Niemczyk, E. Green lacewing (Chrysopa carnea Steph., Neuroptera Chrysopidae) as a predator of the European mite (Panonychus ulmi Koch) on apple trees. iI. the effectiveness of Chrysopa carnea larvae in control of Panonychus ulmi Koch. Fruit Sci. Rep. 1978, 5, 11–20. [Google Scholar]
- Principi, M.; Canard, M. Feeding habits [Chrysopidae]. Ser. Entomol. 1984, 27, 76–92. [Google Scholar]
- Canard, M.; Duelli, P. Predatory behavior of larvae and cannibalism. Biol. Chrysopidae 1984, 27, 92–100. [Google Scholar]
- McEwen, P.K.; New, T.R.; Whittington, A.E. Lacewings in the Crop Environment; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Gaumont, J. Observations sur quelques Chrysopidae (Insectes: Planipennes) prédateurs d’aphides. Ann. De L’université Et De L’apers 1965, 3, 24–32. [Google Scholar]
- Barbosa, P.R.; Michaud, J.; Bain, C.L.; Torres, J.B. Toxicity of three aphicides to the generalist predators Chrysoperla carnea (Neuroptera: Chrysopidae) and Orius insidiosus (Hemiptera: Anthocoridae). Ecotoxicology 2017, 26, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Luna, R.F.; Bestete, L.R.; Torres, J.B.; da Silva-Torres, C.S.A. Predation and behavioral changes in the neotropical lacewing Chrysoperla externa (Hagen)(Neuroptera: Chrysopidae) exposed to lambda-cyhalothrin. Ecotoxicology 2018, 27, 689–702. [Google Scholar] [CrossRef]
- Sun, R.; Jiang, X.; Reichelt, M.; Gershenzon, J.; Pandit, S.S.; Vassão, D.G. Tritrophic metabolism of plant chemical defenses and its effects on herbivore and predator performance. ELife 2019, 8, e51029. [Google Scholar] [CrossRef]
- ROTHSCHILD, M.; Von Euw, J.; Reichstein, T. Cardiac glycosides in a scale insect (Aspidiotus), a ladybird (Coccinella) and a lacewing (Chrysopa). J. Entomol. Ser. AGen. Entomol. 1973, 48, 89–90. [Google Scholar] [CrossRef]
- Firn, R.D.; Jones, C.G. Natural products–a simple model to explain chemical diversity. Nat. Prod. Rep. 2003, 20, 382–391. [Google Scholar] [CrossRef]
- Richards, L.A.; Lampert, E.C.; Bowers, M.D.; Dodson, C.D.; Smilanich, A.M.; Dyer, L.A. Synergistic effects of iridoid glycosides on the survival, development and immune response of a specialist caterpillar, Junonia coenia (Nymphalidae). J. Chem. Ecol. 2012, 38, 1276–1284. [Google Scholar] [CrossRef]
- Brower, L.P.; McEvoy, P.B.; Williamson, K.L.; Flannery, M.A. Variation in cardiac glycoside content of monarch butterflies from natural populations in eastern North America. Science 1972, 177, 426–429. [Google Scholar] [CrossRef]
- Isman, M.; Duffey, S.; Scudder, G. Cardenolide content of some leaf-and stem-feeding insects on temperate North American milkweeds (Asclepias spp.). Can. J. Zool. 1977, 55, 1024–1028. [Google Scholar] [CrossRef]
- Duffey, S.S. Cardiac glycosides and distastefulness: Some observations on the palatability spectrum of butterflies. Science 1970, 169, 78. [Google Scholar] [CrossRef] [PubMed]
- Brower, L.P.; Seiber, J.N.; Nelson, C.J.; Lynch, S.P.; Holland, M.M. Plant-determined variation in the cardenolide content, thin-layer chromatography profiles, and emetic potency of monarch butterflies, Danaus plexippus L. Reared on milkweed plants in California: 2.Asclepias speciosa. J. Chem. Ecol. 1984, 10, 601–639. [Google Scholar] [CrossRef] [PubMed]
- Rasmann, S.; Agrawal, A.A. Latitudinal patterns in plant defense: evolution of cardenolides, their toxicity and induction following herbivory. Ecology Letters 2011, 14, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Vencl, F.V.; Srygley, R.B. Enemy targeting, trade-offs, and the evolutionary assembly of a tortoise beetle defense arsenal. Evol. Ecol. 2013, 27, 237–252. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Allen, W.L.; Sherratt, T.N.; Speed, M.P. Avoiding Attack: The Evolutionary Ecology of Crypsis, Aposematism, and Mimicry; Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- Camara, M.D. Physiological mechanisms underlying the costs of chemical defence in Junonia coenia Hübner (Nymphalidae): A gravimetric and quantitative genetic analysis. Evol. Ecol. 1997, 11, 451–469. [Google Scholar] [CrossRef]
- Skelhorn, J.; Ruxton, G.D. Ecological factors influencing the evolution of insects’ chemical defenses. Behav. Ecol. 2008, 19, 146–153. [Google Scholar] [CrossRef][Green Version]
- Reudler, J.; Lindstedt, C.; Pakkanen, H.; Lehtinen, I.; Mappes, J. Costs and benefits of plant allelochemicals in herbivore diet in a multi enemy world. Oecologia 2015, 179, 1147–1158. [Google Scholar] [CrossRef]
- Zvereva, E.L.; Kozlov, M.V. The costs and effectiveness of chemical defenses in herbivorous insects: A meta-analysis. Ecol. Monogr. 2016, 86, 107–124. [Google Scholar] [CrossRef]
- Pasteels, J.M.; Grégoire, J.-C.; Rowell-Rahier, M. The chemical ecology of defense in arthropods. Annu. Rev. Entomol. 1983, 28, 263–289. [Google Scholar] [CrossRef]
- Munch, J.C.; Krantz Jr, J.C. Phrmacological and chemical studies of the digitalis group. I. Adonis, apocynum and convallaria. J. Am. Pharm. Assoc. 1934, 23, 988–996. [Google Scholar]
- McLAIN, D.K.; SHURE, D.J. Host plant toxins and unpalatability of Neacoryphus bicrucis (Hemiptera: Lygaeidae). Ecol. Entomol. 1985, 10, 291–298. [Google Scholar] [CrossRef]
- Rojas, B.; Burdfield-Steel, E.; Pakkanen, H.; Suisto, K.; Maczka, M.; Schulz, S.; Mappes, J. How to fight multiple enemies: Target-specific chemical defences in an aposematic moth. Proc. R. Soc. B Biol. Sci. 2017, 284, 20171424. [Google Scholar] [CrossRef] [PubMed]
- Tullberg, B.S.; Gamberale-Stille, G.; Solbreck, C. Effects of food plant and group size on predator defence: Differences between two co-occurring aposematic Lygaeinae bugs. Ecol. Entomol. 2000, 25, 220–225. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pokharel, P.; Sippel, M.; Vilcinskas, A.; Petschenka, G. Defense of Milkweed Bugs (Heteroptera: Lygaeinae) against Predatory Lacewing Larvae Depends on Structural Differences of Sequestered Cardenolides. Insects 2020, 11, 485. https://doi.org/10.3390/insects11080485
Pokharel P, Sippel M, Vilcinskas A, Petschenka G. Defense of Milkweed Bugs (Heteroptera: Lygaeinae) against Predatory Lacewing Larvae Depends on Structural Differences of Sequestered Cardenolides. Insects. 2020; 11(8):485. https://doi.org/10.3390/insects11080485
Chicago/Turabian StylePokharel, Prayan, Marlon Sippel, Andreas Vilcinskas, and Georg Petschenka. 2020. "Defense of Milkweed Bugs (Heteroptera: Lygaeinae) against Predatory Lacewing Larvae Depends on Structural Differences of Sequestered Cardenolides" Insects 11, no. 8: 485. https://doi.org/10.3390/insects11080485
APA StylePokharel, P., Sippel, M., Vilcinskas, A., & Petschenka, G. (2020). Defense of Milkweed Bugs (Heteroptera: Lygaeinae) against Predatory Lacewing Larvae Depends on Structural Differences of Sequestered Cardenolides. Insects, 11(8), 485. https://doi.org/10.3390/insects11080485




