Drosophila melanogaster Response to Feeding with Neomycin-Based Medium Expressed in Fluctuating Asymmetry
Abstract
1. Introduction
2. Materials and Methods
2.1. Drosophila melanogaster Culture
2.2. Feeding Experiments
2.3. FA Measurement
2.3.1. Larvae
2.3.2. Imagoes
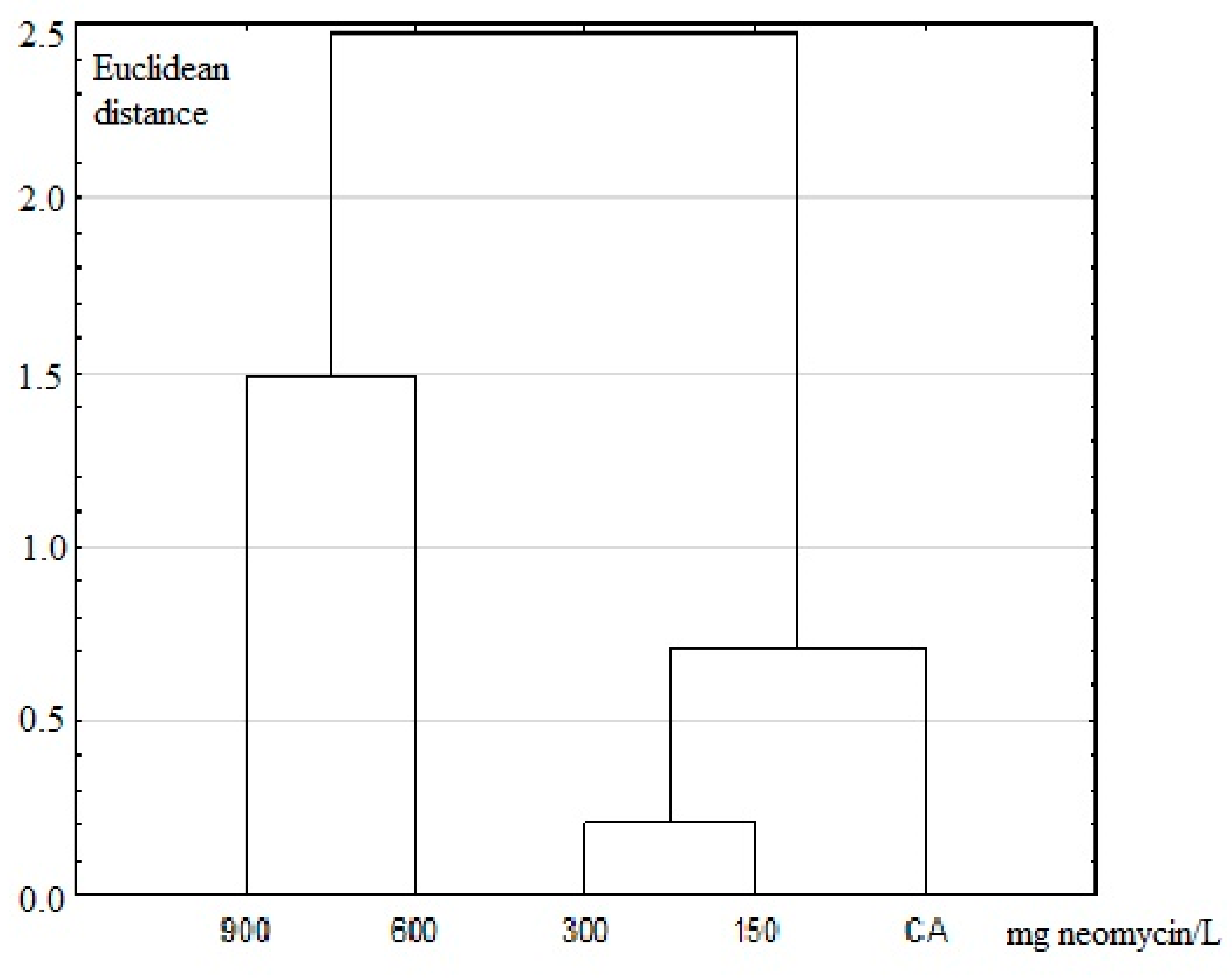
2.4. Biometric and Statistical Analyses
3. Results
3.1. Results of Larvae Fluctuation Asymmetry Measurements
3.2. Results of Imago Fluctuation Asymmetry Measurements
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Cakir, S.; Sarikaya, R. Genotoxicity testing of some organophosphate insecticides in the Drosophila wing spot test. Food Chem. Toxicol. 2005, 43, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Dogan, E.E.; Yesilada, E.; Ozata, L.; Yologlu, S. Genotoxicity Testing of Four Textile Dyes in Two Crosses of Drosophila Using Wing Somatic Mutation and Recombination Test. Drug Chem. Toxicol. 2005, 28, 289–301. [Google Scholar] [CrossRef]
- Ventrella, E.; Adamski, Z.; Chudzińska, E.; Miądowicz-Kobielska, M.; Marciniak, P.; Büyükgüzel, E.; Erdem, M.; Falabella, P.; Scrano, L.; Bufo, S.A. Solanum tuberosum and Lycopersicon esculentum leaf extracts and single metabolites affect development and reproduction of Drosophila melanogaster. PLoS ONE 2016, 11, e0155958. [Google Scholar] [CrossRef] [PubMed]
- Chowański, S.; Chudzińska, E.; Lelario, F.; Ventrella, E.; Marciniak, P.; Miądowicz-Kobielska, M.; Spochacz, M.; Szymczak, M.; Scrano, L.; Bufo, S.A.; et al. Insecticidal properties of Solanum nigrum and Armoracia rusticana extracts on reproduction and development of Drosophila melanogaster. Ecotox. Environ. Saf. 2018, 162, 454–463. [Google Scholar] [CrossRef]
- Singh, P.; House, H.L. Antimicrobials: ‘Safe’ levels in a synthetic diet of an insect, Agria affinis. J. Insect Physiol. 1970, 16, 1769–1782. [Google Scholar] [CrossRef]
- Büyükgüzel, K.; Yazgan, Ş. Effects of antimicrobial agents on survival and development of larvae of Pimpla turionellae L. (Hymenoptera: Ichneumonidae) reared on an artificial diet. Turk. J. Zool. 2002, 26, 111–119. [Google Scholar]
- Büyükgüzel, K. Positive Effects of Some Gyrase Inhibitors on Survival and Development of Pimpla turionellae (Hymenoptera: Ichneumonidae) Larvae Reared on an Artificial Diet. J. Econ. Entomol. 2001, 94, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Büyükgüzel, E.; Kalender, Y. Penicillin-induced oxidative stress: Effects on antioxidative response of midgut tissues in larval instars of G. mellonella. J. Econ. Entomol. 2007, 100, 1533–1541. [Google Scholar] [CrossRef]
- Büyükgüzel, E.; Kalender, Y. Galleria mellonella (L.) survivorship, development and protein content in response to dietary antibiotics. J. Entomol. Sci. 2008, 43, 27–40. [Google Scholar] [CrossRef]
- Büyükgüzel, E.; Kalender, Y. Exposure to streptomycin alters oxidative and antioxidative response in larval midgut tissues of Galleriamellonella. Pestic. Biochem. Physiol. 2009, 94, 112–118. [Google Scholar] [CrossRef]
- Kalghatgi, S.; Spina, C.S.; Costello, J.C.; Liesa, M.; Morones-Ramirez, J.R.; Slomovic, S.; Molina, A.; Shirihai, O.S.; Collins, J.J. Bactericidal Antibiotics Induce Mitochondrial Dysfunction and Oxidative Damage in Mammalian Cells. Sci. Transl. Med. 2013, 5, 192. [Google Scholar] [CrossRef] [PubMed]
- Al-Waili, N.; Salom, K.; Al-Ghamdi, A.; Ansari, M.J. Antibiotic, Pesticide, and Microbial Contaminants of Honey: Human Health Hazards. Sci. World J. 2012, 2012, 930849. [Google Scholar] [CrossRef]
- Ouye, M.T. Effects of antimicrobial agents on microorganisms and pink bollworm development. J. Econ. Entomol. 1962, 55, 854–857. [Google Scholar] [CrossRef]
- Kishaba, A.N.; Henneberry, T.J.; Pangaldan, R.; Tsao, P.H. Effects of mold inhibitors in larval diet on the biology of the cabbage looper. J. Econ. Entomol. 1968, 61, 1189–1194. [Google Scholar] [CrossRef]
- Hedin, P.A.; Maxwe, F.G.; Jenkins, J.N. Insect plant attractants, feeding stimulants, repellents, deterrents, and other related factors affecting insect behavior. In Procedings of the Summer Institute on Biological Control of Plant Insects and Diseases; Maxwell, F.G., Harris, F.A., Eds.; University Press: Jackson, MS, USA, 1974; pp. 494–527. [Google Scholar]
- Costa, H.S.; Thomas, J.H.; Nick, C.T. Effect of antibacterial materials on Bemisia argentifolii (Homoptera: Aleyrodidae) oviposition, growth, survival and sex ratio. J. Econ. Entomol. 1997, 90, 333–339. [Google Scholar] [CrossRef]
- Alverson, J.; Cohen, A.C. Effect of antifungal agents on biological fitness of Lygus hesperus (Heteroptera: Miridae). J. Econ. Entomol. 2002, 95, 256–260. [Google Scholar] [CrossRef]
- Boxall, A.B.A.; Fogg, L.A.; Blackwell, P.A.; Kay, P.; Pemberton, E.J.; Croxford, A. Veterinary medicines in the environment. Rev. Environ. Contam. Toxicol. 2004, 180, 1–91. [Google Scholar]
- Kaul, M.; Pilch, D.S. Thermodynamics of aminoglycoside-rRNA recogniton: The binding of neomycin-class aminoglycosides to the A site of 16 S rRNA. Biochemistry 2002, 41, 7695–7706. [Google Scholar] [CrossRef]
- Kaul, M.; Barbieri, C.M.; Kerrigan, J.E.; Pilch, D.S. Coupling of drug protonation to the specific binding of aminoglycosides to the A site of 16 S rRNA: Elucidation of the number of drug amino groups involved and their identites. J. Mol. Biol. 2003, 326, 1373–1387. [Google Scholar] [CrossRef]
- Carney, D.H.; Scott, D.L.; Gordon, E.A.; LaBelle, E.F. Phosphoinositides in mitogenesis: Neomycin inhibits thrombin-stimulated phosphoinositides turnover and initiation of cell proliferation. Cell 1985, 42, 479–488. [Google Scholar] [CrossRef]
- Arya, D.P.; Coffee, R.L. DNA Triple Helix Stabilization by Aminoglycoside Antibiotics. Bioorg. Med. Chem. Lett. 2000, 10, 1897–1899. [Google Scholar] [CrossRef]
- Gilbert, D.N. Aminoglycosides. In Principles and Practice of Infectious Diseases; Mandell, G.L., Bennett, J.E., Dolin, R., Eds.; Elsevier Cherchill Livingstone: Philadelphia, PA, USA, 2005; pp. 328–356. [Google Scholar]
- Denamur, S.; Van Bambeke, F.; Mingeot-Leclercq, M.P.; Tulkens, P.M. Apoptosis Induced by Aminoglycosides in LLC-PK1 Cells: Comparative Study of Neomycin. Gentamicin, Amikacin, and Isepamicin Using Electroporation. Antimicrob. Agets Chemother. 2008, 52, 2236–2238. [Google Scholar] [CrossRef] [PubMed]
- Rathore, H.S.; Swarup, H. Studies on the effects of neomycin on puffing in Chironomus. Acta Histochem. 1980, 67, 86–94. [Google Scholar] [CrossRef]
- Erdem, M.; Küçük, C.; Büyükgüzel, E.; Büyükgüzel, K. Ingestion of the anti-bacterial agent, gemifloxacin mesylate, leads to increased GST activity and peroxidation products in hemolymph of Galleria mellonella L. (Lepidoptera: Pyralidae), Arch. Insect Biochem. Physiol. 2016, 93, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Büyükgüzel, E.; Kayaoğlu, S. Niklozamidin Galleria mellonella L. (Lepidoptera: Pyralidae)’nın bazı biyolojik ve fizyolojik ozelliklerine etkisi. Turk. Entomol. Derg. 2014, 38, 83–99. [Google Scholar] [CrossRef][Green Version]
- Boxall, A.B.A.; Fogg, L.A.; Kay, P.; Blackwell, P.A.; Pemberton, E.J.; Croxford, A. Prioritisation of veterinary medicines in the UK environment. Toxicol. Lett. 2003, 142, 207–218. [Google Scholar] [CrossRef]
- Badyaev, A.V. Stress-induced variation in evolution: From behavioural plasticity to genetic assimilation. Proc. R. Soc. B 2005, 272, 877–886. [Google Scholar] [CrossRef]
- Polack, M. (Ed.) Developmental Instability: Causes and Consequences; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Soto, I.M.; Carreira, V.P.; Soto, E.M.; Hasson, E. Wing morphology and fluctuating asymmetry depend on the host plant in cactophilic Drosophila. J. Evol. Biol. 2008, 21, 598–609. [Google Scholar] [CrossRef]
- Leary, R.F.; Allendorf, F.W. Fluctuating asymmetry as an indicator of stress: Implication for conservation biology. Trends Ecol. Evol. 1989, 4, 214–217. [Google Scholar] [CrossRef]
- De Coster, G.; Van Dongen, S.; Malaki, P.; Muchane, M.; Alcantara-Exposito, A.; Matheve, H.; Lens, L. Fluctuating Asymmetry and Environmental Stress: Understanding the Role of Trait History. PLoS ONE 2013, 8, e57966. [Google Scholar] [CrossRef]
- Coda, J.A.; Martinez, J.J.; Steinmann, A.R.; Priotto, J.; Gomez, M.D. Fluctuating asymmetry as an indicator of environmental stress in small mammals. Mastozool. Neotrop. 2017, 24, 313–321. [Google Scholar]
- Te Velde, J.H.; Molthoff, C.F.M.; Scharloo, W. The function of anal papillae in salt adaptations of Drosophila melanogaster larvae. J. Evol. Biol. 1988, 2, 139–153. [Google Scholar] [CrossRef]
- Debat, V.; Begin, M.; Legout, H.; David, J.R. Allometric and nonallometric components of Drosophila wing shape respond differently to developmental temperature. Evolution 2003, 57, 2773–2784. [Google Scholar] [CrossRef] [PubMed]
- Debat, V.; Milton, C.C.; Rutherford, S.; Klingenberg, C.P.; Hoffmann, A.A. HSP90 and the quantitative variation of wing shape in Drosophila melanogaster. Evolution 2006, 60, 2529–2538. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.H.; Daborn, P.J.; Hoffmann, A.A.; Takano-Shimizu, T. Environmental Stress-Dependent Effects of Deletions Encompassing Hsp70Ba on Canalization and Quantitative Trait Asymmetry in Drosophila melanogaster. PLoS ONE 2011, 6, e17295. [Google Scholar] [CrossRef]
- Lesch, C.; Goto, A.; Lindgren, M.; Bidla, G.; Dushay, M.S.; Theopold, U. A role for hemolectin in coagulation and immunity in Drosophila melonagaster. Dev. Comp. Immunol. 2007, 31, 1255–1263. [Google Scholar] [CrossRef]
- Rogina, B.; Reenan, R.A.; Nilsen, S.P.; Helfand, S.L. Extended life-span conferred by contransporter gene mutations in Drosophila. Science 2000, 290, 2137–2140. [Google Scholar] [CrossRef]
- Reis, T. Effects of Synthetic Diets Enriched in Specific Nutrients on Drosophila Development, Body Fat, and Lifespan. PLoS ONE 2016, 1, e0146758. [Google Scholar] [CrossRef]
- Roberts, D.B. Drosophila: A Practical Approach; Roberts, D.B., Ed.; IRL Press: Oxford, UK, 1986; p. 19. [Google Scholar]
- Erdem, M.; Büyükgüzel, E.; Büyükgüzel, K. Effect of Dietary Sodium Tetraborate on Adult Longevity and Fecundity of Drosophila melanogaster (Diptera: Drosphilidae). J. Entomol. Sci. 2016, 51, 305–313. [Google Scholar] [CrossRef]
- Güneş, E.; Büyükgüzel, E. Oxidative effects of boric acid on different developmental stages of Drosophila melanogaster Meigen, 1830 (Diptera: Drosophilidae). Turk. Entomol. Derg. 2017, 41, 3–15. [Google Scholar] [CrossRef]
- Büyükgüzel, K.; Yazgan, Ş. Bazı Antibiyotiklerin Endoparazitoid Pimpla turionellae L. (Hymenoptera: Ichneumonidae)’nın yaĢama ve GeliĢimine Etkileri. Turk. J. Zool. 1996, 20, 1–7. [Google Scholar]
- Kozlov, M.V.; Niemelä, P.; Junttila, J. Needle fluctuating asymmetry is a sensitive indicator of pollution impact on Scots pine (Pinus sylvestris). Ecol. Indic. 2002, 1, 271–277. [Google Scholar] [CrossRef]
- Chudzinska, E.; Pawlaczyk, E.M.; Celinski, K.; Diatta, J. Response of Scots pine (Pinus sylvestris L.) to stress induced by different types of pollutants—Testing the fluctuating asymmetry. Water Environ. J. 2014, 28, 533–539. [Google Scholar] [CrossRef]
- Ferguson., G.A.; Takane, Y. Statistical Analysis in Psychology and Education Ed(s).; McGraw Hill: Columbus, OH, USA, 2007. [Google Scholar]
- Chang, X.; Zhai, B.; Liu, X.; Wang, M. Effects of temperature stress and pesticide exposure on fluctuating asymmetry and mortality of Copera annulata (Selys) (Odonata: Zygoptera) larvae. Ecotox. Environ. Saf. 2007, 67, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Zhai, B.; Wang, M.; Wang, B. Relationship between exposure to an insecticide and fluctuating asymmetry in a damselfly (Odonata: Coenagriidae). Hydrobiologia 2007, 586, 213–220. [Google Scholar] [CrossRef]
- Mpho, M.; Holloway, G.J.; Callghan, A. A comparison of the effects of organophosphate insecticide exposure and temperature stress on fluctuating asymmetry and life history traits in Culex quinquefasciatus. Chemosphere 2001, 45, 713–720. [Google Scholar] [CrossRef]
- Antipin, M.I.; Imasheva, A.G. Genetic Variability and Fluctuating Asymmetry of Morphological Traits in Drosophila melanogaster Reared on a Pesticide-Containing Medium. Russ. J. Genet. 2001, 37, 247–252. [Google Scholar] [CrossRef]
- Polak, M.; Opoka, R.; Cartwright, I.L. Response of fluctuating asymmetry to arsenic toxicity: Support for the developmental selection hypothesis. Environ. Pollut. 2002, 118, 19–28. [Google Scholar] [CrossRef]
- Vishalakshi, B.N.; Singh, C. Effect of environmental stress on fluctuating asymmetry in certain morphological traits in Drosophila ananassae: Nutrition and larval crowding. Can. J. Zool. 2008, 86, 427–437. [Google Scholar] [CrossRef]
- Clarke, G.M.; McKenzie, L.J. Fluctuating asymmetry as a quality control ındicator for ınsect mass rearing processes. J. Econ. Entomol. 1992, 85, 2045–2050. [Google Scholar] [CrossRef]
- De Souza Rosa, A.; Teixeira, J.S.G.; Vollet-Neto, A.; Queiroz, E.P.; Blochtein, B.; Pires, C.S.S.; Imperatriz-Fonseca, V.L. Consumption of the neonicotinoid thiamethoxam during the larval stage affects the survival and development of the stingless bee, Scaptotrigona aff. depilis. Apidologie 2016, 47, 729–738. [Google Scholar] [CrossRef]
- Aslan, N.; Büyükgüzel, E.; Büyükgüzel, K. Oxidative effects of gemifloxacin on some biological traits of Drosophila melanogaster (Diptera: Drosophilidae). Environ. Entomol. 2019, 48, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Kastamonuluoğlu, S.; Büyükgüzel, K.; Büyükgüzel, E. The use of dietary antifungal agent terbınafıne in artifıcial diet and its effects on some biological and biochemical parameters of the model organism Galleria mellonella L. J. Econ. Entomol. 2020. [Google Scholar] [CrossRef]
- Üstündağ, G.; Büyükgüzel, K.; Büyükgüzel, E. The effect of niclosamide on certain biological and biochemical properties of Drosophila melanogaster. Eur. J. Biol. 2019, 78, 29–38. [Google Scholar] [CrossRef]
- Sillanpaa, S.; Salminen, J.P.; Eeva, T. Fluctuating asymmetry in great tit nestlings in relation to diet quality, calcium availability and pollution exposure. Sci. Total Environ. 2010, 408, 3303–3309. [Google Scholar] [CrossRef]
- Vijendravarma, R.K.; Narasimha, S.; Kawecki, T.J. Adaptation to larval malnutrition does not affect fluctuating asymmetry in Drosophila melanogaster. Biol. J. Linn. Soc. 2011, 104, 19–28. [Google Scholar] [CrossRef]
- Vijendravarma, R.K.; Narasimha, S.; Kawecki, T.J. Plastic and evolutionary responses of cell size and number to larval malnutrition in Drosophila melanogaster. J. Evol. Biol. 2011, 24, 897–903. [Google Scholar] [CrossRef]
- Møller, A.P.; Cuervo, J.J. Asymmetry, size, and sexual selection: Factors affecting heterogeneity in relationships between asymmetry and sexual selection. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: Oxford, UK, 2003; pp. 262–278. [Google Scholar]
- Tryjanowski, P.; Adamski, Z. Sex differences in fluctuating asymmetry of body traits in chewing lice Docophorulus coarctatus (Phthiraptera: Ischnocera). Parasit. Res. 2007, 101, 1289–1294. [Google Scholar] [CrossRef]
- McLachlan, A.J. Fluctuating Asymmetry in Flies, What Does It Mean? Symmetry 2010, 2, 1099–1107. [Google Scholar] [CrossRef]
- Mead, F.; Williams, A.J. Block of the Ryanodine Receptor Channel by Neomycin Is Relieved at High Holding Potentials. Biophys. J. 2002, 82, 1953–1963. [Google Scholar] [CrossRef][Green Version]
- Mead, F.; Williams, A.J. Ryanodine-inducted structural alterations in the RyR channel suggested by neomycin block. Biophys. J. 2002, 82, 1964–1974. [Google Scholar] [CrossRef]
- Mead, F.; Williams, A.J. Electrostatic mechanisms underlie neomycin block of the cardiac ryanodine receptor channel (RyR2). Biophys. J. 2004, 87, 3814–3825. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Capes, E.M.; Loaiza, R.; Valdivia, H.H. Ryanodine receptors. Skelet. Muscle 2011, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.M.; Scott, K.; Zuker, C.S.; Rubin, G.M. The ryanodine receptor is essential for larval development in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2000, 97, 5942–5947. [Google Scholar] [CrossRef]
- Baumann, O. Distribution of ryanodine receptor Ca2+ channels in insects photoreceptor cells. J. Comp. Neurol. 2000, 421, 347–361. [Google Scholar] [CrossRef]
- Harmanci, C.; Büyükgüzel, K.; Büyükgüzel, E. The Effect of Neomycin on Survival and Development of Pimpla turionellae L. (Hymenoptera: Ichneumonidae) Reared on a Natural Host. J. Econ. Entomol. 2019, 22, 1081–1088. [Google Scholar]
- Sha, S.H.; Schacht, J. Salicylate attenuates gentamicin-inducted ototoxicity. Lab. Investig. 1999, 79, 807–813. [Google Scholar] [PubMed]
- Xie, J.; Talaska, A.E.; Schacht, J. New developments in aminoglycoside therapy and ototoxicity. Hear Res. 2011, 281, 28–37. [Google Scholar] [CrossRef]
- Desa, D.; Nichols, M.G.; Smith, H.J. Aminoglycosides rapidly inhibit NAD(P)H metabolism increasing reactive oxygen species and cochlear cell demise. J. Biomed. Opt. 2018, 24, 051403. [Google Scholar] [CrossRef]
- Gangestad, S.W.; Merriman, L.A.; Thompson, M.E. Men’s oxidative stress, fluctuating asymmetry and physical attractiveness. Anim. Behav. 2010, 80, 1005–1013. [Google Scholar] [CrossRef]
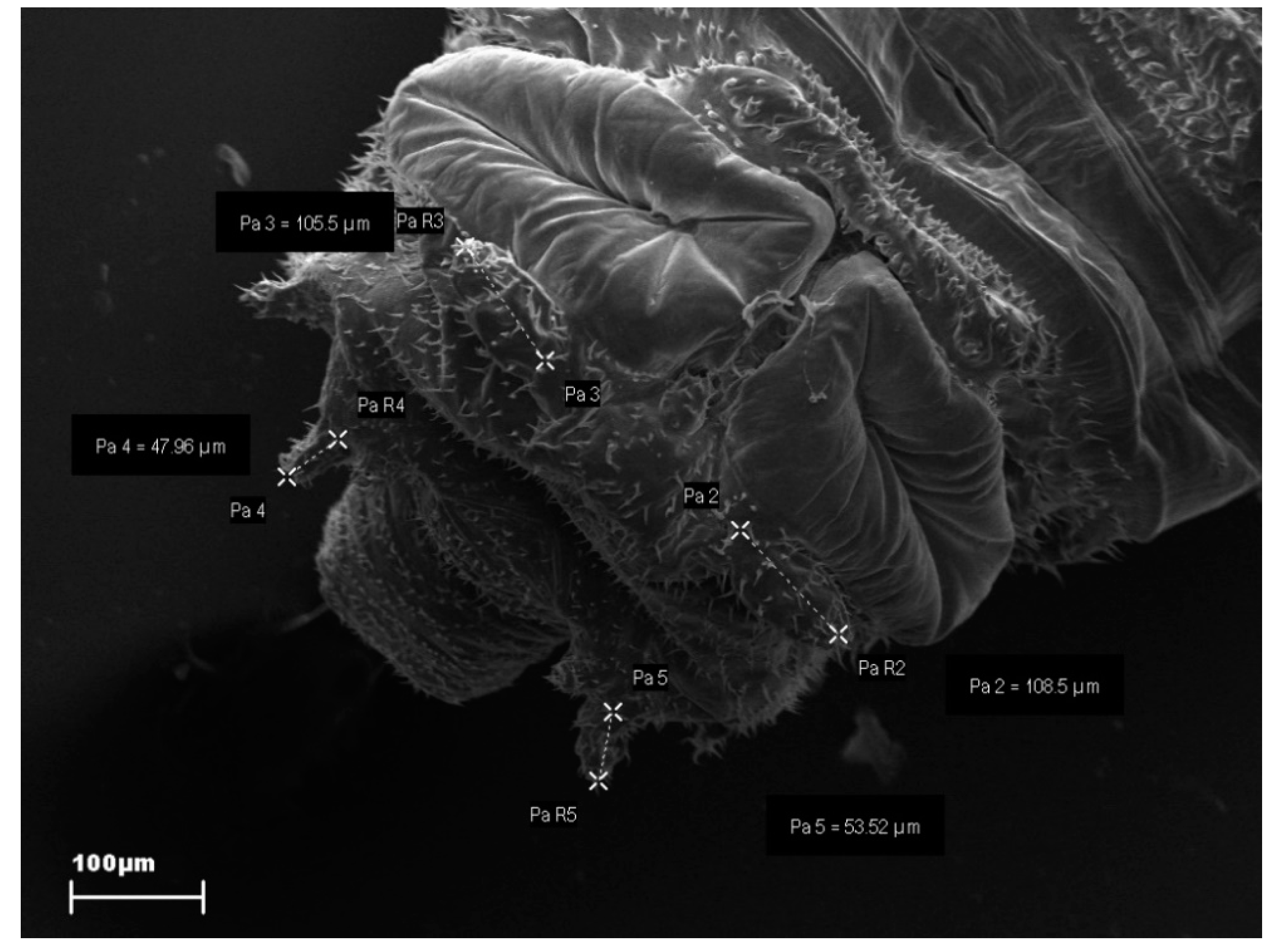






| Anal Papillae | N | Pearson Correlation Coefficient | p-Value |
|---|---|---|---|
| AP1 | 49 | 0.65 | <0.0001 *** |
| AP2 | 53 | 0.71 | <0.0001 *** |
| Anal Papillae | Mean of Longer Appendix | Mean of Shorter Appendix | AP1: F(1,96) AP2: F(1,104) | p-Value |
|---|---|---|---|---|
| AP1 | 98.9 | 90.2 | 4.87 | 0.0297 * |
| AP2 | 63.8 | 56.9 | 11.10 | 0.0011 ** |
| Vein | N | Pearson Correlation Coefficient | p-Value |
|---|---|---|---|
| A | 42 | 0.52 | <0.0001 |
| B | 42 | 0.66 | <0.0001 |
| C | 42 | 0.83 | <0.0001 |
| D | 42 | 0.87 | <0.0001 |
| E | 42 | 0.74 | <0.0001 |
| F | 42 | 0.54 | <0.0001 |
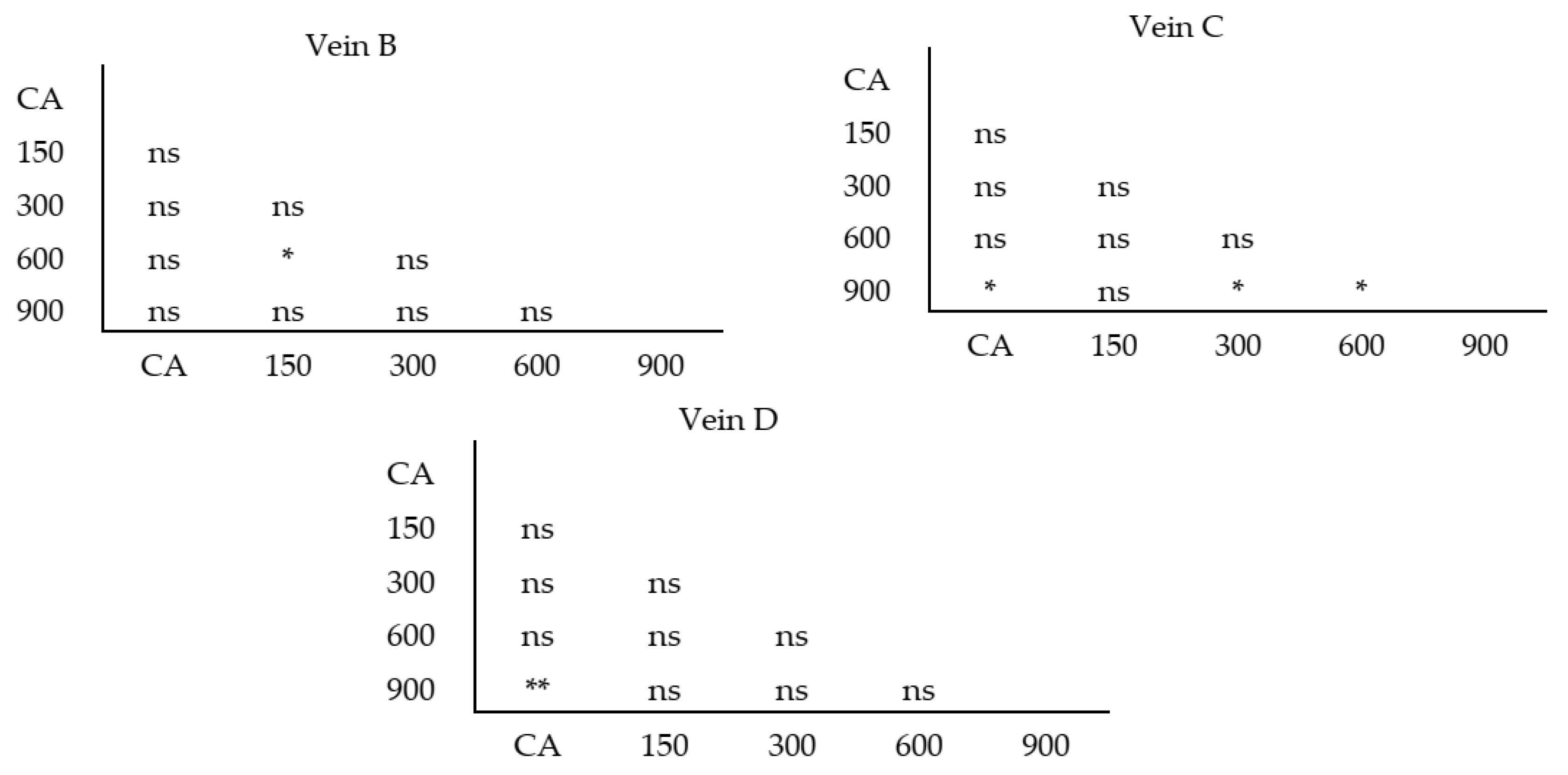
| Vein | Mean of Longer Vein | Mean of Shorter Vein | F(1, 82) | p-Value |
|---|---|---|---|---|
| A | 1352.6 | 1324.6 | 1.27 | 0.263 |
| B | 56.4 | 51.1 | 9.07 | 0.003 ** |
| C | 153.9 | 142.5 | 6.14 | 0.015 * |
| D | 323.2 | 308.6 | 5.65 | 0.019 * |
| E | 929.1 | 915.4 | 0.82 | 0.369 |
| F | 1386.5 | 1373.8 | 0.35 | 0.555 |
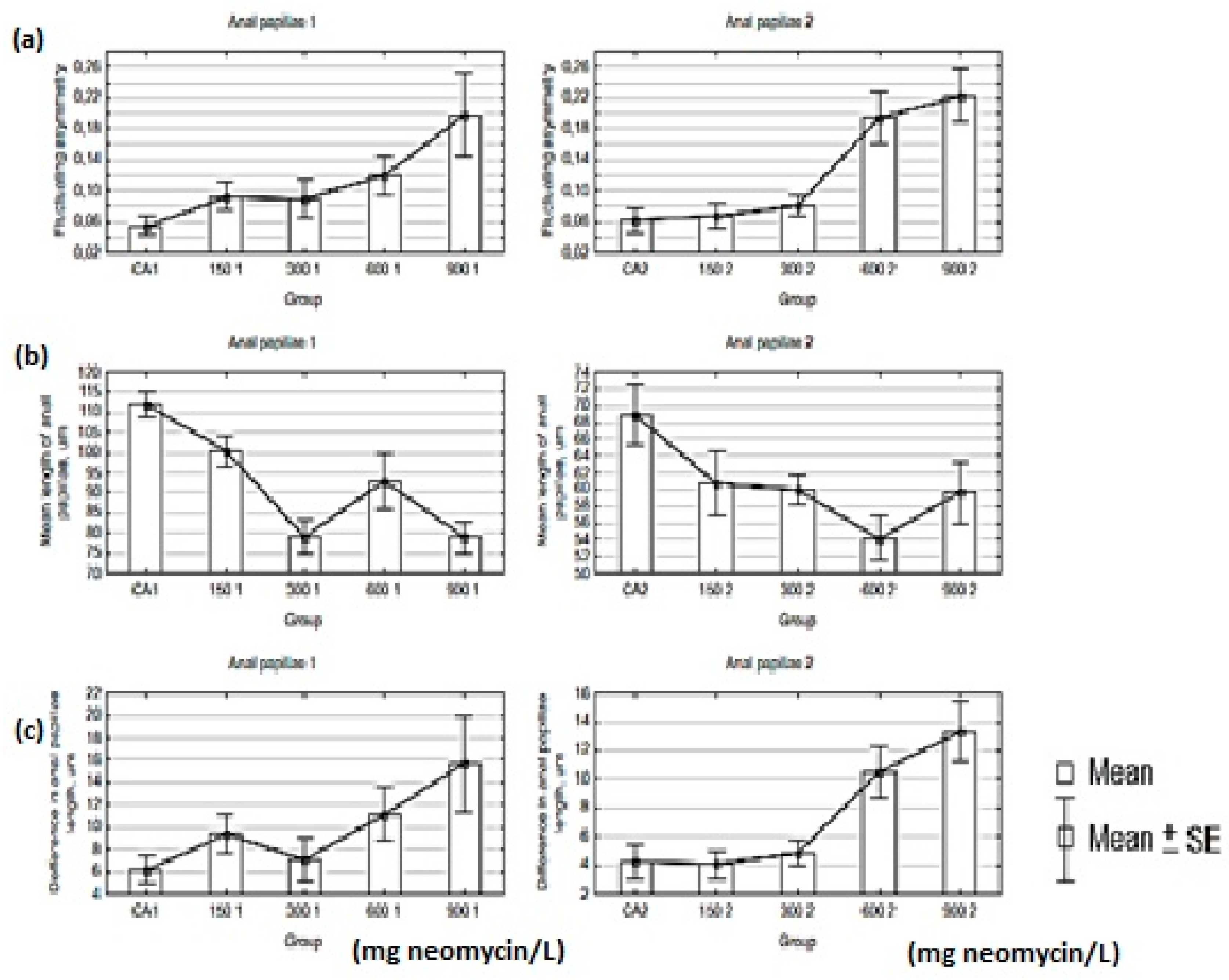
| Anal Papillae | Group | N | Mean | Min. | Max. | Range | SD | V% |
|---|---|---|---|---|---|---|---|---|
| AP 1 | CA | 12 | 0.05481 | 0.0089 | 0.1260 | 0.1170 | 0.039 | 70.56 |
| 150 | 10 | 0.09347 | 0.0232 | 0.2046 | 0.1814 | 0.053 | 56.83 | |
| 300 | 9 | 0.08938 | 0.0085 | 0.1915 | 0.1831 | 0.072 | 80.27 | |
| 600 | 11 | 0.12023 | 0.0235 | 0.3109 | 0.2874 | 0.084 | 70.01 | |
| 900 | 7 | 0.19788 | 0.0116 | 0.3687 | 0.3570 | 0.144 | 72.54 | |
| AP 2 | CA | 9 | 0.06147 | 0.0039 | 0.1568 | 0.1529 | 0.053 | 86.15 |
| 150 | 11 | 0.06655 | 0.0013 | 0.1927 | 0.1914 | 0.053 | 80.01 | |
| 300 | 13 | 0.08154 | 0.0032 | 0.1678 | 0.1646 | 0.052 | 64.30 | |
| 600 | 12 | 0.19467 | 0.0499 | 0.4492 | 0.3994 | 0.116 | 59.48 | |
| 900 | 8 | 0.22331 | 0.0867 | 0.4012 | 0.3145 | 0.098 | 43.77 |
| Vein | Group | N | Mean | Min. | Max. | Range | SD | V% |
|---|---|---|---|---|---|---|---|---|
| B | CA | 12 | 0.09677 | 0.0104 | 0.2169 | 0.2065 | 0.055 | 56.88 |
| 150 | 8 | 0.22371 | 0.0299 | 0.6875 | 0.6576 | 0.222 | 99.44 | |
| 300 | 7 | 0.06302 | 0.0233 | 0.1121 | 0.0889 | 0.033 | 52.27 | |
| 600 | 11 | 0.07752 | 0.0310 | 0.1374 | 0.1064 | 0.040 | 52.00 | |
| 900 | 4 | 0.13546 | 0.0164 | 0.2488 | 0.2325 | 0.107 | 79.35 | |
| C | CA | 12 | 0.05021 | 0.0127 | 0.2807 | 0.2680 | 0.074 | 146.68 |
| 150 | 8 | 0.16775 | 0.0147 | 0.9371 | 0.9224 | 0.315 | 187.71 | |
| 300 | 7 | 0.04436 | 0.0128 | 0.0889 | 0.0760 | 0.024 | 54.01 | |
| 600 | 11 | 0.08178 | 0.0219 | 0.2957 | 0.2738 | 0.083 | 101.79 | |
| 900 | 4 | 0.23740 | 0.0474 | 0.5152 | 0.4678 | 0.202 | 85.26 | |
| D | CA | 12 | 0.04517 | 0.0214 | 0.0691 | 0.0477 | 0.015 | 32.49 |
| 150 | 8 | 0.09850 | 0.0112 | 0.5013 | 0.4901 | 0.165 | 167.91 | |
| 300 | 7 | 0.03594 | 0.0137 | 0.0734 | 0.0598 | 0.025 | 68.86 | |
| 600 | 11 | 0.03677 | 0.0032 | 0.0691 | 0.0658 | 0.020 | 53.87 | |
| 900 | 4 | 0.01693 | 0.0089 | 0.0235 | 0.0146 | 0.007 | 38.47 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Büyükgüzel, K.; Büyükgüzel, E.; Chudzińska, E.; Lewandowska-Wosik, A.; Gaj, R.; Adamski, Z. Drosophila melanogaster Response to Feeding with Neomycin-Based Medium Expressed in Fluctuating Asymmetry. Insects 2020, 11, 378. https://doi.org/10.3390/insects11060378
Büyükgüzel K, Büyükgüzel E, Chudzińska E, Lewandowska-Wosik A, Gaj R, Adamski Z. Drosophila melanogaster Response to Feeding with Neomycin-Based Medium Expressed in Fluctuating Asymmetry. Insects. 2020; 11(6):378. https://doi.org/10.3390/insects11060378
Chicago/Turabian StyleBüyükgüzel, Kemal, Ender Büyükgüzel, Ewa Chudzińska, Anetta Lewandowska-Wosik, Renata Gaj, and Zbigniew Adamski. 2020. "Drosophila melanogaster Response to Feeding with Neomycin-Based Medium Expressed in Fluctuating Asymmetry" Insects 11, no. 6: 378. https://doi.org/10.3390/insects11060378
APA StyleBüyükgüzel, K., Büyükgüzel, E., Chudzińska, E., Lewandowska-Wosik, A., Gaj, R., & Adamski, Z. (2020). Drosophila melanogaster Response to Feeding with Neomycin-Based Medium Expressed in Fluctuating Asymmetry. Insects, 11(6), 378. https://doi.org/10.3390/insects11060378







