Developmental Differences on the Internal Reproductive Systems between the Prediapause and Prereproductive Riptortus pedestris Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects Rearing and Sample Collection
2.2. Dissection and Imaging of Reproductive Organs
2.3. Morphometric Analysis of Different Reproductive Parts
2.4. Data Analysis
3. Results
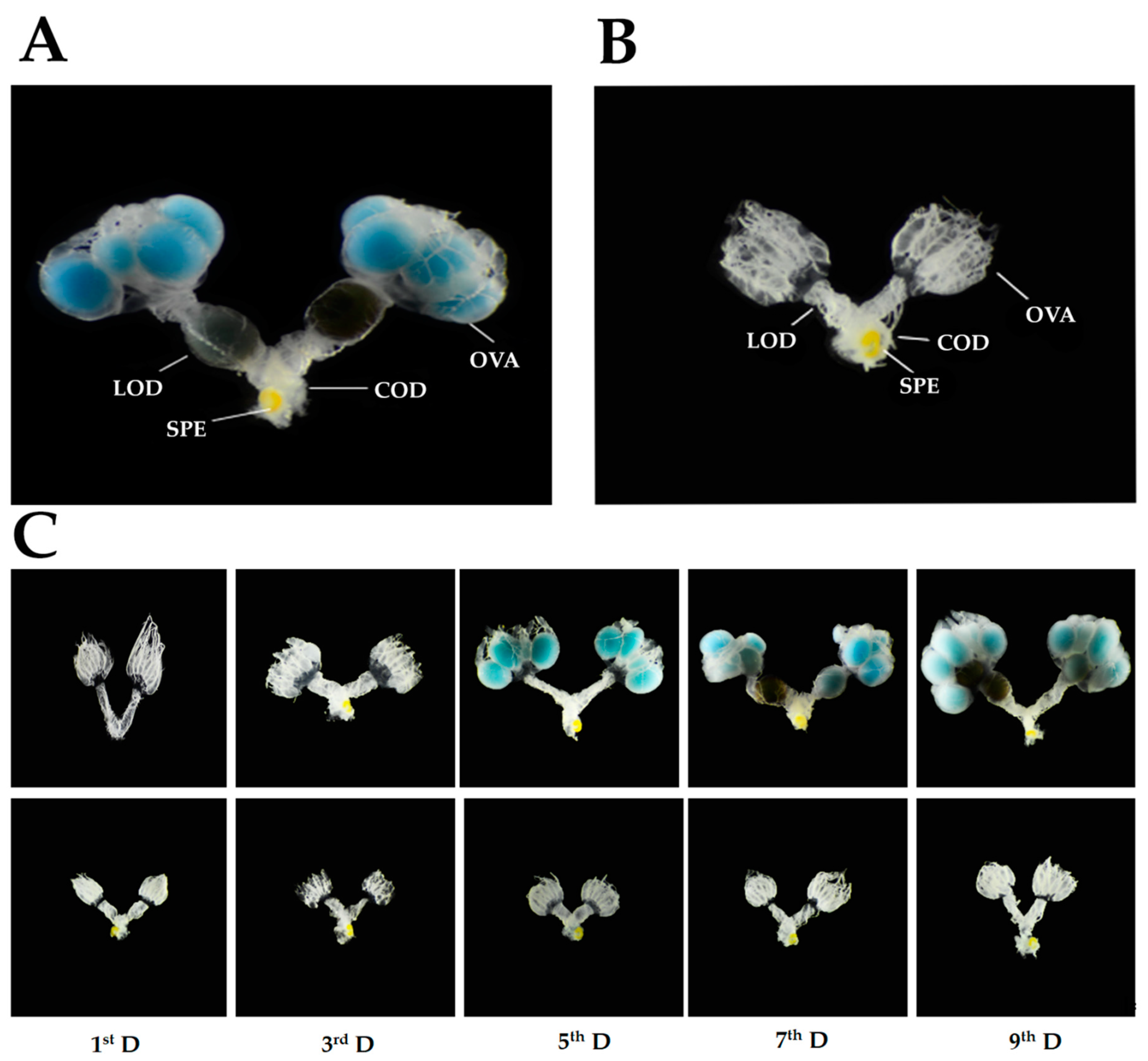
3.1. Development of Internal Reproductive Organs in Prereproductive and Prediapause Females
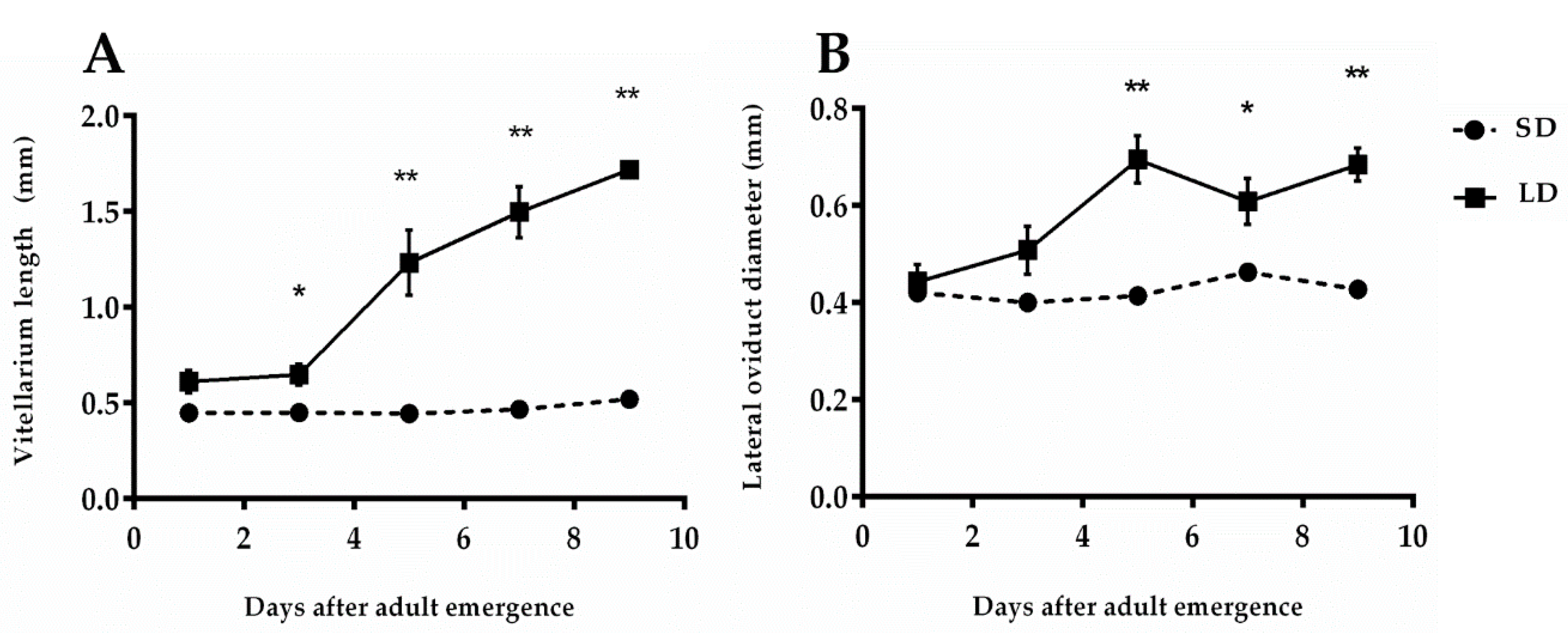
3.1.1. Length of Vitellarium
3.1.2. Diameter of the Lateral Oviduct
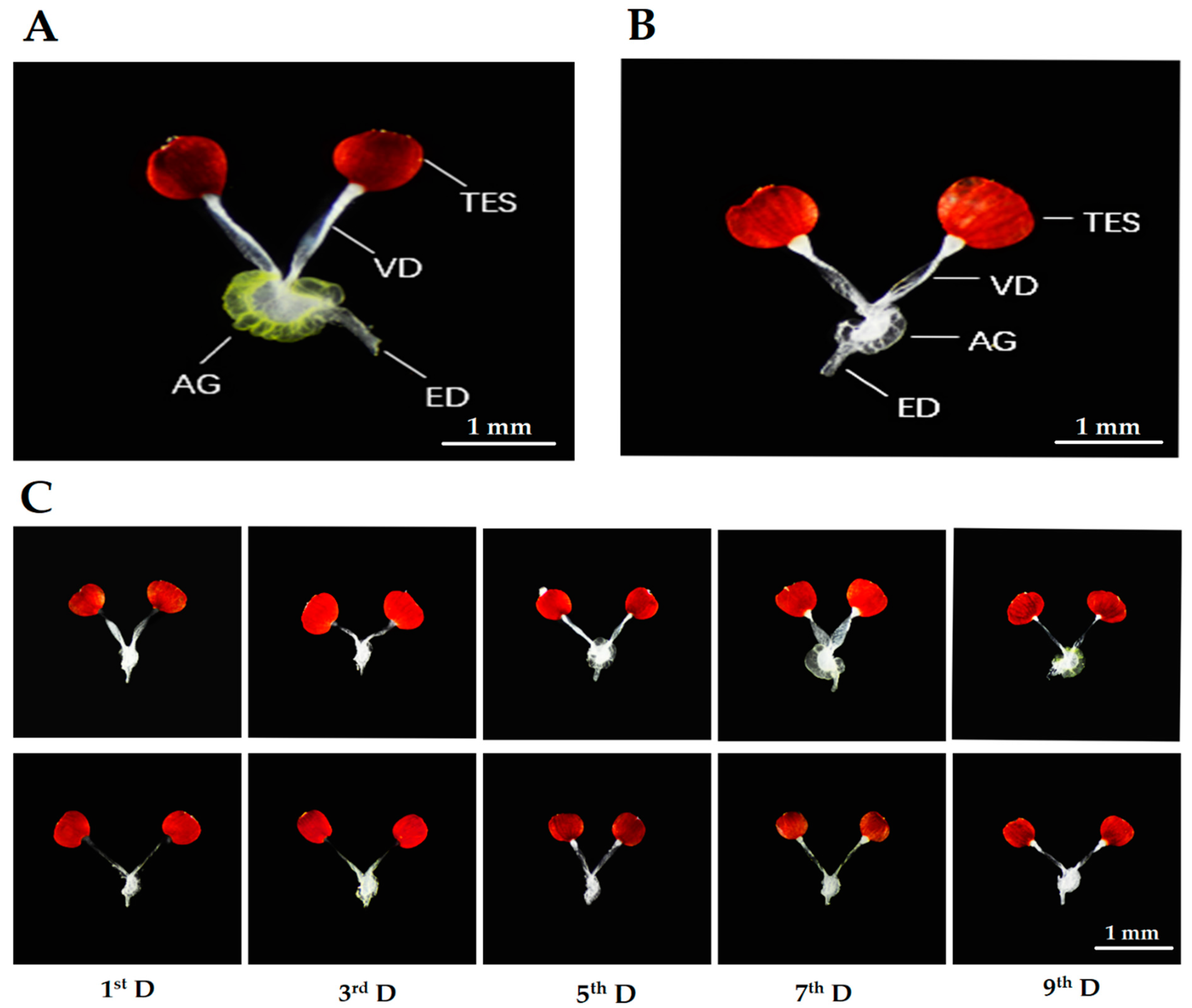
3.2. Development of Internal Reproductive Organs in Prereproductive and Prediapause Males
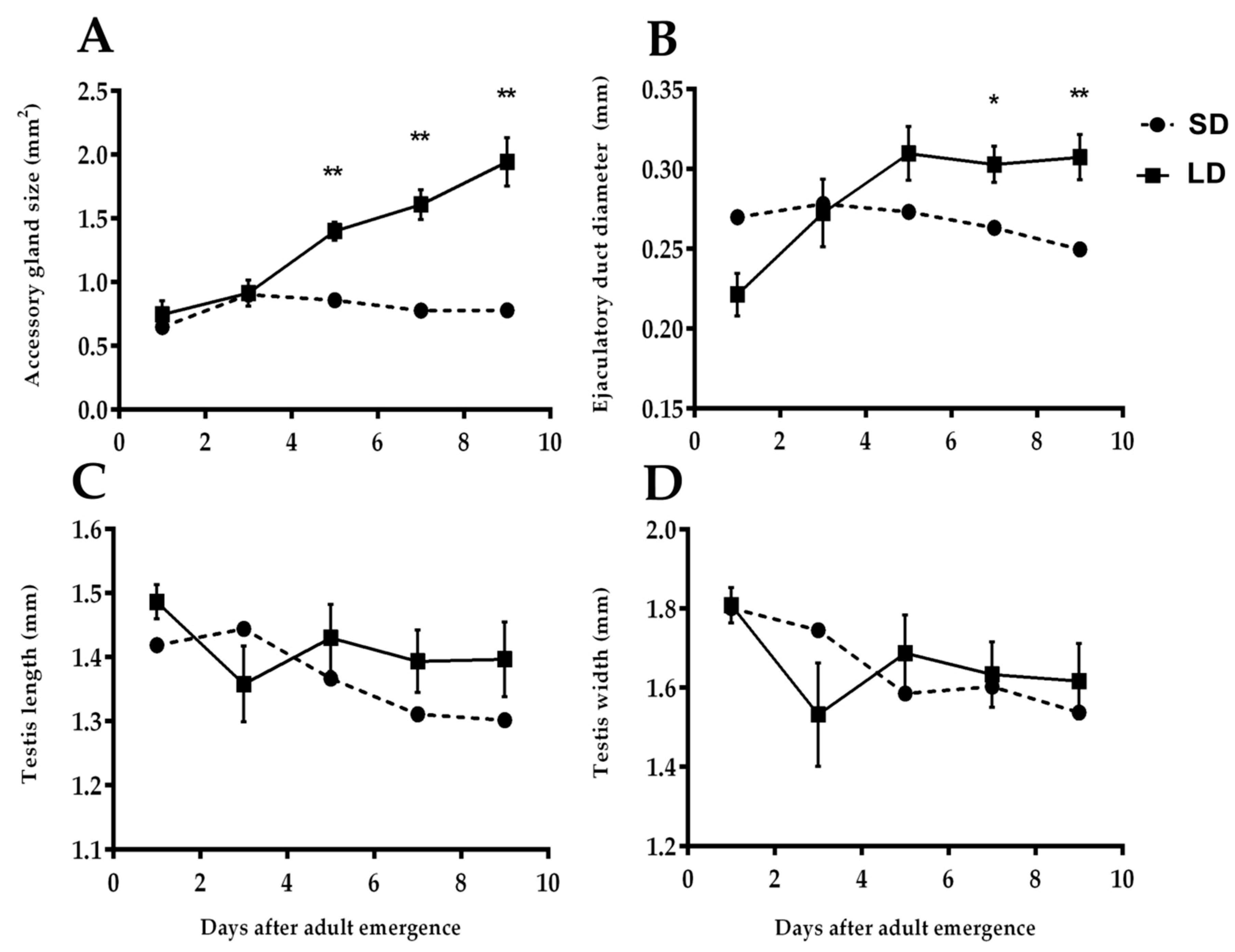
3.2.1. Size and Color of Accessory Gland
3.2.2. Diameter of Ejaculatory Duct
3.2.3. Length and Width of Testes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khaliq, A.; Javed, M.; Sagheer, M.; Sohail, M.; Sohail, M.; Sagheer, M. Environmental effects on insects and their population dynamics. J. Entomol. Zool. Stud. 2014, 1, 1–7. [Google Scholar]
- Rhainds, M. Female mating failures in insects. Entomol. Exp. Appl. 2010, 136, 211–226. [Google Scholar] [CrossRef]
- Gill, H.K.; Goyal, G.; Chahil, G. Insect diapause: A review. J. Agric. Sci. Technol. 2017, 7, 454–473. [Google Scholar]
- Sheikh, A.A.; Rehman, N.; Ritesh, K. Diverse adaptations in insects: A review. J. Entomol. Zool. Stud. 2017, 5, 343–350. [Google Scholar]
- Hahn, D.A.; Denlinger, D.L. Meeting the energetic demands of insect diapause: Nutrient storage and utilization. J. Insect Physiol. 2007, 53, 760–773. [Google Scholar] [CrossRef]
- Koštál, V. Eco-physiological phases of insect diapause. J. Insect Physiol. 2006, 52, 113–127. [Google Scholar] [CrossRef]
- Hodek, I. Controversial aspects of diapause development. Eur. J. Entomol. 2002, 99, 163–173. [Google Scholar] [CrossRef]
- Xue, F.; Spieth, H.R.; Aiqing, L.; Ai, H. The role of photoperiod and temperature in determination of summer and winter diapause in the cabbage beetle, Colaphellus bowringi (Coleoptera: Chrysomelidae). J. Insect Physiol. 2002, 48, 279–286. [Google Scholar] [CrossRef]
- Nakanura, K.; Numata, H. Photoperiodic control of the intensity of diapause and diapause development in the bean bug, Riptortus clavatus (Heteroptera: Alydidae). Eur. J. Entomol. 2000, 97, 19–23. [Google Scholar] [CrossRef]
- Tan, Q.-Q.; Feng, L.; Liu, W.; Zhu, L.; Lei, C.-L.; Wang, X.-P. Differences in the pre-diapause and pre-oviposition accumulation of critical nutrients in adult females of the beetle Colaphellus bowringi. Entomol. Exp. Appl. 2016, 160, 117–125. [Google Scholar] [CrossRef]
- Jung, M.; Lee, D.H. Characterization of overwintering behaviors and sites of bean bug, Riptortus pedestris (Hemiptera: Alydidae), under laboratory and field conditions. Environ. Entomol. 2018, 47, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Kim, E.; Lim, U.T. Seasonal reproductive performance and pre-diapausing mating status of female Riptortus pedestris (Hemiptera: Alydidae) collected in fields. J. Econ. Entomol. 2018, 111, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Numata, H. Photoperiodic sensitivity after diapause termination in the bean bug, Riptortus clavatus Thunberg (Heteroptera: Alydidae). Appl. Entomol. Zool. 1987, 22, 352–357. [Google Scholar] [CrossRef]
- Numata, H.; Hidaka, T. Photoperiodic control of adult diapause in the bean bug, Riptortus clavatus Thunberg (Heteroptera: Coreidae) I. reversible induction and termination of diapause. Appl. Entomol. Zool. 1982, 17, 530–538. [Google Scholar] [CrossRef]
- Numata, H.; Hidaka, T. Photoperiodic control of adult diapause in the bean bug, Riptortus clavatus Thunberg (Heteroptera: Coreidae): III. diapause development and temperature. Appl. Entomol. Zool. 1984, 19, 356–360. [Google Scholar] [CrossRef]
- Ikeno, T.; Numata, H.; Goto, S.G. Circadian clock genes period and cycle regulate photoperiodic diapause in the bean bug Riptortus pedestris males. J. Insect Physiol. 2011, 57, 935–938. [Google Scholar] [CrossRef]
- Hideharu, N.; Toshitaka, H. Morphological and behavioral character of adult diapause and its termination by a juvenile hormone analogue in Riptortus clavatus. In Proceedings of the Regulation of Insect Reproduction IV; Tonner, M., Soldan, T., Bennettova, B., Eds.; Academia: Zinkovy, Czech Republic, 1987; pp. 401–441. [Google Scholar]
- Chapman, R. The Insects: Structure and Function; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Adams, T.S. Morphology of the internal reproductive system of the male and female two-spotted stink bug, Perillus bioculatus (F.) (Heteroptera: Pentatomidae) and the transfer of products during mating. Invertebr. Reprod. Dev. 2001, 39, 45–53. [Google Scholar] [CrossRef]
- Fantinou, A.A.; Perdikis, D.C.; Zota, K.F. Reproductive responses to photoperiod and temperature by diapausing and nondiapausing populations of Sesamia nonagrioides Lef. (Lepidoptera—Noctuidae). Physiol. Entomol. 2004, 29, 169–175. [Google Scholar] [CrossRef]
- Ojima, N.; Ishiguro, S.; An, Y.; Kadosawa, T.; Suzuki, K. Male reproductive maturity and diapause termination in the leaf beetle Gastrophysa atrocyanea. Physiol. Entomol. 2015, 40, 277–283. [Google Scholar] [CrossRef]
- Wang, X.-P.; Zhou, X.-M.; Wang, Y.-Y.; Lei, C.-L. Internal reproductive system and diapausing morphology of the Brassica leaf beetle Phaedoc brassicae blay (Coleoptera: Chrysomelidae: Chrysomelidae). Coleopt. Bull. 2007, 3, 457–462. [Google Scholar] [CrossRef]
- Kubrak, O.I.; Kucerová, L.; Theopold, U.; Nylin, S.; Nässel, D.R. Characterization of reproductive dormancy in male Drosophila melanogaster. Front. Physiol. 2016, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ruhmann, H.; Wensing, K.U.; Neuhalfen, N.; Specker, J.-H.; Fricke, C. Early reproductive success in Drosophila males is dependent on maturity of the accessory gland. Behav. Ecol. 2016, 27, 1859–1868. [Google Scholar]
- Rodrigues, A.R.S.; Serrão, J.E.; Teixeira, V.W.; Torres, J.B.; Teixeira, A.A. Spermatogenesis, changes in reproductive structures, and time constraint associated with insemination in Podisus nigrispinus. J. Insect Physiol. 2008, 54, 1543–1551. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.; Werneck, H.; Malta, J.; Teixeira, A.; Campos, L.; Serrão, J. Post-embryonic development of the seminal vesicle in the stingless bee Melipona quadrifasciata Lepeletier, 1836 (Apidae: Meliponini). Sociobiology 2019, 66, 287–292. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hafeez, A.; Li, B.; Liu, W.; Atiq, M.N.; Wang, X.-P. Developmental Differences on the Internal Reproductive Systems between the Prediapause and Prereproductive Riptortus pedestris Adults. Insects 2020, 11, 347. https://doi.org/10.3390/insects11060347
Hafeez A, Li B, Liu W, Atiq MN, Wang X-P. Developmental Differences on the Internal Reproductive Systems between the Prediapause and Prereproductive Riptortus pedestris Adults. Insects. 2020; 11(6):347. https://doi.org/10.3390/insects11060347
Chicago/Turabian StyleHafeez, Abdul, Bei Li, Wen Liu, Muhammad Nauman Atiq, and Xiao-Ping Wang. 2020. "Developmental Differences on the Internal Reproductive Systems between the Prediapause and Prereproductive Riptortus pedestris Adults" Insects 11, no. 6: 347. https://doi.org/10.3390/insects11060347
APA StyleHafeez, A., Li, B., Liu, W., Atiq, M. N., & Wang, X.-P. (2020). Developmental Differences on the Internal Reproductive Systems between the Prediapause and Prereproductive Riptortus pedestris Adults. Insects, 11(6), 347. https://doi.org/10.3390/insects11060347





