ImergardTMWP: A Non-Chemical Alternative for an Indoor Residual Spray, Effective against Pyrethroid-Resistant Anopheles gambiae (s.l.) in Africa
Abstract
1. Introduction
2. Materials and Methods
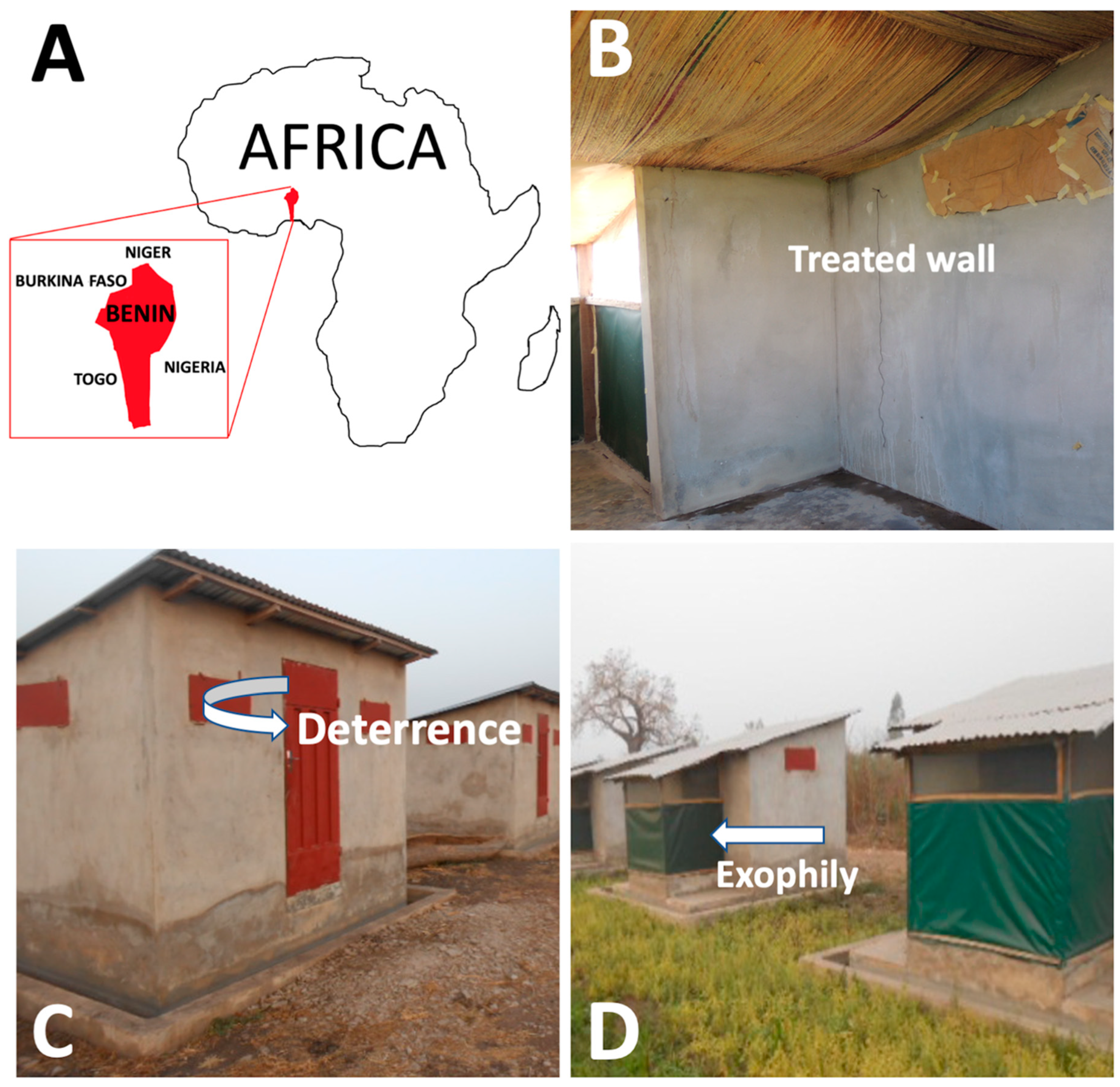
2.1. Study Site and Experimental Huts
2.2. Insecticide Treatments
2.3. Sleepers and Mosquito Collection
2.4. Residual Activity of Insecticide Treatments
2.5. Statistical Analyses
2.6. Ethical Clearance
2.7. Abbreviations
2.8. Data Availabity
3. Results
3.1. Deterrence
3.2. Exophily
3.3. Blood Feeding
3.4. Toxicity
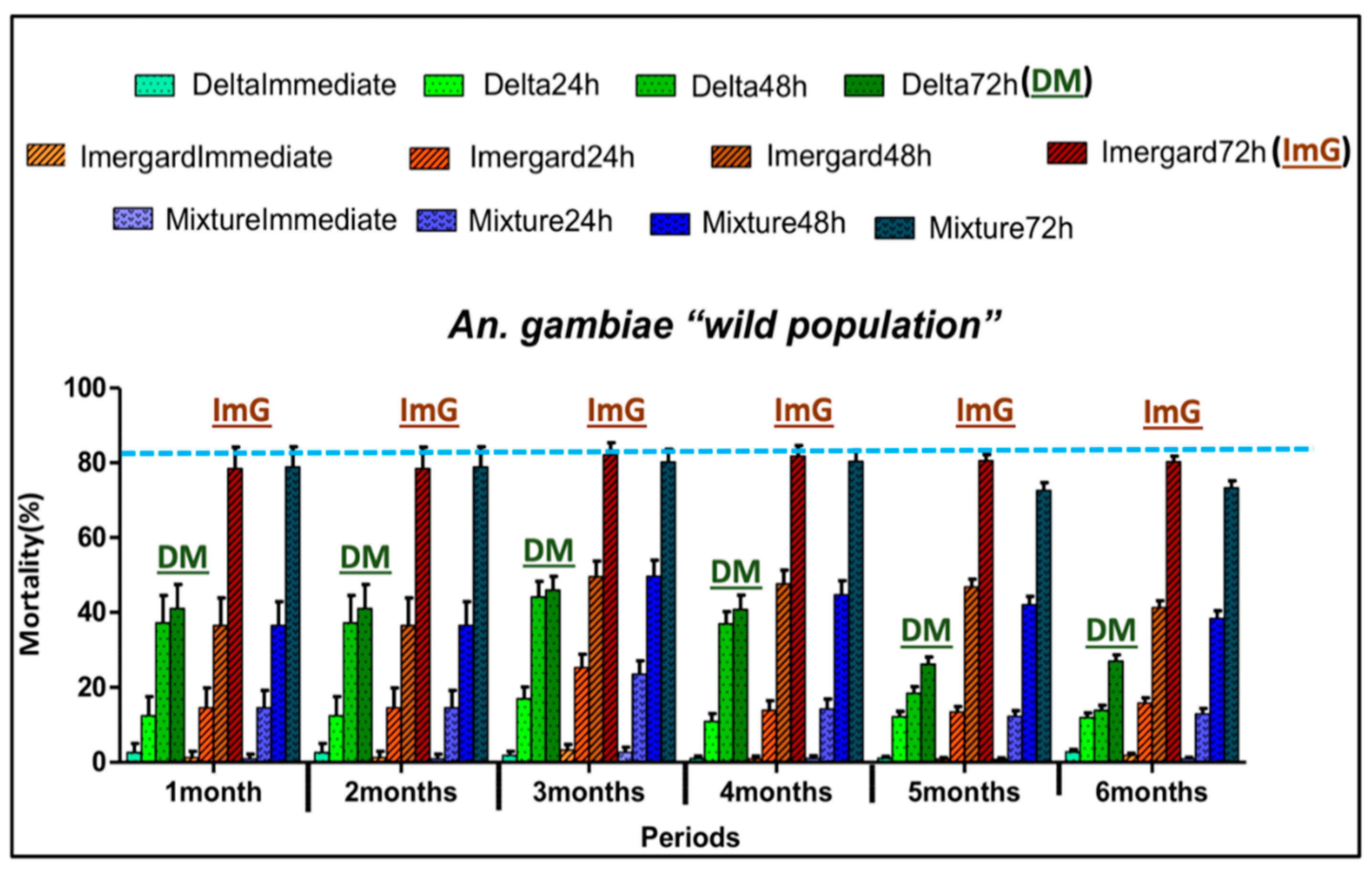
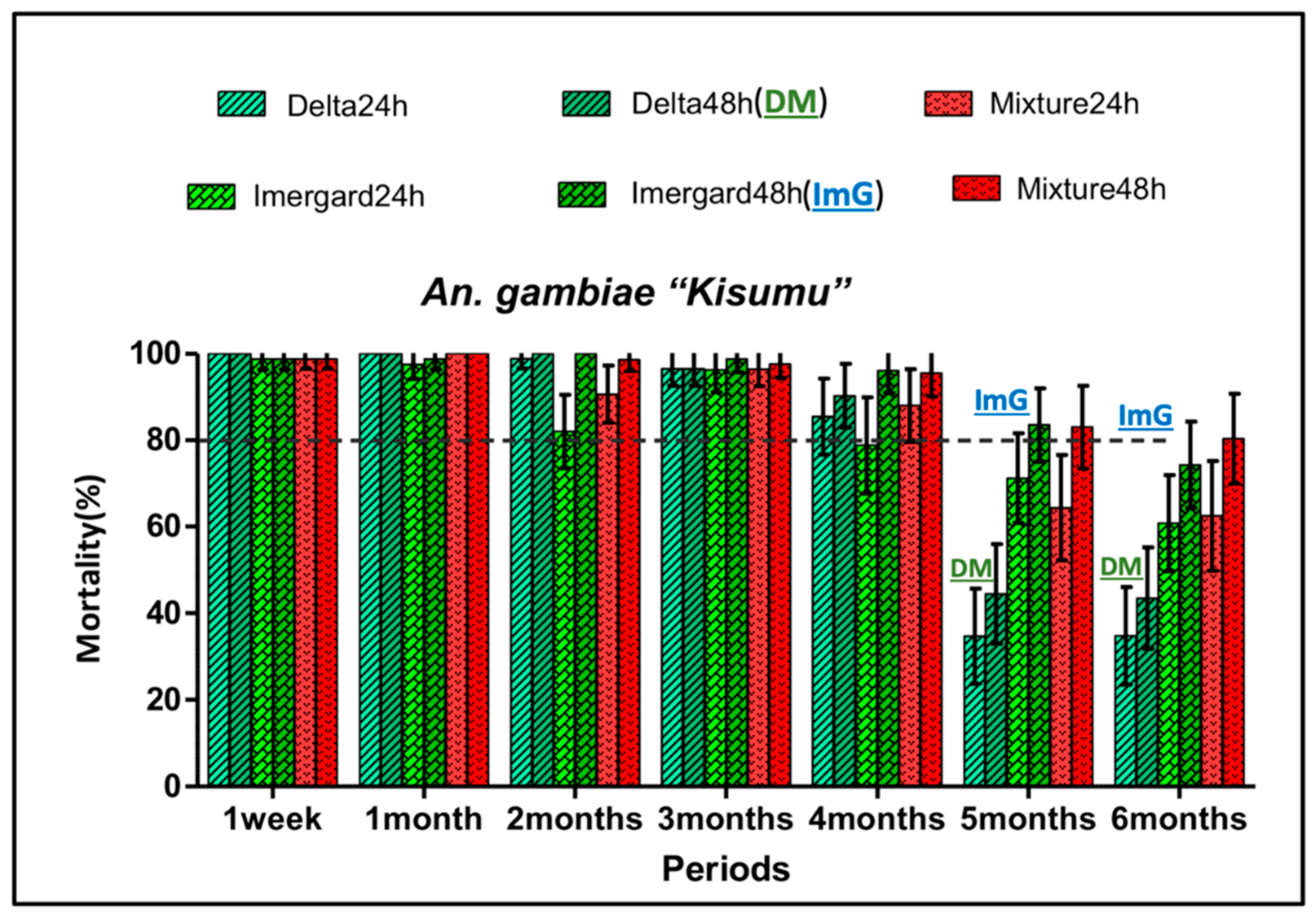
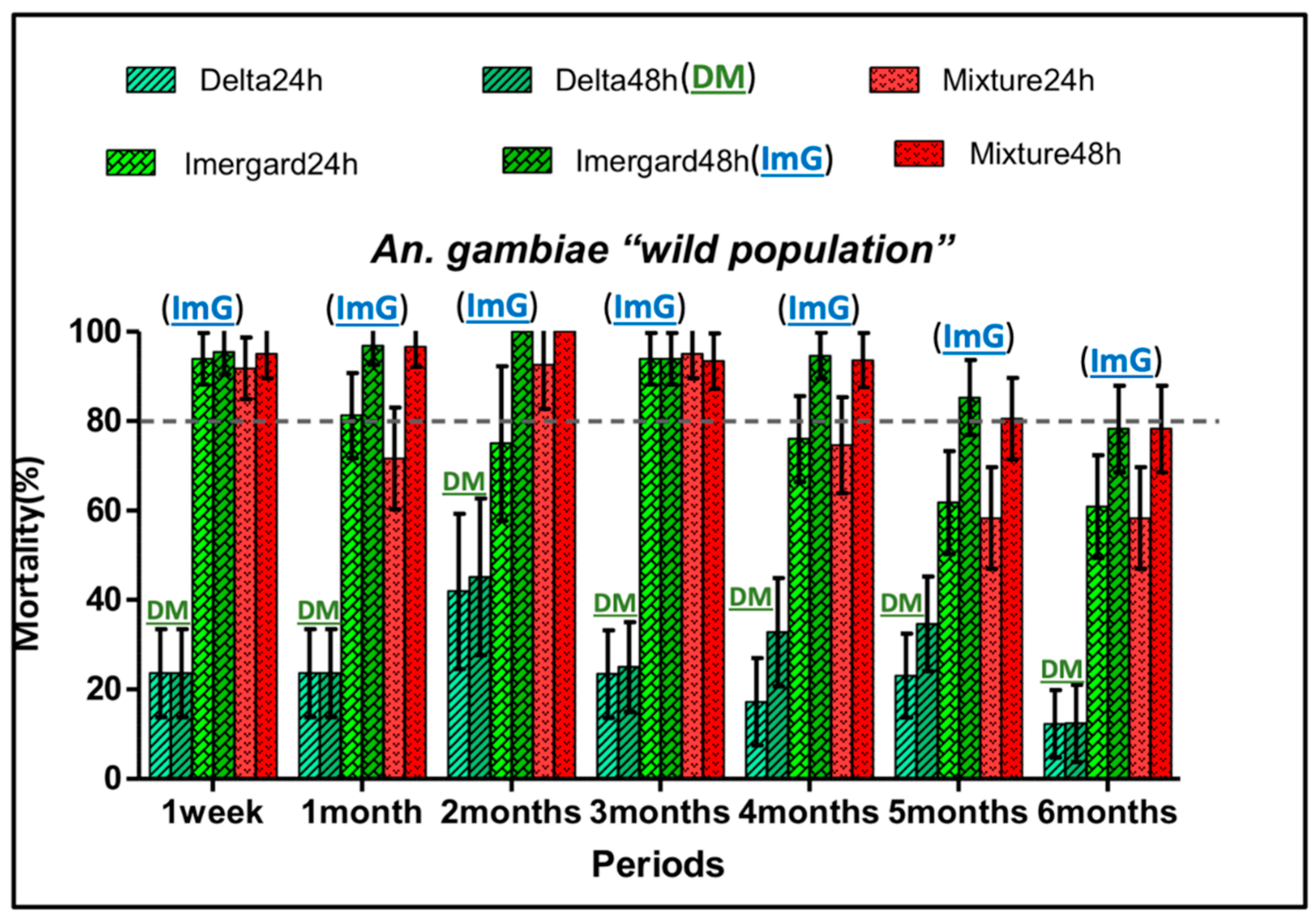
3.5. Insecticide Residual Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. World Malaria Report 2019; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-156572-1. [Google Scholar]
- Mashauri, F.M.; Manjurano, A.; Kinung’hi, S.; Martine, J.; Lyimo, E.; Kishamawe, C.; Ndege, C.; Ramsan, M.M.; Chan, A.; Mwalimu, C.D.; et al. Indoor residual spraying with micro-encapsulated pirimiphos-methyl (Actellic® 300CS) against malaria vectors in the Lake Victoria basin, Tanzania. PLoS ONE 2017, 12, e0176982. [Google Scholar] [CrossRef]
- Mnzava, A.P.; Knox, T.B.; Temu, E.A.; Trett, A.; Fornadel, C.; Hemingway, J.; Renshaw, M. Implementation of the global plan for insecticide resistance management in malaria vectors: Progress, challenges and the way forward. Malar. J. 2015, 14, 173. [Google Scholar] [CrossRef] [PubMed]
- Kitau, J.; Oxborough, R.; Matowo, J.; Mosha, F.; Magesa, S.M.; Rowland, M. Indoor residual spraying with microencapsulated DEET repellent (N, N-diethyl-m-toluamide) for control of Anopheles arabiensis and Culex quinquefasciatus. Parasit. Vectors 2014, 7, 446. [Google Scholar] [CrossRef] [PubMed]
- Dengela, D.; Seyoum, A.; Lucas, B.; Johns, B.; George, K.; Belemvire, A.; Caranci, A.; Norris, L.C.; Fornadel, M. Multi-country assessment of residual bio-efficacy of insecticides used for indoor residual spraying in malaria control on different surface types: Results from program monitoring in 17 PMI/USAID-supported IRS countries. Parasit. Vectors 2018, 11, 71. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Plan for Insecticide Resistance Management in Malaria Vectors (GPIRM); World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Agossa, F.R.; Gnanguenon, V.; Anagonou, R.; Azondékon, R.; Aïzoun, N.; Sovi, A.; Oke-Agbo, F.; Sèzonlin, M.; Akogbéto, M.C. Impact of insecticide resistance on the effectiveness of pyrethroid-based malaria vectors control tools in Benin: Decreased toxicity and repellent effect. PLoS ONE 2015, 10, e0145207. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.L.; Pinder, M.; Bradley, J.; Donnelly, M.J.; Hamid-Adiamoh, M.; Jarju, L.B.; Jawara, M.; Jeffries, D.; Kandeh, B.; Rippon, E.J.; et al. Emergence of knock-down resistance in the Anopheles gambiae complex in the Upper River Region, The Gambia, and its relationship with malaria infection in children. Malar. J. 2018, 17, 205. [Google Scholar] [CrossRef]
- Aïkpon, R.; Sèzonlin, M.; Ossè, R.; Akogbéto, M. Evidence of multiple mechanisms providing carbamate and organophosphate resistance in field An. gambiae population from Atacora in Benin. Parasit. Vectors 2014, 7, 568. [Google Scholar] [CrossRef]
- Hemingway, J.; Ranson, H. Insecticide resistance in insect vectors of human disease. Annu. Rev. Entomol. 2000, 45, 371–391. [Google Scholar] [CrossRef]
- Soko, W.; Chimbari, M.J.; Mukaratirwa, S. Insecticide resistance in malaria-transmitting mosquitoes in Zimbabwe: A review. Infect. Dis. Poverty 2015, 4, 46. [Google Scholar] [CrossRef]
- Athanassiou, C.G.; Vayias, B.J.; Dimizas, C.B.; Kavallieratos, N.G.; Papagregoriou, A.S.; Buchelos, C.T. Insecticidal efficacy of diatomaceous earth against Sitophilus oryzae (L.) (Coleoptera: Curculionidae) and Tribolium confusum du Val (Coleoptera: Tenebrionidae) on stored wheat: Influence of dose rate, temperature and exposure interval. J. Stored Prod. Res. 2005, 41, 47–55. [Google Scholar] [CrossRef]
- El-Aziz, S.E.A. Laboratory and field Evaluation of Kaolin and Bentonite particle films against onion thrips, Thrips tabaci (Lind.) (Thysanoptera: Thripidae) on onion plants. J. Appl. Sci. Res. 2013, 9, 3141–3145. [Google Scholar]
- Alavo, T.B.; Yarou, B.B.; Atachi, P. Field effects of kaolin particle film formulation against major cotton lepidopteran pests in North Benin, West Africa. Int. J. Pest. Manag. 2010, 56, 287–290. [Google Scholar] [CrossRef]
- Saour, G. Efficacy of kaolin particle film and selected synthetic insecticides against pistachio psyllid Agonoscena targionii (Homoptera: Psyllidae) infestation. Crop. Prot. 2005, 24, 711–717. [Google Scholar] [CrossRef]
- Islam, M.S.; Rahman, M.M. Diatomaceous earth induced alterations in the reproductive attributes in the housefly Musca domestica L. (Diptera: Culicidae). Elixir Appl. Zool. 2016, 96, 41241–41244. [Google Scholar]
- Luz, C.; Rodrigues, J.; Rocha, L.F. Diatomaceous earth and oil enhance effectiveness of Metarhizium anisopliae against Triatoma infestans. Acta Trop. 2012, 122, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Maxim, L.D.; Niebo, R.; McConnell, E.E. Perlite toxicology and epidemiology—A review. Inhal. Toxicol. 2014, 26, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Korunic, Z. Diatomaceous Earths, a group of natural insecticides. J. Stored Prod. Res. 1998, 34, 87–97. [Google Scholar] [CrossRef]
- Ngufor, C.; Fongnikin, A.; Rowland, M.; N’Guessan, R. Indoor residual spraying with a mixture of clothianidin (a neonicotinoid insecticide) and deltamethrin provides improved control and long residual activity against pyrethroid resistant Anopheles gambiae sl in Southern Benin. PLoS ONE 2017, 12, e0189575. [Google Scholar] [CrossRef]
- Ngufor, C.; Critchley, J.; Fagbohoun, J.; N’Guessan, R.; Todjinou, D.; Rowland, M. Chlorfenapyr (a pyrrole insecticide) applied alone or as a mixture with alpha-cypermethrin for indoor residual spraying against pyrethroid resistant Anopheles gambiae sl: An experimental hut study in Cove, Benin. PLoS ONE 2016, 11, e0162210. [Google Scholar] [CrossRef]
- Agossa, F.R.; Padonou, G.G.; Koukpo, C.Z.; Zola-Sahossi, J.; Azondekon, R.; Akuoko, O.K.; Ahoga, J.; N’dombidje, B.; Akinro, B.; Jacques, A.; et al. Efficacy of a novel mode of action of an indoor residual spraying product, SumiShield® 50WG against susceptible and resistant populations of Anopheles gambiae (s.l.) in Benin, West Africa. Parasit. Vectors 2018, 11, 293. [Google Scholar] [CrossRef]
- WHO. Guidelines for Testing Mosquito Adulticides for Indoor Residual Spraying and Treatment of Mosquito Nets; World Health Organization: Geneva Switzerland, 2006. [Google Scholar]
- Hougard, J.M.; Martin, T.; Guillet, P.F.; Coosemans, M.; Itoh, T.; Akogbeto, M.; Chandre, F. Preliminary field testing of a long-lasting insecticide-treated hammock against Anopheles gambiae and Mansonia spp. (Diptera: Culicidae) in West Africa. J. Med. Entomol. 2007, 44, 651–655. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gillies, M.T.; De Meillon, B. The Anophelinae of Africa south of the Sahara. S. Afr. Inst. Med. Res. 1968, 54, 1–343. [Google Scholar]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- R Development Core Team. R: A language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Bhatt, S.; Weiss, D.J.; Cameron, E.; Bisanzio, D.; Mappin, B.; Dalrymple, U.; Battle, K.E.; Moyes, C.L.; Henry, A.; Eckhoff, P.A.; et al. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature 2015, 526, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Sherrard-Smith, E.; Griffin, J.T.; Winskill, P.; Corbel, V.; Pennetier, C.; Djenontin, A.; Moore, S.; Richardson, J.H.; Müller, P.; Edi, C.; et al. Systematic review of indoor residual spray efficacy and effectiveness against Plasmodium falciparum in Africa. Nat. Commun. 2018, 9, 4982. [Google Scholar] [CrossRef] [PubMed]
- Akogbeto, M.; Padonou, G.G.; Bankole, H.S.; Gazard, D.K.; Gbedjissi, G.L. Dramatic decrease in malaria transmission after large-scale indoor residual spraying with Bendiocarb in Benin, an area of high resistance of Anopheles gambiae to pyrethroids. Am. J. Trop. Med. Hyg. 2011, 85, 586–593. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aïkpon, R.; Aïzoun, N.; Sovi, A.; Oussou, O.; Govoetchan, R.; Gnaguenon, V.; Oke-Agbo, F.; Osse, R.; Akogbeto, M. Increase of Ace-1 resistance allele in the field population of Anopheles gambiae following a large-scale indoor spraying (IRS) implementation using bendiocarb in Atacora region in Benin, West Africa. J. Cell Anim. Biol. 2014, 8, 15–22. [Google Scholar]
- Oduola, A.O.; Olejebe, J.B.; Ashiegbu, C.O.; Adeogun, A.O.; Otubanjo, O.A.; Awolola, T.S. High level of DDT resistance in the malaria mosquito: Anopheles gambiae s.l. from rural, semi urban and urban communities in Nigeria. J. Rural. Trop. Pub. Health 2010, 9, 114–120. [Google Scholar]
- Ranson, H.; Jensen, B.; Vulule, J.M.; Wang, X.; Hemingway, J.; Collins, F.H. Identification of a point mutation in the voltage-gated sodium channel gene of Kenyan Anopheles gambiae s.s. associated with resistance to DDT and pyrethroids. Ins. Mol. Biol. 2000, 9, 491–497. [Google Scholar] [CrossRef]
- Murray, J.; Eskenazi, B.; Bornman, R.; Gaspar, F.W.; Crause, M.; Obida, M.; Chevrier, J. Exposure to DDT and hypertensive disorders of pregnancy among South African women from an indoor residual spraying region: The VHEMBE study. Environ. Res. 2018, 162, 49–54. [Google Scholar] [CrossRef]
- Eskenazi, B.; An, S.; Rauch, S.A.; Coker, E.S.; Maphula, A.; Obida, M.; Crause, M.; Kogut, K.R.; Bornman, R.; Chevier, J. Prenatal exposure to DDT and pyrethroids for malaria control and child neurodevelopment: The VHEMBE cohort, South Africa. Environ. Health Perspect. 2018, 126, 047004. [Google Scholar] [CrossRef] [PubMed]
- Pan, I.J.; Daniels, J.L.; Goldman, B.D.; Herring, A.H.; Siega-Riz, A.M.; Rogan, W.J. Lactational exposure to polychlorinated biphenyls, dichlorodiphenyltrichloroethane, and dichlorodiphenyldichloroethylene and infant neurodevelopment: An analysis of the pregnancy, infection, and nutrition babies study. Environ. Health Perspect. 2009, 117, 488–494. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gaspar, F.W.; Harley, K.G.; Kogut, K.; Chevrier, J.; Mora, A.M.; Sjodin, A.; Eskenazi, B. Prenatal DDT and DDE exposure and child IQ in the CHAMACOS cohort. Environ. Int. 2015, 85, 206–212. [Google Scholar] [CrossRef]
- Deguenon, J.M.; Riegel, C.; Cloherty-Duvernay, E.R.; Stewart, D.A.; Wand, B.; Gittins, D.; Tihomirov, L.; Apperson, C.S.; McCord, M.G.; Roe, R.M. New mosquitocide derived from volcanic rock. J. Med. Entomol. under review.
- Agossa, F.R.; Aïkpon, R.; Azondekon, R.; Govoetchan, R.; Padonou, G.G.; Oussou, O.; Oke-Agbo, F.; Akogbeto, M.C.A. Efficacy of various insecticides recommended for indoor residual spraying: Pirimiphos methyl, potential alternative to bendiocarb for pyrethroid resistance management in Benin, West Africa. Trans. R. Soc. Trop. Med. Hyg. 2014, 108, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Akogbeto, M.C.; Padonou, G.G.; Gbenou, D.; Irish, S.; Yadouleton, A. Bendiocarb, a potential alternative against pyrethroid resistant Anopheles gambiae in Benin, West Africa. Malar. J. 2010, 9, 204. [Google Scholar] [CrossRef] [PubMed]
- Ebeling, W.; Wagner, R.E. Rapid desiccation of drywood termites with inert sorptive dusts and other substances. J. Econ. Entomol. 1959, 52, 190–207. [Google Scholar] [CrossRef]
- Lockey, K.H. Lipids of the insect cuticle: Origin, composition and function. Comp. Biochem. Physiol. Part B Comp. Biochem. 1988, 89, 595–645. [Google Scholar] [CrossRef]
- Nayar, J.K.; Sauerman, D.M., Jr. The effects of nutrition on survival and fecundity in Florida mosquitoes: Part 1. Utilization of sugar for survival. J. Med. Entomol. 1975, 12, 92–98. [Google Scholar] [CrossRef]
- Nayar, J.K.; Sauerman, D.M., Jr. The effects of nutrition on survival and fecundity in Florida mosquitoes: Part 2. Utilization of a blood meal for survival. J. Med. Entomol. 1975, 12, 99–103. [Google Scholar] [CrossRef]
- Gary, R.E., Jr.; Woodbridge, A.F. Effects of available sugar on the reproductive fitness and vectorial capacity of the malaria vector Anopheles gambiae (Diptera: Culicidae). J. Med. Entomol. 2001, 36, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, P.; Robert, V. Les Anophèles: Biologie, Transmission du Plasmodium et Lutte Antivectorielle; IRD éditions: Marseille, France, 2009; p. 402. [Google Scholar]
- Rúa, G.L.; Quiñones, M.L.; Vélez, I.D.; Zuluaga, J.S.; Rojas, W.; Poveda, G.; Ruiz, D. Laboratory estimation of the effects of increasing temperatures on the duration of gonotrophic cycle of Anopheles albimanus (Diptera: Culicidae). Mem. Inst. Oswaldo Cruz. 2005, 100, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Dana, A.N.; Hong, Y.S.; Kern, M.K.; Hillenmeyer, M.E.; Harker, B.W.; Lobo, N.F.; Hogan, J.R.; Romans, P.; Collins, F.H. Gene expression patterns associated with blood-feeding in the malaria mosquito Anopheles gambiae. BMC Genom. 2005, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Lardeux, F.J.; Tejerina, R.H.; Quispe, V.; Chavez, T.K. A physiological time analysis of the duration of the gonotrophic cycle of Anopheles pseudopunctipennis and its implications for malaria transmission in Bolivia. Malar. J. 2008, 7, 141. [Google Scholar] [CrossRef] [PubMed]
- Djenontin, A.; Chandre, F.; Dabiré, K.R.; Chabi, J.; N’Guessan, R.; Baldet, T.; Akogbeto, M.; Corbel, V. Indoor use of plastic sheeting impregnated with carbamate combined with long-lasting insecticidal mosquito nets for the control of pyrethroid-resistant malaria vectors. Am. J. Trop. Med. Hyg. 2010, 83, 266–270. [Google Scholar] [CrossRef]
- Oxborough, R.M.; Kitau, J.; Jones, R.; Mosha, F.W.; Rowland, M.W. Experimental hut and bioassay evaluation of the residual activity of a polymer-enhanced suspension concentrate (SC-PE) formulation of deltamethrin for IRS use in the control of Anopheles arabiensis. Parasit. Vectors 2014, 7, 454. [Google Scholar] [CrossRef]
- Dunford, J.C.; Estep, A.S.; Waits, C.M.; Richardson, A.G.; Hoel, D.F.; Horn, K.; Walker, T.W.; Blersch, J.S.; Kerce, J.D.; Wirtz, R.A. Evaluation of the long-term efficacy of K-Othrine® PolyZone on three surfaces against laboratory reared Anopheles gambiae in semi-field conditions. Malar. J. 2018, 17, 94. [Google Scholar] [CrossRef]

| Treatment | Months | Total a | Proportion (%) b | 95% CI c | p-Value d |
|---|---|---|---|---|---|
| Control | 1 | 785 | |||
| 2 | 331 | ||||
| 3 | 592 | ||||
| 4 | 796 | ||||
| 5 | 2084 | ||||
| 6 | 2355 | ||||
| Deltamethrin | 1 | 716 | 8.8 | 6.7–10.9 | <0.0001 |
| 2 | 203 | 38.7 | 33.1–44.2 | <0.0001 | |
| 3 | 542 | 8.4 | 6.0–10.8 | <0.0001 | |
| 4 | 838 | - | - | >0.05 | |
| 5 | 1918 | 8.0 | 6.8–9.2 | <0.0001 | |
| 6 | 2539 | - | - | >0.05 | |
| ImergardTMWP | 1 | 645 | 17.8 | 15.0–20.6 | <0.0001 |
| 2 | 204 | 38.4 | 32.8–43.9 | <0.0001 | |
| 3 | 556 | 6.1 | 4.0–8.2 | <0.0001 | |
| 4 | 719 | 9.7 | 7.5–11.8 | <0.0001 | |
| 5 | 2098 | - | - | >0.05 | |
| 6 | 2658 | - | - | >0.05 | |
| DeltaM + ImGe | 1 | 587 | 25.2 | 22.1–28.4 | <0.0001 |
| 2 | 257 | 22.4 | 17.6–27.2 | <0.0001 | |
| 3 | 527 | 11.0 | 8.3–13.7 | <0.0001 | |
| 4 | 678 | 14.8 | 12.2–17.4 | <0.0001 | |
| 5 | 1760 | 15.6 | 13.9–17.2 | <0.0001 | |
| 6 | 2163 | 8.2 | 7.0–9.3 | <0.0001 |
| Treatment | Months | Total b | Proportion (%) a | 95% CI c | p-Value d |
|---|---|---|---|---|---|
| Control | 1 | 785 | 26.9 | 23.8–30.1 | - |
| 2 | 331 | 26.3 | 21.6–31.4 | - | |
| 3 | 592 | 31.6 | 27.9–35.5 | - | |
| 4 | 796 | 30.2 | 33.5–37.0 | - | |
| 5 | 2084 | 34.7 | 32.6–36.8 | - | |
| 6 | 2355 | 33.8 | 31.9–35.8 | - | |
| Deltamethrin | 1 | 716 | 53.2 | 49.5–56.9 | <0.0001 |
| 2 | 203 | 56.2 | 49.0–63.1 | <0.0001 | |
| 3 | 542 | 53.7 | 49.4–58.0 | <0.0001 | |
| 4 | 838 | 56.4 | 53.0–59.8 | <0.0001 | |
| 5 | 1918 | 59.8 | 57.6–62.0 | <0.0001 | |
| 6 | 2539 | 57.1 | 55.1–59.0 | <0.0001 | |
| Imergard | 1 | 645 | 31.0 | 27.4–34.7 | 0.097 |
| 2 | 204 | 34.8 | 28.1–41.6 | 0.045 | |
| 3 | 556 | 31.3 | 27.5–35.3 | 0.966 | |
| 4 | 719 | 33.2 | 29.8–36.8 | 0.216 | |
| 5 | 2098 | 33.6 | 31.5–35.6 | 0.458 | |
| 6 | 2658 | 33.5 | 31.7–35.3 | 0.858 | |
| DeltaM+ ImG e | 1 | 587 | 48.2 | 44.1–52.3 | <0.0001 |
| 2 | 257 | 48.6 | 42.4–54.9 | <0.0001 | |
| 3 | 527 | 48.4 | 44.0–52.8 | <0.0001 | |
| 4 | 678 | 48.7 | 44.8–52.5 | <0.0001 | |
| 5 | 1760 | 49.9 | 47.2–52.0 | <0.0001 | |
| 6 | 2163 | 50.2 | 48.0–52.3 | <0.0001 |
| Treatment | Months | Total b | Proportion (%) a | 95% CI c | p-Value d |
|---|---|---|---|---|---|
| Control | 1 | 785 | 94.3 | 92.4–95.8 | - |
| 2 | 331 | 95.5 | 92.6–97.6 | - | |
| 3 | 592 | 92.4 | 90.0–94.4 | - | |
| 4 | 796 | 95.5 | 93.8–96.8 | - | |
| 5 | 2084 | 97.4 | 96.6–98.0 | - | |
| 6 | 2355 | 96.1 | 95.3–96.9 | - | |
| Deltamethrin | 1 | 716 | 94.8 | 92.9–96.3 | 0.713 |
| 2 | 203 | 97.0 | 93.0–99.0 | 0.496 | |
| 3 | 542 | 95.9 | 93.9–97.4 | 0.016 | |
| 4 | 838 | 96.7 | 95.2–97.8 | 0.270 | |
| 5 | 1918 | 94.2 | 93.0–95.2 | <0.0001 | |
| 6 | 2539 | 92.2 | 91.1–93.2 | <0.0001 | |
| Imergard | 1 | 645 | 89.3 | 86.6–91.6 | 0.0008 |
| 2 | 204 | 98.0 | 94.9–99.6 | 0.186 | |
| 3 | 556 | 93.4 | 90.9–95.3 | 0.612 | |
| 4 | 719 | 95.3 | 93.4–96.7 | 0.956 | |
| 5 | 2098 | 95.5 | 94.5–96.3 | 0.0013 | |
| 6 | 2658 | 91.0 | 90.4–92.5 | <0.0001 | |
| DeltaM+ ImG e | 1 | 587 | 94.2 | 92.0–96.0 | >0.05 |
| 2 | 257 | 98.1 | 95.4–99. 5 | 0.137 | |
| 3 | 527 | 92.2 | 89.6–94.4 | >0.05 | |
| 4 | 678 | 94.2 | 92.2–95.9 | 0.341 | |
| 5 | 1760 | 96.8 | 95.8–97.5 | 0.315 | |
| 6 | 2163 | 95.9 | 95.0–96.7 | 0.723 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deguenon, J.M.; Azondekon, R.; Agossa, F.R.; Padonou, G.G.; Anagonou, R.; Ahoga, J.; N’dombidje, B.; Akinro, B.; Stewart, D.A.; Wang, B.; et al. ImergardTMWP: A Non-Chemical Alternative for an Indoor Residual Spray, Effective against Pyrethroid-Resistant Anopheles gambiae (s.l.) in Africa. Insects 2020, 11, 322. https://doi.org/10.3390/insects11050322
Deguenon JM, Azondekon R, Agossa FR, Padonou GG, Anagonou R, Ahoga J, N’dombidje B, Akinro B, Stewart DA, Wang B, et al. ImergardTMWP: A Non-Chemical Alternative for an Indoor Residual Spray, Effective against Pyrethroid-Resistant Anopheles gambiae (s.l.) in Africa. Insects. 2020; 11(5):322. https://doi.org/10.3390/insects11050322
Chicago/Turabian StyleDeguenon, Jean M., Roseric Azondekon, Fiacre R. Agossa, Gil G. Padonou, Rodrigue Anagonou, Juniace Ahoga, Boris N’dombidje, Bruno Akinro, David A. Stewart, Bo Wang, and et al. 2020. "ImergardTMWP: A Non-Chemical Alternative for an Indoor Residual Spray, Effective against Pyrethroid-Resistant Anopheles gambiae (s.l.) in Africa" Insects 11, no. 5: 322. https://doi.org/10.3390/insects11050322
APA StyleDeguenon, J. M., Azondekon, R., Agossa, F. R., Padonou, G. G., Anagonou, R., Ahoga, J., N’dombidje, B., Akinro, B., Stewart, D. A., Wang, B., Gittins, D., Tihomirov, L., Apperson, C. S., McCord, M. G., Akogbeto, M. C., & Roe, R. M. (2020). ImergardTMWP: A Non-Chemical Alternative for an Indoor Residual Spray, Effective against Pyrethroid-Resistant Anopheles gambiae (s.l.) in Africa. Insects, 11(5), 322. https://doi.org/10.3390/insects11050322





