Spatial Distribution and Flight Patterns of Two Grain Storage Insect Pests, Rhyzopertha dominica (Bostrichidae) and Tribolium castaneum (Tenebrionidae): Implications for Pest Management
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
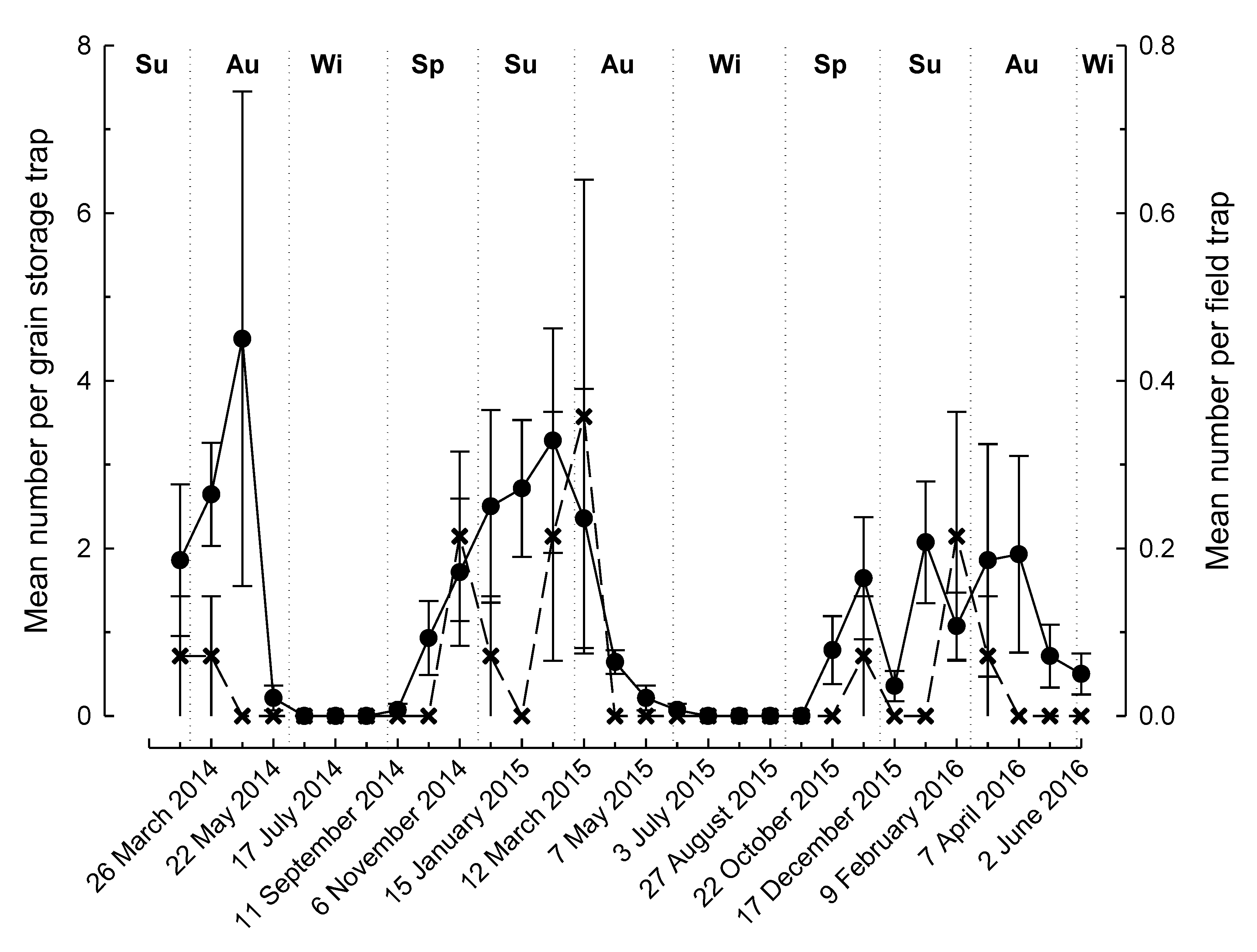
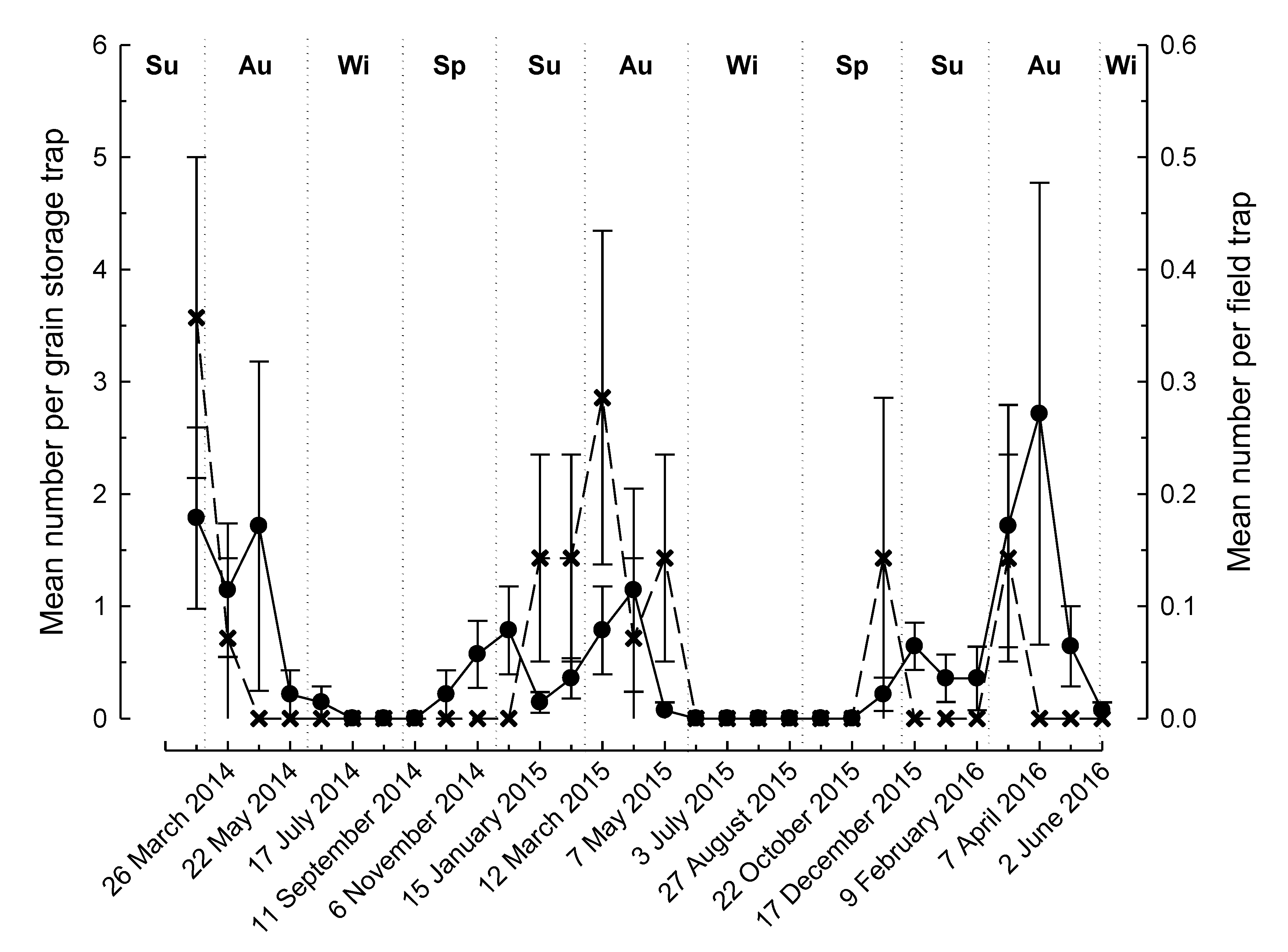
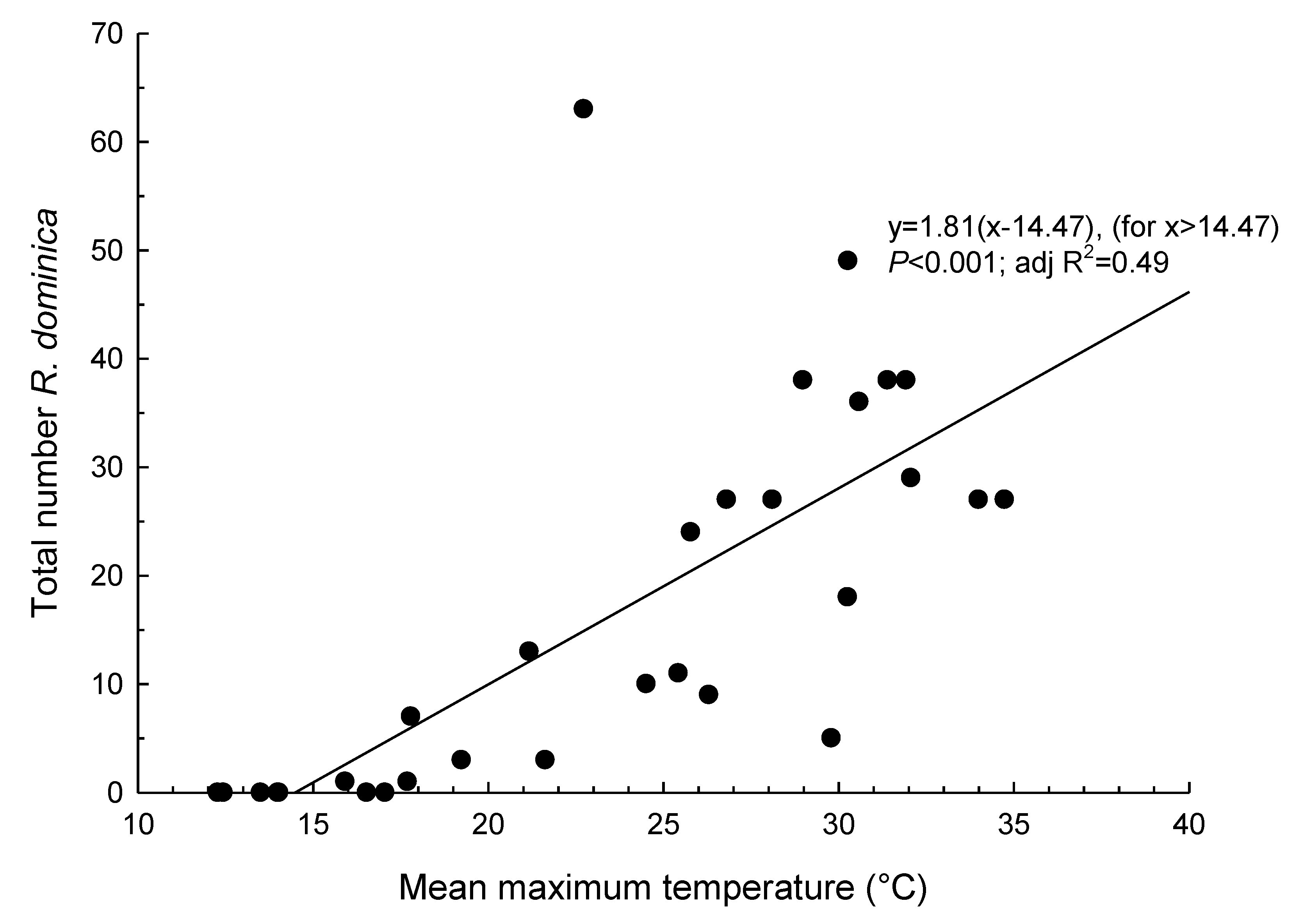
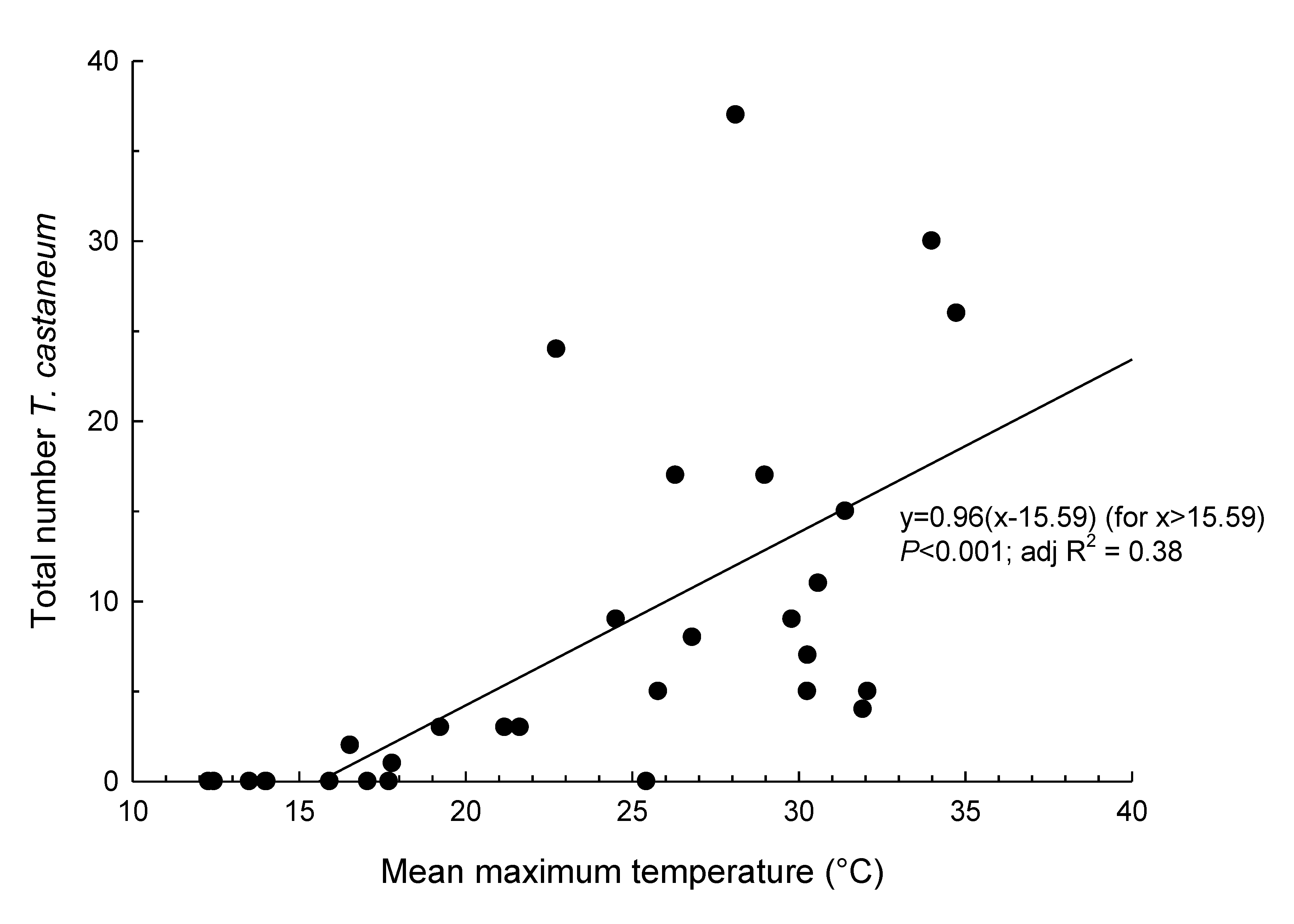
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holloway, J.C.; Falk, M.G.; Emery, R.N.; Collins, P.J.; Nayak, M.K. Resistance to phosphine in Sitophilus oryzae in Australia: A national analysis of trends and frequencies over time and geographical spread. J. Stored Prod. Res. 2016, 69, 129–137. [Google Scholar] [CrossRef]
- Collins, P.J.; Falk, M.G.; Nayak, M.K.; Emery, R.N.; Holloway, J.C. Monitoring resistance to phosphine in the lesser grain borer, Rhyzopertha dominica, in Australia: A national analysis of trends, storage types and geography in relation to resistance detections. J. Stored Prod. Res. 2017, 70, 25–36. [Google Scholar] [CrossRef]
- Daglish, G.J.; Holloway, J.C.; Nayak, M.K. Implications of methoprene resistance for managing Rhyzopertha dominica (F.) in stored grain. J. Stored Prod. Res. 2013, 54, 8–12. [Google Scholar] [CrossRef]
- Nayak, M.K.; Falk, M.G.; Emery, R.N.; Collins, P.J.; Holloway, J.C. An analysis of trends, frequencies and factors influencing the development of resistance to phosphine in the red flour beetle Tribolium castaneum (Herbst) in Australia. J. Stored Prod. Res. 2017, 72, 35–48. [Google Scholar] [CrossRef]
- Nayak, M.K.; Holloway, J.C.; Emery, R.N.; Pavic, H.; Bartlet, J.; Collins, P.J. Strong resistance to phosphine in the rusty grain beetle Cryptolestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae): Its characterisation, a rapid assay for diagnosis and its distribution in Australia. Pest Manag. Sci. 2013, 69, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Lorini, I.; Galley, D.J. Deltamethrin resistance in Rhyzopertha dominica (F.) (Coleoptera: Bostrichidae), a pest of stored grain in Brazil. J. Stored Prod. Res. 1999, 35, 37–45. [Google Scholar] [CrossRef]
- Astuti, L.P.; Mudjiono, G.; Rasminah, S.; Rahardjo, B.T. Susceptibility of milled rice varieties to the lesser grain borer (Rhyzopertha dominica, F). J. Agric. Sci. 2013, 5, 145–149. [Google Scholar] [CrossRef]
- Sánchez-Mariñez, R.I.; Cortez-Rocha, M.O.; Ortega-Dorame, F.; Morales-Valdes, M.; Silveira, M.I. End-use quality of flour from Rhyzopertha dominica infested wheat. Cereal Chem. 1997, 74, 481–483. [Google Scholar] [CrossRef]
- Abdullahi, G.; Muhamad, R.; Dzolkhifli, O.; Sinniah, U.R. Damage potential of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae) on cocoa beans: Effect of initial adult population density and post infestation storage time. J. Stored Prod. Res. 2018, 75, 1–9. [Google Scholar] [CrossRef]
- Javed, M.; Majeed, M.Z.; Khaliq, A.; Arshad, M.; Ahmad, M.H.; Sufyan, M. Quantitative losses in some advanced genotypes of barley incurred by Tribolium castaneum L. (Herbst.). Int. J. Agron. Agric. Res. 2016, 8, 45–50. [Google Scholar]
- Alexander, C.; Kenkel, P. Economics of commodity storage. In Stored Product Protection; Hagstrum, D.W., Phillips, T.W., Cuperus, G., Eds.; Kansas State University: Manhattan, KS, USA, 2012; pp. 305–316. [Google Scholar]
- Jagadeesan, R.; Collins, P.J.; Daglish, G.J.; Ebert, P.R.; Schlipalius, D.I. Phosphine resistance in the rust red flour beetle, Tribolium castaneum (Coleoptera: Tenebrionidae): Inheritance, gene interactions and fitness costs. PLoS ONE 2012, 7, e31582. [Google Scholar] [CrossRef] [PubMed]
- Opit, G.P.; Phillips, T.W.; Aikins, M.J.; Hasan, M.M. Phosphine resistance in Tribolium castaneum and Rhyzopertha dominica from stored wheat in Oklahoma. J. Econ. Entomol. 2012, 105, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Collins, P.J. A new resistance to pyrethroids in Tribolium castaneum (Herbst). Pestic. Sci. 1990, 28, 101–115. [Google Scholar] [CrossRef]
- Daglish, G.J.; Ridley, A.W.; Reid, R.; Walter, G.H. Testing the consistency of spatio-temporal patterns of flight activity in the stored grain beetles Tribolium castaneum (Herbst) and Rhyzopertha dominica (F.). J. Stored Prod. Res. 2017, 72, 68–74. [Google Scholar] [CrossRef]
- Ridley, A.W.; Hereward, J.P.; Daglish, G.J.; Raghu, S.; Collins, P.J. The spatiotemporal dynamics of Tribolium castaneum (Herbst): Adult flight and gene flow. Mol. Ecol. 2011, 20, 1635–1646. [Google Scholar] [CrossRef]
- Ridley, A.W.; Hereward, J.P.; Daglish, G.J.; Raghu, S.; McCulloch, G.A.; Walter, G.H. Flight of Rhyzopertha dominica (Coleoptera: Bostrichidae)—A spatio-temporal analysis with pheromone trapping and population genetics. J. Econ. Entomol. 2016, 109, 2561–2571. [Google Scholar] [CrossRef]
- Gurdasani, K.; Rafter, M.A.; Daglish, G.J.; Walter, G.H. The dispersal flight of Tribolium castaneum—A field test of laboratory generated predictions. J. Stored Prod. Res. 2019, 83, 25–33. [Google Scholar] [CrossRef]
- Semeao, A.A.; Campbell, J.F.; Hutchinson, J.M.S.; Whitworth, R.J.; Sloderbeck, P.E. Spatio-temporal distribution of stored-products insects around food processing and storage facilities. Agric. Ecosyst. Environ. 2013, 165, 151–162. [Google Scholar] [CrossRef]
- Campbell, J.F.; Arbogast, R.T. Stored-product insects in a flour mill: Population dynamics and response to fumigation treatments. Entomol. Exp. Appl. 2004, 112, 217–225. [Google Scholar] [CrossRef]
- Campbell, J.F.; Hagstrum, D.W. Patch exploitation by Tribolium castaneum: Movement patterns, distribution and oviposition. J. Stored Prod. Res. 2002, 38, 55–68. [Google Scholar] [CrossRef]
- Dowdy, A.K.; Howard, R.W.; Seitz, L.M.; McGaughey, W.H. Response of Rhyzopertha dominica (Coleoptera: Bostrichidae) to its aggregation pheromone and wheat volatiles. Environ. Entomol. 1993, 22, 965–970. [Google Scholar] [CrossRef]
- Ahmad, F.; Daglish, G.J.; Ridley, A.W.; Walter, G.H. Responses of Tribolium castaneum to olfactory cues from cotton seeds, the fungi associated with cotton seeds, and cereals. Entomol. Exp. Appl. 2012, 145, 272–281. [Google Scholar] [CrossRef]
- Potter, C. The biology and distribution of Rhyzopertha dominica (Fab.). Trans. Royal Entomol. Soc. Lond. 1935, 83, 449–482. [Google Scholar] [CrossRef]
- Wright, V.F.; Fleming, E.E.; Post, D. Survival of Rhyzopertha dominica (Coleoptera, Bostrichidae) on fruits and seeds collected from woodrat nests in Kansas. J. Kans. Entomol. Soc. 1990, 63, 344–347. [Google Scholar]
- Ahmad, F.; Daglish, G.J.; Ridley, A.W.; Burrill, P.R.; Walter, G.H. Short-range resource location by Tribolium castaneum Herbst (Coleoptera: Tenebrionidae) demonstrates a strong preference for fungi associated with cotton seed. J. Stored Prod. Res. 2013, 52, 21–27. [Google Scholar] [CrossRef]
- Dowdy, A.K.; McGaughey, W.H. Seasonal activity of stored-product insects in and around farm-stored wheat. J. Econ. Entomol. 1994, 87, 1351–1358. [Google Scholar] [CrossRef]
- Edde, P.A.; Phillips, T.W.; Nansen, C.; Payton, M.E. Flight activity of the lesser grain borer, Rhyzopertha dominica F. (Coleoptera: Bostrichidae), in relation to weather. Environ. Ecol. 2006, 35, 616–624. [Google Scholar] [CrossRef]
- Campbell, J.F.; Ching’oma, G.P.; Toews, M.D.; Ramsawany, S.B. Spatial distribution and movement patterns of stored-product pests. In Proceedings of the 9th International Working Conference on Stored-Product Protection, Sao Paulo, Brazil, 15–18 October 2006; Lorini, I., Bacaltchuk, B., Beckel, H., Deckers, D., Sundfeld, E., dos Santos, J.P., Biagi, J.D., Celaro, J.C., Faroni, L.R.D.A., Bortolini, L.d.O.F., Eds.; Brazilian Post-Harvest Association—ABRAPOS: Passo Fundo, Brazil, 2006; pp. 361–370. [Google Scholar]
- Campbell, J.F.; Toews, M.D.; Arthur, F.H.; Arbogast, R.T. Long-term monitoring of Tribolium castaneum in two flour mills: Seasonal patterns and impact of fumigation. J. Econ. Entomol. 2010, 103, 991–1001. [Google Scholar] [CrossRef]
- Fields, P.G.; Van Loon, J.; Dolinski, M.G.; Harris, J.L.; Burkholder, W.E. The distribution of Rhyzopertha dominica (F.) in western Canada. Can. Entomol. 1993, 125, 317–328. [Google Scholar] [CrossRef]
- Holloway, J.C.; Mayer, D.G.; Daglish, G.J. Flight activity of Cryptolestes ferrugineus in southern New South Wales, Australia. J. Pest Sci. 2018, 91, 1353–1362. [Google Scholar] [CrossRef]
- Climate Data Online. Available online: http://www.bom.gov.au/climate/data/index.shtml (accessed on 25 January 2017).
- GenStat. GenStat for Windows; Release 19.1; VSN International Ltd.: Oxford, UK, 2018. [Google Scholar]
- Stevens, M.M. Combining Monitoring and Incursion Surveillance for Grains; Final Report, Project CRC2113; Plant Biosecurity Cooperative Research Centre: Canberra, Australia, 2018. [Google Scholar]
- McKay, T.; White, A.L.; Starkus, L.A.; Arthur, F.H.; Campbell, J.F. Seasonal patterns of stored-product insects at a rice mill. J. Econ. Entomol. 2017, 110, 1366–1376. [Google Scholar] [CrossRef] [PubMed]
- Throne, J.E.; Cline, L.D. Seasonal flight activity and seasonal abundance of selected stored-product Coleoptera around grain storages in South Carolina. J. Agric. Entomol. 1994, 11, 321–338. [Google Scholar]
- Daglish, G.J.; Ridley, A.W.; Hereward, J.P.; Emery, R.N.; Holloway, J.C.; Raghu, S.; Walter, G.H. Investigation of dispersal and spatio-temporal distributions of stored grain insects in Australia using ecological and molecular tools. In Proceedings of the 11th International Working Conference on Stored Product Protection, Chiang Mai, Thailand, 24–28 November 2014; Arthur, F.H., Kengkanpanich, R., Chayaprasert, W., Suthisut, D., Eds.; Kansas State University: Manhattan, NY, USA, 2014; pp. 74–78. [Google Scholar]
- Rajan, T.S.; Muralitharan, V.; Daglish, G.J.; Mohankumar, S.; Rafter, M.A.; Chandrasekaran, S.; Mohan, S.; Vimal, D.; Srivastava, C.; Loganathan, M.; et al. Flight of three major insect pests of stored grain in the monsoonal tropics of India, by latitude, season and habitat. J. Stored Prod. Res. 2018, 76, 43–50. [Google Scholar] [CrossRef]
- Toews, M.D.; Campbell, J.F.; Arthur, F.H.; Ramaswamy, S.B. Outdoor flight activity and immigrations of Rhyzopertha dominica into seed wheat warehouses. Entomol. Exp. Appl. 2006, 121, 73–85. [Google Scholar] [CrossRef]
- Borgmeister, C.; Meikle, W.G.; Scholz, D.; Adda, C.; Degbey, P.; Markham, R.H. Seasonal and weather factors influencing the annual flight cycle of Prostephanus truncatus (Coleoptera: Bostrichidae) and its predator Teretriosoma nigrescens (Coleoptera: Histeridae) in Benin. Bull. Entomol. Res. 1997, 87, 239–246. [Google Scholar] [CrossRef]
- Sinclair, E.R.; Alder, J. Migration of stored-grain insect pests from a small wheat bulk. Aust. J. Exp. Agric. Anim. Husb. 1987, 24, 260–266. [Google Scholar] [CrossRef]
- Barrer, P.M.; Starick, N.T.; Morton, R.; Wright, E.J. Factors influencing initiation of flight by Rhyzopertha dominica (F.) (Coleoptera: Bostrichidae). J. Stored Prod. Res. 1993, 29, 1–5. [Google Scholar] [CrossRef]
- Dowdy, A.K. Flight initiation of lesser grain borer (Coleoptera: Bostrichidae) as influenced by temperature, humidity, and light. J. Econ. Entomol. 1994, 87, 1714–1717. [Google Scholar] [CrossRef]
- Aslam, M.; Hagstrum, D.W.; Dover, B.A. The effect of photoperiod on the flight activity and biology of Rhyzopertha dominica (Coleoptera: Bostrichidae). J. Kans. Entomol. Soc. 1994, 67, 107–115. [Google Scholar]
- Taylor, T.A. On the flight activity of Sitophilus zeamais Motsch. (Coleoptera, Curculionidae) and some other grain-infesting beetles in the field and a store. J. Stored Prod. Res. 1971, 6, 295–306. [Google Scholar] [CrossRef]
- Cox, P.D.; Wakefield, M.E.; Jacob, T.A. The effects of temperature on flight initiation in a range of moths, beetles and parasitoids associated with stored products. J. Stored Prod. Res. 2007, 43, 111–117. [Google Scholar] [CrossRef]
- Wright, E.J.; Morton, R. Daily flight activity of Trogoderma variabile (Coleoptera: Dermestidae) and Rhyzopertha dominica (Coleoptera: Bostrichidae). J. Stored Prod. Res. 1995, 31, 177–184. [Google Scholar] [CrossRef]
- McKay, T.; Bowombe-Toko, M.P.; Starkus, L.A.; Arthur, F.H.; Campbell, J.F. Monitoring Tribolium castaneum (Coleoptera: Tenebrionidae) in rice mills using pheromone-baited traps. J. Econ. Entomol. 2019, 112, 1454–1462. [Google Scholar] [CrossRef]
- Sinclair, E.R.; Haddrell, R.L. Flight of stored products beetles over a grain farming area in southern Queensland. J. Aust. Entomol. Soc. 1985, 24, 9–15. [Google Scholar] [CrossRef]
- Mahroof, R.M.; Edde, P.A.; Robertson, B.; Puckette, J.A.; Phillips, T.W. Dispersal of Rhyzopertha dominica (Coleoptera: Bostrichidae) in different habitats. Environ. Entomol. 2010, 39, 930–938. [Google Scholar] [CrossRef]
- Toon, A.; Daglish, G.J.; Ridley, A.W.; Emery, R.N.; Holloway, J.C.; Walter, G.H. Significant population structure in Australian Cryptolestes ferrugineus and interpreting the potential spread of phosphine resistance. J. Stored Prod. Res. 2018, 77, 219–224. [Google Scholar] [CrossRef]
- Rafter, M.A.; McCulloch, G.A.; Daglish, G.J.; Gurdasani, K.; Walter, G.H. Polyandry, genetic diversity and fecundity of emigrating beetles: Understanding new foci of infestation and selection. J. Pest Sci. 2018, 91, 287–298. [Google Scholar] [CrossRef]
- Daglish, G.J.; Nayak, M.K. Prevalence of resistance to deltamethrin in Rhyzopertha dominica (F.) in eastern Australia. J. Stored Prod. Res. 2018, 78, 45–49. [Google Scholar] [CrossRef]
- Phillips, T.W.; Jiang, X.-L.; Burkholder, W.E.; Phillips, J.K.; Tran, H.Q. Behavioral responses to food volatiles by two species of stored-product coleoptera, Sitophilus oryzae (Curculionidae) and Tribolium castaneum (Tenebrionidae). J. Chem. Ecol. 1993, 19, 723–734. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holloway, J.C.; Daglish, G.J.; Mayer, D.G. Spatial Distribution and Flight Patterns of Two Grain Storage Insect Pests, Rhyzopertha dominica (Bostrichidae) and Tribolium castaneum (Tenebrionidae): Implications for Pest Management. Insects 2020, 11, 715. https://doi.org/10.3390/insects11100715
Holloway JC, Daglish GJ, Mayer DG. Spatial Distribution and Flight Patterns of Two Grain Storage Insect Pests, Rhyzopertha dominica (Bostrichidae) and Tribolium castaneum (Tenebrionidae): Implications for Pest Management. Insects. 2020; 11(10):715. https://doi.org/10.3390/insects11100715
Chicago/Turabian StyleHolloway, Joanne C., Gregory J. Daglish, and David G. Mayer. 2020. "Spatial Distribution and Flight Patterns of Two Grain Storage Insect Pests, Rhyzopertha dominica (Bostrichidae) and Tribolium castaneum (Tenebrionidae): Implications for Pest Management" Insects 11, no. 10: 715. https://doi.org/10.3390/insects11100715
APA StyleHolloway, J. C., Daglish, G. J., & Mayer, D. G. (2020). Spatial Distribution and Flight Patterns of Two Grain Storage Insect Pests, Rhyzopertha dominica (Bostrichidae) and Tribolium castaneum (Tenebrionidae): Implications for Pest Management. Insects, 11(10), 715. https://doi.org/10.3390/insects11100715




