Evaluation of Knockdown Bioassay Methods to Assess Phosphine Resistance in the Red Flour Beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Tribolium castaneum Populations
2.2. Knockdown Definition
2.3. Methods to Conduct and Assess Knockdown Tests
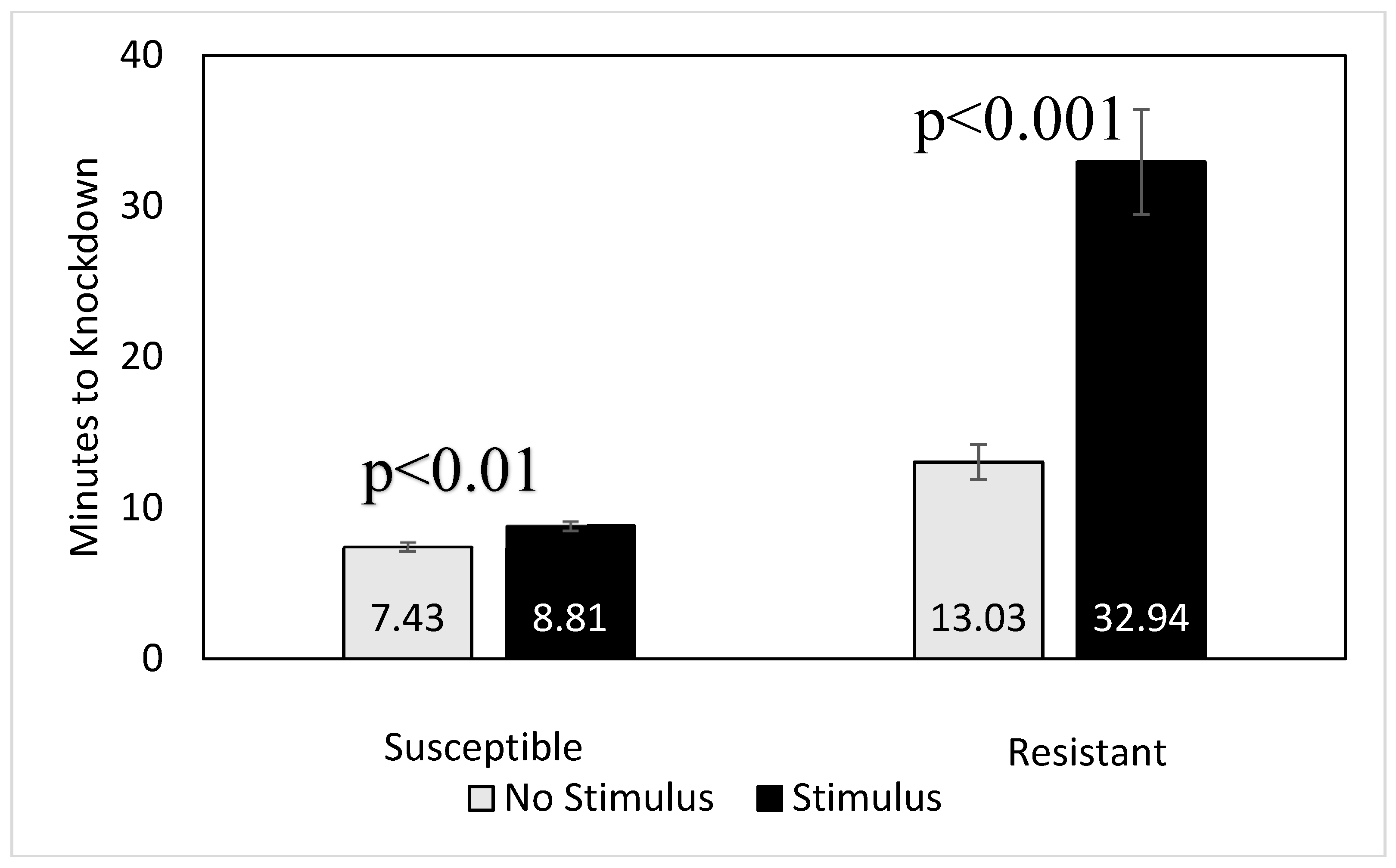
2.4. Evaluation of Physical Disturbance on Beetle Knockdown Time
2.5. Statistics
3. Results
3.1. Variation in Knockdown Times among Populations with Different Resistance Levels
3.2. Effect of Physical Disturbance on Knockdown Times of Single Beetles
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hagstrum, D.W.; Phillips, T.W.; Cuperus, G.W. (Eds.) Stored Product Protection; Kansas State University: Manhattan, NY, USA, 2012. [Google Scholar]
- Cuperus, G.W.; Prickett, C.K.; Bloome, P.D.; Pitts, J.T. Insect Populations in aerated and unaerated stored wheat in Oklahoma. J. Kans. Entomol. Soc. 1986, 59, 620–627. [Google Scholar]
- Schlipalius, D.I.; Valmas, N.; Tuck, A.G.; Jagadeesan, R.; Ma, L.; Kaur, R.; Goldinger, A.; Anderson, C.; Kuang, K.; Zuryn, K.; et al. A core metabolic enzyme mediates resistance to phosphine gas. Science 2012, 338, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Champ, B.R.; Dyte, C.E. Report of the FAO Global Survey of Pesticide Susceptibility of Stored Grain Pests; FAO Plant Production and Protection Series: Rome, Italy, 1976. [Google Scholar]
- Zettler, J.L.; Halliday, W.R.; Arthur, H. Phosphine resistance in insects infesting stored peanuts in the southeastern United States. J. Econ. Entomol. 1989, 82, 1508–1511. [Google Scholar] [CrossRef]
- Zettler, J.L.; Cuperus, G.W. Pesticide resistance in Tribolium castaneum (Coleoptera: Tenebrionidae) and Rhyzopertha dominica (Coleoptera: Bostrichidae) in wheat. J. Econ. Entomol. 1990, 83, 1677–1681. [Google Scholar] [CrossRef]
- Opit, J.P.; Phillips, T.W.; Aikins, M.J.; Hasan, M.M. Phosphine resistance in Tribolium castaneum and Rhyzopertha dominica from stored wheat in Oklahoma. J. Econ. Entomol. 2012, 105, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Cato, A.J.; Elliott, B.; Nayak, M.K.; Phillips, T.W. Geographic variation in phosphine resistance among North American populations of the red flour beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J. Econ. Entomol. 2017, 110, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Nayak, M.K.; Daglish, G.J.; Phillips, T.W. Managing resistance to chemical treatments in stored products pests. Stewart Post-Harvest Rev. 2015, 1, 2. [Google Scholar] [CrossRef]
- Anonymous. Recommended Methods for the Detection and Measurement of Resistance of Agricultural Pests to Pesticides-Tentative Method for Adults of Some Major Pest Species of Stored Cereals, with Methyl Bromide and Phosphine; FAO Method Number 16; Plant Production and Protection Div.: Rome, Italy, 1975; Volume 23, pp. 12–25. [Google Scholar]
- Winks, R.G. The toxicity of phosphine to adults of Tribolium castaneum (Herbst): Time as a dosage factor. J. Stored Prod. Res. 1984, 18, 159–169. [Google Scholar] [CrossRef]
- Reichmuth, C. A Quick Test to Determine Phosphine Resistance in Stored-Products Research; GASGA Newsletter: Rome, Italy, 1991; Volume 15, pp. 14–15. [Google Scholar]
- Reichmuth, C. Uptake of phosphine by stored-product pest insects during fumigation. In Proceedings of the 6th International Working Conference on Stored-Prod, Canberra, Australia, 17–23 April 1994; Volume 1, pp. 157–162. [Google Scholar]
- Bang, Y.H.; Telford, H.S. Effect of sub lethal doses of fumigants on stored-grain insects. In Technical Bulletin 50; Washington Agricultural Experiment Station: Pullman, WA, USA, 1966; pp. 1–22. [Google Scholar]
- Bell, C.H.; Savvidou, N.; Mills, K.A.; Bradberry, S.; Barlow, M.L. A same-day test for detecting resistance to phosphine. In Proceedings of the 6th International Working Conference on Stored-Prod, Canberra, Australia, 17–23 April 1994; Volume 1, pp. 41–44. [Google Scholar]
- Cao, Y.; Wang, D. Relationship between phosphine resistance and narcotic knockdown in Tribolium castaneum (Herbst), Sitophilus oryzae (L.), and S. zeamais (Motsch). In Proceedings of the Conference Controlled Atmosphere and Fumigation in Stored-products, Adelaide, Australia, 1–4 August 2000; pp. 609–616. [Google Scholar]
- Collins, P.J. Resistance to grain protectants and fumigants in insect pests of stored products in Australia. In Proceedings of the Australian Post-Harvest Technical Conference, Canberra, Australia, 26–29 May 1998; pp. 55–57. [Google Scholar]
- Chen, Z.; Schlipalius, D.; Opit, G.; Subramanyam, B.; Phillips, T.W. Diagnostic molecular markers for phosphine resistance in U.S. populations of Tribolium castaneum and Rhyzopertha dominica. PLoS ONE 2015, 10, e0121343. [Google Scholar] [CrossRef] [PubMed]
- Nayak, M.K.; Collins, P.J.; Holloway, J.C.; Emery, R.N.; Pavic, H.; Bartlet, J. Strong resistance to phosphine in the rusty grain beetle, Cryptolestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae): Its characterization, a rapid assay for diagnosis and its distribution in Australia. Pest Manag. Sci. 2013, 69, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Nayak, M.K.; Kaur, R.; Jagadeesan, R.; Pavic, H.; Phillips, T.W.; Daglish, G.J. Development of a quick knock down test for diagnosing resistance to phosphine in rice weevil, Sitophilus oryzae, a major pest of stored products. J. Econ. Entomol. 2019, in press. [Google Scholar] [CrossRef]
- Steuerwald, R.; Dierks-Lange, H.; Schmitt, S. Rapid bioassay for determining the phosphine tolerance. In Proceedings of the 9th International Conference on Stored-Product Protection, Sao Paulo, Brazil, 15–18 October 2006; pp. 306–311. [Google Scholar]
- Athanassiou, C.G.; Kavallieratos, N.G.; Brabec, D.L.; Oppert, B.; Guedes, R.N.C.; Campbell, J.F. From immobilization to recovery: Towards the development of a rapid diagnostic indicator for phosphine resistance. J. Stored Prod. Res. 2019, 80, 28–33. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org/ (accessed on 15 May 2019).
- Hori, M.; Kasaishi, Y. Development of a new assay method for quickly evaluating phosphine resistance of the cigarette beetle, Lasioderma serricorne (Fabricius) (Coleoptera: Anobiidae), based on knockdown of the adult beetles. Appl. Entomol. Zool. 2005, 40, 99–104. [Google Scholar] [CrossRef][Green Version]


| Population 1 & Resistance Status | FAO % Resist. 2 | Single KT (± SE) 3,4,5 | 50% KT (± SE) 3,4,5 | 100% KT (± SE) 3,4,5 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Susceptible | ||||||||||
| USDA Lab Susceptible | 0 | 8.46 | ± | 0.66 A,a | 5.19 | ± | 0.21 A,b | 9.13 | ± | 0.46 A,a |
| Excelsior Springs | 0 | 10.82 | ± | 1.04 A,a | 6.13 | ± | 0.48 A,a | 17.05 | ± | 3.01 AB,b |
| Calgary, Alberta | 0 | 9.24 | ± | 0.42 A,a | 7.27 | ± | 0.11 A,a | 16.23 | ± | 1.21 AB,b |
| Davis, Calif. | 0 | 14.13 | ± | 0.84 AB,a | 9.16 | ± | 0.84 AB,a | 34.83 | ± | 7.42 AB,b |
| Arbuckle, Calif. | 0 | 14.37 | ± | 1.35 AB,a | 10.00 | ± | 0.30 B,a | 31.28 | ± | 3.12 AB,b |
| Williams, Calif. | 0 | 10.91 | ± | 1.01 A,a | 10.05 | ± | 0.53 B,a | 26.16 | ± | 2.15 AB,b |
| Weak Resistance | ||||||||||
| Thailand | 15 | 14.62 | ± | 2.00 AB,a | 7.89 | ± | 0.29 AB,a | 29.17 | ± | 3.92 AB,b |
| Abilene, Kansas | 39 | 16.14 | ± | 1.21 AB,a | 11.50 | ± | 0.61 BC,a | 37.30 | ± | 3.84 AB,b |
| Russell, Kansas | 41 | 16.82 | ± | 1.38 AB,a | 14.38 | ± | 0.42 CD,a | 62.78 | ± | 9.12 BC,b |
| Strong Resistance | ||||||||||
| Minneapolis, Kansas | 89 | 24.47 | ± | 2.35 B,a | 16.98 | ± | 1.04 D,a | 100.20 | ± | 11.77 C,b |
| Mitchell, Kansas | 93 | 19.32 | ± | 1.54 AB,a | 14.55 | ± | 0.87 CD,a | 172.15 | ± | 24.72 D,b |
| Red Level, Alabama | 100 | 52.70 | ± | 1.54 AB,a | 24.33 | ± | 2.05 E,a | 194.13 | ± | 22.13 D,b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cato, A.; Afful, E.; Nayak, M.K.; Phillips, T.W. Evaluation of Knockdown Bioassay Methods to Assess Phosphine Resistance in the Red Flour Beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Insects 2019, 10, 140. https://doi.org/10.3390/insects10050140
Cato A, Afful E, Nayak MK, Phillips TW. Evaluation of Knockdown Bioassay Methods to Assess Phosphine Resistance in the Red Flour Beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Insects. 2019; 10(5):140. https://doi.org/10.3390/insects10050140
Chicago/Turabian StyleCato, Aaron, Edwin Afful, Manoj K. Nayak, and Thomas W. Phillips. 2019. "Evaluation of Knockdown Bioassay Methods to Assess Phosphine Resistance in the Red Flour Beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae)" Insects 10, no. 5: 140. https://doi.org/10.3390/insects10050140
APA StyleCato, A., Afful, E., Nayak, M. K., & Phillips, T. W. (2019). Evaluation of Knockdown Bioassay Methods to Assess Phosphine Resistance in the Red Flour Beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Insects, 10(5), 140. https://doi.org/10.3390/insects10050140





