Diversity, Daily Activity Patterns, and Pollination Effectiveness of the Insects Visiting Camellia osmantha, C. vietnamensis, and C. oleifera in South China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. The Diversity and Daily Activity of Insects Visiting C. osmantha, C. vietnamensis, and C. oleifera
2.3. Floral Traits of C. osmantha, C. vietnamensis, and C. oleifera
2.4. Nectar Volumes and Sugar Concentrations of C. osmantha, C. vietnamensis, and C. oleifera Flowers
2.5. Effect of Insect Pollination on the Fruit Production Rates of C. osmantha, C. vietnamensis, and C. oleifera
2.6. Statistical Analyses
3. Results
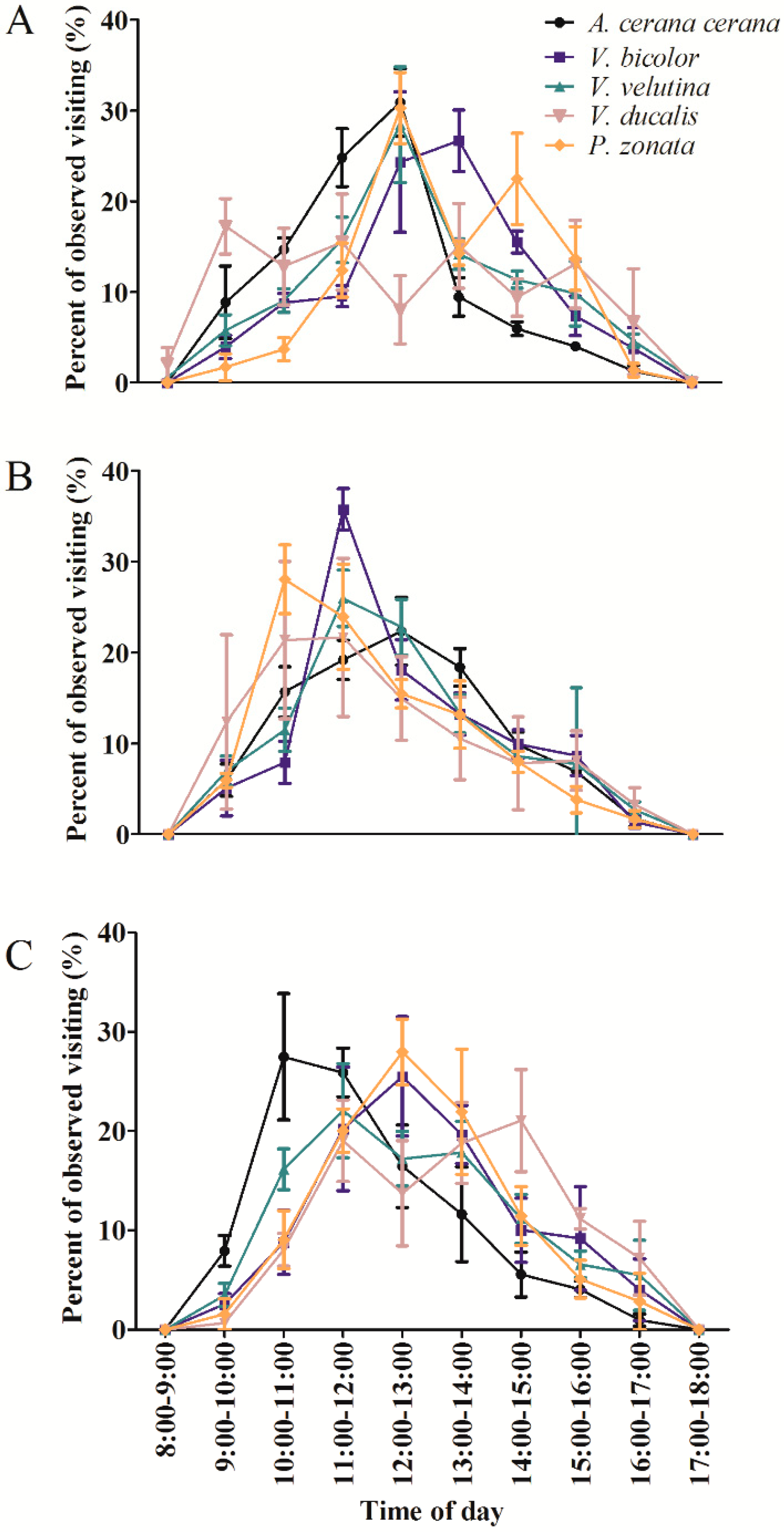
3.1. The Diversity and Daily Activity of Insects Visiting C. osmantha, C. vietnamensis, and C. oleifera
3.2. Floral Traits of C. osmantha, C. vietnamensis, and C. oleifera
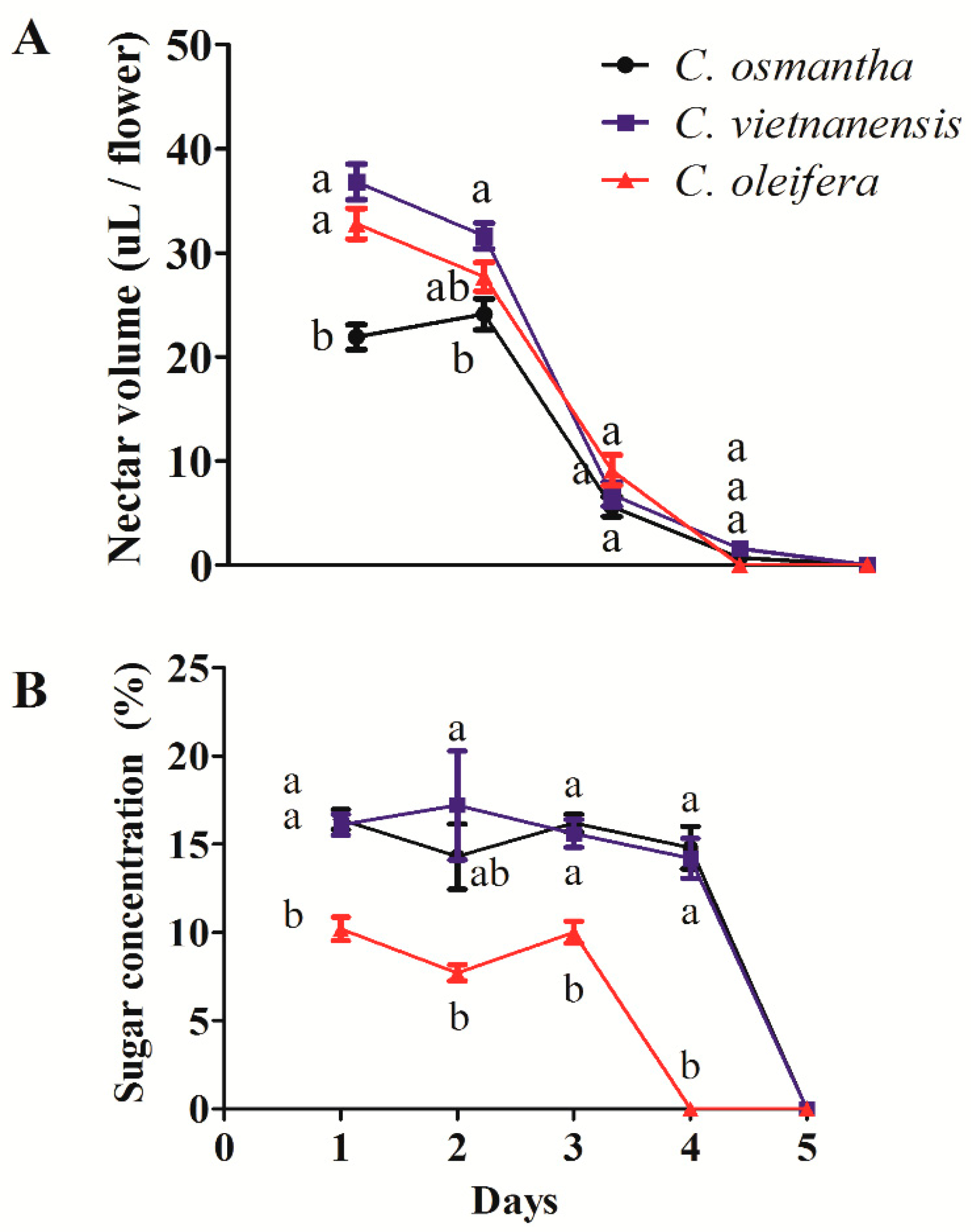
3.3. Nectar Volumes and Sugar Concentrations in C. osmantha, C. vietnamensis, and C. oleifera Flowers
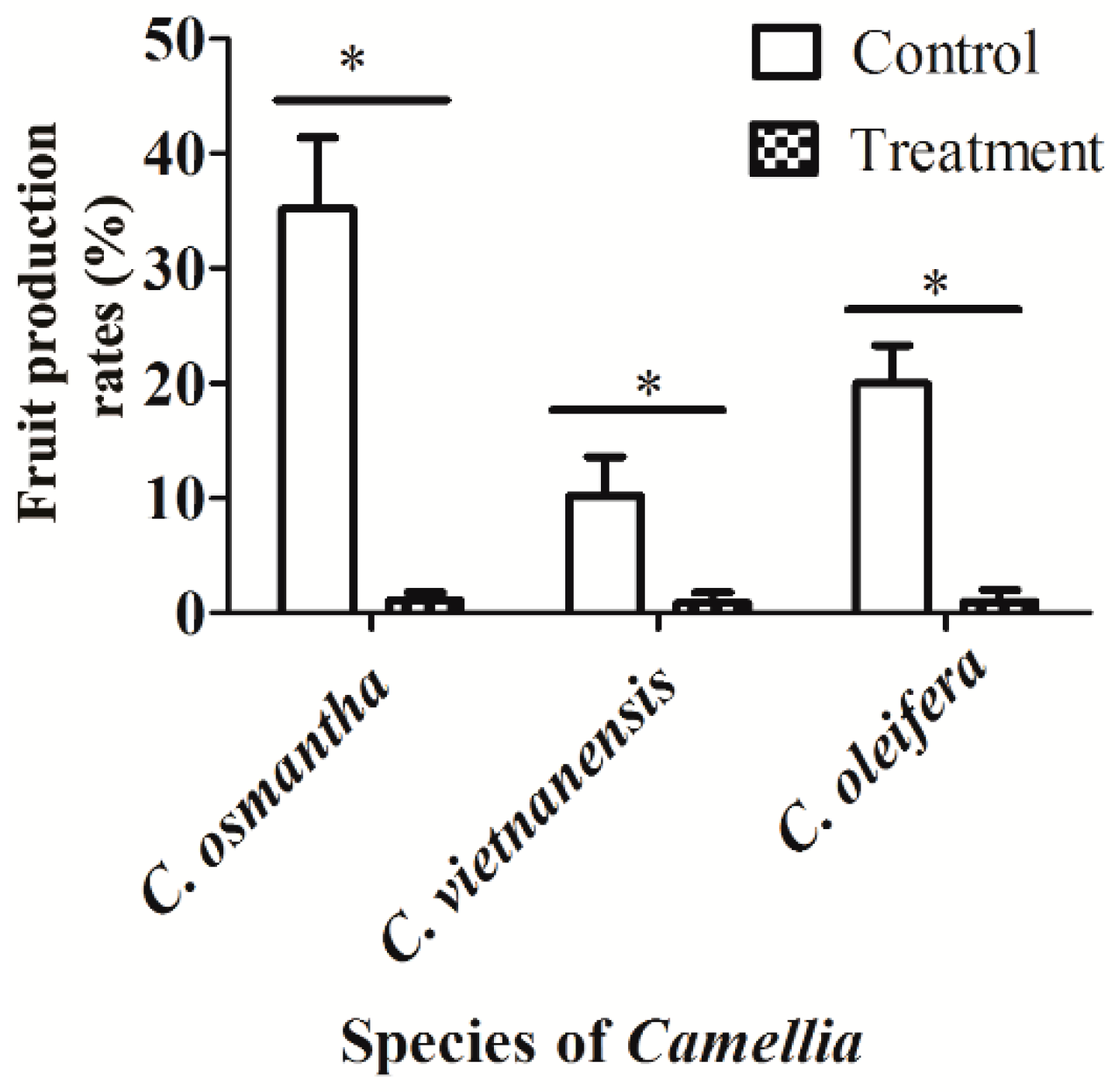
3.4. Effect of Insect Pollination on the Fruit Production Rates of C. osmantha, C. vietnamensis, and C. oleifera
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klein, A.M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. Lond. B: Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Inouye, D.W.; Larson, B.M.H.; Ssymank, A.; Kevan, P.G. Flies and flowers III: Ecology of foraging and pollination. J. Pollinat. Ecol. 2015, 16, 115–133. [Google Scholar]
- Rader, R.; Bartomeus, I.; Garibaldi, L.A.; Garratt, M.P.D.; Howlett, B.G.; Winfree, R.; Cunningham, S.A.; Mayfield, M.M.; Arthur, A.D.; Andersson, G.K.; et al. Non-bee insects are important contributors to global crop pollination. Proc. Natl. Acad. Sci. USA 2016, 113, 146–151. [Google Scholar] [CrossRef]
- New, T.R. Classic Themes: Pollination Mutualisms of Insects and Plants. In Mutualisms and Insect Conservation; New, T.R., Ed.; Springer: Cham, Switzerland, 2017; pp. 37–62. [Google Scholar]
- Harder, L.D.; Johnson, S.D. Darwin’s beautiful contrivances: Evolutionary and functional evidence for floral adaptation. New Phytol. 2009, 183, 530–545. [Google Scholar] [CrossRef]
- Caruso, C.M.; Eisen, K.E.; Martin, R.A.; Sletvold, N. A meta-analysis of the agents of selection on floral traits. Evolution 2019, 73, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Conner, J.K.; Rush, S. Effects of flower size and number on pollinator visitation to wild radish, Raphanus raphanistrum. Oceologia 1996, 105, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Howlett, B.G.; Evans, L.J.; Kendall, L.K.; Rader, R.; McBrydie, H.M.; Read, S.F.J.; Cutting, B.T.; Robson, A.; Pattemore, D.E.; Willcox, B.K. Surveying insect flower visitors to crops in New Zealand and Australia. BioRxiv 2018, 373126. [Google Scholar] [CrossRef]
- Robards, K.; Prenzler, P.; Ryan, D.; Zhong, H. Camellia oil and tea oil. In Gourmet and Health-Promoting Specialty Oils; Moreau, R.A., Kamal-Eldin, A., Eds.; AOCS Press: Urbana, IL, USA, 2009; pp. 313–343. [Google Scholar]
- Ruter, J.M. Nursery production of tea oil camellia under different light levels. In Trends in New Crops, New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, VA, USA, 2002; pp. 222–224. [Google Scholar]
- Wang, X.Q.; Zeng, Q.M.; Verardo, V.; Contreras, M.M. Fatty acid and sterol composition of tea seed oils: Their comparison by the “FancyTiles” approach. Food Chem. 2017, 233, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.Y.; He, B.; Gu, P.; Su, T.J.; Zhu, C.D. Discussion on current situation and research direction of pollination insects of Camellia oleifera. J. Environ. Entomol. 2017, 39, 213–220. [Google Scholar]
- Wei, W.; Li, X.Y.; Wei, X.; Lu, W.; Yang, X.H.; Zheng, X.L. Review of species, nesting and pollination behaviors of pollinating insects in Camellia spp. Guangxi For. Sci. 2017, 46, 98–101. [Google Scholar]
- Xie, Z.H.; Chen, X.M.; Qiu, J.S. Reproductive failure of Camellia oleifera in the Plateau region of China due to a shortage of legitimate pollinators. Int. J. Agric. Biol. 2013, 15, 458–464. [Google Scholar]
- Ma, J.L.; Ye, H.; Ye, C.X. A new species of Camellia sect. Paracamellia. Guihaia 2012, 32, 753–755. [Google Scholar]
- Zhou, S.; Zhu, J.H.; Xiao, J.Z.; Xiang, H.G.; Shi, Y.F. Distant crossing trial with oiltea camellia. Econ. For. Res. 2001, 19, 20–25. [Google Scholar]
- Zhou, H.; Wang, C.Z.; Ye, J.Z.; Chen, H.X. New triterpene saponins from the seed cake of Camellia oleifera and their cytotoxic activity. Phytochem. Lett. 2014, 8, 46–51. [Google Scholar] [CrossRef]
- Su, R.; Dong, Y.; Dong, K.; He, S.Y. The toxic honey plant Camellia oleifera. J. Apic. Res. 2012, 51, 277–279. [Google Scholar] [CrossRef]
- Luo, J.; Zhao, C.J.; Huang, H.Y.; Jiang, X.J. Variety investigation on pollinating insects of sasanqua in Guangxi. Guangxi For. Sci. 2014, 43, 61–65. [Google Scholar]
- Huang, H.Y.; Luo, J.; Jiang, X.J.; Zhao, C.J.; Li, G.H. The investigation on hymenoptera pollination insect of Camellia oleifera in Guangxi Province. J. Fujian For. Sci. Tech. 2014, 41, 79–83. [Google Scholar]
- Deng, Y.Y.; Yuan, X.L.; Luo, Y.B. The role of native bees on the reproductive success of Camellia oleifera in Hunan Province, central south China. Acta Ecol. Sin. 2010, 30, 4427–4436. [Google Scholar]
- He, X.Y.; Cai, S.P.; Xiong, Y.; Han, G.Y.; Chen, Y.D.; Huang, L.R.; Wu, Q.R. Investigation on the pollination insect species and their foraging behaviors on Camellia oleifera in Fujian Province. J. Fujian For. Sci. Tech. 2010, 37, 1–5, 30. [Google Scholar]
- Han, N.L.; You, H.L. Study on the utilization of Colletes gigas Cockerell. Sci. Silvae Sin. 1979, 3, 215–218. [Google Scholar]
- Zhao, S.W.; Zheng, Q.Z.; Liu, Z.Z. Studies on the fruiting production rates of Camellia oleifera improved by Apis cerana cerana Fabricius and developing the nectar source. For. Sci. Tech. 1985, 4, 16–19. [Google Scholar]
- Li, T.S. Economic Insect Fauna of China. Fasc. 30, Hymenoptera: Vespoidea; Science Press: Beijing, China, 1985; pp. 1–159. [Google Scholar]
- Fan, Z.D. Animal Fauna of China. Fasc. 6, Diptera: Calliphoridae; Science Press: Beijing, China, 1997; pp. 1–707. [Google Scholar]
- Huang, C.M.; Cheng, X.Y. Animal Fauna of China. Fasc. 50, Diptera: Syrphidae; Science Press: Beijing, China, 2012; pp. 1–852. [Google Scholar]
- Herrera, C.M. Daily patterns of pollinator activity, differential pollinating effectiveness, and floral resource availability, in a sumrner-flowering Mediterranean shrub. Oikos 1990, 58, 277–288. [Google Scholar] [CrossRef]
- Katsuhara, K.R.; Kitamura, S.; Ushimaru, A. Functional significance of petals as landing sites in fungus-gnat pollinated flowers of Mitella pauciflora (Saxifragaceae). Funct. Ecol. 2017, 31, 1193–1200. [Google Scholar] [CrossRef]
- Iwata, T.; Nagasaki, O.; Ishii, H.S.; Ushimaru, A. Inflorescence architecture affects pollinator behaviour and mating success in Spiranthes sinensis (Orchidaceae). New Phytol. 2012, 193, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Huber, F.K.; Kaiser, R.; Sauter, W.; Schiestl, F.P. Floral scent emission and pollinator attraction in two species of Gymnadenia (Orchidaceae). Oecologia 2005, 142, 564–575. [Google Scholar] [CrossRef] [PubMed]
- Grison-pigé, L.; Bessière, J.M.; Hossaert-McKey, M. Specific attraction of fig-pollinating wasps: Role of volatile compounds released by tropical figs. J. Chem. Ecol. 2002, 28, 283–295. [Google Scholar] [CrossRef]
- Marden, J.M. Remote perception of floral nectar by bumblebees. Oecologia 1984, 64, 232–240. [Google Scholar] [CrossRef]
- Goulson, D.; Stout, J.C.; Langley, J.; Hughes, W.H.O. Identity and function of scent marks deposited by foraging bumblebees. J. Chem. Ecol. 2000, 26, 2897–2911. [Google Scholar] [CrossRef]
- Jones, E.I.; Dornhaus, A. Predation risk makes bees reject rewarding flowers and reduce foraging activity. Behav. Ecol. Sociobiol. 2011, 65, 1505–1511. [Google Scholar] [CrossRef]
- Pierrot, L.M.; Schlindwein, C. Variation in daily flight activity and foraging patterns in colonies of uruçu—Melipona scutellaris Latreille (Apidae: Meliponini). Rev. Bras. Zool. 2003, 20, 565–571. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Grozinger, C.M.; Tooker, J.F. Bumble bees exhibit daily behavioral patterns in pollen foraging. Arthropod-Plant Interact. 2014, 8, 273–283. [Google Scholar] [CrossRef]


| Order | Family | Species | Observed Number | ||
|---|---|---|---|---|---|
| C. osmantha | C. vietnamensis | C. oleifera | |||
| Hymenoptera | Apidae | Apis cerana cerana Fabricius | 412 | 416 | 380 |
| Halictidae | Lasioglossum sp. | 0 | 0 | 1 | |
| Vespidae | Vespa bicolor Fabricius | 244 | 347 | 408 | |
| V. velutina Lepeletier | 350 | 457 | 430 | ||
| V. ducalis Smith | 109 | 162 | 230 | ||
| V. mocsaryana du Buysson | 1 | 6 | 9 | ||
| V. velutina nigrithorax du Buysson | 7 | 6 | 22 | ||
| V. affinis L. | 15 | 35 | 29 | ||
| Ichneumonidae | Xanthopimpla pedator Fabricius | 0 | 0 | 1 | |
| Formicidae | Dolichoderus sibiricus Emery | 1 | 0 | 0 | |
| Diptera | Syrphidae | Phytomia zonata Fabricius | 165 | 177 | 158 |
| Eristalis cerealis Fabricius | 12 | 14 | 10 | ||
| Episyrphus balteatus de Geer | 9 | 12 | 12 | ||
| Syrphus ribesii L. | 5 | 8 | 12 | ||
| Eristalinus arvorum Fabricius | 4 | 6 | 7 | ||
| Calliphoridae | Calliphora vicina Robineau-Desvoidy | 3 | 0 | 5 | |
| Chrysomya megacephala Fabricius | 7 | 16 | 15 | ||
| Graphomya maculate Scopoli | 3 | 5 | 8 | ||
| Neomyia timorensis Robineau-Desvoidy | 0 | 0 | 1 | ||
| Lepidoptera | Satyridae | Melanitis leda L. | 1 | 0 | 0 |
| Junonia almana L. | 0 | 1 | 0 | ||
| Pieridae | Pieris rapae L. | 1 | 0 | 0 | |
| Arctiidae | Syntomoides imaon Cramer | 2 | 1 | 0 | |
| Coleoptera | Coccinellidae | Propylaea japonica Thunberg | 2 | 3 | 2 |
| Species | Number of Insects Visiting on Flowers (Visits/Day) ξ | ||
|---|---|---|---|
| C. osmantha | C. vietnamensis | C. oleifera | |
| A. cerana cerana | 51.8 ± 10.5 a | 100.5 ± 35.4 a | 95.0 ± 26.6 a |
| V. bicolor | 43.3 ± 9.3 b | 36.5 ± 13.4 b | 102.0 ± 23.9 a |
| V. velutina | 117.3 ± 19.1 a | 80.3 ± 20.0 a | 107.5 ± 40.8 a |
| V. ducalis | 15.3 ± 3.6 b | 14.0 ± 4.4 b | 57.5 ± 15.1 a |
| P. zonata | 26.8 ± 7.5 a | 59.0 ± 20.1 a | 37.0 ± 7.3 a |
| Species | Percentage of Insects Visiting on Flowers (%) ξ | ||
|---|---|---|---|
| 1 Flower | 2 Flowers | ≥3 Flowers | |
| A. cerana cerana | 17.2 ± 5.4 b (154) | 11.2 ± 2.2 b (103) | 71.6 ± 6.8 a (732) |
| V. bicolor | 27.5 ± 7.6 ab (290) | 12.5 ± 4.5 b (88) | 60.0 ± 7.1 a (474) |
| V. velutina | 28.2 ± 10.4 ab (436) | 11.0 ± 2.7 b (140) | 60.8 ± 7.9 a (817) |
| V. ducalis | 41.4 ± 3.4 a (85) | 13.6 ± 2.4 b (28) | 45.0 ± 2.9 a (96) |
| P. zonata | 42.5 ± 9.7 a (198) | 15.6 ± 1.5 a (80) | 42.0 ± 8.6 a (215) |
| Floral Traits | C. osmantha | C. vietnamensis | C. oleifera |
|---|---|---|---|
| Diameter of corolla (mm) | 63.3 ± 1.3 c | 97.8 ± 2.4 a | 86.1 ± 1.1 b |
| Height of stamens (mm) | 10.5 ± 0.3 c | 15.3 ± 0.5 a | 13.8 ± 0.3 b |
| Diameter of stamens (mm) | 14.4 ± 0.6 c | 25.4 ± 1.0 a | 16.8 ± 0.5 b |
| Height of pistils (mm) | 11.6 ± 0.3 b | 15.3 ± 0.5 a | 15.3 ± 0.2 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, W.; Wu, H.; Li, X.; Wei, X.; Lu, W.; Zheng, X. Diversity, Daily Activity Patterns, and Pollination Effectiveness of the Insects Visiting Camellia osmantha, C. vietnamensis, and C. oleifera in South China. Insects 2019, 10, 98. https://doi.org/10.3390/insects10040098
Wei W, Wu H, Li X, Wei X, Lu W, Zheng X. Diversity, Daily Activity Patterns, and Pollination Effectiveness of the Insects Visiting Camellia osmantha, C. vietnamensis, and C. oleifera in South China. Insects. 2019; 10(4):98. https://doi.org/10.3390/insects10040098
Chicago/Turabian StyleWei, Wei, Haipan Wu, Xueyuan Li, Xing Wei, Wen Lu, and Xialin Zheng. 2019. "Diversity, Daily Activity Patterns, and Pollination Effectiveness of the Insects Visiting Camellia osmantha, C. vietnamensis, and C. oleifera in South China" Insects 10, no. 4: 98. https://doi.org/10.3390/insects10040098
APA StyleWei, W., Wu, H., Li, X., Wei, X., Lu, W., & Zheng, X. (2019). Diversity, Daily Activity Patterns, and Pollination Effectiveness of the Insects Visiting Camellia osmantha, C. vietnamensis, and C. oleifera in South China. Insects, 10(4), 98. https://doi.org/10.3390/insects10040098




