The Effects of a Bacterial Endotoxin on Behavior and Sensory-CNS-Motor Circuits in Drosophila melanogaster
Abstract
1. Introduction
2. Materials and Methods
2.1. Behaviors
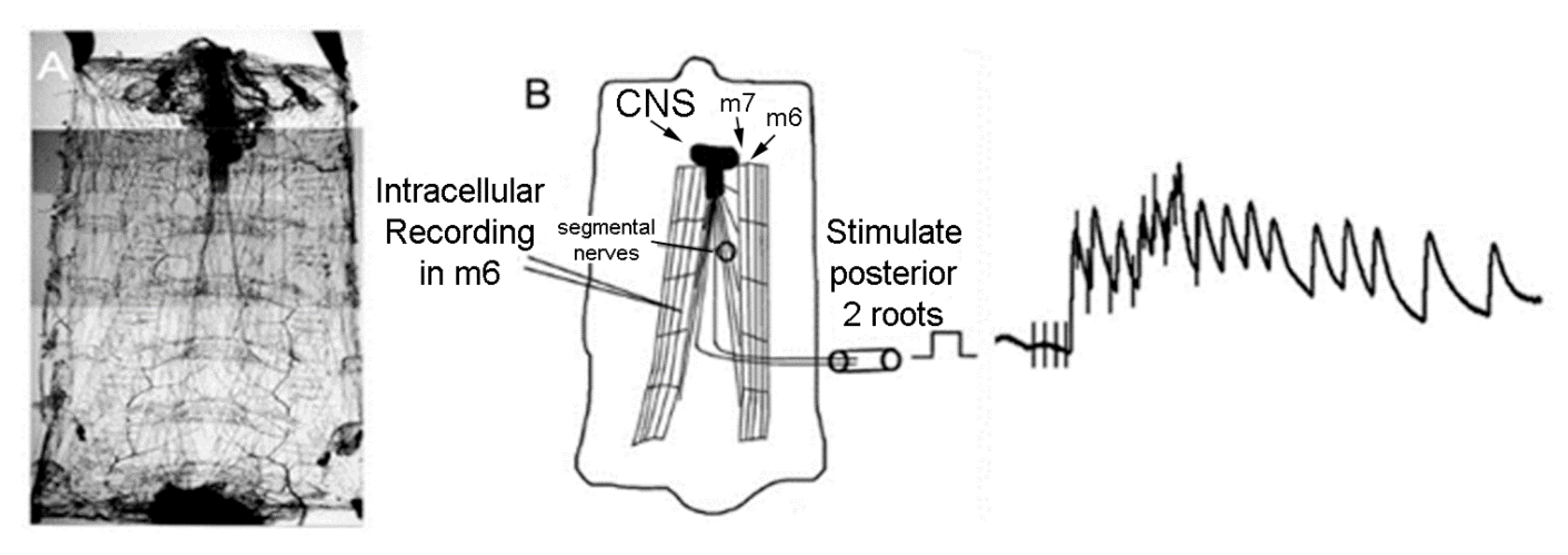
2.2. Electrophysiology
3. Results
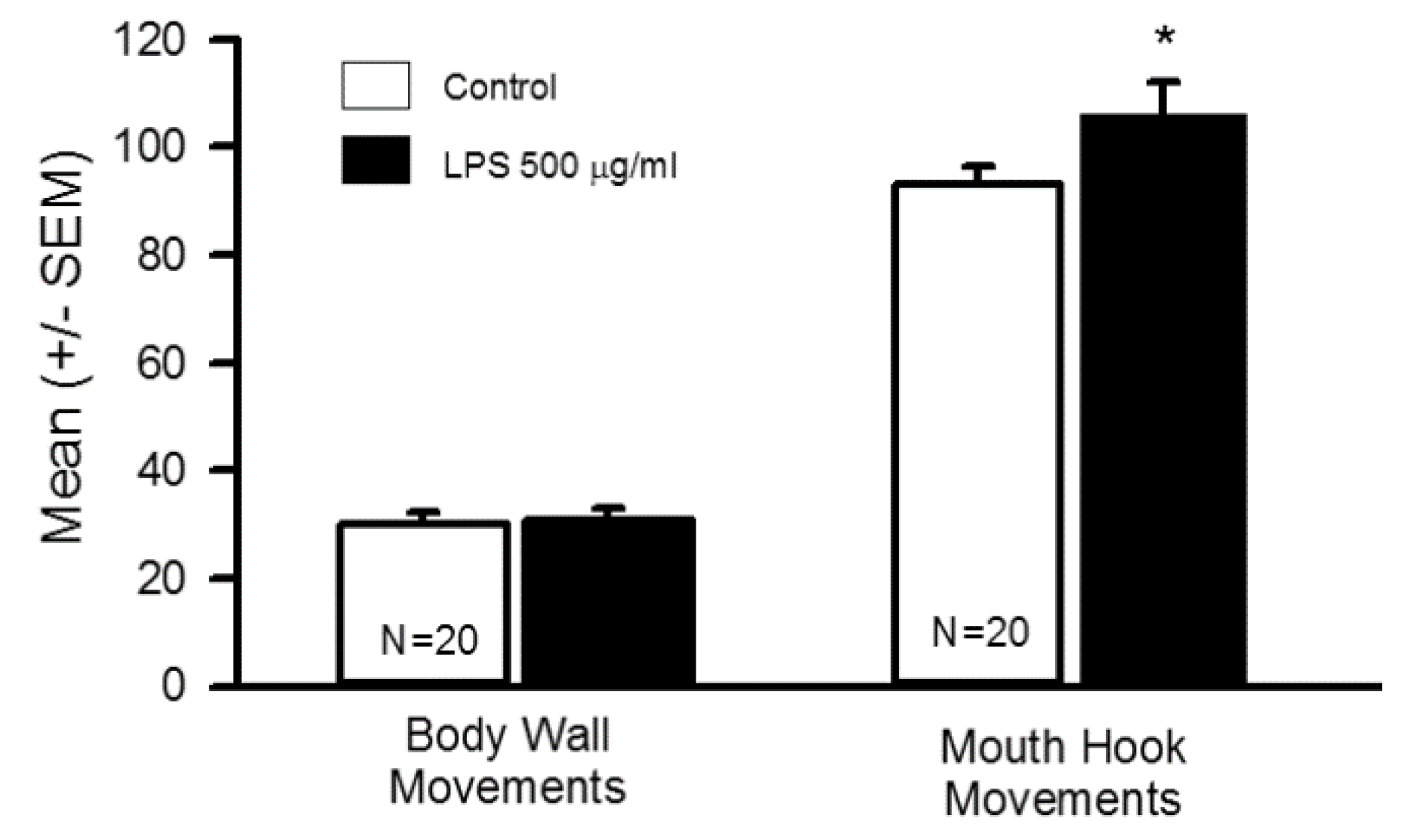
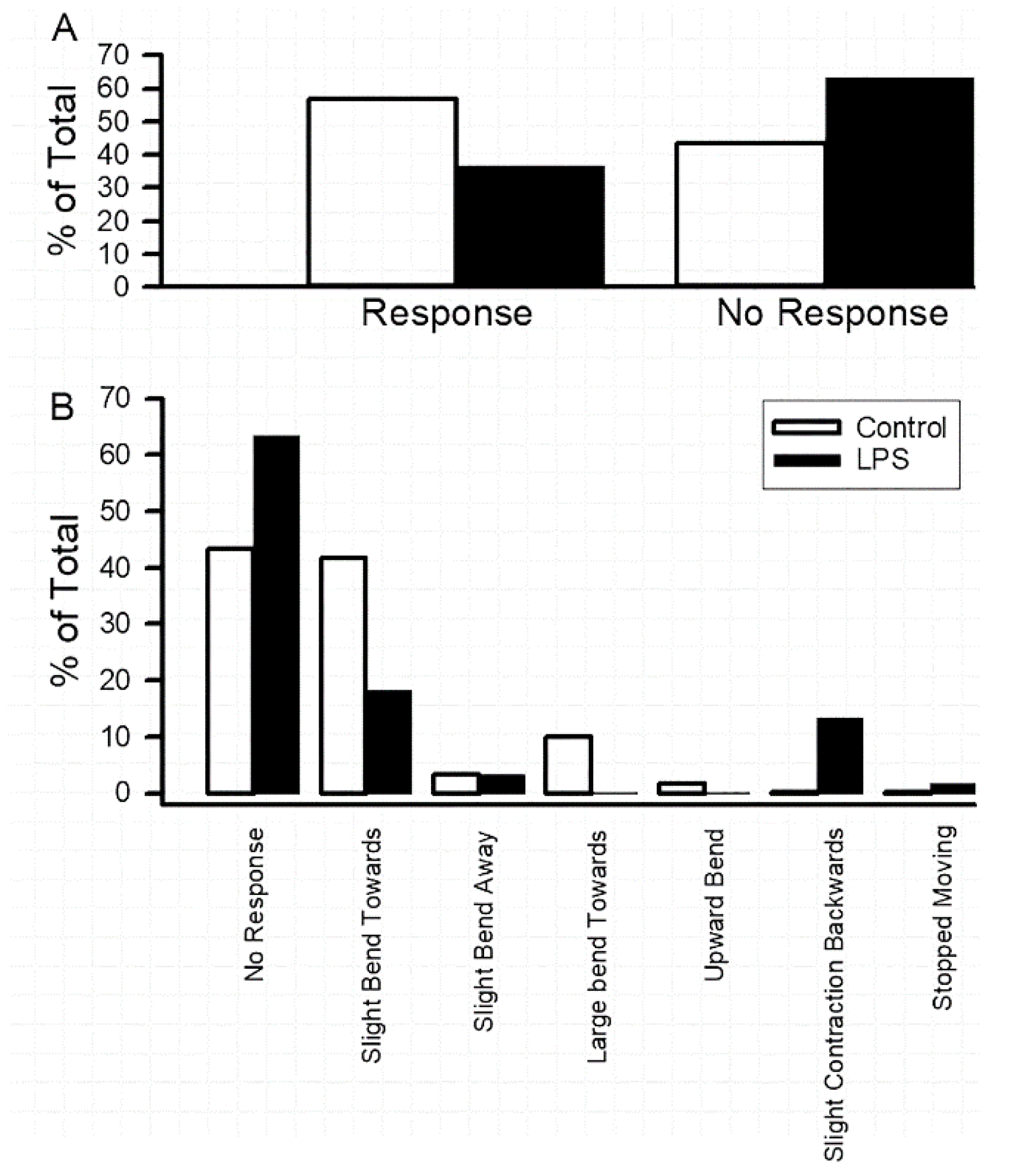
3.1. Impact of Oral Supplementation of LPS on Larval Locomotion and Feeding
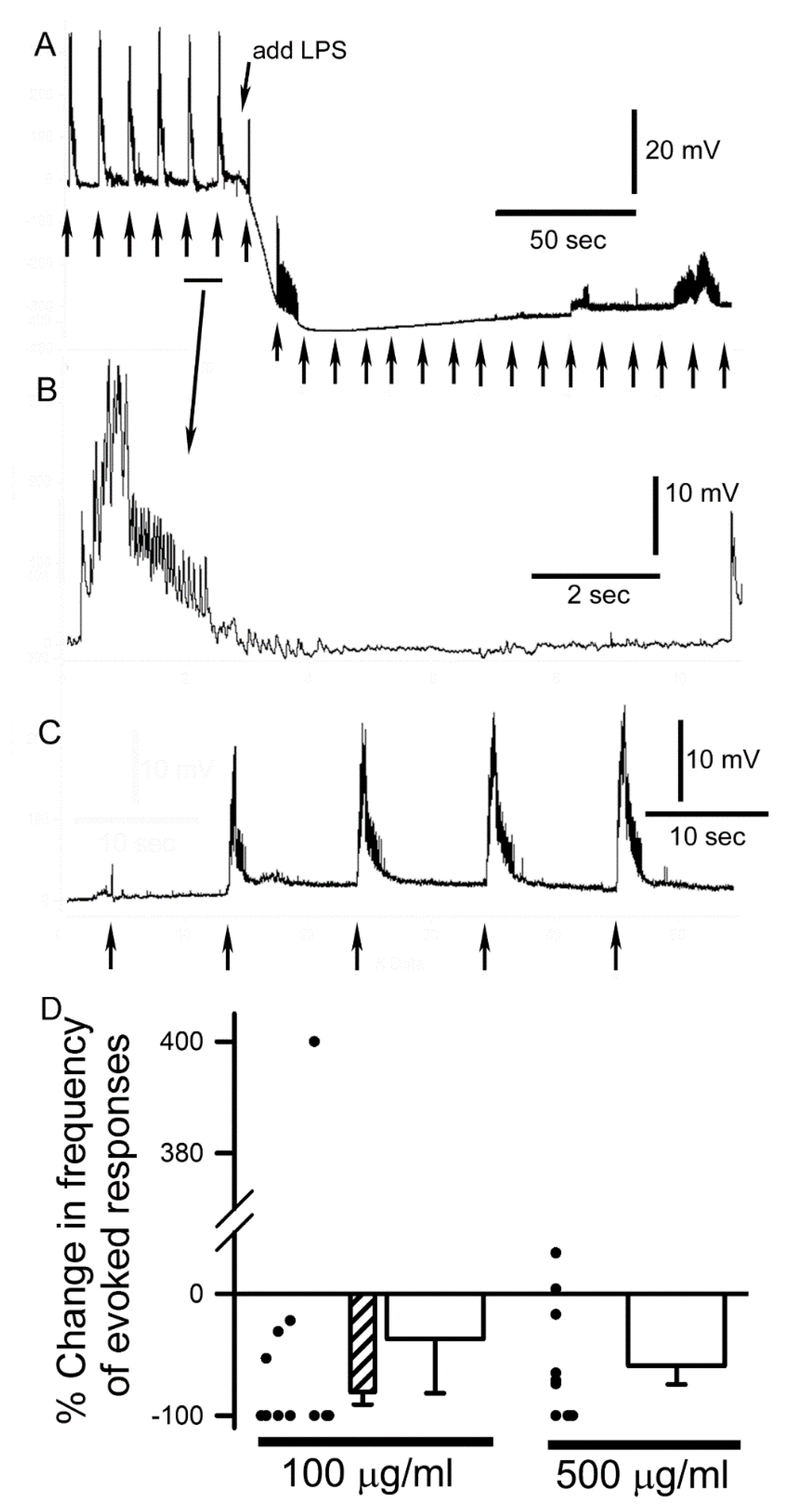
3.2. LPS Modulation of Sensorimotor Circuit Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Data Provided
Funding
Acknowledgments
Conflicts of Interest
References
- CDC Statistics. 2017. Available online: https://www.cdc.gov/nchs/fastats/leading-causes-of-death.htm (accessed on 22 July 2018).
- Da Silva Correia, J.; Soldau, K.; Christen, U.; Tobias, P.S.; Ulevitch, R.J. Lipopolysaccharide is in close proximity to each of the proteins in its membrane receptor complex. transfer from CD14 to TLR4 and MD-2. J. Biol. Chem. 2001, 276, 21129–21135. [Google Scholar] [CrossRef] [PubMed]
- Park, B.S.; Lee, J.O. Recognition of lipopolysaccharide pattern by TLR4 complexes. Exp. Mol. Med. 2013, 45, e66. [Google Scholar] [CrossRef]
- Rodríguez-Ramos, T.; Carpio, Y.; Bolívar, J.; Gómez, L.; Estrada, M.P.; Pendón, C. Nitric oxide synthase-dependent immune response against gram negative bacteria in a crustacean, Litopenaeus vannamei. Fish Shellfish Immunol. 2016, 50, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.Q.; Meng, J.H.; Gao, J.; Xu, Y.H.; Wang, X.W. Identification of a crustacean β-1,3-glucanase related protein as a pattern recognition protein in antibacterial response. Fish Shellfish Immunol. 2018, 80, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Kinoshita, K.; Ashida, M. Purification of a peptidoglycan recognition protein from hemolymph of the silkworm, Bombyx mori. J. Biol. Chem. 1996, 271, 13854–13860. [Google Scholar] [CrossRef]
- Steiner, H. Peptidoglycan recognition proteins: On and off switches for innate immunity. Immunol. Rev. 2004, 198, 83–96. [Google Scholar] [PubMed]
- Kaneko, T.; Goldman, W.E.; Mellroth, P.; Steiner, H.; Fukase, K.; Kusumoto, S.; Harley, W.; Fox, A.; Golenbock, D.; Silverman, N. Monomeric and polymeric gram-negative peptidoglycan but not purified LPS stimulate the Drosophila IMD pathway. Immunity 2004, 20, 637–649. [Google Scholar] [CrossRef]
- Lai, A.G.; Aboobaker, A.A. Comparative genomic analysis of innate immunity reveals novel and conserved components in crustacean food crop species. BMC Genom. 2017, 18, 389. [Google Scholar] [CrossRef] [PubMed]
- Werner, T.; Liu, G.; Kang, D.; Ekengren, S.; Steiner, H.; Hultmark, D. A family of peptidoglycan recognition proteins in the fruit fly Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2000, 97, 13772–13777. [Google Scholar] [CrossRef]
- Harris, N.; Fetter, R.D.; Brasier, D.J.; Tong, A.; Davis, G.W. Molecular interface of neuronal innate immunity, synaptic vesicle stabilization, and presynaptic homeostatic plasticity. Neuron 2018, 100, 1163–1179. [Google Scholar] [CrossRef]
- Soldano, A.; Alpizar, Y.A.; Boonen, B.; Franco, L.; Lopez-Requena, A.; Liu, G.; Mora, N.; Yaksi, E.; Voets, T.; Vennekens, R.; et al. Gustatory-mediated avoidance of bacterial lipopolysaccharides via TRPA1 activation in Drosophila. Elife 2016, 14, e13133. [Google Scholar] [CrossRef] [PubMed]
- Boonen, B.; Alpizar, Y.A.; Meseguer, V.M.; Talavera, K. TRP Channels as Sensors of Bacterial Endotoxins. Toxins 2018, 10, 326. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Rodríguez, M.; de la Fuente, C.; García-Durillo, M.; García-Rodríguez, C.; Villalobos, C.; Núñez, L. Aging and amyloid β oligomers enhance TLR4 expression, LPS-induced Ca2+ responses, and neuron cell death in cultured rat hippocampal neurons. J. Neuroinflamm. 2017, 14, 24. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Zhou, R.; Yan, H.; Yin, H.; Wu, X.; Tan, Y.; Li, L. Metabotropic glutamate receptor 5 modulates calcium oscillation and innate immune response induced by lipopolysaccharide in microglial cell. Neuroscience 2014, 281, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Yu, S.; Li, Y.; Li, P.; Liu, F. Direct binding of Toll-like receptor 4 to ionotropic glutamate receptor N-methyl-D-aspartate subunit 1 induced by lipopolysaccharide in microglial cells N9 and EOC20. Int. J. Mol. Med. 2018, 41, 1323–1330. [Google Scholar] [CrossRef]
- Parnas, I.; Reinhold, R.; Fine, J. Synaptic transmission in the crayfish: Increased release of transmitter substance by bacterial endotoxin. Science 1971, 171, 1153–1155. [Google Scholar] [CrossRef]
- Person, R.J. Bacterial lipopolysaccharide depresses spontaneous, evoked, and ionophore-induced transmitter release at the neuromuscular junction. J. Neurosci. Res. 1979, 4, 105–114. [Google Scholar] [CrossRef]
- Person, R.J. Depression of spontaneous and ionophore-induced neurotransmitter release by Salmonella. Experientia 1979, 35, 801–802. [Google Scholar] [CrossRef]
- Jo, J.H.; Park, E.J.; Lee, J.K.; Jung, M.W.; Lee, C.J. Lipopolysaccharide inhibits induction of long-term potentiation and depression in the rat hippocampal CA1 area. Eur. J. Pharmacol. 2001, 422, 69–76. [Google Scholar] [CrossRef]
- Dasari, S.; Cooper, R.L. Modulation of sensory to motor circuits by serotonin, octopamine, and dopamine in semi-intact Drosophila larva. Neurosci. Res. 2004, 48, 221–227. [Google Scholar] [CrossRef]
- Majeed, Z.R.; Abdeljaber, E.; Soveland, R.; Cornwell, K.; Bankemper, A.; Koch, F.; Cooper, R.L. Modulatory action by the serotonergic system: Behavior and neurophysiology in Drosophila melanogaster. Neural Plast. 2016, 2016, 7291438. [Google Scholar] [CrossRef]
- Anyagaligbo, O.; Bernard, J.; Greenhalgh, A.; Cooper, R.L. The effects of bacterial endotoxin (LPS) on cardiac function in a medicinal blow fly (Phaenicia sericata) and a fruit fly (Drosophila melanogaster). Comp. Biochem. Physiol. C 2019, 217, 15–24. [Google Scholar] [CrossRef]
- Cooper, R.L.; McNabb, M.; Nadolski, J. The effects of a bacterial endotoxin LPS on synaptic transmission at the neuromuscular junction. Heliyon 2019, 5, e01430. [Google Scholar] [CrossRef]
- Neckameyer, W.S. Multiple roles for dopamine in Drosophila development. Dev. Biol. 1996, 176, 209–219. [Google Scholar] [CrossRef]
- Li, H.; Harrison, D.; Jones, G.; Jones, D.; Cooper, R.L. Alterations in development, behavior, and physiology in Drosophila larva that have reduced ecdysone production. J. Neurophysiol. 2001, 85, 98–104. [Google Scholar] [CrossRef]
- Iwaya, A.; Nakagawa, S.; Iwakura, N.; Taneike, I.; Kurihara, M.; Kuwano, T.; Gondaira, F.; Endo, M.; Hatakeyama, K.; Yamamoto, T. Rapid and quantitative detection of blood Serratia marcescens by a real-time PCR assay: Its clinical application and evaluation in a mouse infection model. FEMS Microbiol. Lett. 2005, 248, 163–170. [Google Scholar] [CrossRef][Green Version]
- Titlow, J.S.; Rice, J.; Majeed, Z.R.; Holsopple, E.; Biecker, S.; Cooper, R.L. Anatomical and genotype-specific mechanosensory responses in Drosophila melanogaster larvae. Neurosci. Res. 2014, 83, 54–63. [Google Scholar] [CrossRef]
- Stewart, B.A.; Atwood, H.L.; Renger, J.J.; Wang, J.; Wu, C.F. Improved stability of Drosophila larval neuromuscular preparation in haemolymph-like physiological solutions. J. Comp. Physiol. A 1994, 175, 179–191. [Google Scholar] [CrossRef]
- De Castro, C.; Titlow, J.; Majeed, Z.R.; Cooper, R.L. Analysis of various physiological salines for heart rate, CNS function, and synaptic transmission at neuromuscular junctions in Drosophila melanogaster larvae. J. Comp. Physiol. A 2014, 200, 83–92. [Google Scholar] [CrossRef]
- Lee, J.Y.; Bhatt, D.; Chung, W.Y.; Cooper, R.L. Furthering pharmacological and physiological assessment of the glutamatergic receptors at the Drosophila neuromuscular junction. Comp. Biochem. Physiol. C 2009, 150, 546–555. [Google Scholar] [CrossRef]
- Exton, M.S. Infection induced anorexia: Active host defense strategy. Appetite 1997, 29, 369–383. [Google Scholar] [CrossRef]
- Goldsworthy, G. Locusts as model organisms in which to study immunogen-induced anorectic behaviour. J. Insect Physiol. 2010, 56, 991–997. [Google Scholar] [CrossRef]
- Sullivan, K.; Fairn, E.; Adamo, S.A. Sickness behaviour in the cricket Gryllus texensis: Comparison with animals across phyla. Behav. Processes 2016, 128, 134–143. [Google Scholar] [CrossRef]
- Buchner, E. Gene Expressed in the adult brain of Drosophila and effects of their mutations on behavior-survey of transmitter-related and 2nd messenger-related genes. J. Neurogenet. 1991, 7, 153–192. [Google Scholar] [CrossRef]
- Gorczyca, M.G.; Budnik, V.; White, K.; Wu, C.F. Dual muscarinic and nicotinic action on a motor program in Drosophila. J. Neurobiol. 1991, 22, 391–404. [Google Scholar] [CrossRef]
- Rohrbough, J.; Broadie, K. Electrophysiological analysis of synaptic transmission in central neurons of Drosophila larvae. J. Neurophysiol. 2002, 88, 847–860. [Google Scholar] [CrossRef]
- Su, H.; O’Dowd, D. Fast synaptic currents in Drosophila mushroom body Kenyon cells are mediated by alpha-bungarotoxin-sensitive nicotinic acetylcholine receptors and picrotoxin-sensitive GABA receptors. J. Neurosci. 2003, 8, 9246–9253. [Google Scholar] [CrossRef]
- Titlow, J.S.; Cooper, R.L. Glutamatergic synthesis, recycling, and receptor pharmacology at Drosophila and crustacean neuromuscular junctions. In Biochemical Approaches for Glutamatergic Neurotransmission, Neuromethods; Parrot, S., Denoroy, L., Eds.; Humana Press: New York, NY, USA, 2018; Volume 130, pp. 263–291. [Google Scholar]
- Takagawa, K.; Salvattera, P. Analysis of choline acetyltransferase protein in temperature sensitive mutant flies using newly generated monoclonal antibody. Neurosci. Res. 1996, 24, 237–243. [Google Scholar] [CrossRef]
- Schwaerzel, M.; Monastirioti, M.; Scholz, H.; Friggi-Grelin, F.; Birman, S.; Heisenberg, M. Dopamine and octopamine differentiate between aversive and appetitive olfactory memories in Drosophila. J. Neurosci. 2003, 23, 10495–10502. [Google Scholar] [CrossRef]
- Sinakevitch, I.; Strausfeld, N.J. Comparison of octopamine-like immunoreactivity in the brains of the fruit fly and blow fly. J. Comp. Neurol. 2006, 494, 460–475. [Google Scholar] [CrossRef]
- Honjo, K.; Furukubo-Tokunaga, K. Distinctive neuronal networks and biochemical pathways for appetitive and aversive memory in Drosophila larvae. J. Neurosci. 2009, 29, 852–862. [Google Scholar] [CrossRef]
- Huser, A.; Eschment, M.; Güllü, N.; Collins, K.A.; Böpple, K.; Pankevych, L.; Rolsing, E.; Thum, A.S. Anatomy and behavioral function of serotonin receptors in Drosophila melanogaster larvae. PLoS ONE 2017, 12, e0181865. [Google Scholar] [CrossRef]
- Gao, F.; Liu, Z.; Ren, W.; Jiang, W. Acute lipopolysaccharide exposure facilitates epileptiform activity via enhanced excitatory synaptic transmission and neuronal excitability in vitro. Neuropsychiatr. Dis. Treat. 2014, 10, 1489–1495. [Google Scholar] [CrossRef]
- Abudara, V.; Roux, L.; Dallérac, G.; Matias, I.; Dulong, J.; Mothet, J.P.; Rouach, N.; Giaume, C. Activated microglia impairs neuroglial interaction by opening Cx43 hemichannels in hippocampal astrocytes. Glia 2014, 63, 795–811. [Google Scholar] [CrossRef]
- Zhang, J.; Malik, A.; Choi, H.B.; Ko, R.W.; Dissing-Olesen, L.; MacVicar, B.A. Microglial CR3 activation triggers long-term synaptic depression in the hippocampus via NADPH oxidase. Neuron 2014, 82, 195–207. [Google Scholar] [CrossRef]
- Sullivan, M.N.; Earley, S. TRP channel Ca(2+) sparklets: Fundamental signals underlying endothelium-dependent hyperpolarization. Am. J. Physiol. Cell Physiol. 2013, 305, C999–C1008. [Google Scholar] [CrossRef]
- Gottar, M.; Gobert, V.; Michel, T.; Belvin, M.; Duyk, G.; Hoffmann, J.A.; Ferrandon, D.; Royet, J. The Drosophila immune response against Gram-negative bacteria is mediated by a peptidoglycan recognition protein. Nature 2002, 416, 640–644. [Google Scholar] [CrossRef]
- Takehana, A.; Katsuyama, T.; Yano, T.; Oshima, Y.; Takada, H.; Aigaki, T.; Kurata, S. Overexpression of a pattern-recognition receptor, peptidoglycanrecognition protein-LE, activates imd/relishmediated antibacterial defense and the prophenoloxidase cascade in Drosophila larvae. Proc. Natl. Acad. Sci. USA 2002, 99, 13705–13710. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Istas, O.; Greenhalgh, A.; Cooper, R. The Effects of a Bacterial Endotoxin on Behavior and Sensory-CNS-Motor Circuits in Drosophila melanogaster. Insects 2019, 10, 115. https://doi.org/10.3390/insects10040115
Istas O, Greenhalgh A, Cooper R. The Effects of a Bacterial Endotoxin on Behavior and Sensory-CNS-Motor Circuits in Drosophila melanogaster. Insects. 2019; 10(4):115. https://doi.org/10.3390/insects10040115
Chicago/Turabian StyleIstas, Oscar, Abigail Greenhalgh, and Robin Cooper. 2019. "The Effects of a Bacterial Endotoxin on Behavior and Sensory-CNS-Motor Circuits in Drosophila melanogaster" Insects 10, no. 4: 115. https://doi.org/10.3390/insects10040115
APA StyleIstas, O., Greenhalgh, A., & Cooper, R. (2019). The Effects of a Bacterial Endotoxin on Behavior and Sensory-CNS-Motor Circuits in Drosophila melanogaster. Insects, 10(4), 115. https://doi.org/10.3390/insects10040115





