Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Inert Dust
2.2. Essential Oils and Chemical Analyses
2.3. Essential Oils Sensory Characterization
2.4. Laboratory Toxicity Bioassays
2.5. Medium-Scale Toxicity Bioassays
2.6. Statistics and Data Analyses
3. Results
3.1. Essential Oils Composition
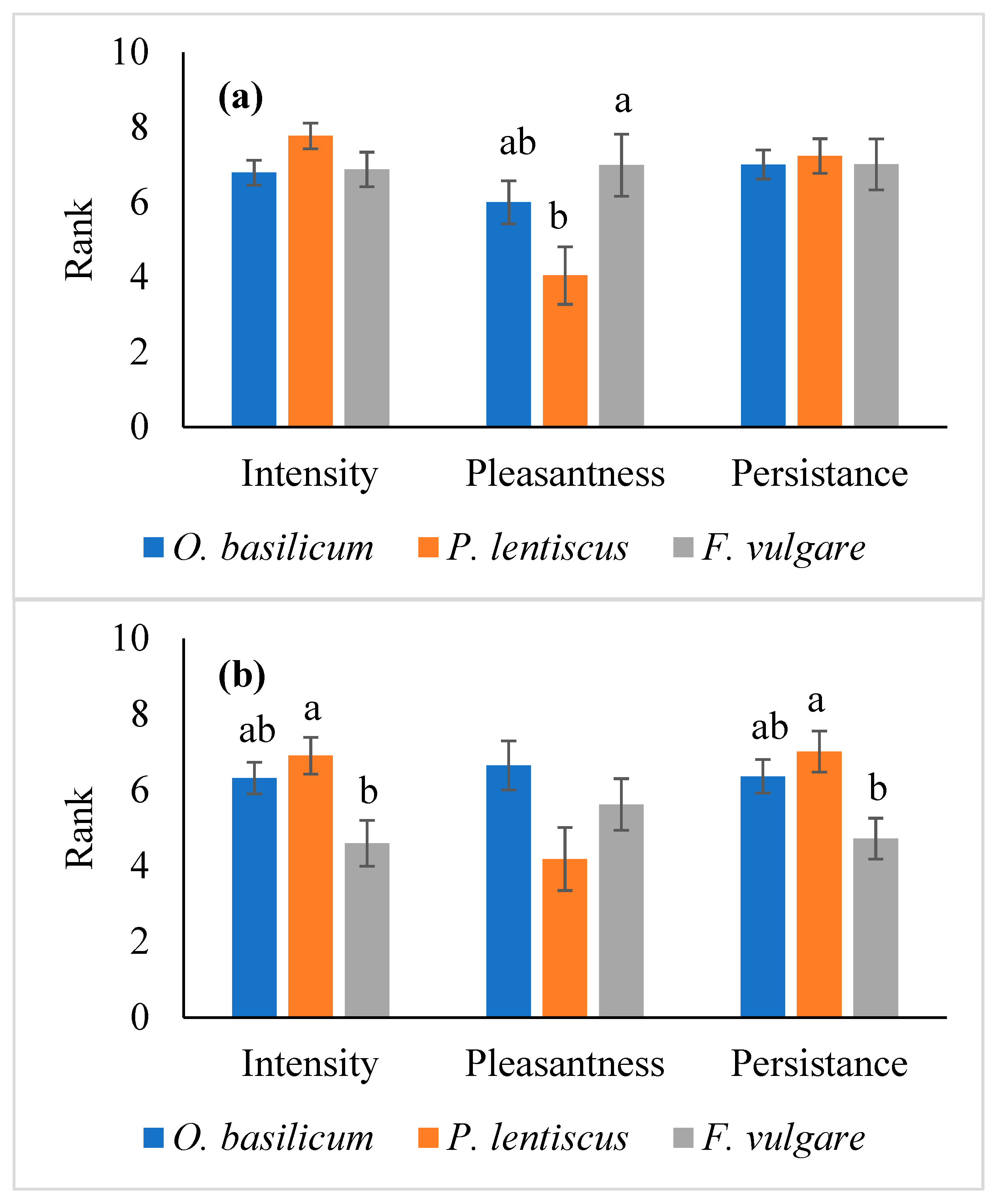
3.2. Essential Oils Sensory Characterization
3.3. Laboratory Toxicity Bioassays
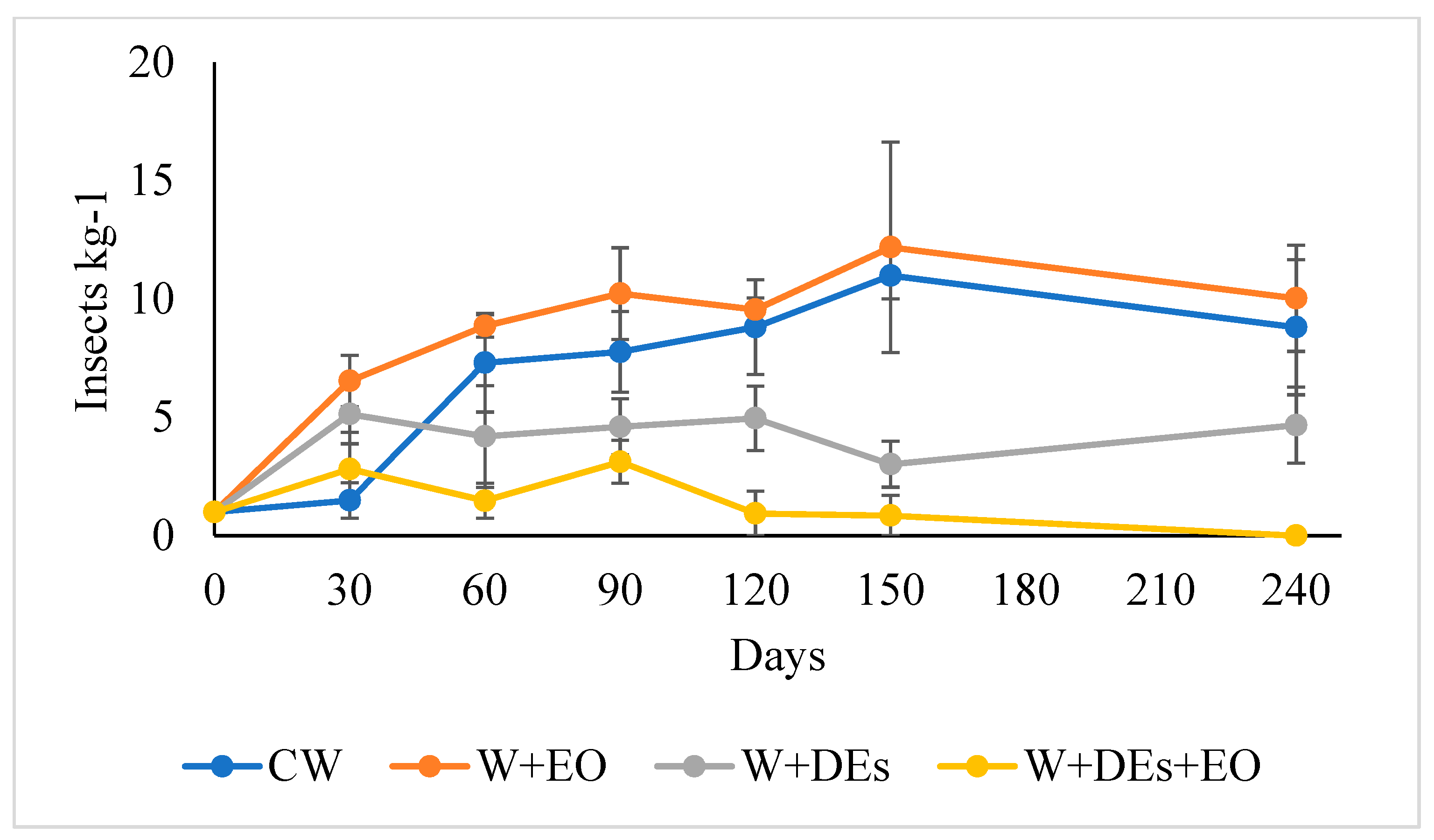
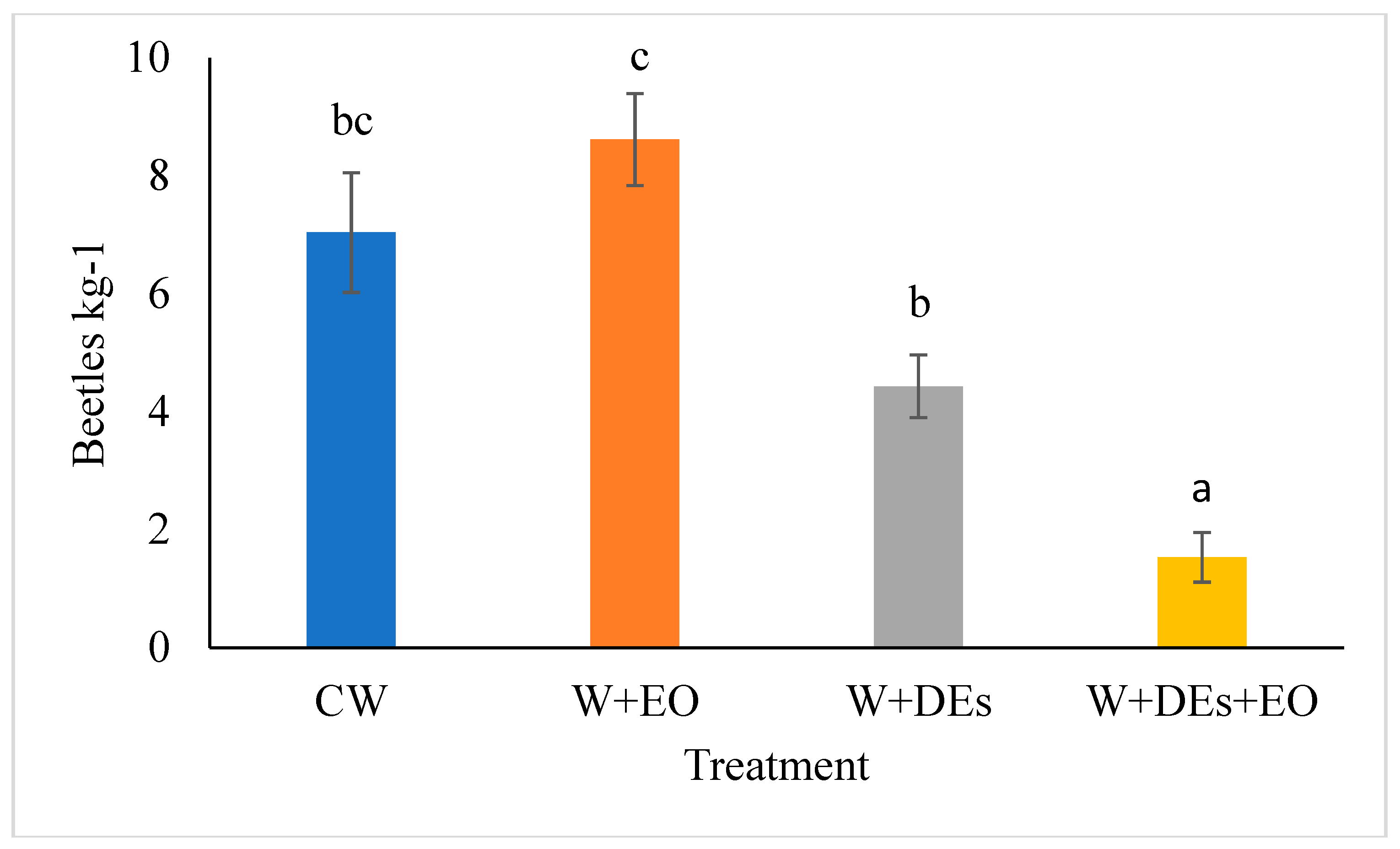
3.4. Medium-Scale Toxicity Bioassays
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hernandez Nopsa, J.F.; Daglish, G.J.; Hagstrum, D.W.; Leslie, J.F.; Phillips, T.W.; Scoglio, C.; Thomas-Sharma, S.; Walter, G.H.; Garrett, K.H. Ecological Networks in Stored Grain: Key Postharvest Nodes for Emerging Pests, Pathogens, and Mycotoxins. BioScience 2015, 65, 985–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cox, P.D. Potential for using semiochemicals to protect stored products from insect infestation. J. Stored Prod. Res. 2004, 40, 1–25. [Google Scholar]
- Athanassiou, C.G.; Kavallieratos, N.G.; Meletsis, C.M. Insecticidal effect of three diatomaceous earth formulations, applie alone or in combination, against three stored- product beetle species on wheat and maize. J. Stored Prod. Res. 2007, 43, 330–334. [Google Scholar] [CrossRef]
- Riudavets, J.; Pons, M.J.; Gabarra, R.; Castañé, C.; Alomar, O.; Vega, L.F.; Guri, S. The toxicity effects of atmospheres with high content of carbon dioxide with addition of sulphur dioxide on two stored-product pest species: Sitophilus oryzae and Tribolium confusum. J. Stored Prod. Res. 2014, 57, 58–62. [Google Scholar] [CrossRef]
- Campolo, O.; Verdone, M.; Laudani, F.; Malacrinò, A.; Chiera, E.; Palmeri, V. Response of four stored products insects to a structural heat treatment in a flourmill. J. Stored Prod. Res. 2013, 54, 54–58. [Google Scholar] [CrossRef]
- Campolo, O.; Malacrinò, A.; Zappalà, L.; Laudani, F.; Chiera, E.; Serra, D.; Russo, M.; Palmeri, V. Fumigant bioactivity of five citrus essential oils against Tribolium confusum. Phytoparasitica 2014, 42, 223–233. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Bedini, S.; Flamini, G.; Girardi, J.; Cosci, F.; Conti, B. Not just for beer: Evaluation of spent hops (Humulus lupulus) as a source of eco-friendly repellents for insect pests of stored foods. J. Pest. Sci. 2015, 88, 583–592. [Google Scholar] [CrossRef]
- Bedini, S.; Flamini, G.; Cosci, F.; Ascrizzi, R.; Benelli, G.; Conti, B. Cannabis sativa and Humulus lupulus essential oils as novel control tools against the invasive mosquito Aedes albopictus and fresh water snail Physella acuta. Ind. Crops Prod. 2016, 85, 318–323. [Google Scholar] [CrossRef]
- Bedini, S.; Flamini, G.; Cosci, F.; Ascrizzi, R.; Echeverria, M.C.; Guidi, L.; Landi, M.; Lucchi, A.; Conti, B. Artemisia spp. essential oils against the disease-carrying blowfly Calliphora vomitoria. Parasites Vector 2017, 10, 80. [Google Scholar]
- Conti, B.; Benelli, G.; Leonardi, M.; Afifi, F.U.; Cervelli, C.; Profeti, R.; Pistelli, L.; Canale, A. Repellent effect of Salvia dorisiana, S. longifolia, and S. sclarea (Lamiaceae) essential oils against the mosquito Aedes albopictus Skuse (Diptera: Culicidae). Parasitol. Res. 2012, 111, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils from seven aromatic plants grown in Colombia against Sitophilus zeamais Motschulsky (Coleoptera). J. Stored Prod. Res. 2009, 45, 212–214. [Google Scholar] [CrossRef]
- Bedini, S.; Flamini, G.; Ascrizzi, R.; Venturi, F.; Ferroni, G.; Bader, A.; Girardi, J.; Conti, B. Essential oils sensory quality and their bioactivity against the mosquito Aedes albopictus. Sci. Rep. 2018, 8, 17857. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.A.; Khan, A.A. Use of diatomaceous earth for the management of stored-product pests. Int. J. Pest Manag. 2014, 60, 100–113. [Google Scholar] [CrossRef]
- Ziaee, M.; Moharramipour, S.; Francikowski, J. The synergistic effects of Carum copticum essential oil on diatomaceous earth against Sitophilus granarius and Tribolium confusum. J. Asia-Pac. Entomol. 2014, 17, 817–822. [Google Scholar] [CrossRef]
- Yang, F.-L.; Liang, G.-W.; Xu, Y.-J.; Lu, Y.-Y.; Zeng, L. Diatomaceous earth enhances the toxicity of garlic, Allium sativum, essential oil against stored-product pests. J. Stored Prod. Res. 2010, 46, 118–123. [Google Scholar] [CrossRef]
- Khorrami, F.; Valizadegan, O.; Forouzan, M.; Soleymanzade, A. The antagonistic/synergistic effects of some medicinal plant essential oils, extracts and powders combined with Diatomaceous earth on red flour beetle, Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). Arch. Phytopathol Plant Prot. 2018, 51, 685–695. [Google Scholar] [CrossRef]
- Bougherra-Nehaoua, H.H.; Bedini, S.; Cosci, F.; Flamini, G.; Belhamel, K.; Conti, B. Enhancing the insecticidal efficacy of inert dusts against stored food insect pest by the combined action with essential oils. Integr. Prot. Stored Prod. IOBC-WPRS Bull. 2015, 111, 31–38. [Google Scholar]
- Islam, M.S.; Hasan, M.M.; Lei, C.; Mucha-Pelzer, T.; Mewis, I.; Ulrichs, C. Direct and admixture toxicity of diatomaceous earth and monoterpenoids against the storage pests Callosobruchus maculatus (F.) and Sitophilus oryzae (L.). J. Pest Sci. 2010, 83, 105–112. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy; Allured: Carol Stream, NC, USA, 1995. [Google Scholar]
- Davies, N.W. Gas chromatographic retention indexes of monoterpenes and sesquiterpenes on methyl silicone and Carbowax 20M phases. J. Chromatogr. 1990, 503, 1–24. [Google Scholar] [CrossRef]
- Jennings, W.; Shibamoto, T. Qualitative Analysis of Flavor and Fragrance Volatiles by Glass Capillary Chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Masada, Y. Analysis of Essential Oils by Gas Chromatography and Mass Spectrometry; J. Wiley & Sons: New York, NY, USA, 1976. [Google Scholar]
- Stenhagen, E.; Abrahamsson, S.; McLafferty, F.W. Registry of Mass Spectral Data; J. Wiley & Sons: New York, NY, USA, 1974. [Google Scholar]
- Swigar, A.A.; Silverstein, R.M. Monoterpenes; Aldrich Chemical Company: Milwaukee, WI, USA, 1981. [Google Scholar]
- Sanmartin, C.; Venturi, F.; Macaluso, M.; Nari, A.; Quartacci, M.F.; Sgherri, C.; Flamini, G.; Taglieri, I.; Ascrizzi, R.; Andrich, G.; et al. Preliminary Results About the Use of Argon and Carbon Dioxide in the Extra Virgin Olive Oil (EVOO) Storage to Extend Oil Shelf Life: Chemical and Sensorial Point of View. Eur. J. Lipid Sci. Technol. 2018, 120, 1800156. [Google Scholar] [CrossRef]
- Venturi, F.; Sanmartin, C.; Taglieri, I.; Xiaoguo, Y.; Quartacci, M.F.; Sgherri, C.; Andrich, G.; Zinnai, A. A kinetic approach to describe the time evolution of red wine as a function of packaging conditions adopted: Influence of closure and storage position. Food Packag. Shelf Life 2017, 13, 44–48. [Google Scholar] [CrossRef]
- Sun, Y.P.; Johnson, E.R. Analysis of joint action of insecticides against house flies. J. Econ. Entomol. 1960, 53, 887–892. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Upadhyay, S. Repellent and insecticidal activities of Hyptis suaveolens (Lamiaceae) leaf essential oil against four stored-grain coleopteran pests. Int. J. Trop. Insect Sci. 2009, 29, 219–228. [Google Scholar] [CrossRef]
- Bougherra, H.H.; Bedini, S.; Flamini, G.; Cosci, F.; Belhamel, K.; Conti, B. Pistacia lentiscus essential oil has repellent effect against three major insect pests of pasta. Ind. Crops Prod. 2015, 63, 249–255. [Google Scholar] [CrossRef]
- Gutierrez, J.; Barry-Ryan, C.; Bourk, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol. 2008, 124, 91–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Díez, J.A.; Falco, V.; Fraqueza, M.J.; Patarata, L. Chemical characterization and antimicrobialproperties of herbs and spices essential oils against pathogens and spoilage bacteria associated to dry-cured meat products. J. Essent. Oil Res. 2017, 29, 117–125. [Google Scholar] [CrossRef]
- Worwood, V.A. The Complete Book of Essential Oils and Aromatherapy, Revised and Expanded: Over 800 Natural, Nontoxic, and Fragrant Recipes to Create Health, Beauty, and Safe Home and Work Environments; New World Library: Novato, CA, USA, 2016. [Google Scholar]
- Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Bachrouch, O.; Jemaa, J.M.B.; Talou, T.; Marzouk, B.; Abderraba, M. Fumigant toxicity of Pistacia lentiscus essential oil against Tribolium castaneum and Lasioderma serricorne. Bull. Insectol. 2010, 63, 129–135. [Google Scholar]
- Keita, S.M.; Vincent, C.; Schmit, J.P.; Arnason, J.T.; Bélanger, A. Efficacy of essential oil of Ocimum basilicum L. and O. gratissimum L. applied as an insecticidal fumigant and powder to control Callosobruchus maculatus (Fab.) [Coleoptera: Bruchidae]. J. Stored Prod. Res. 2001, 37, 339–349. [Google Scholar] [CrossRef]
- Shaaya, E.; Kostjukovski, M.; Eilberg, J.; Sukprakarn, C. Plant oils as fumigants and contact insecticides for the control of stored-product insects. J. Stored Prod. Res. 1997, 33, 7–15. [Google Scholar] [CrossRef]
- Zhao, N.N.; Zhou, L.; Liu, Z.L.; Du, S.S.; Deng, Z.W. Evaluation of toxicities of some common spices essential oils from China against Liposcelis bostrychophila. Food Control 2012, 26, 486–490. [Google Scholar] [CrossRef]
- Seada, M.A.; Arab, R.A.; Adel, I.; Seif, A.I. Bioactivity of essential oils of basil, fennel, and geranium against Sitophilus oryzae and Callosobruchus maculatus: Evaluation of repellency, progeny production and residual activity. Egypt. J. Exp. Biol. (Zool.) 2016, 12, 1–12. [Google Scholar]
- Al-Sinjari, S.H.S.; Al-Attar, H.J. Toxic Effects of Essential oils of Elattaria cardamomum L. and Lambda-Cyhalothrin on Tribolium confusum (Duval). SJUOZ 2015, 3, 15–26. [Google Scholar] [CrossRef]
- Finch, S. Chemical attraction of plant feeding insects to plants. In Applied Biology; Coaker, T.H., Ed.; Academic Press: London, UK, 1980; Volume 5, pp. 67–143. [Google Scholar]
- Ahmad, S. Herbivorous Insects: Host-Seeking Behaviour and Mechanisms; Academic Press: New York, NY, USA, 1983; 257p. [Google Scholar]
- Górski, R. Effectiveness of natural essential oils in the monitoring of greenhouse whitefly (Trialeurodes vaporariorum Westwood). Folia Hortic. 2004, 16, 183–187. [Google Scholar]
- Athanassiou, C.G.; Vayias, B.J.; Dimizas, C.B.; Kavallieratos, N.G.; Papagregoriou, A.S.; Buchelos, C.T. Insecticidal efficacy of diatomaceous earth against Sitophilus oryzae (L.) (Coleoptera: Curculionidae) and Tribolium confusum du Val (Coleoptera: Tenebrionidae) on stored wheat: Influence of dose rate, temperature and exposure interval. J. Stored Prod. Res. 2005, 41, 47–55. [Google Scholar] [CrossRef]
- Wigglesworth, L.A. Flexure and Torsion of a Circular Shaft with two Cracks. Proc. Lond. Math. Soc. 1942, 15, 20–36. [Google Scholar] [CrossRef]
- Beament, J.W.L. The cuticular lipoids of insects. J. Exp. Biol. 1945, 21, 115–131. [Google Scholar]
- Malia, H.A.E.; Rosi-Denadai, C.A.; Guedes, N.M.P.; Martins, G.F.; Guedes, R.N.C. Diatomaceous earth impairment of water balance in the maize weevil, Sitophilus zeamais. J. Pest Sci 2016, 89, 945–954. [Google Scholar] [CrossRef]
| Chemical Classes | Relative Abundance (%) | ||
|---|---|---|---|
| Pl | Ob | Fv | |
| Monoterpene hydrocarbons | 91.2 | 2.8 | 13.6 |
| Oxygenated monoterpenes | 3.8 | 8.7 | 2.5 |
| Sesquiterpene hydrocarbons | 1.5 | 6.4 | 0.1 |
| Oxygenated sesquiterpenes | 0.4 | 1.6 | - |
| Diterpene hydrocarbons | 0.2 | - | - |
| Phenylpropanoids | 0.3 | 80.1 | 81.6 |
| Non-terpene derivatives | 1.0 | - | 2.3 |
| Total identified (%) | 98.3 | 99.7 | 100.0 |
| O. basilicum | P. lentiscus | F. vulgare | ||||
|---|---|---|---|---|---|---|
| EO | EO + wheat | EO | EO + wheat | EO | EO + wheat | |
| Vegetative odors | mint | mint | mint | mint | anis | anis |
| thyme | thyme | thyme | thyme | floral | floral | |
| caper | caper | caper | caper | fennel | fennel | |
| licorice | licorice | calamint | calamint | licorice | licorice | |
| calamint | calamint | |||||
| Spicy odors | balsamic | balsamic | balsamic | balsamic | sweet | sweet |
| spices | spices | spices | resin | balsamic | balsamic | |
| resin | resin | resin | spices | spices | ||
| Off-flavors | chemical | |||||
| Treatment | CTC a | LC50 b | Slope | Intercept | χ2 (df) | Sig. c |
|---|---|---|---|---|---|---|
| Des d | 36.47 (24.84–46.72) a | 1.783 ± 0.404 | −2.785 ± 0.687 | 1.912 (1) | 0.167 | |
| Ob e | 43.42 (35.08–52.32) | 2.447 ± 0.422 | −4.007 ± 0.718 | 0.036 (1) | 0.849 | |
| ObDEs f | 1.36 | 26.78 (19.23–32.82) | 2.643 ± 0.478 | −3.774 ± 0.782 | 0.007 (1) | 0.934 |
| Pl g | 36.36 (26.42–51.76) | 0.945 ± 0.197 | −1.475 ± 0.307 | 1.089 (3) | 0.780 | |
| PlDEs h | 1.47 | 24.79 (19.57–29.11) | 3.843 ± 0.707 | −5.358 ± 1.072 | 0.627 (1) | 0.429 |
| Fv i | 77.59 (58.92–120.12) | 2.104 ± 0.707 | −3.976 ± 1.309 | 0.033 (1) | 0.860 | |
| FvDEs l | 3.35 | 23.18 (13.14–29.88) | 2.669 ± 0.741 | −3.644 ± 1.141 | 0.003 (1) | 0.960 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pierattini, E.C.; Bedini, S.; Venturi, F.; Ascrizzi, R.; Flamini, G.; Bocchino, R.; Girardi, J.; Giannotti, P.; Ferroni, G.; Conti, B. Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects. Insects 2019, 10, 114. https://doi.org/10.3390/insects10040114
Pierattini EC, Bedini S, Venturi F, Ascrizzi R, Flamini G, Bocchino R, Girardi J, Giannotti P, Ferroni G, Conti B. Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects. Insects. 2019; 10(4):114. https://doi.org/10.3390/insects10040114
Chicago/Turabian StylePierattini, Erika Carla, Stefano Bedini, Francesca Venturi, Roberta Ascrizzi, Guido Flamini, Rossella Bocchino, Jessica Girardi, Paolo Giannotti, Giuseppe Ferroni, and Barbara Conti. 2019. "Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects" Insects 10, no. 4: 114. https://doi.org/10.3390/insects10040114
APA StylePierattini, E. C., Bedini, S., Venturi, F., Ascrizzi, R., Flamini, G., Bocchino, R., Girardi, J., Giannotti, P., Ferroni, G., & Conti, B. (2019). Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects. Insects, 10(4), 114. https://doi.org/10.3390/insects10040114









