Local and Landscape Drivers of Carabid Activity, Species Richness, and Traits in Urban Gardens in Coastal California
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Local and Landscape Factors
2.3. Carabid Sampling, Identification, and Traits
2.4. Statistical Analysis
3. Results
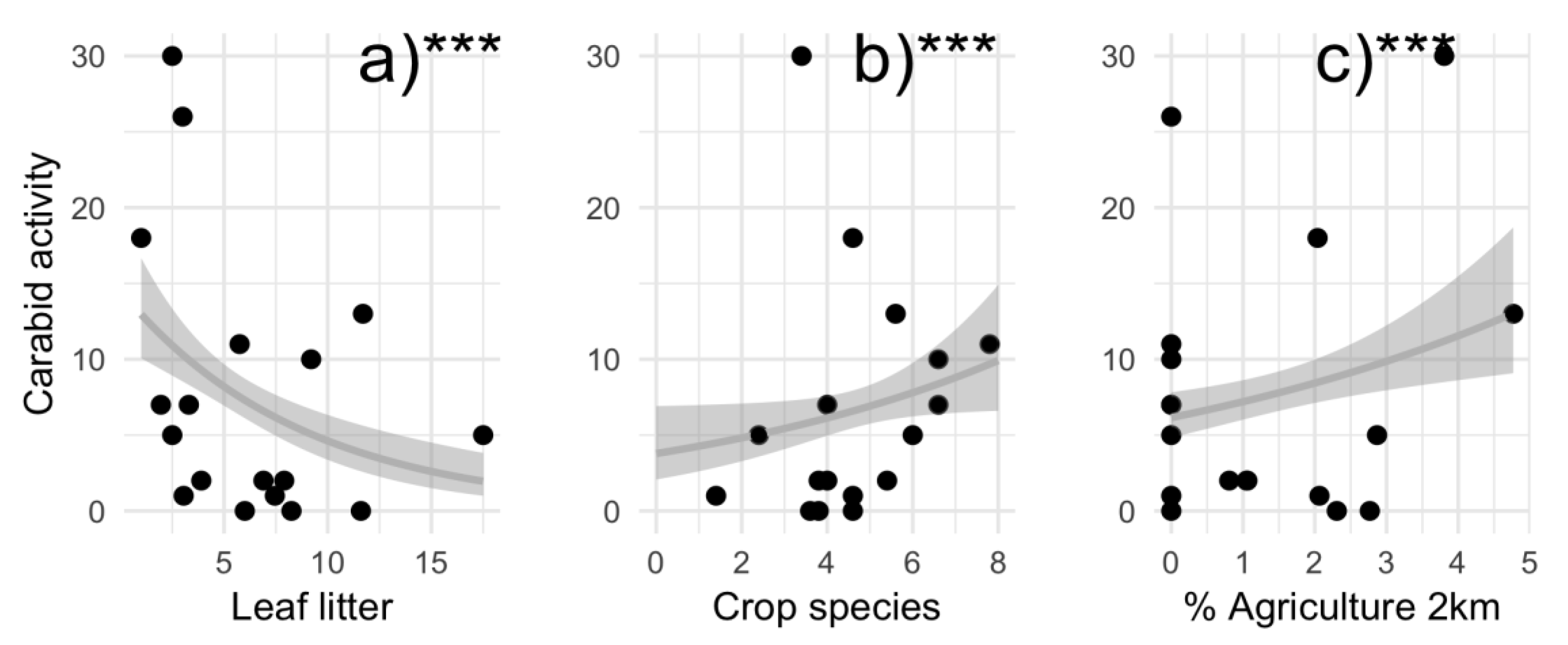
3.1. Local and Landscape Drivers of Carabid Activity and Richness
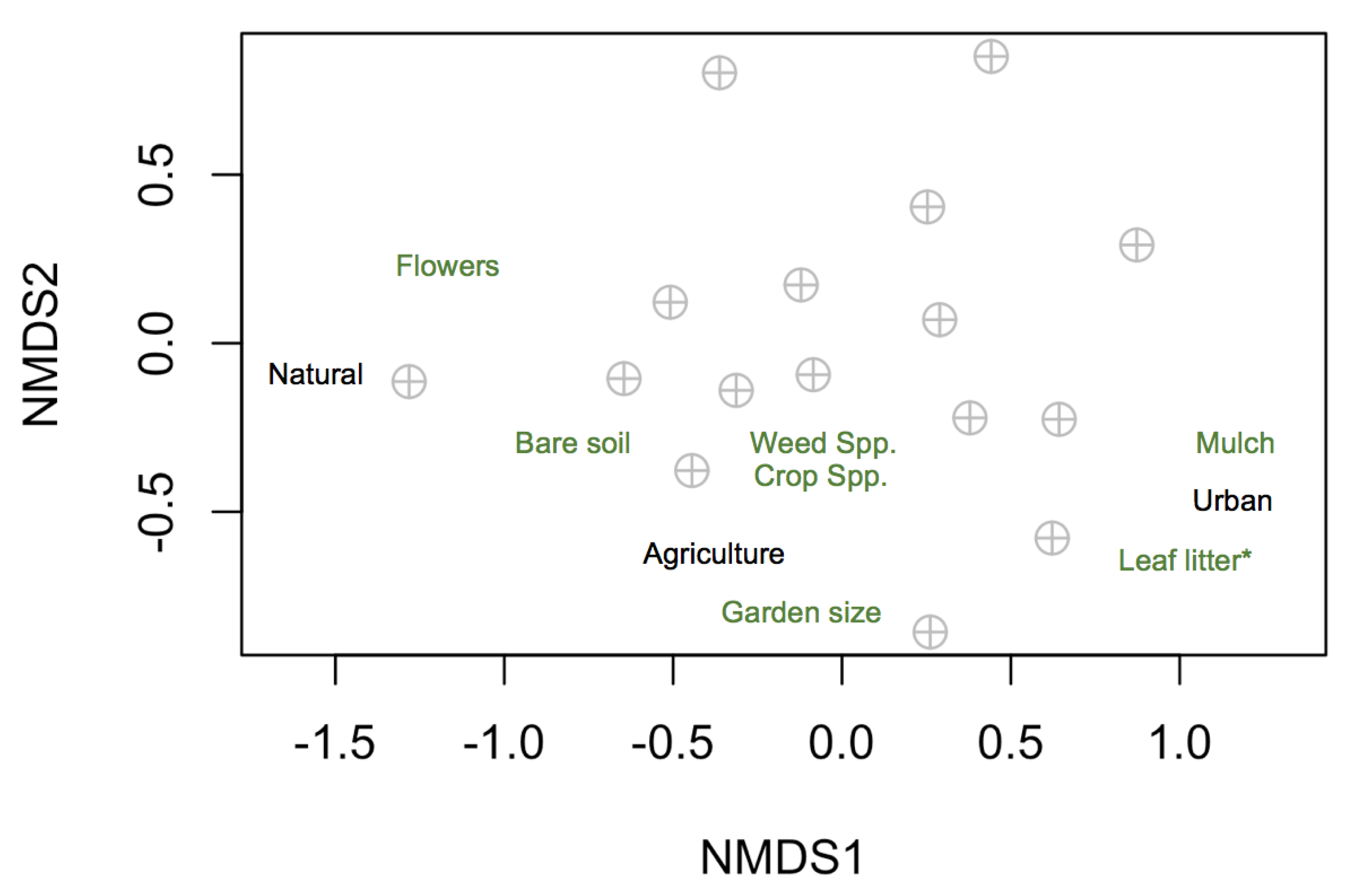
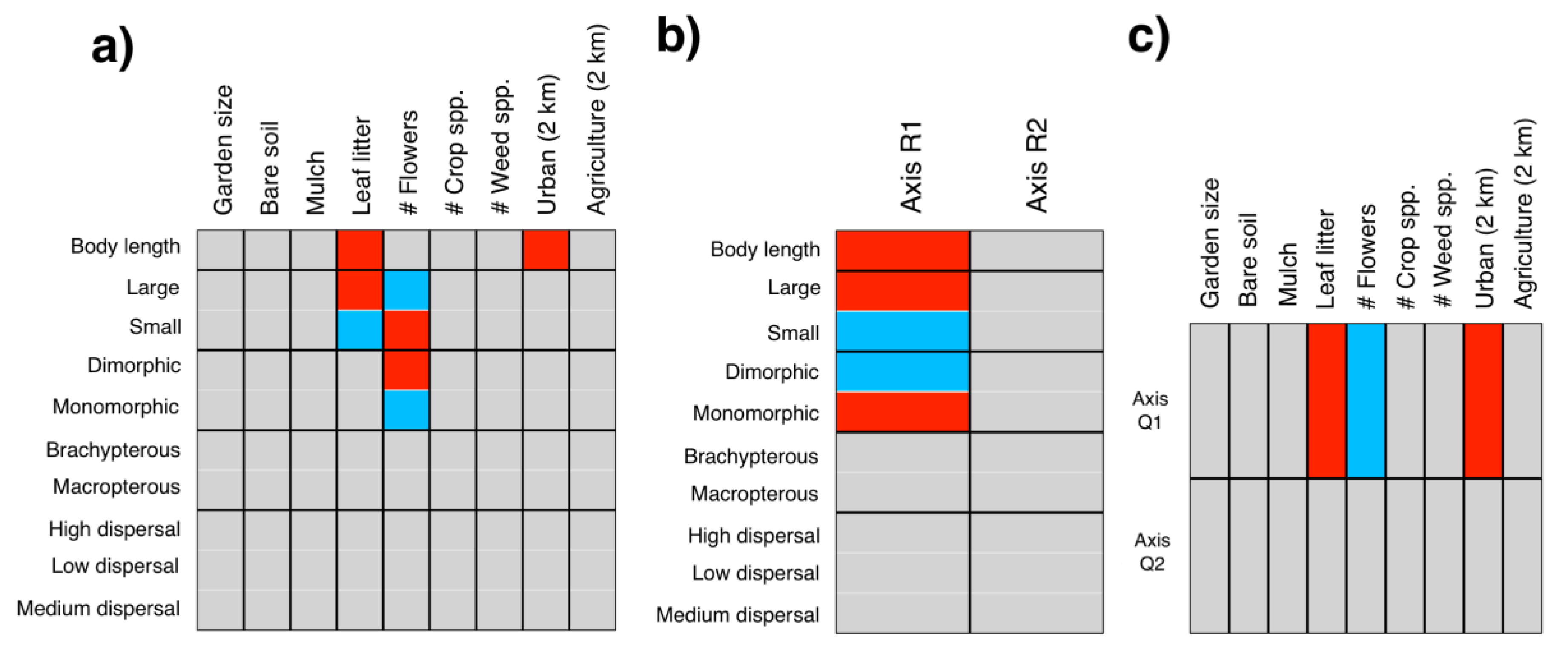
3.2. Local and Landscape Drivers of Community and Trait Composition
4. Discussion
4.1. Local and Landscape Drivers of Carabid Activity and Richness
4.2. Local and Landscape Drivers of Community and Trait Composition
4.3. Implications for Pest Control in Urban Gardens
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alberti, M. Maintaining ecological integrity and sustaining ecosystem function in urban areas. Curr. Opin. Environ. Sustain. 2010, 2, 178–184. [Google Scholar] [CrossRef]
- McKinney, M.L. Urbanization, Biodiversity, and Conservation. The impacts of urbanization on native species are poorly studied, but educating a highly urbanized human population about these impacts can greatly improve species conservation in all ecosystems. Bioscience 2002, 52, 883–890. [Google Scholar] [CrossRef]
- Aronson, M.F.; Handel, S.N.; La Puma, I.P.; Clemants, S.E. Urbanization promotes non-native woody species and diverse plant assemblages in the New York metropolitan region. Urban Ecosyst. 2015, 18, 31–45. [Google Scholar] [CrossRef]
- Lizée, M.-H.; Mauffrey, J.-F.; Tatoni, T.; Deschamps-Cottin, M. Monitoring urban environments on the basis of biological traits. Ecol. Indic. 2011, 11, 353–361. [Google Scholar] [CrossRef]
- Egerer, M.H.; Arel, C.; Otoshi, M.D.; Quistberg, R.D.; Bichier, P.; Philpott, S.M. Urban arthropods respond variably to changes in landscape context and spatial scale. J. Urban Ecol. 2017, 3, 1–10. [Google Scholar] [CrossRef]
- Smith, R.M.; Thompson, K.; Hodgson, J.G.; Warren, P.H.; Gaston, K.J. Urban domestic gardens (IX): Composition and richness of the vascular plant flora, and implications for native biodiversity. Biol. Conserv. 2006, 129, 312–322. [Google Scholar] [CrossRef]
- Sperling, C.D.; Lortie, C.J. The importance of urban backgardens on plant and invertebrate recruitment: A field microcosm experiment. Urban Ecosyst. 2010, 13, 223–235. [Google Scholar] [CrossRef]
- Angold, P.; Sadler, J.P.; Hill, M.O.; Pullin, A.; Rushton, S.; Austin, K.; Small, E.; Wood, B.; Wadsworth, R.; Sanderson, R. Biodiversity in urban habitat patches. Sci. Total Environ. 2006, 360, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Savard, J.-P.L.; Clergeau, P.; Mennechez, G. Biodiversity concepts and urban ecosystems. Landsc. Urban Plan. 2000, 48, 131–142. [Google Scholar] [CrossRef]
- Smith, R.M.; Gaston, K.J.; Warren, P.H.; Thompson, K. Urban domestic gardens (V): Relationships between landcover composition, housing and landscape. Landsc. Ecol. 2005, 20, 235–253. [Google Scholar] [CrossRef]
- Bennett, A.B.; Gratton, C. Local and landscape scale variables impact parasitoid assemblages across an urbanization gradient. Landsc. Urban Plan. 2012, 104, 26–33. [Google Scholar] [CrossRef]
- Jha, S.; Kremen, C. Urban land use limits regional bumble bee gene flow. Mol. Ecol. 2013, 22, 2483–2495. [Google Scholar] [CrossRef]
- Philpott, S.M.; Cotton, J.; Bichier, P.; Friedrich, R.L.; Moorhead, L.C.; Uno, S.; Valdez, M. Local and landscape drivers of arthropod abundance, richness, and trophic composition in urban habitats. Urban Ecosyst. 2014, 17, 513–532. [Google Scholar] [CrossRef]
- Sattler, T.; Duelli, P.; Obrist, M.; Arlettaz, R.; Moretti, M. Response of arthropod species richness and functional groups to urban habitat structure and management. Landsc. Ecol. 2010, 25, 941–954. [Google Scholar] [CrossRef]
- Petchey, O.L.; Gaston, K.J. Functional diversity: Back to basics and looking forward. Ecol. Lett. 2006, 9, 741–758. [Google Scholar] [CrossRef] [PubMed]
- Quistberg, R.D.; Bichier, P.; Philpott, S.M. Landscape and local correlates of bee abundance and species richness in urban gardens. Environ. Entomol. 2016, 45, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Horváth, R.; Magura, T.; Tóthmérész, B. Ignoring ecological demands masks the real effect of urbanization: A case study of ground-dwelling spiders along a rural–urban gradient in a lowland forest in Hungary. Ecol. Res. 2012, 27, 1069–1077. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Conversion from environmental filtering to randomness as assembly rule of ground beetle assemblages along an urbanization gradient. Sci. Rep. 2018, 8, 16992. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, N.E. Ecology of urban arthropods: A review and a call to action. Ann. Entomol. Soc. Am. 2000, 93, 825–835. [Google Scholar] [CrossRef]
- Hawksworth, D.L.; Bull, A.T. (Eds.) Arthropod Diversity and Conservation; Springer: Dordrecht, The Netherlands, 2006; Volume 1, ISBN 1-4020-5204-9. [Google Scholar]
- Jones, E.L.; Leather, S.R. Invertebrates in urban areas: A review. Eur. J. Entomol. 2012, 109, 463–478. [Google Scholar] [CrossRef]
- McIntyre, N.E.; Rango, J.; Fagan, W.F.; Faeth, S.H. Ground arthropod community structure in a heterogeneous urban environment. Landsc. Urban Plan. 2001, 52, 257–274. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, D.J. Carabid beetle assemblages along urban to rural gradients: A review. Landsc. Urban Plan. 2009, 92, 65–71. [Google Scholar] [CrossRef]
- Landis, D.A.; Menalled, F.D.; Costamagna, A.C.; Wilkinson, T.K. Manipulating plant resources to enhance beneficial arthropods in agricultural landscapes. Weed Sci. 2005, 53, 902–908. [Google Scholar] [CrossRef]
- Kotze, D.J.; Brandmayr, P.; Casale, A.; Dauffy-Richard, E.; Dekoninck, W.; Koivula, M.J.; Lövei, G.L.; Mossakowski, D.; Noordijk, J.; Paarmann, W. Forty years of carabid beetle research in Europe–from taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. ZooKeys 2011, 100, 55–148. [Google Scholar] [CrossRef] [PubMed]
- Lövei, G.L.; Sunderland, K.D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 1996, 41, 231–256. [Google Scholar] [CrossRef]
- Burel, F. Effect of landscape structure and dynamics on species diversity in hedgerow networks. Landsc. Ecol. 1992, 6, 161–174. [Google Scholar] [CrossRef]
- De Vries, H.; Den Boer, P.; Van Dijk, T.S. Ground beetle species in heathland fragments in relation to survival, dispersal, and habitat preference. Oecologia 1996, 107, 332–342. [Google Scholar] [CrossRef]
- Elek, Z.; Kovács, B.; Aszalós, R.; Boros, G.; Samu, F.; Tinya, F.; Ódor, P. Taxon-specific responses to different forestry treatments in a temperate forest. Sci. Rep. 2018, 8, 16990. [Google Scholar] [CrossRef]
- Chiverton, P.; Sotherton, N. The effects of beneficial arthropods of the exclusion of herbicides from cereal crop edges. J. Appl. Ecol. 1991, 28, 1027–1039. [Google Scholar] [CrossRef]
- Desender, K.; Turin, H. Loss of habitats and changes in the composition of the ground and tiger beetle fauna in four West European countries since 1950 (Coleoptera: Carabidae, Cicindelidae). Biol. Conserv. 1989, 48, 277–294. [Google Scholar] [CrossRef]
- Fournier, E.; Loreau, M. Effects of newly planted hedges on ground-beetle diversity (Coleoptera, Carabidae) in an agricultural landscape. Ecography 1999, 22, 87–97. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, J.; Ashworth, A.; Brandmayr, P.; Desender, K.; New, T.; Penev, L.; Samways, M.; Spence, J. The search for common anthropogenic impacts on biodiversity: A global network. J. Insect Conserv. 2000, 4, 3–9. [Google Scholar] [CrossRef]
- Jelaska, L.; Durbes˘ ić, P. Comparison of the body size and wing form of carabid species (Coleoptera: Carabidae) between isolated and continuous forest habitats. Ann. Société Entomol. Fr. 2009, 45, 327–338. [Google Scholar] [CrossRef][Green Version]
- Liebherr, J. General Patterns in West Indian Insects, and Graphical Biogeographic Analysis of Some Circum-caribbean Platynus Beetles (Carabidae). Syst. Biol. 1988, 37, 385–409. [Google Scholar] [CrossRef]
- Lindroth, C.H. An analysis of the carabid beetle fauna of the refugium. In The Kodiak Island Refugium; Karlstrom, T., Ball, G., Eds.; Ryerson Press: Toronto, ON, Canada, 1969; Volume XIII, pp. 195–210. [Google Scholar]
- Delgado de la Flor, Y.A.; Burkman, C.E.; Eldredge, T.K.; Gardiner, M.M. Patch and landscape-scale variables influence the taxonomic and functional composition of beetles in urban greenspaces. Ecosphere 2017, 8, e02007. [Google Scholar] [CrossRef]
- Fournier, E.; Loreau, M. Activity and satiation state in Pterostichus melanarius: An experiment in different agricultural habitats. Ecol. Entomol. 2001, 26, 235–244. [Google Scholar] [CrossRef]
- Alaruikka, D.; Kotze, D.J.; Matveinen, K.; Niemelä, J. Carabid beetle and spider assemblages along a forested urban–rural gradient in southern Finland. J. Insect Conserv. 2002, 6, 195–206. [Google Scholar] [CrossRef]
- Gaublomme, E.; Hendrickx, F.; Dhuyvetter, H.; Desender, K. The effects of forest patch size and matrix type on changes in carabid beetle assemblages in an urbanized landscape. Biol. Conserv. 2008, 141, 2585–2596. [Google Scholar] [CrossRef]
- Magura, T.; Tóthmérész, B.; Elek, Z. Changes in carabid beetle assemblages as Norway spruce plantations age. Community Ecol. 2006, 7, 1–12. [Google Scholar] [CrossRef]
- Niemelä, J.; Kotze, D.J.; Venn, S.; Penev, L.; Stoyanov, I.; Spence, J.; Hartley, D.; De Oca, E.M. Carabid beetle assemblages (Coleoptera, Carabidae) across urban-rural gradients: An international comparison. Landsc. Ecol. 2002, 17, 387–401. [Google Scholar] [CrossRef]
- Russell, M.C.; Lambrinos, J.; Records, E.; Ellen, G. Seasonal shifts in ground beetle (Coleoptera: Carabidae) species and functional composition maintain prey consumption in Western Oregon agricultural landscapes. Biol. Control 2017, 106, 54–63. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Does urbanization decrease diversity in ground beetle (Carabidae) assemblages? Glob. Ecol. Biogeogr. 2010, 19, 16–26. [Google Scholar] [CrossRef]
- Ulrich, W.; Zalewski, M. Abundance and co-occurrence patterns of core and satellite species of ground beetles on small lake islands. Oikos 2006, 114, 338–348. [Google Scholar] [CrossRef]
- Bolund, P.; Hunhammar, S. Ecosystem services in urban areas. Ecol. Econ. 1999, 29, 293–301. [Google Scholar] [CrossRef]
- Niemelä, J. Ecology and urban planning. Biodivers. Conserv. 1999, 8, 119–131. [Google Scholar] [CrossRef]
- Lin, B.B.; Philpott, S.M.; Jha, S. The future of urban agriculture and biodiversity-ecosystem services: Challenges and next steps. Basic Appl. Ecol. 2015, 16, 189–201. [Google Scholar] [CrossRef]
- Gardiner, M.M.; Prajzner, S.P.; Burkman, C.E.; Albro, S.; Grewal, P.S. Vacant land conversion to community gardens: Influences on generalist arthropod predators and biocontrol services in urban greenspaces. Urban Ecosyst. 2014, 17, 101–122. [Google Scholar] [CrossRef]
- Hartley, D.J.; Koivula, M.J.; Spence, J.R.; Pelletier, R.; Ball, G.E. Effects of urbanization on ground beetle assemblages (Coleoptera, Carabidae) of grassland habitats in western Canada. Ecography 2007, 30, 673–684. [Google Scholar] [CrossRef]
- Jin, S.; Yang, L.; Danielson, P.; Homer, C.; Fry, J.; Xian, G. A comprehensive change detection method for updating the National Land Cover Database to circa 2011. Remote Sens. Environ. 2013, 132, 159–175. [Google Scholar] [CrossRef]
- Sadler, J.P.; Small, E.C.; Fiszpan, H.; Telfer, M.G.; Niemela, J. Investigating environmental variation and landscape characteristics of an urban-rural gradient using woodland carabid assemblages. J. Biogeogr. 2006, 33, 1126–1138. [Google Scholar] [CrossRef]
- Weller, B.; Ganzhorn, J.U. Carabid beetle community composition, body size, and fluctuating asymmetry along an urban-rural gradient. Basic Appl. Ecol. 2004, 5, 193–201. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Spake, R.; Barsoum, N.; Newton, A.C.; Doncaster, C.P. Drivers of the composition and diversity of carabid functional traits in UK coniferous plantations. For. Ecol. Manag. 2016, 359, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Varet, M.; Burel, F.; Lafage, D.; Pétillon, J. Age-dependent colonization of urban habitats: A diachronic approach using carabid beetles and spiders. Anim. Biol. 2013, 63, 257–269. [Google Scholar] [CrossRef]
- Calcagno, V.; de Mazancourt, C. glmulti: An R package for easy automated model selection with (generalized) linear models. J. Stat. Softw. 2010, 34, 1–29. [Google Scholar] [CrossRef]
- Breheny, P.; Burchett, W. Visualization of regression models using visreg. R J. 2013, 9, 56–71. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Simpson, G.; Solymos, P.; Heny, M.; Stevens, H.; et al. Vegan: Community ecology package. R package version 2.3-4. 2018. Available online: http:// CRAN.R-project.org/package=vegan (accessed on 8 July 2018).
- Dray, S.; Dufour, A.-B. The ade4 package: Implementing the duality diagram for ecologists. J. Stat. Softw. 2007, 22, 1–20. [Google Scholar] [CrossRef]
- Dray, S.; Choler, P.; Dolédec, S.; Peres-Neto, P.R.; Thuiller, W.; Pavoine, S.; ter Braak, C.J. Combining the fourth-corner and the RLQ methods for assessing trait responses to environmental variation. Ecology 2014, 95, 14–21. [Google Scholar] [CrossRef]
- Dray, S.; Legendre, P. Testing the species traits–environment relationships: The fourth-corner problem revisited. Ecology 2008, 89, 3400–3412. [Google Scholar] [CrossRef]
- Legendre, P.; Gallagher, E.D. Ecologically meaningful transformations for ordination of species data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Belaoussoff, S.; Kevan, P.G.; Murphy, S.; Swanton, C. Assessing tillage disturbance on assemblages of ground beetles (Coleoptera: Carabidae) by using a range of ecological indices. Biodivers. Conserv. 2003, 12, 851–882. [Google Scholar] [CrossRef]
- Paoletti, M.; Bressan, M.; Edwards, C. Soil invertebrates as bioindicators of human disturbance. Crit. Rev. Plant Sci. 1996, 15, 21–62. [Google Scholar] [CrossRef]
- Hagley, E.; Holliday, N.; Barber, D. Laboratory studies of the food preferences of some orchard carabids (Coleoptera: Carabidae). Can. Entomol. 1982, 114, 431–437. [Google Scholar] [CrossRef]
- Pausch, R.D. Observations on the biology of the seed corn beetles, Stenolophus comma and Stenolophus lecontei. Ann. Entomol. Soc. Am. 1979, 72, 24–28. [Google Scholar] [CrossRef]
- Eversham, B.C.; Roy, D.B.; Telfer, M.G. Urban, industrial and other manmade sites as analogues of natural habitats for Carabidae. Ann. Zool. Fenn. 1996, 33, 149–156. [Google Scholar]
- Bolger, D.T.; Suarez, A.V.; Crooks, K.R.; Morrison, S.A.; Case, T.J. Arthropods in Urban Habitat Fragments in Southern California: Area, Age, and Edge Effects. Ecol. Appl. 2000, 10, 1230–1248. [Google Scholar] [CrossRef]
- Collinge, S.K.; Palmer, T.M. The influences of patch shape and boundary contrast on insect response to fragmentation in California grasslands. Landsc. Ecol. 2002, 17, 647–656. [Google Scholar] [CrossRef]
- Roume, A.; Deconchat, M.; Raison, L.; Balent, G.; Ouin, A. Edge effects on ground beetles at the woodlot–field interface are short-range and asymmetrical. Agric. For. Entomol. 2011, 13, 395–403. [Google Scholar] [CrossRef]
- Magura, T. Ignoring functional and phylogenetic features masks the edge influence on ground beetle diversity across forest-grassland gradient. For. Ecol. Manag. 2017, 384, 371–377. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L.; Tóthmérész, B. Edge responses are different in edges under natural versus anthropogenic influence: A meta-analysis using ground beetles. Ecol. Evol. 2017, 7, 1009–1017. [Google Scholar] [CrossRef]
- Bilde, T.; Toft, S. Prey consumption and fecundity of the carabid beetle Calathus melanocephalus on diets of three cereal aphids: High consumption. Pedobiologia 1999, 43, 422–429. [Google Scholar]
- Honek, A.; Martinkova, Z.; Jarosik, V. Ground beetles (Carabidae) as seed predators. Eur. J. Entomol. 2013, 100, 531–544. [Google Scholar] [CrossRef]
- Frank, S.D.; Shrewsbury, P.M.; Denno, R.F. Plant versus prey resources: Influence on omnivore behavior and herbivore suppression. Biol. Control 2011, 57, 229–235. [Google Scholar] [CrossRef]
- Kielty, J.; Allen-Williams, L.; Underwood, N. Prey preferences of six species of Carabidae (Coleoptera) and one Lycosidae (Araneae) commonly found in UK arable crop fields. J. Appl. Entomol. 1999, 123, 193–200. [Google Scholar] [CrossRef]
- Lang, A.; Gsödl, S. Prey vulnerability and active predator choice as determinants of prey selection: A carabid beetle and its aphid prey. J. Appl. Entomol. 2001, 125, 53–61. [Google Scholar] [CrossRef]
- Dudás, P.; Gedeon, C.; Menyhárt, L.; Ambrus, G.; Tóth, F. The effect of mulching on the abundance and diversity of ground beetle assemblages in two Hungarian potato fields. Columella J. Agric. Environ. Sci. 2016, 3, 45–53. [Google Scholar] [CrossRef]
- Magura, T.; Tóthmérész, B.; Molnár, T. A species-level comparison of occurrence patterns in carabids along an urbanisation gradient. Landsc. Urban Plan. 2008, 86, 134–140. [Google Scholar] [CrossRef]
- Cárcamo, H.A.; Niemalä, J.K.; Spence, J.R. Farming and ground beetles: Effects of agronomic practice on populations and community structure. Can. Entomol. 1995, 127, 123–140. [Google Scholar] [CrossRef]
- Mitchell, B. Ecology of two carabid beetles, Bembidion lampros (Herbst) and Trechus quadristriatus (Schrank). J. Anim. Ecol. 1963, 32, 289–299. [Google Scholar] [CrossRef]
- Koivula, M.; Punttila, P.; Haila, Y.; Niemelä, J. Leaf litter and the small-scale distribution of carabid beetles (Coleoptera, Carabidae) in the boreal forest. Ecography 1999, 22, 424–435. [Google Scholar] [CrossRef]
- Ogai, T.; Kenta, T. The effects of vegetation types and microhabitats on carabid beetle community composition in cool temperate Japan. Ecol. Res. 2016, 31, 177–188. [Google Scholar] [CrossRef]
- Small, E.C.; Sadler, J.P.; Telfer, M.G. Carabid beetle assemblages on urban derelict sites in Birmingham, UK. J. Insect Conserv. 2002, 6, 233–246. [Google Scholar] [CrossRef]
- Tyler, G. Differences in abundance, species richness, and body size of ground beetles (Coleoptera: Carabidae) between beech (Fagus sylvatica L.) forests on Podzol and Cambisol. For. Ecol. Manag. 2008, 256, 2154–2159. [Google Scholar] [CrossRef]
- Magura, T.; Tóthmérész, B.; Molnár, T. Changes in carabid beetle assemblages along an urbanisation gradient in the city of Debrecen, Hungary. Landsc. Ecol. 2004, 19, 747–759. [Google Scholar] [CrossRef]
- Šustek, Z. Changes in body size structure of carabid communities (Coleoptera, Carabidae) along an urbanisation gradient. Biológia Bratisl. 1987, 42, 145–156. [Google Scholar]
- Gerisch, M.; Dziock, F.; Schanowski, A.; Ilg, C.; Henle, K. Community resilience following extreme disturbances: The response of ground beetles to a severe summer flood in a Central European lowland stream. River Res. Appl. 2012, 28, 81–92. [Google Scholar] [CrossRef]
- Oberholtzer, L.; Dimitri, C.; Pressman, A. Urban agriculture in the United States: Characteristics, challenges, and technical assistance needs. J. Ext. 2014, 52, 6FEA1. [Google Scholar]
- Brewer, M.; Elliott, N. Biological control of cereal aphids in North America and mediating effects of host plant and habitat manipulations. Annu. Rev. Entomol. 2004, 49, 219–242. [Google Scholar] [CrossRef]
- Kromp, B. Carabid beetles in sustainable agriculture: A review on pest control efficacy, cultivation impacts and enhancement. Agric. Ecosyst. Environ. 1999, 74, 187–228. [Google Scholar] [CrossRef]
- Stiling, P.; Cornelissen, T. What makes a successful biocontrol agent? A meta-analysis of biological control agent performance. Biol. Control 2005, 34, 236–246. [Google Scholar] [CrossRef]
- Sunderland, K.D.; Vickerman, G.P. Aphid feeding by some polyphagous predators in relation to aphid density in cereal fields. J. Appl. Ecol. 1980, 17, 389–396. [Google Scholar] [CrossRef]
- Symondson, W.; Sunderland, K.; Greenstone, M. Can generalist predators be effective biocontrol agents? Annu. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef] [PubMed]
- Riddick, E.W.; Mills, N.J. Potential of adult carabids (Coleoptera: Carabidae) as predators of fifth-instar codling moth (Lepidoptera: Tortricidae) in apple orchards in California. Environ. Entomol. 1994, 23, 1338–1345. [Google Scholar] [CrossRef]
- Ball, S.; Woodcock, B.; Potts, S.; Heard, M. Size matters: Body size determines functional responses of ground beetle interactions. Basic Appl. Ecol. 2015, 16, 621–628. [Google Scholar] [CrossRef]
- Desender, K. Flight muscle development and dispersal in the life cycle of carabid beetles: Patterns and processes. Bull. Inst. R. Sci. Nat. Belg. Entomol. 2000, 70, 13–31. [Google Scholar]
- Statzner, B.; Beche, L. Can biological invertebrate traits resolve effects of multiple stressors on running water ecosystems? Freshw. Biol. 2010, 55, 80–119. [Google Scholar] [CrossRef]
| Species | No. Indiv. | No. Sites | Length (mm) | Size § | Wing Morph-Ology §§ | Dispersal Ability ¶¶ |
|---|---|---|---|---|---|---|
| Amara (Amara) aenea (DeGeer) | 2 | 1 | 9 | L | M | M |
| Amara (Amara) littoralis (Dejean) | 5 | 4 | 8.8 | L | M | H |
| Amara (Celia) californica californica (Dejean) | 1 | 1 | 11 | L | M | M |
| Amara (Zezea) scitula (Zimmermann) | 1 | 1 | 11 | L | M | M |
| Anisodactylus californicus (Dejan) | 1 | 1 | 12 | L | M | M |
| Axinopalpus biplagiatus (Dejean) | 3 | 2 | 3.3 | S | M | H |
| Bembidion (Neja) ambiguum (Dejean) | 2 | 1 | 3.5 | S | M | H |
| Bradycellus (Liocellus) nitidus (Dejean) | 3 | 2 | 5 | S | M | H |
| Bradycellus (Stenocellus) nubifer (LeConte) | 4 | 3 | 4.5 | S | M | H |
| Calathus ruficollis ruficollis (Dejean) | 5 | 2 | 10.2 | L | B | L |
| Chlaenius (Chlaeniellus) tricolor vigilans (Say) | 3 | 1 | 15 | L | M | M |
| Harpalus (Pseudoophonus) pensylvanicus (DeGeer) | 8 | 2 | 17.5 | L | M | M |
| Laemostenus complanatus (Dejean) | 32 | 2 | 16.2 | L | M | M |
| Microlestes nigrinus (Mannerheim) | 3 | 2 | 3.7 | S | D | M |
| Notiobia (Anisotarsus) terminata (Say) | 3 | 2 | 12 | L | M | M |
| Poecilus (Poecilus) cursitor (LeConte) | 1 | 1 | 11 | L | M | M |
| Pterostichus (Bothriopterus) lustrans (LeConte) | 4 | 2 | 12.3 | L | M | M |
| Pterostichus (Hypherpes) californicus (Dejean) | 14 | 5 | 16.6 | L | B | L |
| Pterostichus (Hypherpes) vicinus (Mannerheim) | 3 | 2 | 16 | L | M | M |
| Trechus (Trechus) obtusus (Erichson) | 51 | 7 | 4.1 | S | D | M |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Philpott, S.M.; Albuquerque, S.; Bichier, P.; Cohen, H.; Egerer, M.H.; Kirk, C.; Will, K.W. Local and Landscape Drivers of Carabid Activity, Species Richness, and Traits in Urban Gardens in Coastal California. Insects 2019, 10, 112. https://doi.org/10.3390/insects10040112
Philpott SM, Albuquerque S, Bichier P, Cohen H, Egerer MH, Kirk C, Will KW. Local and Landscape Drivers of Carabid Activity, Species Richness, and Traits in Urban Gardens in Coastal California. Insects. 2019; 10(4):112. https://doi.org/10.3390/insects10040112
Chicago/Turabian StylePhilpott, Stacy M., Simone Albuquerque, Peter Bichier, Hamutahl Cohen, Monika H. Egerer, Claire Kirk, and Kipling W. Will. 2019. "Local and Landscape Drivers of Carabid Activity, Species Richness, and Traits in Urban Gardens in Coastal California" Insects 10, no. 4: 112. https://doi.org/10.3390/insects10040112
APA StylePhilpott, S. M., Albuquerque, S., Bichier, P., Cohen, H., Egerer, M. H., Kirk, C., & Will, K. W. (2019). Local and Landscape Drivers of Carabid Activity, Species Richness, and Traits in Urban Gardens in Coastal California. Insects, 10(4), 112. https://doi.org/10.3390/insects10040112





