Lethal Effects of High Temperatures on Brown Marmorated Stink Bug Adults before and after Overwintering
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Lethal High Temperatures with Short Exposure Times
2.3. Nutrient Index and H. halys Mortality after High-Temperature Exposures
2.4. Data Analysis
3. Results
3.1. Lethal High Temperatures
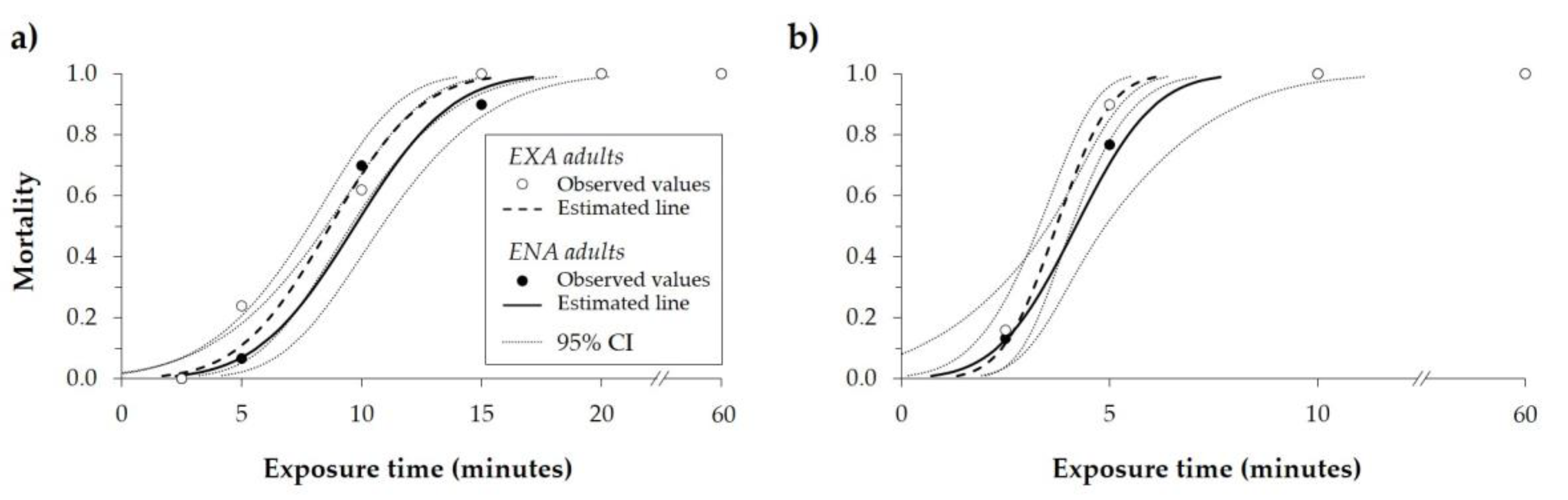
3.2. Exposure Times at High Temperatures
3.3. Weight and Nutrient Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kenis, M.; Auger-Rozenberg, M.-A.; Roques, A.; Timms, L.; Péré, C.; Cock, M.J.W.; Settele, J.; Augustin, S.; Lopez-Vaamonde, C. Ecological effects of invasive alien insects. Biol. Invasions 2009, 11, 21–45. [Google Scholar] [CrossRef]
- Kenis, M.; Branco, M. Impact of alien terrestrial arthropods in Europe. Chapter 5. BioRisk 2010, 4, 51–71. [Google Scholar] [CrossRef] [Green Version]
- Pimentel, D.; Zuniga, R.; Morrison, D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 2005, 52, 273–288. [Google Scholar] [CrossRef]
- Kenis, M.; Roques, A.; Santini, A.; Liebhold, A.M. Impact of non-native invertebrates and pathogens on market forest tree resources. In Impact of Biological Invasions on Ecosystem Services; Vilà, M., Hulme, P.E., Eds.; Invading Nature—Springer Series in Invasion Ecology; Springer: Cham, Switzerland, 2017; Volume 12, pp. 103–117. ISBN 9783319451213. [Google Scholar]
- Leskey, T.C.; Nielsen, A.L. Impact of the invasive brown marmorated stink bug in North America and Europe: History, biology, ecology, and management. Annu. Rev. Entomol. 2018, 63, 599–618. [Google Scholar] [CrossRef]
- Wermelinger, B.; Wyniger, D.; Forster, B. First records of an invasive bug in Europe: Halyomorpha halys Stål (Heteroptera: Pentatomidae), a new pest on woody ornamentals and fruit trees? Mitteilungen Schweiz. Entomol. Ges. 2008, 81, 1–8. [Google Scholar]
- Haye, T.; Gariepy, T.; Hoelmer, K.; Rossi, J.-P.; Streito, J.-C.; Tassus, X.; Desneux, N. Range expansion of the invasive brown marmorated stinkbug, Halyomorpha halys: An increasing threat to field, fruit and vegetable crops worldwide. J. Pest Sci. 2015, 88, 665–673. [Google Scholar] [CrossRef]
- Vétek, G.; Károlyi, B.; Mészáros, Á.; Horváth, D.; Korányi, D. The invasive brown marmorated stink bug (Halyomorpha halys) is now widespread in Hungary. Entomol. Gen. 2018, 38, 3–14. [Google Scholar] [CrossRef]
- Maistrello, L.; Dioli, P.; Bariselli, M. Trovata una cimice esotica dannosa per i frutteti. Agricoltura 2013, 6, 67–68. [Google Scholar]
- Maistrello, L.; Dioli, P.; Bariselli, M.; Mazzoli, G.L.; Giacalone-Forini, I. Citizen science and early detection of invasive species: Phenology of first occurrences of Halyomorpha halys in Southern Europe. Biol. Invasions 2016, 18, 3109–3116. [Google Scholar] [CrossRef]
- Bariselli, M.; Bugiana, R.; Maistrello, L. Distribution and damage caused by Halyomorpha halys in Italy. EPPO Bull. 2016, 46, 332–334. [Google Scholar] [CrossRef]
- Inkley, D.B. Characteristics of home invasion by the brown marmorated stink bug (Hemiptera: Pentatomidae). J. Entomol. Sci. 2012, 47, 125–130. [Google Scholar] [CrossRef]
- Cambridge, J.; Payenski, A.; Hamilton, G.C. The distribution of overwintering brown marmorated stink bugs (Hemiptera: Penatatomidae) in college dormitories. Fla. Entomol. 2015, 98, 1257–1259. [Google Scholar] [CrossRef]
- Nicoli Aldini, R.; Fraschini, A. Gestione della difesa antiparassitaria in un’industria conserviera del nord Italia. In Programma e Riassunti, Proceedings of the X Simposio—La Difesa Antiparassitaria nelle Industrie Alimentari e la Protezione degli Alimenti, Piacenza, Italy, 20–22 September 2017; Accademia Nazionale Italiana di Entomologia: Piacenza, Italy, 2017; p. 47. [Google Scholar]
- Acebes-Doria, A.L.; Morrison, W.R.; Short, B.D.; Rice, K.B.; Bush, H.G.; Kuhar, T.P.; Duthie, C.; Leskey, T.C. Monitoring and biosurveillance tools for the brown marmorated stink bug, Halyomorpha halys (Stål) (Hemiptera: Pentatomidae). Insects 2018, 9, 82. [Google Scholar] [CrossRef] [PubMed]
- Nixon, L.J.; Morrison, W.R.; Rice, K.B.; Brockerhoff, E.G.; Leskey, T.C.; Guzman, F.; Khrimian, A.; Goldson, S.; Rostás, M. Identification of volatiles released by diapausing brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). PLoS ONE 2018, 13, e0191223. [Google Scholar] [CrossRef] [PubMed]
- Nixon, L.J.; Tabb, A.; Morrison, W.R.; Rice, K.B.; Brockerhoff, E.G.; Leskey, T.C.; van Koten, C.; Goldson, S.; Rostás, M. Volatile release, mobility, and mortality of diapausing Halyomorpha halys during simulated shipping movements and temperature changes. J. Pest Sci. 2019, 92, 633–641. [Google Scholar] [CrossRef]
- Burges, H.D.; Burrell, N.J. Cooling bulk grain in the British climate to control storage insects and to improve keeping quality. J. Sci. Food Agric. 1964, 15, 32–50. [Google Scholar] [CrossRef]
- Chapman, R.N. Insects infesting stored food products. Minn. Bull. 1932, 198, 1–83. [Google Scholar]
- Dean, G.A. Heat as a means of controlling mill insects. J. Econ. Entomol. 1911, 4, 142–161. [Google Scholar] [CrossRef]
- Dean, G.A. Further data on heat as a means of controlling mill insects. J. Econ. Entomol. 1913, 6, 40–55. [Google Scholar] [CrossRef]
- Fields, P.G.; White, N.D.G. Alternatives to methyl bromide treatments for stored-product and quarantine insects. Annu. Rev. Entomol. 2002, 47, 331–359. [Google Scholar] [CrossRef]
- Phillips, T.W.; Throne, J.E. Biorational approaches to managing stored-product insects. Annu. Rev. Entomol. 2010, 55, 375–397. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.D.; Johnson, J.A.; Winter, D.A. History and use of heat in pest control: A review. Int. J. Pest Manag. 2011, 57, 267–289. [Google Scholar] [CrossRef]
- Kells, S.A.; Goblirsch, M.J. Temperature and time requirements for controlling bed bugs (Cimex lectularius) under commercial heat treatment conditions. Insects 2011, 2, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Nayak, M.K.; Collins, P.J.; Throne, J.E.; Wang, J.-J. Biology and management of Psocids infesting stored products. Annu. Rev. Entomol. 2014, 59, 279–297. [Google Scholar] [CrossRef] [PubMed]
- Neven, L.G. Physiological responses of insects to heat. Postharvest Biol. Technol. 2000, 21, 103–111. [Google Scholar] [CrossRef]
- Hammond, D. Heat Treatment for Insect Control: Developments and Applications; Woodhead Publishing Series in Food Science, Technology and Nutrition: Number 241; Woodhead Publishing: Amsterdam, The Netherlands, 2015; pp. 1–120. ISBN 9780857097767. [Google Scholar]
- Mangan, R.L.; Hallman, G.J. Temperature treatments for quarantine security: New approaches for fresh commodities. In Temperature Sensitivity in Insects and Application in Integrated Pest Management; Hallman, G.J., Denlinger, D.L., Eds.; Westview Press: Boulder, CO, USA, 1998; pp. 201–236. ISBN 0813389909. [Google Scholar]
- Cira, T.M.; Venette, R.C.; Aigner, J.; Kuhar, T.; Mullins, D.E.; Gabbert, S.E.; Hutchison, W.D. Cold tolerance of Halyomorpha halys (Hemiptera: Pentatomidae) across geographic and temporal scales. Environ. Entomol. 2016, 45, 484–491. [Google Scholar] [CrossRef]
- Cira, T.M.; Koch, R.L.; Burkness, E.C.; Hutchison, W.D.; Venette, R.C. Effects of diapause on Halyomorpha halys (Hemiptera: Pentatomidae) cold tolerance. Environ. Entomol. 2018, 47, 997–1004. [Google Scholar] [CrossRef]
- Lowenstein, D.M.; Walton, V.M. Halyomorpha halys (Hemiptera: Pentatomidae) winter survival, feeding activity, and reproduction rates based on episodic cold shock and winter temperature regimes. J. Econ. Entomol. 2018, 111, 1210–1218. [Google Scholar] [CrossRef]
- Aigner, J.D.; Kuhar, T.P. Lethal high temperature extremes of the brown marmorated stink bug (Hemiptera: Pentatomidae) and efficacy of commercial heat treatments for control in expert shipping cargo. J. Agric. Urban Entomol. 2016, 32, 1–6. [Google Scholar] [CrossRef]
- Watanabe, M.; Arakawa, R.; Shinagawa, Y.; Okazawa, T. Overwintering flight of brown-marmorated stink bug, Halyomorpha mista to the buildings. Jpn. J. Sanit. Zool. 1994, 45, 25–31. [Google Scholar] [CrossRef]
- Lee, D.-H.; Cullum, J.P.; Anderson, J.L.; Daugherty, J.L.; Beckett, L.M.; Leskey, T.C. Characterization of overwintering sites of the invasive brown marmorated stink bug in natural landscapes using human surveyors and detector canines. PLoS ONE 2014, 9, e91575. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.L.; Chen, S.; Fleischer, S.J. Coupling developmental physiology, photoperiod, and temperature to model phenology and dynamics of an invasive heteropteran, Halyomorpha halys. Front. Physiol. 2016, 7, 165. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.L.; Fleischer, S.; Hamilton, G.C.; Hancock, T.; Krawczyk, G.; Lee, J.C.; Ogburn, E.; Pote, J.M.; Raudenbush, A.; Rucker, A.; et al. Phenology of brown marmorated stink bug described using female reproductive development. Ecol. Evol. 2017, 7, 6680–6690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skillman, V.P.; Wiman, N.G.; Lee, J.C. Nutrient declines in overwintering Halyomorpha halys populations. Entomol. Exp. Appl. 2018, 166, 778–789. [Google Scholar] [CrossRef]
- Skillman, V.P.; Wiman, N.G.; Lee, J.C. Monitoring nutrient status of Brown Marmorated Stink Bug adults and nymphs on Summer Holly. Insects 2018, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Costi, E.; Haye, T.; Maistrello, L. Biological parameters of the invasive brown marmorated stink bug, Halyomorpha halys, in southern Europe. J. Pest Sci. 2017, 90, 1059–1067. [Google Scholar] [CrossRef]
- Funayama, K. Importance of apple fruits as food for the brown-marmorated stink bug, Halyomorpha halys (Stål) (Heteroptera: Pentatomidae). Appl. Entomol. Zool. 2004, 39, 617–623. [Google Scholar] [CrossRef]
- SAS Institute. PROC User’s Manual, 6th ed.; SAS Institute: Cary, NC, USA, 2016; Available online: http://support.sas.com/documentation/cdl/en/indbug/68442/PDF/default/indbug.pdf (accessed on 26 August 2019).
- Robertson, J.L.; Savin, N.E.; Preisler, H.K.; Russell, R.M. Bioassays with Arthropods, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2007; pp. 1–224. ISBN 9781420004045. [Google Scholar]
- Bergh, J.C.; Morrison III, W.R.; Joseph, S.V.; Leskey, T.C. Characterizing spring emergence of adult Halyomorpha halys using experimental overwintering shelters and commercial pheromone traps. Entomol. Exp. Appl. 2017, 162, 336–345. [Google Scholar] [CrossRef]
- Lee, D.-H.; Leskey, T.C. Flight behavior of foraging and overwintering brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Bull. Entomol. Res. 2015, 105, 566–573. [Google Scholar] [CrossRef]
- Lee, D.-H.; Short, B.D.; Joseph, S.V.; Bergh, J.C.; Leskey, T.C. Review of the biology, ecology, and management of Halyomorpha halys (Hemiptera: Pentatomidae) in China, Japan, and the Republic of Korea. Environ. Entomol. 2013, 42, 627–641. [Google Scholar] [CrossRef]
- Loeschcke, V.; Krebs, R.A.; Barker, J.S.F. Genetic variation for resistance and acclimation to high temperature stress in Drosophila buzzatii. Biol. J. Linn. Soc. 1994, 52, 83–92. [Google Scholar] [CrossRef]
- Chown, S.L.; Jumbam, K.R.; Sørensen, J.G.; Terblanche, J.S. Phenotypic variance, plasticity and heritability estimates of critical thermal limits depend on methodological context. Funct. Ecol. 2009, 23, 133–140. [Google Scholar] [CrossRef]
- Overgaard, J.; Kristensen, T.N.; Sørensen, J.G. Validity of thermal ramping assays used to assess thermal tolerance in arthropods. PLoS ONE 2012, 7, e32758. [Google Scholar] [CrossRef] [PubMed]
- Terblanche, J.S.; Hoffmann, A.A.; Mitchell, K.A.; Rako, L.; le Roux, P.C.; Chown, S.L. Ecologically relevant measures of tolerance to potentially lethal temperatures. J. Exp. Biol. 2011, 214, 3713–3725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haye, T.; Abdallah, S.; Gariepy, T.; Wyniger, D. Phenology, life table analysis and temperature requirements of the invasive brown marmorated stink bug, Halyomorpha halys, in Europe. J. Pest Sci. 2014, 87, 407–418. [Google Scholar] [CrossRef]
- Ingels, C.A.; Daane, K.M. Phenology of brown marmorated stink bug in a California urban landscape. J. Econ. Entomol. 2018, 111, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Perkins, S.E.; Alexander, L.V. On the measurement of heat waves. J. Clim. 2013, 156, 4500–4517. [Google Scholar] [CrossRef]
- Perkins, S.E.; Alexander, L.V.; Nairn, J.R. Increasing frequency, intensity and duration of observed global heatwaves and warm spells. Geophys. Res. Lett. 2012, 39, L20714. [Google Scholar] [CrossRef]
- WMO Statement on the State of the Global Climate in 2018. Available online: https://library.wmo.int/doc_num.php?explnum_id=5789 (accessed on 26 August 2019).
- European Heatwave Sets New Temperature Records. Available online: https://public.wmo.int/en/media/news/european-heatwave-sets-new-temperature-records (accessed on 26 August 2019).
- 2019 Starts with Extreme, High-Impact Weather. Available online: https://public.wmo.int/en/media/news/2019-starts-extreme-high-impact-weather (accessed on 26 August 2019).
- Kriticos, D.J.; Kean, J.M.; Phillips, C.B.; Senay, S.D.; Acosta, H.; Haye, T. The potential global distribution of the brown marmorated stink bug, Halyomorpha halys, a critical threat to plant biosecurity. J. Pest Sci. 2017, 90, 1033–1043. [Google Scholar] [CrossRef]
- Zhu, G.; Bu, W.; Gao, Y.; Liu, G. Potential geographic distribution of brown marmorated stink bug invasion (Halyomorpha halys). PLoS ONE 2012, 7, e31246. [Google Scholar] [CrossRef]
- Zobel, E.S.; Hooks, C.R.R.; Dively, G.P. Seasonal abundance, host suitability, and feeding injury of the brown marmorated stink bug, Halyomorpha halys (Heteroptera: Pentatomidae), in selected vegetables. J. Econ. Entomol. 2016, 109, 1289–1302. [Google Scholar] [CrossRef] [PubMed]
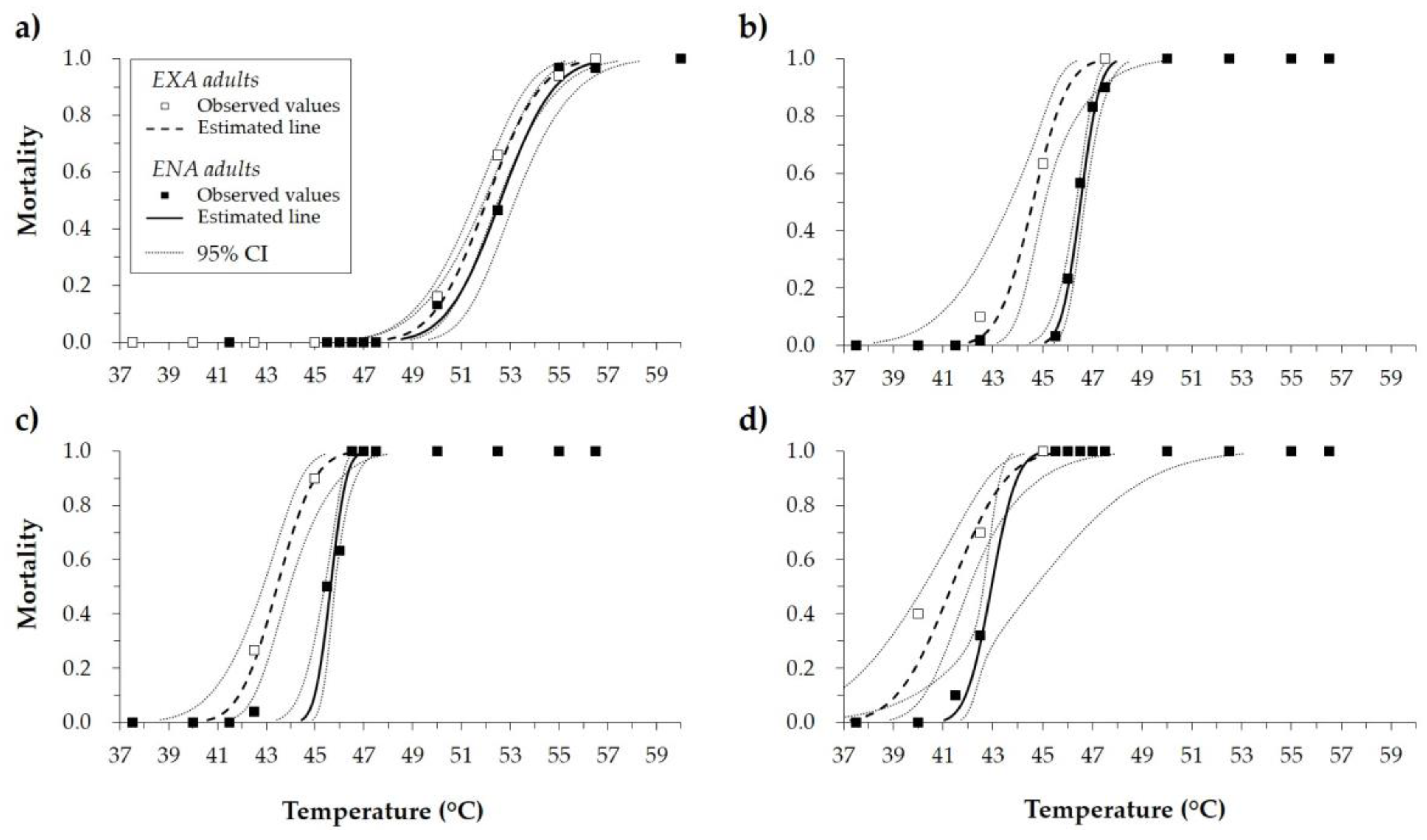
| Adults’ State | Exposure Time (Minutes) | n | LT50 (°C) * | 95% CILT50 (°C) | LT99 (°C) * | 95% CILT99 (°C) | Intercept | SEIntercept | Slope | SESlope | Χ2 ** | df | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | Lower | Upper | |||||||||||||
| EXA | 2.5 | 310 | 52.04 | b | 51.54 | 52.50 | 56.07 | b | 55.22 | 57.39 | −30.01 | 3.70 | 0.58 | 0.07 | 32.94 | 29 |
| ENA | 2.5 | 400 | 52.61 | a | 51.94 | 53.20 | 56.66 | a | 55.68 | 58.30 | −73.15 | 4.51 | 1.55 | 0.09 | 26.16 | 35 |
| EXA | 15.0 | 310 | 44.60 | b | 43.69 | 45.09 | 47.17 | b | 46.33 | 49.81 | −40.47 | 11.49 | 0.91 | 0.25 | 8.77 | 29 |
| ENA | 15.0 | 400 | 46.53 | a | 46.36 | 46.69 | 47.95 | a | 47.62 | 48.51 | −76.34 | 10.92 | 1.64 | 0.23 | 24.66 | 35 |
| EXA | 30.0 | 310 | 43.44 | b | 42.86 | 43.96 | 46.29 | b | 45.44 | 48.01 | −35.44 | 6.81 | 0.82 | 0.16 | 10.97 | 29 |
| ENA | 30.0 | 400 | 45.64 | a | 45.38 | 45.81 | 46.83 | a | 46.52 | 47.53 | −89.54 | 19.04 | 1.96 | 0.41 | 14.17 | 35 |
| EXA | 60.0 | 310 | 41.34 | b | 40.23 | 42.05 | 45.39 | a | 44.26 | 47.94 | −23.74 | 5.31 | 0.57 | 0.13 | 11.01 | 29 |
| ENA | 60.0 | 400 | 42.95 | a | 42.62 | 44.70 | 44.83 | b | 43.78 | 53.05 | −53.09 | 20.83 | 1.24 | 0.49 | 4.61 | 35 |
| Adults’ State | Temperature (°C) | n | MT50 (Minutes) * | 95% CIMT50 (Minutes) | MT99 (Minutes) * | 95% CIMT99 (Minutes) | Intercept | SEIntercept | Slope | SESlope | Χ2 ** | df | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | Lower | Upper | |||||||||||||
| EXA | 47.5 | 400 | 8.67 | a | 7.80 | 9.54 | 15.65 | b | 14.06 | 18.13 | −2.89 | 0.38 | 0.33 | 0.04 | 33.28 | 43 |
| ENA | 47.5 | 310 | 9.69 | a | 8.53 | 10.83 | 17.14 | a | 15.22 | 20.39 | −3.03 | 0.48 | 0.31 | 0.05 | 22.14 | 28 |
| EXA | 50.0 | 400 | 3.72 | a | 3.31 | 4.09 | 6.10 | b | 5.50 | 7.12 | −3.64 | 0.61 | 0.98 | 0.15 | 9.05 | 34 |
| ENA | 50.0 | 310 | 4.19 | a | 3.53 | 4.90 | 7.67 | a | 6.40 | 11.18 | −2.80 | 0.72 | 0.67 | 0.17 | 2.43 | 28 |
| Source of Variation | 15 min | 30 min | 60 min | ||||||
|---|---|---|---|---|---|---|---|---|---|
| F Value | df | p-Value | F Value | df | p-Value | F Value | df | p-Value | |
| Status (dead or alive) | 0.10 | 1; 210 | 0.7517 | 0.38 | 1; 210 | 0.5404 | 0.17 | 1; 210 | 0.6850 |
| Temperature (Temp) | 3.47 | 1; 210 | 0.0639 | 2.47 | 1; 210 | 0.1173 | 1.20 | 1; 210 | 0.2739 |
| Status × Temp | 0.50 | 1; 210 | 0.4804 | 0.66 | 1; 210 | 0.4173 | 0.00 | 1; 210 | 0.9954 |
| Sex | 92.77 | 1; 210 | <0.0001 | 54.81 | 1; 210 | <0.0001 | 46.79 | 1; 210 | <0.0001 |
| Status × Sex | 0.31 | 1; 210 | 0.5793 | 1.47 | 1; 210 | 0.2273 | 0.27 | 1; 210 | 0.6033 |
| Sex × Temp | 0.46 | 1; 210 | 0.4968 | 0.27 | 1; 210 | 0.6066 | 0.83 | 1; 210 | 0.3621 |
| Status × Sex × Temp | 0.14 | 1; 210 | 0.7090 | 0.73 | 1; 210 | 0.3950 | 0.18 | 1; 210 | 0.6732 |
| Overwintering state (OS) | 22.12 | 1; 210 | <0.0001 | 13.88 | 1; 210 | 0.0003 | 16.35 | 1; 210 | <0.0001 |
| Status × OS | 0.08 | 1; 210 | 0.7759 | 0.41 | 1; 210 | 0.5202 | 0.30 | 1; 210 | 0.5815 |
| OS × Temp | 0.11 | 1; 210 | 0.7391 | 0.45 | 1; 210 | 0.5037 | 1.29 | 1; 210 | 0.2579 |
| Status × OS × Temp | 0.51 | 1; 210 | 0.4763 | 0.21 | 1; 210 | 0.6442 | 1.86 | 1; 210 | 0.1744 |
| Sex × OS | 0.76 | 1; 210 | 0.3844 | 1.84 | 1; 210 | 0.1765 | 1.37 | 1; 210 | 0.2439 |
| Status × Sex × OS | 0.07 | 1; 210 | 0.7867 | 0.18 | 1; 210 | 0.6746 | 0.62 | 1; 210 | 0.4324 |
| Sex × OS × Temp | 0.93 | 1; 210 | 0.3366 | 0.69 | 1; 210 | 0.4071 | 0.24 | 1; 210 | 0.6227 |
| Status × Sex × OS × Temp | 0.03 | 1; 210 | 0.8633 | 0.27 | 1; 210 | 0.6029 | 0.01 | 1; 210 | 0.9118 |
| Source of Variation | 15 min | 30 min | 60 min | ||||||
|---|---|---|---|---|---|---|---|---|---|
| F Value | df | p-Value | F Value | df | p-Value | F Value | df | p-Value | |
| Status (dead or alive) | 0.14 | 1; 210 | 0.7120 | 0.00 | 1; 210 | 0.9877 | 0.69 | 1; 210 | 0.4066 |
| Temperature (Temp) | 1.94 | 1; 210 | 0.1652 | 2.55 | 1; 210 | 0.1118 | 0.98 | 1; 210 | 0.3223 |
| Status × Temp | 0.16 | 1; 210 | 0.6930 | 0.84 | 1; 210 | 0.3595 | 0.02 | 1; 210 | 0.9024 |
| Sex | 1.34 | 1; 210 | 0.2481 | 2.87 | 1; 210 | 0.0915 | 3.88 | 1; 210 | 0.0501 |
| Status × Sex | 0.03 | 1; 210 | 0.8519 | 1.65 | 1; 210 | 0.1999 | 1.93 | 1; 210 | 0.1657 |
| Sex × Temp | 0.94 | 1; 210 | 0.3343 | 1.42 | 1; 210 | 0.2353 | 0.28 | 1; 210 | 0.6002 |
| Status × Sex × Temp | 0.67 | 1; 210 | 0.4144 | 0.17 | 1; 210 | 0.6766 | 0.13 | 1; 210 | 0.7237 |
| Overwintering state (OS) | 22.5 | 1; 210 | <0.0001 | 17.77 | 1; 210 | <0.0001 | 20.34 | 1; 210 | <0.0001 |
| Status × OS | 0.02 | 1; 210 | 0.8758 | 0.90 | 1; 210 | 0.3431 | 1.03 | 1; 210 | 0.3110 |
| OS × Temp | 0.29 | 1; 210 | 0.5913 | 0.81 | 1; 210 | 0.3701 | 0.49 | 1; 210 | 0.4832 |
| Status × OS × Temp | 0.32 | 1; 210 | 0.5732 | 0.62 | 1; 210 | 0.4313 | 0.19 | 1; 210 | 0.6646 |
| Sex × OS | 0.38 | 1; 210 | 0.5361 | 2.70 | 1; 210 | 0.1016 | 1.80 | 1; 210 | 0.1814 |
| Status × Sex × OS | 0.01 | 1; 210 | 0.9237 | 0.42 | 1; 210 | 0.5154 | 0.28 | 1; 210 | 0.5991 |
| Sex × OS × Temp | 3.88 | 1; 210 | 0.0501 | 1.80 | 1; 210 | 0.1817 | 0.53 | 1; 210 | 0.4692 |
| Status × Sex × OS × Temp | 0.00 | 1; 210 | 0.9530 | 0.00 | 1; 210 | 0.979 | 0.46 | 1; 210 | 0.5000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scaccini, D.; Duso, C.; Pozzebon, A. Lethal Effects of High Temperatures on Brown Marmorated Stink Bug Adults before and after Overwintering. Insects 2019, 10, 355. https://doi.org/10.3390/insects10100355
Scaccini D, Duso C, Pozzebon A. Lethal Effects of High Temperatures on Brown Marmorated Stink Bug Adults before and after Overwintering. Insects. 2019; 10(10):355. https://doi.org/10.3390/insects10100355
Chicago/Turabian StyleScaccini, Davide, Carlo Duso, and Alberto Pozzebon. 2019. "Lethal Effects of High Temperatures on Brown Marmorated Stink Bug Adults before and after Overwintering" Insects 10, no. 10: 355. https://doi.org/10.3390/insects10100355





