Microbial Control of Invasive Forest Pests with Entomopathogenic Fungi: A Review of the Current Situation
Abstract
1. Introduction
2. Invasive Forest Insect Pests
2.1. Cypress Aphid
2.2. Eucalyptus Weevil
2.3. Gypsy Moth
2.4. Asian Longhorned Beetle
2.5. Emerald Ash Borer
2.6. Oak Lace Bug
3. Microbial Control of Invasive Pests with Entomopathogenic Fungi
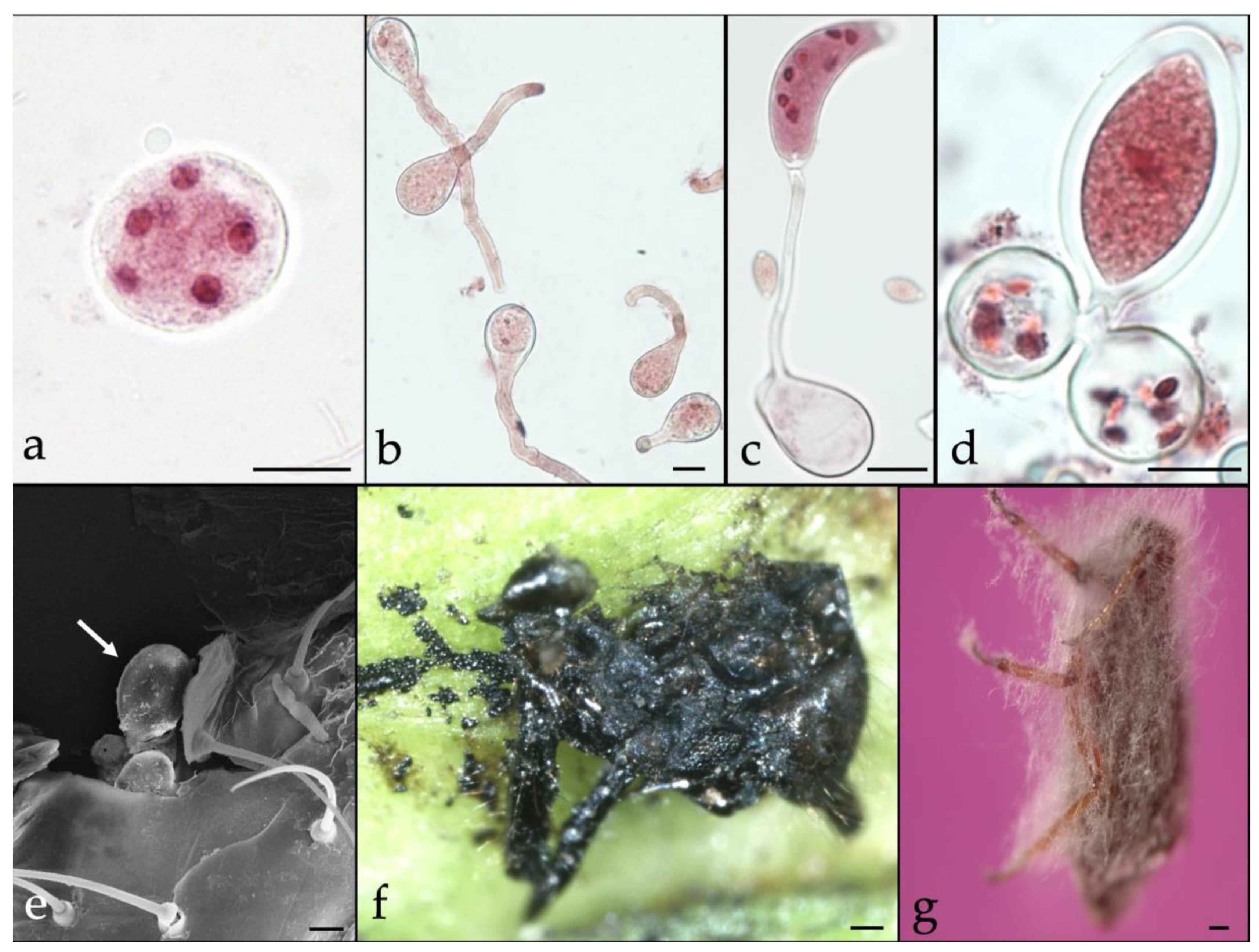
3.1. Cypress Aphid Control
3.2. Eucalyptus Weevil Control
3.3. Gypsy Moth Control
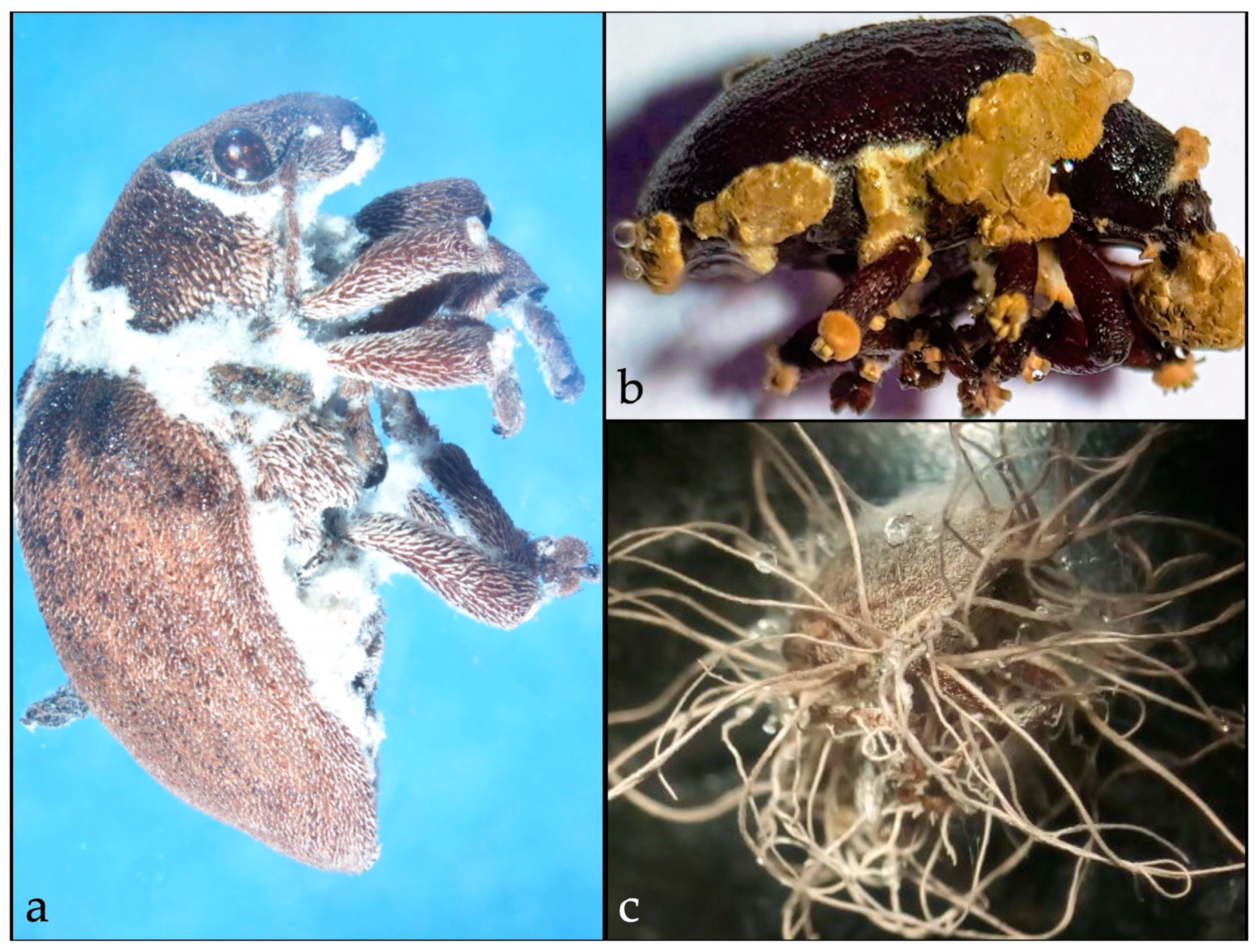
3.4. Asian Longhorned Beetle Control
3.5. Emerald Ash Borer Control
3.6. Oak Lace Bug Control
4. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Dara, S.K. The new integrated pest management paradigm for the modern age. J. IPM 2019, 10, 12. [Google Scholar] [CrossRef]
- Wraight, S.; Inglis, G.; Goettel, M. Fungi. In Field Manual of Techniques in Invertebrate Pathology, 2nd ed.; Lacey, L., Kaya, H., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 223–248. [Google Scholar]
- Samson, R.; Evans, H.; Latgé, J.P. Atlas of Entomopathogenic Fungi, 1st ed.; Springer: Berlin/Heidelberg, Germany, 1988; p. 187. [Google Scholar]
- Goettel, M.; Inglis, G.; Wraight, S. Fungi. In Field Manual of Techniques in Invertebrate Pathology, 1st ed.; Lacey, L., Kaya, H., Eds.; Kluwer Academic Publishers: London, UK, 2000; pp. 255–282. [Google Scholar]
- Hajek, A. Ecology of terrestrial fungal entomopathogens. In Advances in Microbial Ecology, 1st ed.; Jones, G.J., Ed.; Springer: New York, NY, USA, 1997; Volume 15, pp. 193–249. [Google Scholar]
- Charnley, A.; Collins, S. Entomopathogenic fungi and their role in pest control. In The Mycota IV, Environmental and Microbial Relationships, 2nd ed.; Kubicek, C.P., Druzhinina, I.S., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 159–187. [Google Scholar]
- Roy, H.; Brodie, E.; Chandler, D.; Goettel, M.; Pell, J.; Wajnberg, E.; Vega, F. Deep space and hidden depths: Understanding the evolution and ecology of fungal entomopathogens. BioControl 2010, 55, 1–6. [Google Scholar] [CrossRef]
- Humber, R. Seeking stability for research and applied uses of entomopathogenic fungi as biological control agents. J. Asia Pac. Entomol. 2016, 19, 1019–1025. [Google Scholar] [CrossRef]
- Augustyniuk-Kram, A.; Kram, K.J. Entomopathogenic fungi as an important natural regulator of insect outbreaks in forests (Review). In Forest Ecosystems-More than Just Trees; Blanco, J.A., Lo, Y.H., Eds.; InTech: Rijeka, Croatia, 2012; pp. 265–294. [Google Scholar] [CrossRef]
- Liebhold, A. Forest pest management in a changing world. Int. J. Pest Manag. 2012, 58, 289–295. [Google Scholar] [CrossRef]
- Liebhold, A.; Tobin, P. Population ecology of insect invasions and their management. Annu. Rev. Entomol. 2008, 53, 387–408. [Google Scholar] [CrossRef] [PubMed]
- Kenis, M.; Auger-Rozenberg, M.; Roques, A.; Timms, L.; Péré, C.; Cock, M.; Settele, J.; Augustin, S.; Lopez-Vaamonde, C. Ecological effects of invasive alien insects. Biol. Invasions 2009, 11, 21–45. [Google Scholar] [CrossRef]
- Brockerhoff, E.; Barratt, B.; Beggs, J.; Fagan, L.; Malcolm, K.; Phillips, C.; Vink, C. Impacts of exotic invertebrates on New Zealand’s indigenous species and ecosystems. N. Z. J. Ecol. 2010, 34, 158–174. [Google Scholar]
- Watson, G.; Voegtlin, D.; Murphy, S.; Fottit, R. Biogeography of the Cinara cupressi complex (Hemiptera: Aphididae) on Cupressaceae, with description of a pest species introduced into Africa. Bull. Entomol. Res. 1999, 89, 271–283. [Google Scholar] [CrossRef]
- Blackman, R.; Eastop, V. Aphids on the World’s Trees: An Identification and Information Guide, 1st ed.; CAB International: England, UK, 1994; p. 986. [Google Scholar]
- FAO. Global Review of Forest Pests and Diseases, FAO Forestry Paper 156; Food and Agriculture Organization of the United Nations: Rome, Italy, 2009; p. 222. [Google Scholar]
- Montalva, C.; Rojas, E.; Ruiz, C.; Lanfranco, D. El pulgón del ciprés en Chile: Una revisión de la situación actual y antecedentes del control biológico. Bosque 2010, 31, 81–88. [Google Scholar] [CrossRef]
- Carter, C.; Maslen, N. Conifer Lachnids in Britain (Forestry Commission Bulletin); Her Majesty’s Stationery Office: Richmond, UK, 1982; Volume 58, 75p. [Google Scholar]
- Chilima, C. Cinara Cupressi: A Pest of Mulanje cedar and Cypress Trees in Malawi; FRIM Report Nº 89009; Forestry Research Institute of Malawi: Zomba, Malawi, 1989; p. 8. [Google Scholar]
- Ciesla, W.M. The Cypress aphid, Cinara cupressi (Buckton) in Africa. In FAO Exotic Aphid Pests of Conifers: A Crisis in African Forestry, Workshop proceedings, Muguga, Kenya, 3–6 June 1991; Ciesla, W.M., Ed.; Kenya Forestry Research Institute: Muguga, Kenya, 1991; p. 160. [Google Scholar]
- Eskiviski, E.; Agostini, J.; Toloza, R.; Coll, O. Daños Producidos por el Pulgón del Pino Cinara Atlantica W. (Hemiptera: Aphididae) en Plantas Jóvenes de Pinus Taeda L; XI Jornadas Técnicas Forestales y Ambientales; INTA Montecarlo: Montecarlo, Argentina, 2005; p. 6.
- Penteado, S.; Trentini, R.; Tadeo, E.; Reis, W. Ocorrencia, distribucicao, dano e controle de Pulgoes do genero Cinara em Pinus spp. no Brasil. Floresta 2000, 30, 55–64. [Google Scholar] [CrossRef]
- Orondo, S.; Day, R. Cypress aphid (Cinara cupressi) damage to a cypress (Cupressus lusitanica) stand in Kenya. Int. J. Pest Manag. 1994, 40, 141–144. [Google Scholar] [CrossRef]
- Murphy, S.; Nair, K.; Sharma, J. Status and impact of invasive conifer aphid pests in Africa. In Impact of Diseases and Insect Pests in Tropical Forests, Proceedings of the IUFRO Symposium, Peechi, India, 23–26 November 1993; Nair, K., Sharma, J., Varma, R., Eds.; Kerala Forest Research Institute and Forestry Research Support Programme for Asia and the Pacific: Bangkok, Thailand, 1996; pp. 289–297. [Google Scholar]
- Estay, S. Invasive insects in the Mediterranean forests of Chile. In Insects and Diseases of Mediterranean Forest Systems; Paine, T.D., Lieutier, F., Eds.; Springer: Cham, Switzerland, 2016; pp. 379–396. [Google Scholar] [CrossRef]
- Hechenleitner, P.; Gardner, M.; Thonas, T.; Echeverría, C.; Escobar, B.; Brownless, P.; Martínez, C. Plantas Amenazadas del centro-sur de Chile. Distribución, Conservación y Propagación, 1st ed.; Universidad Austral de Chile y Real Jardín Botánico de Edimburgo: Valdivia, Chile, 2005; p. 188. [Google Scholar]
- Jeger, M.; Bragard, C.; Caffier, D.; Candresse, T.; Chatzivassiliou, E.; Dehnen-Schmutz, K.; Gilioli, G.; Jaques-Miret, J.A.; MacLeod, A.; Navajas-Navarro, M.; et al. Pest categorisation of the Gonipterus scutellatus species complex. EFSA J. 2018, 16, 5107. [Google Scholar] [CrossRef]
- Mansilla-Vázquez, J.P. Presencia sobre Eucalyptus globulus Labill de Gonipterus scutellatus Gyll. (Col.Curculionidae) en Galicia. Bol. San. Veg. Plagas 1992, 18, 547–554. [Google Scholar]
- Mally, C.W. The Eucalyptus Snout-beetle. (Gonipterus scutellatus, Gyll.). J. Dep. Agric. S. Afr. 1924, 9, 415–442. [Google Scholar]
- Tooke, F.G.C. The Eucalyptus Snout beetle, Gonipterus scutellatus Gyll. A Study of its Ecology and Control by biological means. In Entomology Memoirs. Department of Agriculture and Forestry, Union of South Africa Union of South Africa; Department of Agricultural Technical Services: Pretoria, South Africa, 1955; p. 282. [Google Scholar]
- Álvarez, M.F.; Lorenzo, H.; Rodriguez, J.R.; Picos, J. Workflow to Improve the Forest Management of Eucalyptus Globulus Stands Affected by Gonipterus Scutellatus in Galicia (Spain) Using Remote Sensing and GIS. 11th SPIE International Symposium on Remote Sensing; SPIE: Maspalomas, Spain, 2004; Volume 5574, pp. 372–383. [Google Scholar] [CrossRef]
- Reis, A.R.; Ferreira, L.; Tomé, M.; Araujo, C.; Branco, M. Efficiency of biological control of Gonipterus platensis (Coleoptera: Curculionidae) by Anaphes nitens (Hymenoptera: Mymaridae) in cold areas of the Iberian Peninsula: Implications for defoliation and wood production in Eucalyptus globulus. For. Ecol. Manag. 2012, 270, 216–222. [Google Scholar] [CrossRef]
- Pinkard, E.A.; Baillie, C.; Patel, V.; Mohammed, C.L. Effects of fertilizing with nitrogen and phosphorus on growth and crown condition of Eucalyptus globulus Labill. Experiencing insect defoliation. For. Ecol. Manag. 2006, 231, 131–137. [Google Scholar] [CrossRef]
- McNamara, D.G. EPPO’s perspective on the gypsy moth in Europe. In Proceedings of the 1995 Annual Gypsy Moth Review, Traverse City, MI, USA, 5–8 November 1995; Department of Agriculture: Lansing, MI, USA, 1996; pp. 60–65. [Google Scholar]
- McManus, M.; Csóka, G. History and impact of gypsy moth in North America and comparison to recent outbreaks in Europe. Acta Silv. Lign. Hung. 2007, 3, 47–64. [Google Scholar]
- Tobin, P.C.; Blackburn, L.M. Slow the Spread: A National Program to Manage the Gypsy Moth; USDA Forest Service General Technical Report NRS-6; USDA Forest Service, Northern Research Station: Newtown Square, PA, USA, 2007; p. 109. [Google Scholar]
- Leuschner, W.A.; Young, J.A.; Walden, S.A.; Ravlin, F.W. Potential benefits of slowing the gypsy moth’s spread. South J. Appl. For. 1996, 20, 65–73. [Google Scholar] [CrossRef][Green Version]
- Doane, C.C.; McManus, M.L. The Gypsy Moth: Research toward Integrated Pest Management; USDA Forest Service Technical Bulletin 1584; Government Printing Office: Washington DC, USA, 1981; p. 514.
- Elkinton, J.S.; Liebhold, A.M. Population dynamics of gypsy moth in North America. Annu. Rev. Entomol. 1990, 35, 571–596. [Google Scholar] [CrossRef]
- Peterson, T.; Williams, R.; Chen, G. Modeled global invasive potential of Asian gypsy moths, Lymantria dispar. Entomol. Exp. Appl. 2007, 125, 39–44. [Google Scholar] [CrossRef]
- Paini, D.R.; Mwebaze, P.; Kuhnert, P.M.; Kriticos, D.J. Global establishment threat from a major forest pest via international shipping: Lymantria dispar. Sci. Rep. 2018, 8, 13723. [Google Scholar] [CrossRef] [PubMed]
- Aukema, J.E.; Leung, B.; Kovacs, K.; Chivers, C.; Britton, K.O.; Englin, J.; Frankel, S.J.; Haight, R.G.; Holmes, T.P.; Liebhold, A.M.; et al. Economic impacts of non-native forest insects in the continental United States. PLoS ONE 2011, 6, e24587. [Google Scholar] [CrossRef] [PubMed]
- Twery, M.J. Effects of defoliation by gypsy moth. In Proceedings of USDA Interagency Gypsy Moth Research Review 1990; Gottschalk, K.W., Twery, M.J., Smith, S.I., Eds.; General Technical Report NE-146; USDA Forest Service, Northeastern Forest Experiment Station: East Windsor, CT, USA, 1990; pp. 27–39. [Google Scholar]
- Fajvan, M.; Wood, J.M. Stand structure and development after gypsy moth defoliation in the Appalachian Plateau. For. Ecol. Manag. 1996, 89, 79–88. [Google Scholar] [CrossRef]
- Haack, R.A.; Hérard, F.; Sun, J.; Turgeon, J.J. Managing invasive populations of Asian longhorned beetle and citrus longhorned beetle: A worldwide perspective. Annu. Rev. Entomol. 1981, 55, 521–546. [Google Scholar] [CrossRef] [PubMed]
- Meng, P.S.; Hoover, K.; Keena, M.A. Asian longhorned beetle (Coleoptera: Cerambycidae), an introduced pest of maple and other hardwood trees in North America and Europe. J. Integr. Pest Manag. 2015, 6, 4. [Google Scholar] [CrossRef]
- Hsiao, K.J. Forest entomology in China: A general review. Crop Prot. 1982, 1, 359–367. [Google Scholar] [CrossRef]
- Williams, D.W.; Lee, H.P.; Kim, I.K. Distribution and abundance of Anoplophora glabripennis (Coleoptera: Cerambycidae) in natural Acer stands in South Korea. Environ. Entomol. 2004, 33, 540–545. [Google Scholar] [CrossRef]
- Pan, H.Y. Review of the Asian Longhorned Beetle: Research, Biology, Distribution and Management in China; For. Dep. Work Pap. FBS/6E; FAO: Rome, Italy, 2005. [Google Scholar]
- Smith, M.T.; Turgeon, J.J.; de Groot, P.; Gasman, B. Asian longhorned beetle, Anoplophora glabripennis (Motschulsky): Lessons learned and opportunities to improve the process of eradication and management. Am. Entomol. 2008, 55, 21–25. [Google Scholar] [CrossRef]
- Tomiczek, C.; Krehan, H.; Menschhorn, P. Dangerous Asian Longhorn Beetle found in Austria: A new threat for our trees. Allg. Forst Z. Waldwirtsch. Umw. 2002, 57, 52–54. [Google Scholar]
- Benker, U.; Bogel, C.; Blaschke, M. Black longicorn required tree felling: Asian longhorned beetle was found in the rural district of Passau. Allg. Forst Z. Waldwirtsch. Umweltvorsorge 2004, 59, 1112. [Google Scholar]
- Cocquempot, C.; Prost, M.; Carmignac, D. Interceptions et introductions en France de Longicornes asiatiques: Cas des Anoplophora glabripennis (Motschulsky) et chinensis (Förster) (Coleoptera Cerambycidae). Bull. Mens. Soc. Linnéenne Lyon 2003, 72, 273–278. [Google Scholar] [CrossRef]
- Morall, L. Insect pests on trees and shrubs in forests and rural areas in 2010. Vakbl. Nat. Bos Landsch. 2011, 8, 23–27. [Google Scholar]
- Forster, B.; Wermelinger, B. First records and reproductions of the Asian longhorned beetle Anoplophora glabripennis (Motschulsky) (Coleoptera, Cerambycidae) in Switzerland. Mitt. Schweiz. Entomol. Ges. 2012, 85, 267–275. [Google Scholar]
- Straw, N.A.; Fielding, N.J.; Tilbury, C.; Williams, D.T.; Inward, D. Host plant selection and resource utilisation by Asian longhorn beetle Anoplophora glabripennis (Coleoptera: Cerambycidae) in southern England. Forestry 2014, 88, 84–95. [Google Scholar] [CrossRef]
- Haack, R.A.; Law, K.R.; Mastro, V.C.; Ossenbruggen, H.S.; Raimo, B.J. New York’s battle with the Asian long-horned beetle. J. For. 1997, 95, 11–15. [Google Scholar]
- Colombo, M.; Limonta, L. Anoplophora malasiaca Thomson (Coleoptera Cerambycidae Lamiinae Lamiini) in Europe. Boll. Zool. Agrar. E Bachic. 2001, 33, 65–68. [Google Scholar]
- Hérard, F.; Ciampitti, M.; Maspero, M.; Krehan, H.; Benker, U.; Boegel, C.; Schrage, R.; Bouhot-Delduc, L.; Bialooki, P. Anoplophora spp. in Europe: Infestations and management process. EPPO Bull. 2006, 36, 470–474. [Google Scholar] [CrossRef]
- Hérard, F.; Maspero, M.; Ramualde, N.; Jucker, C.; Colombo, M.; Ciampitti, M.; Cavagna, B. Anoplophora glabripennis infestation (col.: Cerambycidae) in Italy. EPPO Bull. 2009, 39, 146–152. [Google Scholar] [CrossRef]
- Wermelinger, B. First report of the Citrus longhorned beetle in Switzerland. Der Gart. 2006, 46, 2–4. [Google Scholar]
- Vukadin, A.; Hrašovec, B. Anoplophora chinensis (Forster) in Croatia. Forstsch. Aktuell 2008, 44, 23–24. [Google Scholar]
- Lingafelter, S.W.; Hoebeke, E.R. Revision of the Genus Anoplophora (Coleoptera: Cerambycidae); The Entomological Society of Washington: Washington, DC, USA, 2002; p. 236. [Google Scholar]
- Haack, R.A.; Jendek, E.; Liu, H.; Marchant, K.R.; Petrice, T.R.; Poland, T.M.; Ye, H. The emerald ash borer: A new exotic pest in North America. Newsl. Mich. Entomol. Soc. 2002, 47, 1–5. [Google Scholar]
- USDA Forest Service. Available online: http://www.emeraldashborer.info/index.php (accessed on 13 February 2018).
- Herms, D.A.; McCullough, D.G. Emerald ash borer invasion of North America: History, biology, ecology, impacts, and management. Ann. Rev. Entomol. 2014, 59, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Orlova-Bienkowskaja, M.J.; Volkovitsh, M.G. Range expansion of Agrilus convexicollis in European Russia expedited by the invasion of the emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae). Biol. Invasions 2015, 17, 537–544. [Google Scholar] [CrossRef]
- Valenta, V.; Moser, D.; Kapeller, S.; Essl, F. A new forest pest in Europe: A review of Emerald ash borer (Agrilus planipennis) invasion. J. Appl. Entomol. 2017, 141, 507–526. [Google Scholar] [CrossRef]
- Smitley, D.; Davis, T.; Rebek, E. Progression of ash canopy thinning and dieback outward from the initial infestation of emerald ash borer (Coleoptera: Buprestidae) in southeastern Michigan. J. Econ. Entomol. 2008, 101, 1643–1650. [Google Scholar] [CrossRef]
- Cappaert, D.; Mccullough, D.G.; Poland, T.M.; Siegert, N.W. Emerald ash borer in North America a research and regulatory challenge. Am. Entomol. 2005, 51, 152–165. [Google Scholar] [CrossRef]
- Flower, C.E.; Knight, K.S.; Rebbeck, J.; Gonzalez-Meler, M.A. The relationship between the emerald ash borer (Agrilus planipennis) and ash (Fraxinus spp.) tree decline: Using visual canopy condition assessments and leaf isotope measurements to assess pest damage. For. Ecol. Manag. 2013, 303, 143–147. [Google Scholar] [CrossRef]
- Knight, K.S.; Brown, J.P.; Long, R.P. Factors affecting the survival of ash trees (Fraxinus spp.) infested by emerald ash borer (Agrilus planipennis). Biol. Invasions 2013, 15, 371–383. [Google Scholar] [CrossRef]
- McKenney, D.W.; Pedlar, J.; Yemshanov, D.; Lyons, D.B.; Campbell, K.L.; Lawerence, K. Estimates of the potential cost of emerald ash borer (Agrilus planipennis Fairmaire) in Canadian municipalities. Arboric. Urban For. 2012, 38, 81–91. [Google Scholar]
- Kovacs, K.F.; Haight, R.G.; McCullough, D.G.; Mercader, R.J.; Siegert, N.W.; Liebhold, A.M. Cost of potential emerlad ash borer damage in US communities, 2009–2019. Ecol. Econ. 2010, 69, 569–578. [Google Scholar] [CrossRef]
- Gandhi, J.K.J.; Herms, D.A. Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol. Invasions 2010, 12, 389–405. [Google Scholar] [CrossRef]
- Mutun, S.; Ceyhan, Z.; Sözen, C. Invasion by the oak lace bug, Corythucha arcuata (Say) (Heteroptera: Tingidae), in Turkey. Turk. J. Zool. 2009, 33, 263–268. [Google Scholar] [CrossRef]
- Dobreva, M.; Simov, N.; Georgiev, G.; Mirchev, P.; Georgieva, M. First record of Corythucha arcuata (Say) (Heterotera: Tingidae) on the Balkan Peninsula. Acta Zool. Bulg. 2013, 65, 409–412. [Google Scholar]
- Hrašovec, B.; Posarić, D.; Lukić, I.; Pernek, M. First record of oak lace bug (Corythucha arcuata) in Croatia. Šumarski List 137 2013, 9–10, 499–503. [Google Scholar]
- Pap, P.; Drekić, M.; Poljaković-Pajnik, L.; Marković, M.; Vasić, V. Forest health monitoring in Vojvodina in 2015. Topola 2015, 195–196, 117–133. [Google Scholar]
- Bernardinelli, I.; Zandigiacomo, P. First record of the oak lace bug Corythucha arcuata (Say) (Heteroptera, Tingidae) in Europe. Inf. Fitopatol. 2000, 12, 47–49. [Google Scholar]
- Bernardinelli, I. Distribution of the oak lace bug Corythucha arcuata (Say) in northern Italy (Heteroptera, Tingidae). Redia 2000, 83, 157–162. [Google Scholar]
- Mutun, S. First report of the oak lace bug, Corythucha arcuata (Say, 1832) (Heteroptera: Tingidae) from Bolu, Turkey. Isr. J. Zool. 2003, 49, 323–324. [Google Scholar]
- Forster, B.; Giacalone, I.; Moretti, M.; Dioli, P.; Wermelinger, B. Die amerikanische Eichennetzwanze Corythucha arcuata (Say) (Heteroptera, Tingidae) hat die Südschweitz erreicht. Mitt. Schweiz. Entomol. Ges. 2005, 78, 317–323. [Google Scholar]
- Chireceanu, C.; Teodoru, A.; Chiriloaie, A. New records of the oak lace bug Corythucha arcuata (Say, 1832) (Hemiptera: Tingidae) in Southern Romania. Acta Zool. Bulg. 2017, 9, 297–299. [Google Scholar]
- Connell, W.A.; Beacher, J.H. Life History and Control of the Oak Lace Bug; Delaware Agricultural Experiment Station Bulletin, No. 265; University of Delaware Agricultural Experiment Station: Newark, DE, USA, 1947; p. 28. [Google Scholar]
- Bernardielli, I. Potential host plants of Corythucha arcuata (Het., Tingidae) in Europe: A laboratory study. J. Appl. Entomol. 2006, 130, 480–484. [Google Scholar] [CrossRef]
- Mutun, S. Corythucha ciliata, a new Platanus pest in Turkey. Phytoparasitica 2009, 37, 65–66. [Google Scholar] [CrossRef]
- Simov, N.; Grozeva, S.; Langourov, M.; Georgieva, M.; Mirchev, P.; Georgiev, G. Rapid expansion of the Oak lace bug Corythucha arcuata (Say, 1832) (Hemiptera: Tingidae) in Bulgaria. Hist. Nat. Bulg. 2018, 27, 51–55. [Google Scholar]
- Drake, C.J.; Ruhoff, F.A. Lace Bugs of the World: A Catalog (Hemiptera: Tingidae); United States National Museum Bull. No. 243; Smithsonian Institution: Washington DC, USA, 1965; p. 634. [Google Scholar]
- Drew, W.A.; Arnold, D.C. Tingoidea of Oklahoma (Hemiptera). Proc. Okla. Acad. Sci. 1977, 57, 29–31. [Google Scholar]
- Eilenberg, J.; Hajek, A.; Lomer, C. Suggestions for unifying the terminology in biological control. BioControl 2001, 46, 387–400. [Google Scholar] [CrossRef]
- Pell, J.K.; Eilenberg, J.; Hajek, A.E.; Steinkraus, D.C. Biology, ecology and pest management potential of Entomophthorales. In Fungi as Biocontrol Agents; Butt, T.M., Jackson, C., Magan, N., Eds.; CABI Publishing: Wallingfort, UK, 2001; pp. 71–153. [Google Scholar]
- Shah, P.; Pell, J. Entomopathogenic fungi as biological control agents. Appl. Microbiol. Biotechnol. 2003, 61, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Humber, R.A. Entomophthoromycota: A new phylum and reclassification for entomophthoroid fungi. Mycotaxon 2012, 120, 477–492. [Google Scholar] [CrossRef]
- Strasser, H.; Vey, A.; Butt, T.M. Are there any risks in using entomopathogenic fungi for pest control, with particular reference to the bioactive metabolites of Metarhizium, Tolypocladium and Beauveria species? Biocontrol Sci. Technol. 2000, 10, 717–735. [Google Scholar] [CrossRef]
- Tucker, D.L.; Beresford, C.H.; Sigler, L.; Rogers, K. Disseminated Beauveria bassiana infection in a patient with acute lymphoblastic leukemia. J. Clin. Microbiol. 2004, 42, 5412–5414. [Google Scholar] [CrossRef] [PubMed]
- Westwood, G.S.; Huang, S.W.; Keyhani, N.O. Allergens of the entomopathogenic fungus Beauveria bassiana. Clin. Mol. Allergy 2005, 3, 1–8. [Google Scholar] [CrossRef]
- Henke, M.O.; de Hoog, G.S.; Gross, U.; Zimmermann, G.; Kraemer, D.; Weig, M. Human deep tissue infection with an entomopathogenic Beauveria species. J. Clin. Microbiol. 2002, 40, 2698–2702. [Google Scholar] [CrossRef] [PubMed]
- Patil, S. Development of Entomopathogenic Fungi Based Biopesticide Technology; Lambert Academic Publishing: Latvia, European Union, 2011; p. 188. [Google Scholar]
- Bass, C.; Puinean, A.M.; Zimmer, C.T.; Denholm, I.; Field, L.M.; Foster, S.P.; Gutbrod, O.; Nauen, R.; Slater, R.; Williamson, M.S. The evolution of insecticide resistance in the peach potato aphid, Myzus persicae. Insect Biochem. Mol. Biol. 2014, 51, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Montalva, C.; Barta, M.; Rojas, E.; Valenzuela, E. Neozygites osornensis sp. nov., a new fungal species causing mortality to the cypress aphid Cinara cupressi in Chile. Mycologia 2013, 105, 661–669. [Google Scholar] [CrossRef]
- Loureiro, E.; Oliveira, N.; Wilcken, C.; Filho, A. Patogenicidade de Verticillium lecanii ao pulgão do pinus. Rev. Árvore 2004, 28, 765–770. [Google Scholar] [CrossRef]
- Pereira, M.; Chiarello, S. Seleção de isolados de Verticillium lecanii para o controle de Cinara atlantica. Pesqui Agropecuária Bras. 2005, 40, 1141–1144. [Google Scholar] [CrossRef]
- Montalva, C.; Valenzuela, E.; Barta, M.; Rojas, E.; Arismendi, N.; Rodrigues, J.; Humber, R. Lecanicillium attenuatum isolates affecting the invasive cypress aphid (Cinara cupressi) in Chile. BioControl 2017, 62, 625–637. [Google Scholar] [CrossRef]
- Barta, M.; Cagáň, Ľ. Aphid-pathogenic Entomophthorales (their taxonomy, biology and ecology). Biologia 2006, 61, 543–616. [Google Scholar] [CrossRef]
- Montalva, C.; Barta, M.; Rojas, E.; Gutiérrez, M.; Valenzuela, E. Neozygites species associated with aphids in Chile: Current status and new reports. Mycotaxon 2014, 129, 233–245. [Google Scholar] [CrossRef]
- Montalva, C.; Luz, C.; Humber, R. Neozygites osornensis (Neozygitales: Neozygitaceae) Infecting Cinara sp. (Hemiptera: Aphididae) in Brazil. Neotrop. Entomol. 2016, 45, 227–230. [Google Scholar] [CrossRef]
- Hountondji, F.C.C.; Lomer, C.J.; Hanna, R.; Cherry, A.J.; Dara, S.K. Field evaluation of Brazilian isolates of Neozygites floridana (Entomophthorales: Neozygitaceae) for the microbial control of cassava green mite in Benin, West Africa. Biocontrol Sci. Technol. 2002, 12, 361–370. [Google Scholar] [CrossRef]
- Echeverri-Molina, D.; Santolamazza-Carbone, S. Toxicity of synthetic and biological insecticides against adults of the Eucalyptus snout-beetle Gonipterus scutellatus Gyllenhal (Coleoptera: Curculionidae). J. Pest Sci. 2010, 83, 297–305. [Google Scholar] [CrossRef]
- Hajek, A.E.; van Frankenhuyzen, K. Use of entomopathogens against forest pests. In Microbial Control of Insect and Mite Pests: From Theory to Practice; Lacey, L.A., Ed.; Elsevier: Cambridge, MA, USA, 2017; pp. 313–330. [Google Scholar]
- Gencer, D.; Bayramoglu, Z.; Nalcacioglu, R.; Kleespies, R.G.; Demirbag, Z.; Demir, I. Characterisation of three Alphabaculovirus isolates from the gypsy moth, Lymantria dispar dispar (Lepidoptera: Erebidae), in Turkey. Biocontrol Sci. Technol. 2018, 28, 107–121. [Google Scholar] [CrossRef]
- Blackburn, L.M.; Hajek, A.E. Gypsy Moth Larval Necropsy Guide; General Technical Report NRS-179; USDA Forest Service, Northern Research Station: Newtown Square, PA, USA, 2018; p. 30. [Google Scholar] [CrossRef]
- Soper, R.S.; Shimazu, M.; Humber, R.A.; Ramos, M.E.; Hajek, A.E. Isolation and characterization of Entomophaga maimaiga sp. nov., a fungal pathogen of gypsy moth, Lymantria dispar, from Japan. J. Invertebr. Pathol. 1988, 51, 229–241. [Google Scholar] [CrossRef]
- Speare, A.T.; Colley, R.H. The Artificial Use of the Brown-Tail Fungus in Massachusetts, with Practical Suggestions for Private Experiment, and a Brief Note on a Fungous Disease of the Gypsy Caterpillar; Wright and Potter Printing Co.: Boston, MA, USA, 1912. [Google Scholar]
- Andreadis, T.G.; Weseloh, R.M. Discovery of Entomophaga maimaiga in North American gypsy moth, Lymantria dispar. Proc. Natl. Acad. Sci. USA 1990, 87, 2461–2465. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.; Milgroom, M.G.; Hajek, A.E. Genetic diversity in the gypsy moth fungal pathogen Entomophaga maimaiga from founder populations in North America and source populations in Asia. Mycol. Res. 2005, 109, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Hajek, A.E. Introduction of the entomopathogenic fungus Entomophaga maimaiga into North America. In Biological Control: A Global Perspective; Vincent, C., Goettel, M.S., Lazarovits, G., Eds.; CABI Publishing: Wallingford, UK, 2007; pp. 53–62. [Google Scholar]
- Tobin, P.C.; Hajek, A.E. Release, establishment, and initial spread of the fungal pathogen Entomophaga maimaiga in island populations of Lymantria dispar. Biol. Control 2012, 63, 31–39. [Google Scholar] [CrossRef]
- Pilarska, D.; McManus, M.; Hajek, A.; Herard, F.; Vega, F.; Pilarski, P.; Markova, G. Introduction of the entomopathogenic fungus Entomophaga maimaiga Hum., Shim. & Sop. (Zygomycetes: Entomophtorales) to a Lymantria dispar (L.) (Lepidoptera: Lymantriidae) population in Bulgaria. J. Pest Sci. 2000, 73, 125–126. [Google Scholar]
- Pilarska, D.; McManus, M.; Pilarski, P.; Georgiev, G.; Mirchev, P.; Linde, A. Monitoring the establishment and prevalence of the fungal entomopathogen Entomophaga maimaiga in two Lymantria dispar L. populations in Bulgaria. J. Pest Sci. 2006, 79, 63–67. [Google Scholar] [CrossRef]
- Pilarska, D.; Todorov, M.; Pilarski, P.; Djorova, V.; Solter, L.; Georgiev, G. Bioassays for detection of the entomopathogenic fungus Entomophaga maimaiga (Entomophthorales: Entomophthoraceae) in soil from different sites in Bulgaria. Acta Zool. Bulg. 2013, 65, 173–177. [Google Scholar]
- Georgiev, G.; Mirchev, P.; Rossnev, B.; Petkov, P.; Georgieva, M.; Pilarska, D.; Golemansky, V.; Pilarski, P.; Hubenov, Z. Potential of Entomophaga maimaiga for suppressing Lymantria dispar outbreaks in Bulgaria. Cr. Acad. Bulg. Sci. 2013, 66, 1025–1032. [Google Scholar]
- Kereselidze, M.; Pilarska, D.; Hajek, A.; Jensen, A.B.; Linde, A. First record of Entomophaga maimaiga Humber, Shimazu & Soper (Entomophthorales: Entomophthoraceae) in Georgia. Biocontrol Sci. Technol. 2011, 21, 1375–1380. [Google Scholar] [CrossRef]
- Georgiev, G.; Mirchev, P.; Georgieva, M.; Rossnev, B.; Petkov, P.; Matova, M.; Kitanova, S. First record of entomopathogenic fungus Entomophaga maimaiga Humber, Shimazu and Soper (Entomophthorales: Entomophthoraceae) in Lymantria dispar (Linnaeus) (Lepidoptera: Lymantriidae) in Turkey. Acta Zool. Bulg. 2012, 64, 123–127. [Google Scholar]
- Tabaković-Tošić, M.; Georgiev, G.; Mirchev, P.; Tošić, D.; Golubović-Ćurguz, V. Entomophaga maimaiga–new entomopathogenic fungus in the Republic of Serbia. Afr. J. Biotechnol. 2012, 34, 8571–8577. [Google Scholar] [CrossRef]
- Hrašovec, B.; Pernek, M.; Lukić, I.; Milotić, M.; Diminić, D.; Franjević, M.; Hajek, A.; Linde, A.; Pilarska, D. First record of the pathogenic fungus Entomophaga maimaiga Humber, Shimazu, and Soper (Entomophthorales: Entomophthoraceae) within an outbreak populations of Lymantria dispar (Lepidoptera: Erebidae) in Croatia. Period. Biol. 2013, 115, 379–383. [Google Scholar]
- Csóka, G.Y.; Hirka, A.; Szőcs, L.; Hajek, A.E. First occurrence of the entomopathogenic fungus, Entomophaga maimaiga Humber, Shimazu and Soper (Entomophthorales: Entomophthoraceae) in Hungarian gypsy moth (Lymantria dispar) populations. Növényvédelem 2014, 50, 257–262. [Google Scholar]
- Zúbrik, M.; Barta, M.; Pilarska, D.; Goertz, D.; Úradník, M.; Galko, J.; Vakula, J.; Gubka, A.; Rell, S.; Kunca, A. First record of Entomophaga maimaiga (Entomophthorales: Entomophthoraceae) in Slovakia. Biocontrol. Sci. Technol. 2014, 24, 710–714. [Google Scholar] [CrossRef]
- Zúbrik, M.; Hajek, A.; Pilarska, D.; Spilda, I.; Georgiev, G.; Hrasovec, B.; Hirka, A.; Goertz, D.; Hoch, G.; Barta, M.; et al. The potential for Entomophaga maimaiga to regulate gypsy moth Lymantria dispar (L.) (Lepidoptera: Erebidae) in Europe. J. Appl. Entomol. 2016, 140, 565–579. [Google Scholar] [CrossRef]
- Dubois, T.; Li, Z.; Jaifu, H.; Hajek, A.E. Efficacy of fiber bands impregnated with Beauveria brongniartii cultures against the Asian longhorned beetle, Anoplophora glabripennis (Coleoptera: Cerambycidae). Biol. Control 2004, 31, 320–328. [Google Scholar] [CrossRef]
- Hajek, A.E.; Lund, J.; Smith, M.T. Reduction in fitness of female Asian longhorned beetle (Anoplophora glabripennis) infected with Metarhizium anisopliae. J. Invertebr. Pathol. 2008, 98, 198–205. [Google Scholar] [CrossRef]
- Ugine, T.A.; Jenkins, N.E.; Gardescu, S.; Hajek, A.E. Comparing fungal band formulations for Asian longhorned beetle biological control. J. Invertebr. Pathol. 2013, 113, 240–246. [Google Scholar] [CrossRef]
- Shanley, R.P.; Keena, M.; Wheeler, M.M.; Leland, J.; Hajek, A.E. Evaluating the virulence and longevity of non-woven fiber bands impregnated with Metarhizium anisopliae against Asian longhorned beetle, Anoplophora glabripennis (Coleoptera: Cerambycidae). Biol. Conserv. 2009, 50, 94–102. [Google Scholar] [CrossRef]
- Fisher, J.J.; Castrillo, L.A.; Donzelli, B.G.; Hajek, A.E. Starvation and imidacloprid exposure influence immune response by Anoplophora glabripennis (Coleoptera: Cerambycidae) to a fungal pathogen. J. Econ. Entomol. 2017, 110, 1451–1459. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.J.; Hajek, A.E. Influence of mating and age on susceptibility of the beetle Anoplophora glabripennis to the fungal pathogen Metarhizium brunneum. J. Invertebr. Pathol. 2016, 136, 142–148. [Google Scholar] [CrossRef]
- Fisher, J.J.; Hajek, A.E. Thermoregulatory behavior and fungal infection of Anoplophora glabripennis (Coleoptera: Cerambycidae). Environ. Entomol. 2014, 43, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Goble, T.A.; Rehner, S.A.; Long, S.J.; Gardescu, S.; Hajek, A.E. Comparing virulence of North American Beauveria brongniartii and commercial pathogenic fungi against Asian longhorned beetles. Biol. Conserv. 2014, 72, 91–97. [Google Scholar] [CrossRef]
- Kashio, T.; Ujiye, T. Evaluation of the use of entomogenous fungus, Beauveria tenella, isolated from the yellow spotted longicorn beetle, Psacothea hilaris for the biological control of white spotted longicorn beetle Anoplophora malasiaca. Proc. Assoc. Plant Prot. Kyushu 1988, 34, 190–193. [Google Scholar] [CrossRef]
- Higuchi, T.; Saika, T.; Senda, S.; Mizobata, T.; Kawata, Y.; Nagai, J. Development of biorational pest control formulation against longicorn beetles using a fungus, Beauveria brongniartii (Sacc.). Petch. J. Biosci. Bioeng. 1997, 84, 236–243. [Google Scholar] [CrossRef]
- Liu, H.; Bauer, L.S. Microbial control of emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae) with Beauveria bassiana strain GHA: Greenhouse and field trials. Biol. Conserv. 2008, 45, 124–132. [Google Scholar] [CrossRef]
- Castrillo, L.A.; Griggs, M.H.; Liu, H.; Bauer, L.S.; Vandenberg, J.D. Assessing deposition and persistence of Beauveria bassiana GHA (Ascomycota: Hypocreales) applied for control of the emeralad ash borer, Agrilus planipennis (Coleoptera: Buprestidae) in a commercial tree nursery. Biol. Conserv. 2010, 54, 61–67. [Google Scholar] [CrossRef]
- Lyons, D.B.; Lavallée, R.; Keyi-Poku, G.; Van Frankenhuyzen, K.; Johny, S.; Guertin, C.; Francese, J.A.; Jones, G.C.; Blais, M. Towards the development of an autocontamination trap system to manage populations of emerald ash borer (Coleoptera: Buprestidae) with the native entomopathogenic fungus, Beauveria bassiana. J. Econ. Entomol. 2012, 105, 1929–1939. [Google Scholar] [CrossRef]
- Johny, S.; Kyei-Poku, G.; Gauthier, D.; van Frankenhuyzen, K. Isolation and characterisation of Isaria farinosa and Purpureocillium lilacinum associated with emerald ash borer, Agrilus planipennis in Canada. Biocontrol Sci. Technol. 2012, 22, 723–732. [Google Scholar] [CrossRef]
- Lyons, B.; van Frankenhuyzen, K.; Kyei-Pokua, G.; Johny, S.; Guertin, C.; Lavallée, R.; Jones, G.C.; Blais, M. The use of fluorescent powders to track autocontamination of emerald ash borer (Coleoptera: Buprestidae) by the entomopathogen Beauveria bassiana (Ascomycota: Hypocreales). Biocontrol Sci. Technol. 2016, 26, 1113–1128. [Google Scholar] [CrossRef]
- Sparks, B.; Braman, S.K.; Nair, S. Control of Lace Bugs on Ornamental Plants; The University of Georgia, UGA Extension, Bulletin No. 1102; The University of Georgia: Athens, GA, USA, 2015; p. 2. [Google Scholar]
- Sönmez, E.; Demirbağ, Z.; Demir, I. Pathogenicity of selected entomopathogenic fungal isolates against the oak lace bug, Corythucha arcuata Say (Hemiptera: Tingidae), under controlled conditions. Turk. J. Agric. For. 2016, 40, 715–722. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dara, S.K.; Montalva, C.; Barta, M. Microbial Control of Invasive Forest Pests with Entomopathogenic Fungi: A Review of the Current Situation. Insects 2019, 10, 341. https://doi.org/10.3390/insects10100341
Dara SK, Montalva C, Barta M. Microbial Control of Invasive Forest Pests with Entomopathogenic Fungi: A Review of the Current Situation. Insects. 2019; 10(10):341. https://doi.org/10.3390/insects10100341
Chicago/Turabian StyleDara, Surendra K., Cristian Montalva, and Marek Barta. 2019. "Microbial Control of Invasive Forest Pests with Entomopathogenic Fungi: A Review of the Current Situation" Insects 10, no. 10: 341. https://doi.org/10.3390/insects10100341
APA StyleDara, S. K., Montalva, C., & Barta, M. (2019). Microbial Control of Invasive Forest Pests with Entomopathogenic Fungi: A Review of the Current Situation. Insects, 10(10), 341. https://doi.org/10.3390/insects10100341





