Amylose Inclusion Complexes as Emulsifiers for Garlic and Asafoetida Essential Oils for Mosquito Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation and Spray Drying of Amylose Complexes
2.2. Preparation of Emulsions
2.3. Bioassays
2.4. Determination of Droplet Numbers and Diameter
3. Results
3.1. Toxicity of Essential Oil Emulsions
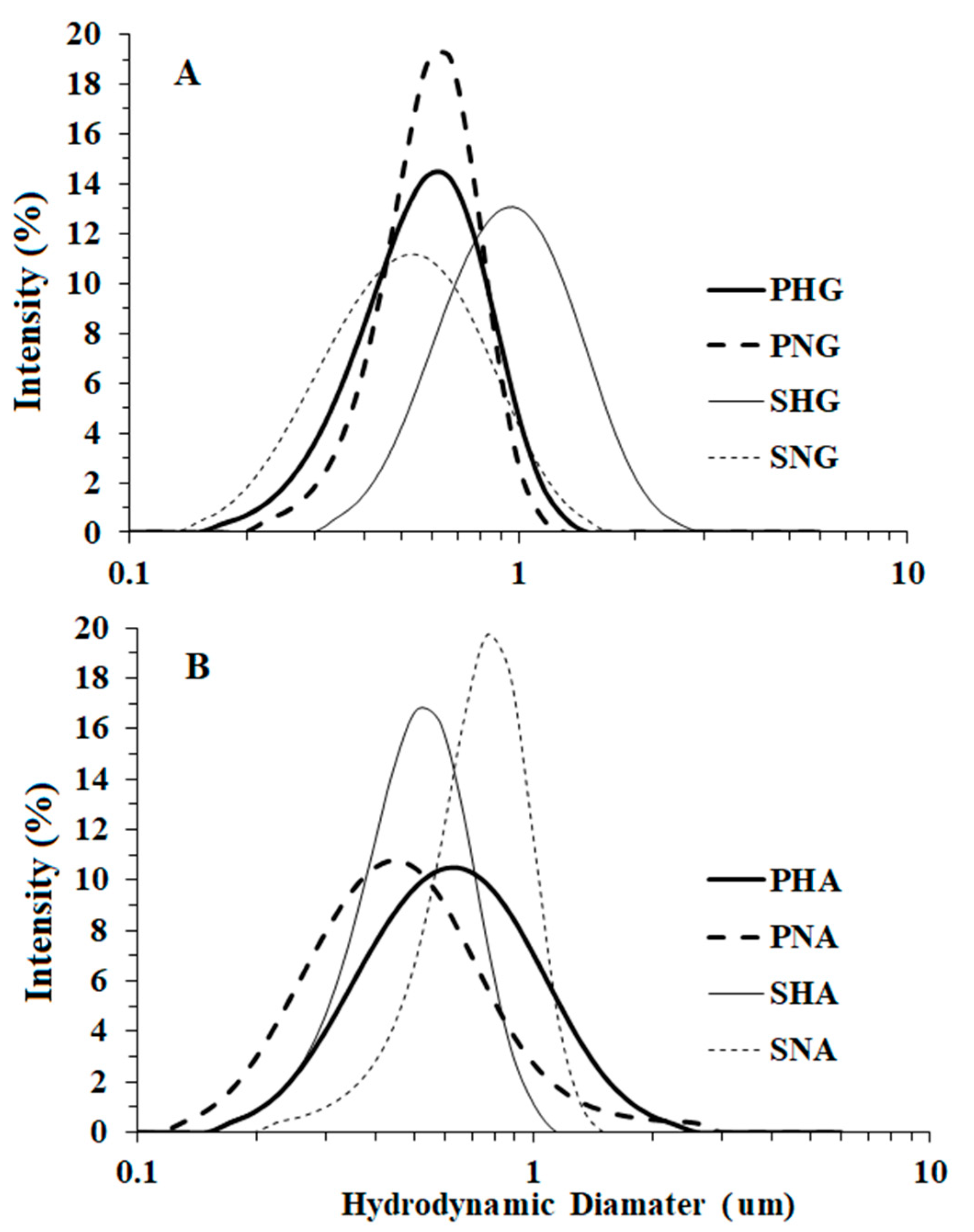
3.2. Droplet Numbers, Size, and Distribution
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Tholl, D. Terpene synthases and the regulation, diversity and biological roles of terpene metabolism. Curr. Opin. Plant Biol. 2006, 9, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Langenheim, J.H. Higher-plant terpenoids-a phytocentric overview of their ecological roles. J. Chem. Ecol. 1994, 20, 1223–1280. [Google Scholar] [CrossRef]
- Aharoni, A.; Jongsma, M.A.; Bouwmeester, H.J. Volatile science? Metabolic engineering of terpenoids in plants. Trends Plant Sci. 2005, 10, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Batish, D.R.; Singh, H.P.; Kohli, S.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. For. Ecol. Manag. 2008, 256, 2166–2174. [Google Scholar] [CrossRef]
- Bhat, S.K.; Kempraj, V. Biocidal potential of clove oils against Aedes albopictus—A comparative study. Afr. J. Biotechnol. 2009, 8, 6933–6937. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Waomar, M. Biological effects of essential oils-A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Manimaran, A.; Cruz, M.M.; Muthu, C.; Vincent, S.; Ignacimuthu, S. Larvicidal and knockdown effects of some essential oils against Culex quinquefasciatus Say, Aedes aegypti (L.) and Anopheles stephensi (Liston). Adv. Biosci. Biotech. 2012, 3, 855–862. [Google Scholar] [CrossRef][Green Version]
- Muturi, E.J.; Ramirez, J.L.; Doll, K.M.; Bowman, M.J. Combined toxicity of three essential oils against Aedes aegypti (Diptera: Culicidae) larvae. J. Med. Entomol. 2017, 54, 1684–1691. [Google Scholar] [CrossRef]
- Warikoo, R.; Wahab, N.; Kumar, S. Oviposition-altering and ovicidal potentials of five essential oils against female adults of the dengue vector, Aedes aegypti L. Parasitol. Res. 2011, 109, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Waliwitiya, R.; Kennedy, C.J.; Lowenberger, C.A. Larvicidal and oviposition-altering activity of monoterpenoids, trans-anithole and rosemary oil to the yellow fever mosquito Aedes aegypti (Diptera: Culicidae). Pest. Manag. Sci. 2009, 65, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Noosidum, A.; Chareonviriyaphap, T.; Chandrapatya, A. Synergistic repellent and irritant effect of combined essential oils on Aedes aegypti (L.) mosquitoes. J. Vector Ecol. 2014, 39, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Gokulakrishnan, J.; Kuppusamy, E.; Shanmugam, D.; Appavu, A.; Kaliyamoorthi, K. Pupicidal and repellent activities of Pogostemon cablin essential oil chemical compounds against medically important human vector mosquitoes. Asia Pac. J. Trop. Dis. 2013, 3, 26–31. [Google Scholar] [CrossRef]
- McAllister, J.C.; Adams, M.F. Mode of action for natural products isolated from essential oils of two trees is different from available mosquito adulticides. J. Med. Entomol. 2010, 47, 1123–1126. [Google Scholar] [CrossRef] [PubMed]
- Tong, F.; Bloomquist, J.R. Plant essential oils affect the toxicities of carbaryl and permethrin against Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2013, 50, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.D.; Norris, E.J.; Kimber, M.J.; Bartholomay, L.C.; Coats, J.R. Essential oils enhance the toxicity of permethrin against Aedes aegypti and Anopheles gambiae. Med. Vet. Entomol. 2017, 31, 55–62. [Google Scholar] [CrossRef]
- Martin, A.; Varona, S.; Navarrete, A.; Cocero, M.J. Encapsulation and co-precipitation processes with supercritical fluids: Applications with essential oils. Open Chem. Eng. J. 2010, 4, 31–41. [Google Scholar] [CrossRef]
- Marinopoulou, A.; Papastergiadis, E.; Raphaelides, S.N.; Kontominas, M.G. Morphological characteristics, oxidative stability and enzymic hydrolysis of amylose-fatty acid complexes. Carbohydr. Polym. 2016, 141, 106–115. [Google Scholar] [CrossRef]
- Maes, C.; Bouquillon, S.; Fauconnier, M.L. Encapsulation of essential oils for the development of biosourced pesticides with controlled release: A review. Molecules 2019, 24, 2539. [Google Scholar] [CrossRef]
- McMclements, D.J. Food Emulsions, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 2–26. [Google Scholar]
- Schramm, L.L.; Stasiuk, E.N.; Marangoni, G.D. Surfactants and their applications. Annu. Rep. Prog. Chem. Sect. C Phys. Chem. 2003, 99, 3–48. [Google Scholar] [CrossRef]
- Donsì, F.; Annunziata, M.; Vincensi, M.; Ferrari, G. Design of nanoemulsion-based delivery systems of natural antimicrobials: Effect of the emulsifier. J. Biotechnol. 2012, 159, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, E. Hydrocolloids at interfaces and the influence on the properties of dispersed systems. Food Hydrocol. 2003, 17, 25–39. [Google Scholar] [CrossRef]
- Dickinson, E.; Galazka, V.B.; Anderson, D.M.W. Emulsifying behavior of gum-arabic. Part 2: Effect of the gum molecular-weight on the emulsion droplet-size distribution. Carbohydr. Polym. 1991, 14, 385–392. [Google Scholar] [CrossRef]
- Matos, M.; Marefati, A.; Gutierrez, G.; Wahlgren, M.; Rayner, M. Comparative emulsifying properties of octenyl succinic anhydride (OSA)-modified starch: Granular form vs. dissolved state. PLoS ONE 2016, 11, e0160140. [Google Scholar] [CrossRef] [PubMed]
- Carlson, T.L.G.; Larsson, K.; Dinhnguyen, N.; Krog, N. Study of the amylose-monoglyceride complex by Raman-Spectroscopy. Starke 1979, 31, 222–224. [Google Scholar] [CrossRef]
- Nimz, O.; Gessler, K.; Uson, I.; Sheldrick, G.M.; Saenger, W. Inclusion complexes of V-amylose with undecanoic acid and dodecanol at atomic resolution: X-ray structures with cycloamylose containing 26 D-glucoses (cyclohexalcosaose) as host. Carbohydr. Res. 2004, 339, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Obiro, W.C.; Ray, S.S.; Emmambux, M.N. V-amylose structural characteristics, methods of preparation, significance, and potential applications. Food Rev. Int. 2012, 28, 412–438. [Google Scholar] [CrossRef]
- Immel, S.; Lichtenthaler, F.W. The hydrophobic topographies of amylose and its blue iodine complex. Starch-Starke 2000, 52, 1–8. [Google Scholar] [CrossRef]
- Zhu, F. Interactions between starch and phenolic compound. Trends Food Sci. Tech. 2015, 43, 129–143. [Google Scholar] [CrossRef]
- Eller, F.J.; Hay, W.T.; Kirker, G.T.; Mankowski, M.E.; Sellling, G.W. Hexadecyl ammonium chloride amylose inclusion complex to emulsify cedarwood oil and treat wood against termites and wood-decay fungi. Int. Biodeterior. Biodegrad. 2018, 129, 95–101. [Google Scholar] [CrossRef]
- Klem, R.E.; Brogly, D.A. Method for selecting the optimum starch binder preparation system. Pulp Pap. 1981, 55, 98–103. [Google Scholar]
- Fanta, G.F.; Kenar, J.A.; Byars, J.A.; Felker, F.C.; Shogren, R.L. Properties of aqueous dispersions of amylose-sodium palmitate complexes prepared by steam jet cooking. Carbohydr. Polym. 2010, 81, 645–651. [Google Scholar] [CrossRef]
- Fanta, G.F.; Kenar, J.A.; Felker, F.C. Preparation and properties of amylose complexes prepared from hexadecylamine and its hydrochloride salt. Carbohydr. Polym. 2013, 98, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Hay, W.T.; Fanta, G.F.; Felker, F.C.; Peterson, S.C.; Skory, C.D.; Hojilla-Evangelista, M.P.; Biresaw, G.; Selling, G.W. Emulsification properties of amylose-fatty sodium salt inclusion complexes. Food Hydrocol. 2019, 90, 490–499. [Google Scholar] [CrossRef]
- De Fenoyl, L.; Hirel, D.; Perez, E.; Lecomte, S.; Morvan, E.; Delample, M. Interfacial activity and emulsifying behaviour of inclusion complexes between helical polysaccharides and flavouring molecules resulting from non-covalent interactions. Food Res. Int. 2018, 105, 801–811. [Google Scholar] [CrossRef]
- Kimbaris, A.C.; Kioulos, E.; Koliopoulos, G.; Polissiou, M.G.; Michaelakis, A. Coactivity of sulfide ingredients: A new perspective of the larvicidal activity of garlic essential oil against mosquitoes. Pest Manag. Sci. 2009, 65, 249–254. [Google Scholar] [CrossRef]
- Muturi, E.J.; Ramirez, J.L.; Zilkowski, B.; Flor-Weiler, L.B.; Rooney, A.P. Ovicidal and larvicidal effects of garlic and asafoetida essential oils against West Nile virus vectors. J. Insect Sci. 2018, 18, 43. [Google Scholar] [CrossRef]
- Fanta, G.F.; Shogren, R.L.; Salch, J.H. Steam jet cooking of high-amylose starch fatty acid mixtures. An investigation of complex formation. Carbohydr. Polym. 1999, 38, 1–6. [Google Scholar] [CrossRef]
- Hay, W.T.; Behle, R.W.; Fanta, G.F.; Felker, F.C.; Peterson, S.C.; Selling, G.W. Effect of spray drying on the properties of amylose-hexadecylammonium chloride inclusion complexes. Carbohydr. Polym. 2017, 157, 1050–1056. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Robertson, J.L.; Preisler, H.K. Pesticide Bioassays with Arthropods; CRC Press, Inc.: Boca Raton, FL, USA, 1992. [Google Scholar]
- Pearce, K.N.; Kinsella, J.E. Emulsifying properties of proteins-evaluation of a turbidimetric technique. J. Agric. Food Chem. 1978, 26, 716–723. [Google Scholar] [CrossRef]
- Montefuscoli, A.R.; Werdin Gonzalez, J.O.; Palma, S.D.; Ferrero, A.A.; Fernandez Band, B. Design and development of aqueous nanoformulations for mosquito control. Parasitol. Res. 2014, 113, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.; Xia, T.; Madler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Okonogi, S.; Chaiyana, W. Enhancement of anti-cholinesterase activity of Zingiber cassumunar essential oil using a microemulsion technique. Drug Discov. Ther. 2012, 6, 249–255. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Boudko, D.Y.; Moroz, L.L.; Linser, P.J.; Trimarchi, J.R.; Smith, P.J.; Harvey, W.R. In situ analysis of pH gradients in mosquito larvae using non-invasive, self-referencing, pH-sensitive microelectrodes. J. Exp. Biol. 2001, 204, 691–699. [Google Scholar] [PubMed]
- Hay, W.T.; Byars, J.A.; Fanta, G.F.; Selling, G.W. Rheological characterization of solutions and thin films made from amylose-hexadecylammonium chloride inclusion complexes and polyvinyl alcohol. Carbohydr. Polym. 2017, 161, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Chanamai, R.; McClements, D.J. Comparison of gum arabic, modified starch, and whey protein isolate as emulsifiers: Influence of pH, CaCl and temperature. J. Food Sci. 2002, 67, 120–125. [Google Scholar] [CrossRef]
- Kruyt, H.R. Colloid Science; Elsevier Pu. Co.: Amsterdam, The Netherlands, 1952. [Google Scholar]
- Napper, D.H. Polymeric Stabilization of Dispersions; Academic Press: London, UK, 1983. [Google Scholar]
- Khandpur, A.K.; Forster, S.; Bates, F.S.; Hamley, I.W.; Ryan, A.J.; Bras, W.; Almdal, K.; Mortensen, K. Polyisoprene-polystyrene diblock copolymer phase diagram near the order-disorder transition. Macromolecules 1995, 28, 8796–8806. [Google Scholar] [CrossRef]
- Wurm, F.; Kilbinger, A.F. Polymeric janus particles. Angew. Chem. Int. Ed. 2009, 48, 8412–8421. [Google Scholar] [CrossRef]
- Enan, E. Insecticidal activity of essential oils: Octopaminergic sites of action. Comp. Biochem. Physiol. C Toxicol. Pharmcol. 2001, 130, 325–337. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Essential oils as ecofriendly biopesticides? Challenges and constraints. Trends Plant Sci. 2016, 21, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Tak, J.H.; Isman, M.B. Enhanced cuticular penetration as the mechanism for synergy of insecticidal constituents of rosemary essential oil in Trichoplusia ni. Sci. Rep. 2015, 5, 12690. [Google Scholar] [CrossRef] [PubMed]
- Junnila, A.; Revay, E.E.; Muller, G.C.; Kravchenko, V.; Qualls, W.A.; Xue, R.D.; Allen, S.A.; Beier, J.C.; Schlein, Y. Efficacy of attractive toxic sugar baits (ATSB) against Aedes albopictus with garlic oil encapsulated in beta-cyclodextrin as the active ingredient. Acta Trop. 2015, 152, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Duarte, J.L.; Amado, J.R.R.; Oliveira, A.E.M.F.M.; Cruz, R.A.S.; Ferreira, A.M.; Souto, R.N.P.; Falcao, D.Q.; Carvalho, J.C.T.; Fernandes, C.P. Evaluation of larvicidal activity of a nanoemulsion of Rosmarinus officinalis essential oil. Rev. Bras. Farmacogn. 2015, 25, 189–192. [Google Scholar] [CrossRef]
- Oliveira, A.E.; Duarte, J.L.; Amado, J.R.; Cruz, R.A.; Rocha, C.F.; Souto, R.N.; Ferreira, R.M.; Santos, K.; da Conceicao, E.C.; de Oliveira, L.A.; et al. Development of a larvicidal nanoemulsion with Pterodon emarginatus Vogel oil. PLoS ONE 2016, 11, e0145835. [Google Scholar] [CrossRef] [PubMed]
- Sugumar, S.; Clarke, S.K.; Nirmala, M.J.; Tyagi, B.K.; Mukherjee, A.; Chandrasekaran, N. Nanoemulsion of eucalyptus oil and its larvicidal activity against Culex quinquefasciatus. Bull. Entomol. Res. 2014, 104, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Lacey, L.A. Bacillus thuringiensis serovariety israelensis and Bacillus sphaericus for mosquito control. J. Am. Mosq. Control Assoc. 2007, 23, 133–163. [Google Scholar] [CrossRef]
- Loke, S.R.; Andy-Tan, W.A.; Benjamin, S.; Lee, H.L.; Sofian-Azirun, M. Susceptibility of field-collected Aedes aegypti (L.) (Diptera: Culicidae) to Bacillus thuringiensis israelensis and temephos. Trop. Biomed. 2010, 27, 493–503. [Google Scholar]
- Gomez, A.; Seccacini, E.; Zerba, E.; Licastro, S. Comparison of the insecticide susceptibilities of laboratory strains of Aedes aegypti and Aedes albopictus. Mem. Inst. Oswaldo Cruz 2011, 106, 993–996. [Google Scholar] [CrossRef]
- Stalinski, R.; Tetreau, G.; Gaude, T.; Despres, L. Pre-selecting resistance against individual Bti Cry toxins facilitates the development of resistance to the Bti toxins cocktail. J. Invertebr. Pathol. 2014, 119, 50–53. [Google Scholar] [CrossRef]
- Tetreau, G.; Bayyareddy, K.; Jones, C.M.; Stalinski, R.; Riaz, M.A.; Paris, M.; David, J.P.; Adang, M.J.; Despres, L. Larval midgut modifications associated with Bti resistance in the yellow fever mosquito using proteomic and transcriptomic approaches. BMC Genom. 2012, 13, 248. [Google Scholar] [CrossRef] [PubMed]
- Tetreau, G.; Stalinski, R.; David, J.P.; Despres, L. Increase in larval gut proteolytic activities and Bti resistance in the dengue fever mosquito. Arch. Insect Biochem. Physiol. 2013, 82, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.S.; Shin, E.H.; Yoo, D.H.; Ahn, Y.J. Enhanced toxicity of binary mixtures of Bacillus thuringiensis subsp. israelensis and three essential oil major constituents to wild Anopheles sinensis (Diptera: Culicidae) and Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2014, 51, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Ribeiro, A.M.; de Melo Carrasco, L.D. Cationic antimicrobial polymers and their assemblies. Int. J. Mol. Sci. 2013, 14, 9906–9946. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Label | n | LC50 (95% CI) | Chi-Square | LDRs at LC50 (95% CI) |
|---|---|---|---|---|---|
| Garlic | Garlic | 180 | 7.95 (7.19–8.66) | 29.84 | |
| Garlic/Amylose-Hex-Am powder | PHG | 180 | 6.62 (6.23–6.98) | 5.63 | 1.20 (1.14–1.26) |
| Garlic/Sodium palmitate powder | PNG | 180 | 7.24 (6.31–8.08) | 22.40 | 1.10 (1.04–1.16) |
| Garlic/Amylose-Hex-Am solution | SHG | 180 | 5.97 (5.68–6.24) | 4.18 | 1.33 (1.26–1.41) |
| Garlic/Sodium palmitate solution | SNG | 180 | 6.29 (5.70–6.83) | 7.79 | 1.26 (1.19–1.34) |
| Asafoetida | Asafoetida | 180 | 10.57 (8.74–12.88) | 81.68 | |
| Asafoetida/Amylose-Hex-Am powder | PHA | 180 | 9.03 (8.53–9.51) | 8.66 | 1.17 (1.12–1.22) |
| Asafoetida/Sodium palmitate powder | PNA | 180 | 9.90 (9.15–10.62) | 14.78 | 1.07 (1.02–1.12) |
| Asafoetida/Amylose-Hex-Am solution | SHA | 180 | 8.11 (7.67–8.54) | 8.75 | 1.30 (1.25–1.36) |
| Asafoetida/Sodium palmitate solution | SNA | 180 | 9.43 (8.99–9.88) | 5.12 | 1.12 (1.07–1.18) |
| Treatment | Label | Concentration (Particles/mL) (02/21/18) | Concentration (Particles/mL) (8/17/18) |
|---|---|---|---|
| Garlic/Amylose-Hex-Am powder | PHG | 1.11 × 109 | 3.22 × 109 |
| Garlic/Sodium palmitate powder | PNG | 2.52 × 109 | 2.54 × 109 |
| Garlic/Amylose-Hex-Am solution | SHG | - | 9.55 × 109 |
| Garlic/Sodium palmitate solution | SNG | - | 3.48 × 109 |
| Asafoetida/Amylose-Hex-Am powder | PHA | 1.79 × 109 | 4.18 × 109 |
| Asafoetida/Sodium palmitate powder | PNA | 3.96 × 109 | 2.33 × 109 |
| Asafoetida/Amylose-Hex-Am solution | SHA | - | 4.34 × 109 |
| Asafoetida/Sodium palmitate solution | SNA | - | 2.73 × 109 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muturi, E.J.; Hay, W.T.; Behle, R.W.; Selling, G.W. Amylose Inclusion Complexes as Emulsifiers for Garlic and Asafoetida Essential Oils for Mosquito Control. Insects 2019, 10, 337. https://doi.org/10.3390/insects10100337
Muturi EJ, Hay WT, Behle RW, Selling GW. Amylose Inclusion Complexes as Emulsifiers for Garlic and Asafoetida Essential Oils for Mosquito Control. Insects. 2019; 10(10):337. https://doi.org/10.3390/insects10100337
Chicago/Turabian StyleMuturi, Ephantus J., William T. Hay, Robert W. Behle, and Gordon W. Selling. 2019. "Amylose Inclusion Complexes as Emulsifiers for Garlic and Asafoetida Essential Oils for Mosquito Control" Insects 10, no. 10: 337. https://doi.org/10.3390/insects10100337
APA StyleMuturi, E. J., Hay, W. T., Behle, R. W., & Selling, G. W. (2019). Amylose Inclusion Complexes as Emulsifiers for Garlic and Asafoetida Essential Oils for Mosquito Control. Insects, 10(10), 337. https://doi.org/10.3390/insects10100337





