Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance
Abstract
1. Introduction
2. Materials and Methods
2.1. Survival Assay
2.2. Pathogen Tolerance Assay
2.3. Statistical Analysis
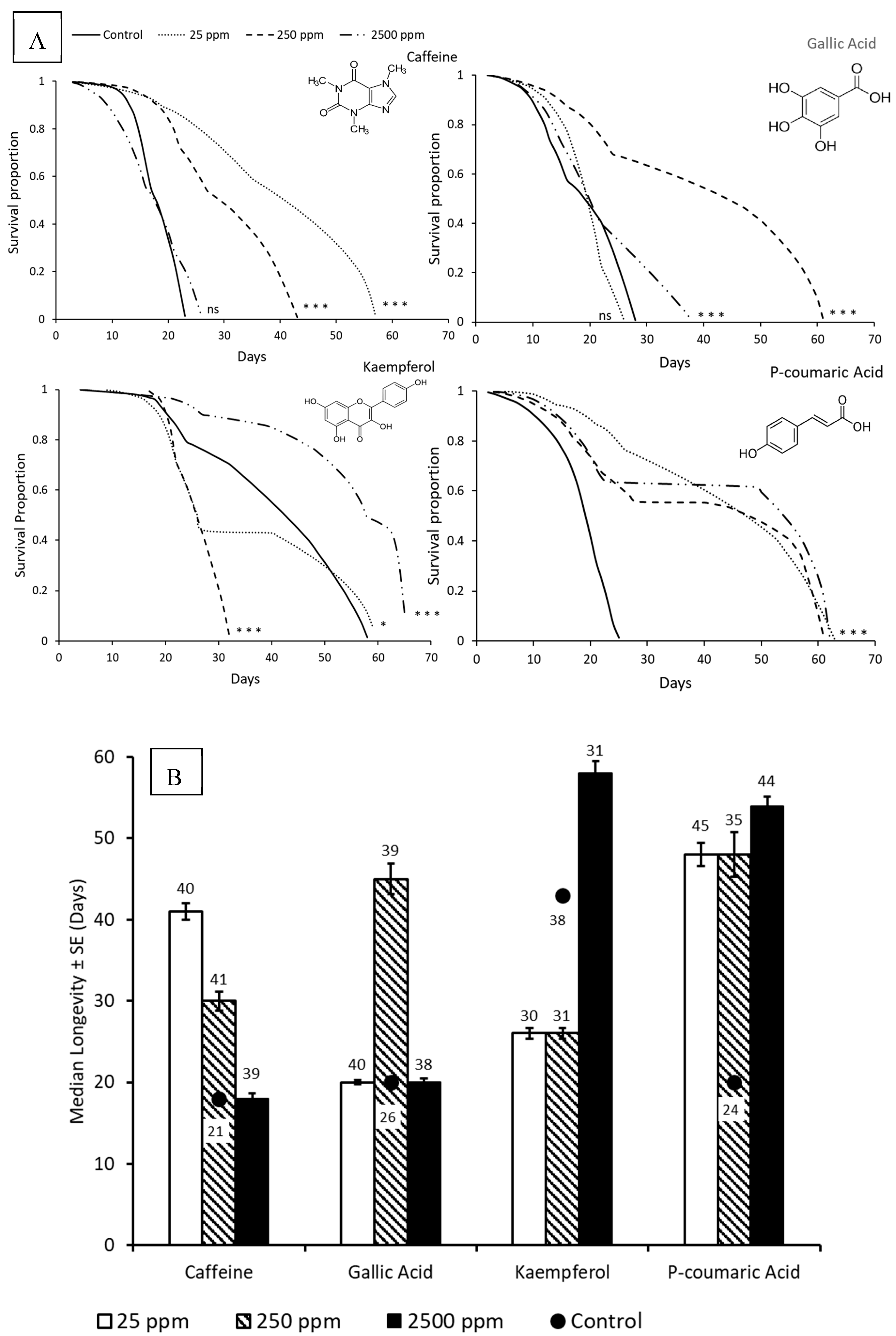
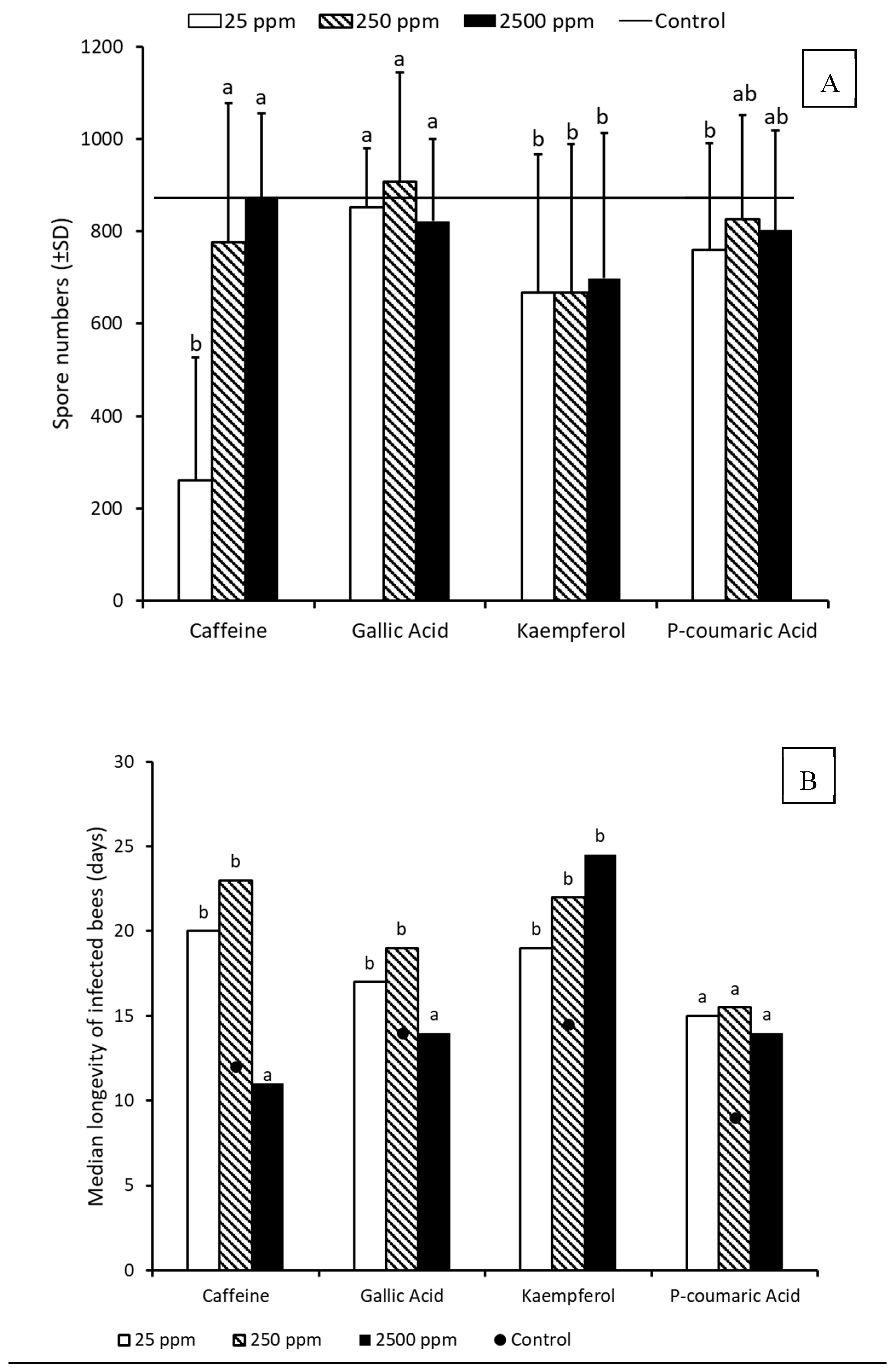
3. Results
3.1. Survival
3.2. Pathogen Tolerance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347. [Google Scholar] [CrossRef] [PubMed]
- Naug, D. Nutritional stress due to habitat loss may explain recent honeybee colony collapses. Biol. Conserv. 2009, 142, 2369–2372. [Google Scholar] [CrossRef]
- Gallant, A.L.; Euliss, N.H., Jr.; Browning, Z. Mapping large-area landscape suitability for honey bees to assess the influence of land-use change on sustainability of national pollination services. PLoS ONE 2014, 9, e99268. [Google Scholar] [CrossRef] [PubMed]
- Brodschneider, R.; Crailsheim, K. Nutrition and health in honey bees. Apidologie 2010, 41, 278–294. [Google Scholar] [CrossRef]
- Scofield, H.N.; Mattila, H.R. Honey Bee Workers That Are Pollen Stressed as Larvae Become Poor Foragers and Waggle Dancers as Adults. PLoS ONE 2015, 10, e0121731. [Google Scholar] [CrossRef] [PubMed]
- Requier, F.; Odoux, J.F.; Henry, M.; Bretagnolle, V. The carry-over effects of pollen shortage decrease the survival of honeybee colonies in farmlands. J. Appl. Ecol. 2017, 54, 1161–1170. [Google Scholar] [CrossRef]
- Simone-Finstrom, M.; Borba, R.; Wilson, M.; Spivak, M. Propolis Counteracts Some Threats to Honey Bee Health. Insects 2017, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.H.; Wu, W.Y.; Berenbaum, M.R. Impacts of dietary phytochemicals in the presence and absence of pesticides on longevity of honey bees (Apis mellifera). Insects 2017, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Bronstein, J.L. Our Current Understanding of Mutualism. Q. Rev. Boil. 1994, 69, 31–51. [Google Scholar] [CrossRef]
- Erler, S.; Moritz, R.F.A. Pharmacophagy and pharmacophory: Mechanisms of self-medication and disease prevention in the honeybee colony (Apis mellifera). Apidologie 2016, 47, 389–411. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Suchail, S.; Brunet, J.-L.; Alaux, C. Influence of pollen nutrition on honey bee health: Do pollen quality and diversity matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef] [PubMed]
- Baker, H.G. Non-sugar chemical constituents of nectar. Apidologie 1977, 8, 349–356. [Google Scholar] [CrossRef]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Boil. Lett. 2010. [Google Scholar] [CrossRef] [PubMed]
- Kather, R.; Drijfhout, F.P.; Martin, S.J. Task Group Differences in Cuticular Lipids in the Honey Bee Apis mellifera. J. Chem. Ecol. 2011, 37, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Ricigliano, V.A.; Fitz, W.; Copeland, D.C.; Mott, B.M.; Maes, P.; Floyd, A.S.; Dockstader, A.; Anderson, K.E. The impact of pollen consumption on honey bee (Apis mellifera) digestive physiology and carbohydrate metabolism. Arch. Insect Biochem. Physiol. 2017, 96, e21406. [Google Scholar] [CrossRef] [PubMed]
- DeGroot, A. Qualitative amino acid requirements of the honeybee. Acta Physiol. Pharmacol. Neerlandica 1954, 3, 433–434. [Google Scholar]
- Sagili, R.R.; Pankiw, T. Effects of protein-constrained brood food on honey bee (Apis mellifera L.) pollen foraging and colony growth. Behav. Ecol. Sociobiol. 2007, 61, 1471–1478. [Google Scholar] [CrossRef]
- Richardson, L.L.; Adler, L.S.; Leonard, A.S.; Andicoechea, J.; Regan, K.H.; Anthony, W.E.; Manson, J.S.; Irwin, R.E. Secondary metabolites in floral nectar reduce parasite infections in bumblebees. Proc. R. Soc. B 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.S.; Otterstatter, M.C.; Thomson, J.D. Consumption of a nectar alkaloid reduces pathogen load in bumble bees. Oecologia 2010, 162, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Honey constituents up-regulate detoxification and immunity genes in the western honey bee Apis mellifera. Proc. Natl. Acad. Sci. USA 2013, 110, 8842–8846. [Google Scholar] [CrossRef] [PubMed]
- Doetterl, S.; Vereecken, N.J. The chemical ecology and evolution of bee-flower interactions: A review and perspectives. Can. J. Zool. 2010, 88, 668–697. [Google Scholar] [CrossRef]
- Stevenson, P.C.; Nicolson, S.W.; Wright, G.A. Plant secondary metabolites in nectar: Impacts on pollinators and ecological functions. Funct. Ecol. 2017, 31, 65–75. [Google Scholar] [CrossRef]
- Irwin, R.E.; Cook, D.; Richardson, L.L.; Manson, J.S.; Gardner, D.R. Secondary Compounds in Floral Rewards of Toxic Rangeland Plants: Impacts on Pollinators. J. Agric. Food Chem. 2014, 62, 7335–7344. [Google Scholar] [CrossRef] [PubMed]
- Palmer-Young, E.C.; Tozkar, C.Ö.; Schwarz, R.S.; Chen, Y.; Irwin, R.E.; Adler, L.S.; Evans, J.D. Nectar and Pollen Phytochemicals Stimulate Honey Bee (Hymenoptera: Apidae) Immunity to Viral Infection. J. Econ. Entomol. 2017, 110, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Lodesani, M.; Maistrello, L. Effect of thymol and resveratrol administered with candy or syrup on the development of Nosema ceranae and on the longevity of honeybees (Apis mellifera L.) in laboratory conditions. Apidologie 2010, 41, 141–150. [Google Scholar] [CrossRef]
- Arismendi, N.; Vargas, M.; López, M.D.; Barría, Y.; Zapata, N. Promising antimicrobial activity against the honey bee parasite Nosema ceranae by methanolic extracts from Chilean native plants and propolis. J. Apic. Res. 2018, 1–14. [Google Scholar] [CrossRef]
- Keckes, S.; Gasic, U.; Velickovic, T.C.; Milojkovic-Opsenica, D.; Natic, M.; Tesic, Z. The determination of phenolic profiles of Serbian unifloral honeys using ultra-high-performance liquid chromatography/high resolution accurate mass spectrometry. Food Chem. 2013, 138, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Pyrzynska, K.; Biesaga, M. Analysis of phenolic acids and flavonoids in honey. Trends Anal. Chem. 2009, 28, 893–902. [Google Scholar] [CrossRef]
- Carayon, J.-L.; Téné, N.; Bonnafé, E.; Alayrangues, J.; Hotier, L.; Armengaud, C.; Treilhou, M. Thymol as an alternative to pesticides: Persistence and effects of Apilife Var on the phototactic behavior of the honeybee Apis mellifera. Environ. Sci. Pollut. Res. 2014, 21, 4934–4939. [Google Scholar] [CrossRef] [PubMed]
- Parachnowitsch, A.L.; Manson, J.S.; Sletvold, N. Evolutionary ecology of nectar. Ann. Bot. 2018. [Google Scholar] [CrossRef] [PubMed]
- Parachnowitsch, A.L.; Manson, J.S. The chemical ecology of plant-pollinator interactions: Recent advances and future directions. Curr. Opin. Insect Sci. 2015, 8, 41–46. [Google Scholar] [CrossRef]
- Arathi, H.S.; Bjostad, L.; Bernklau, E. Metabolomic analysis of pollen from honey bee hives and from canola flowers. Metabolomics 2018, 14, 86. [Google Scholar] [CrossRef]
- Detzel, A.; Wink, M. Attraction, deterrence or intoxication of bees (Apis mellifera) by plant allelochemicals. Chemoecology 1993, 4, 8–18. [Google Scholar] [CrossRef]
- Nicolson, S.W. Bee Food: The Chemistry and Nutritional Value of Nectar, Pollen and Mixtures of the Two. Afr. Zool. 2011, 46, 197–204. [Google Scholar] [CrossRef]
- Baker, H.G.; Baker, I. Studies of nectar-constitution and pollinator-plant coevolution. Coevol. Anim. Plants 1975, 100, 591–600. [Google Scholar]
- Wright, G.A.; Baker, D.D.; Palmer, M.J.; Stabler, D.; Mustard, J.A.; Power, E.F.; Borland, A.M.; Stevenson, P.C. Caffeine in Floral Nectar Enhances a Pollinator’s Memory of Reward. Science 2013, 339, 1202–1204. [Google Scholar] [CrossRef] [PubMed]
- Strachecka, A.; Krauze, M.; Olszewski, K.; Borsuk, G.; Paleolog, J.; Merska, M.; Chobotow, J.; Bajda, M.; Grzywnowicz, K. Unexpectedly strong effect of caffeine on the vitality of western honeybees (Apis mellifera). Biochemistry (Mosc.) 2014, 79, 1192–1201. [Google Scholar] [CrossRef] [PubMed]
- Arathi, H.S.; Burns, I.; Spivak, M. Ethology of Hygienic Behaviour in the Honey Bee Apis mellifera L. (Hymenoptera: Apidae): Behavioural Repertoire of Hygienic Bees. Ethology 2000, 106, 365–379. [Google Scholar] [CrossRef]
- Arathi, H.S.; Spivak, M. Influence of colony genotypic composition on the performance of hygienic behaviour in the honeybee, Apis mellifera L. Anim. Behav. 2001, 62, 57–66. [Google Scholar] [CrossRef]
- Arathi, H.S.; Ho, G.; Spivak, M. Inefficient task partitioning among nonhygienic honeybees, Apis mellifera L., and implications for disease transmission. Anim. Behav. 2006, 72, 431–438. [Google Scholar] [CrossRef]
- Jack, C.J.; Lucas, H.M.; Webster, T.C.; Sagili, R.R. Colony Level Prevalence and Intensity of Nosema ceranae in Honey Bees (Apis mellifera L.). PLoS ONE 2016, 11, e0163522. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Chen, Y.P.; diPrisco, G.; Pettis, J.; Williams, V. Bee cups: Single-use cages for honey bee experiments. J. Apic. Res. Bee World 2009, 48, 300–302. [Google Scholar] [CrossRef]
- Singaravelan, N.; Nee’man, G.; Inbar, M.; Izhaki, I. Feeding Responses of Free-flying Honeybees to Secondary Compounds Mimicking Floral Nectars. J. Chem. Ecol. 2005, 31, 2791–2804. [Google Scholar] [CrossRef] [PubMed]
- Mayack, C.; Naug, D. Energetic stress in the honeybee Apis mellifera from Nosema ceranae infection. J. Invertebr. Pathol. 2009, 100, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Martín-Hernández, R.; Meana, A.; Prieto, L.; Martínez Salvador, A.; Garrido-Bailón, E.; Higes, M. Outcome of colonization of Apis mellifera by Nosema ceranae. Appl. Environ. Microbiol. 2007, 73, 6331–6338. [Google Scholar] [CrossRef] [PubMed]
- Higes, M.; Garcia-Palencia, P.; Martin-Hernandez, R.; Meana, A. Experimental infection of Apis mellifera honeybees with Nosema ceranae (Microsporidia). J. Invertebr. Pathol. 2007, 94, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Meana, A.; Martín-Hernández, R.; Higes, M. The reliability of spore counts to diagnose Nosema ceranae infections in honey bees. J. Apic. Res. 2010, 49, 212–214. [Google Scholar] [CrossRef]
- Malone, L.A.; Gatehouse, H.S. Effects of Nosema apis infection on honey bee (Apis mellifera) digestive proteolytic enzyme activity. J. Invertebr. Pathol. 1998, 71, 169–174. [Google Scholar] [CrossRef]
- Fries, I.; Chauzat, M.-P.; Chen, Y.-P.; Doublet, V.; Genersch, E.; Gisder, S.; Higes, M.; McMahon, D.P.; Martín-Hernández, R.; Natsopoulou, M.; et al. Standard methods for Nosema research. J. Apic. Res. 2013, 52, 1–28. [Google Scholar] [CrossRef]
- Kessler, A.; Halitschke, R. Testing the potential for conflicting selection on floral chemical traits by pollinators and herbivores: Predictions and case study. Funct. Ecol. 2009, 23, 901–912. [Google Scholar] [CrossRef]
- Gegear, R.J.; Manson, J.S.; Thomson, J.D. Ecological context influences pollinator deterrence by alkaloids in floral nectar. Ecol. Lett. 2007, 10, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.M.; Mao, W.; Pollock, H.S.; Niu, G.; Schuler, M.A.; Berenbaum, M.R. Ecologically Appropriate Xenobiotics Induce Cytochrome P450s in Apis mellifera. PLoS ONE 2012, 7, e31051. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. A dietary phytochemical alters caste-associated gene expression in honey bees. Sci. Adv. 2015, 1, e1500795. [Google Scholar] [CrossRef] [PubMed]
- Hendriksma, H.P.; Oxman, K.L.; Shafir, S. Amino acid and carbohydrate tradeoffs by honey bee nectar foragers and their implications for plant–pollinator interactions. J. Insect Physiol. 2014, 69, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Biller, O.M.; Adler, L.S.; Irwin, R.E.; McAllister, C.; Palmer-Young, E.C. Possible Synergistic Effects of Thymol and Nicotine against Crithidia bombi Parasitism in Bumble Bees. PLoS ONE 2015, 10, e0144668. [Google Scholar] [CrossRef] [PubMed]
- Adler, L.S. The ecological significance of toxic nectar. Oikos 2000, 91, 409–420. [Google Scholar] [CrossRef]
- Simone-Finstrom, M.; Evans, J.D.; Spivak, M. Resin collection and social immunity in honey bees. Evolution 2009, 63, 3016–3022. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Schmitt, A.J.; Thomas, J.B.; Carter, C.J. Review: Nectar biology: From molecules to ecosystems. Plant Sci. 2017, 262, 148–164. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.R.V.; Roth, C.L.; Carlson, B.L.; Smart, M.D. Land-use change reduces habitat suitability for supporting managed honey bee colonies in the Northern Great Plains. Proc. Natl. Acad. Sci. USA 2016, 113, 10430–10435. [Google Scholar] [CrossRef] [PubMed]
| Phytochemicals | Classification | Chemical Structure |
|---|---|---|
| Caffeine | Alkaloid | 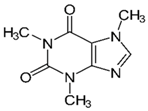 |
| Gallic acid | Flavonol | 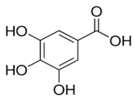 |
| Kaempferol | Phenolic acid |  |
| p-coumaric acid | Phenolic acid | 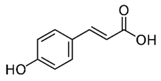 |
| Source of Variation | df | MSS | F | p |
|---|---|---|---|---|
| Direct effects | ||||
| Model | 1 | 3586.66 | 166,225.64 | <0.0001 |
| Phytochemical | 3 | 1.53 | 24.61 | <0.0001 |
| Concentrations | 2 | 1.32 | 14.01 | 0.002 |
| Colony | 4 | 0.02 | 0.23 | 0.9 |
| Interaction effects | 6 | |||
| Phytochemical × Concentration | 6 | 1.15 | 16.92 | <0.0001 |
| Phytochemical × Colony | 12 | 0.06 | 0.91 | 0.5 |
| Concentration × Colony | 8 | 0.09 | 1.39 | 0.3 |
| Phytochemical × Concentration × Colony | 24 | 0.07 | 1.35 | 0.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernklau, E.; Bjostad, L.; Hogeboom, A.; Carlisle, A.; H. S., A. Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance. Insects 2019, 10, 14. https://doi.org/10.3390/insects10010014
Bernklau E, Bjostad L, Hogeboom A, Carlisle A, H. S. A. Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance. Insects. 2019; 10(1):14. https://doi.org/10.3390/insects10010014
Chicago/Turabian StyleBernklau, Elisa, Louis Bjostad, Alison Hogeboom, Ashley Carlisle, and Arathi H. S. 2019. "Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance" Insects 10, no. 1: 14. https://doi.org/10.3390/insects10010014
APA StyleBernklau, E., Bjostad, L., Hogeboom, A., Carlisle, A., & H. S., A. (2019). Dietary Phytochemicals, Honey Bee Longevity and Pathogen Tolerance. Insects, 10(1), 14. https://doi.org/10.3390/insects10010014





