In-Situ Synthesis and Characterization of Biodegradable Estolides via Epoxidation from Canola Biodiesel
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Analytical Techniques and Methods
- η = effective composition to biodegradation coefficient (1 for natural esters and 0.8 for renewable based diester and polyol ester)
- Ca = Fraction of non-aromatics (n-pentene)
- Cc = Fraction of ester (diethyl ether)
- t = time (i.e., 28 days)
- B(t) = Biodegradability of estolides in 28 days
- B(1) = 0.01
2.3. Catalyst Preparation
2.4. Catalyst Characterization Methods
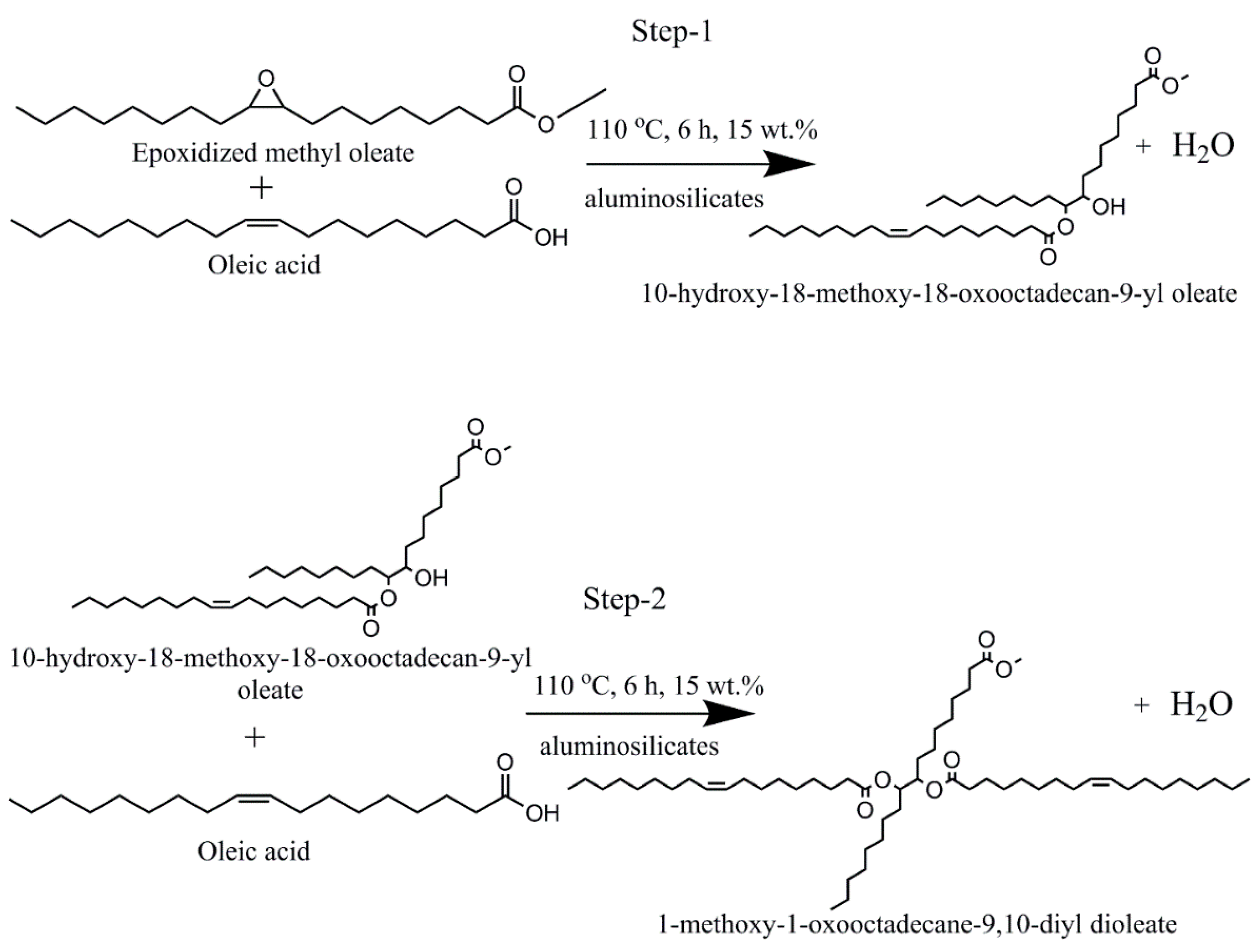
2.5. Estolides Synthesis Procedure
3. Results and Discussion
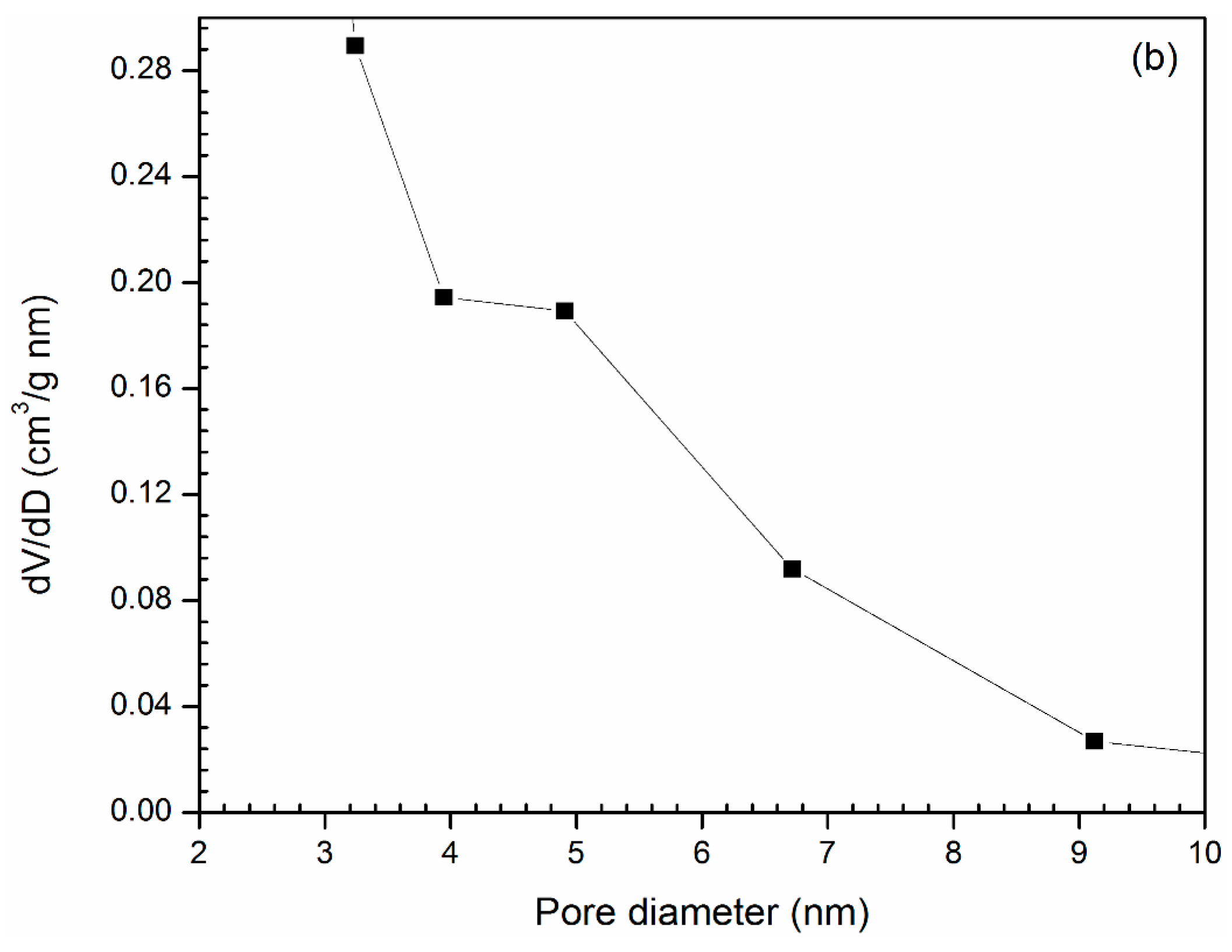
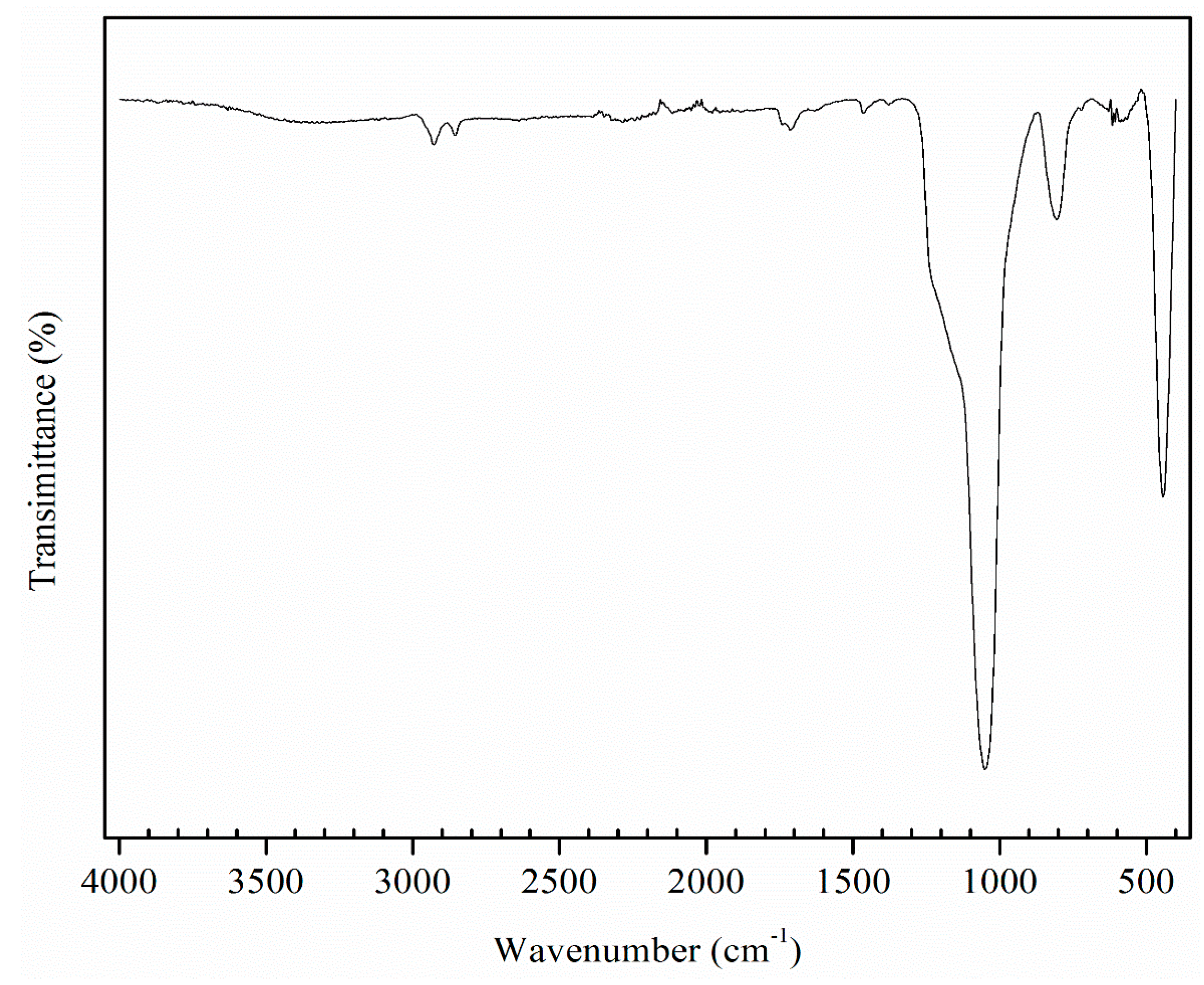
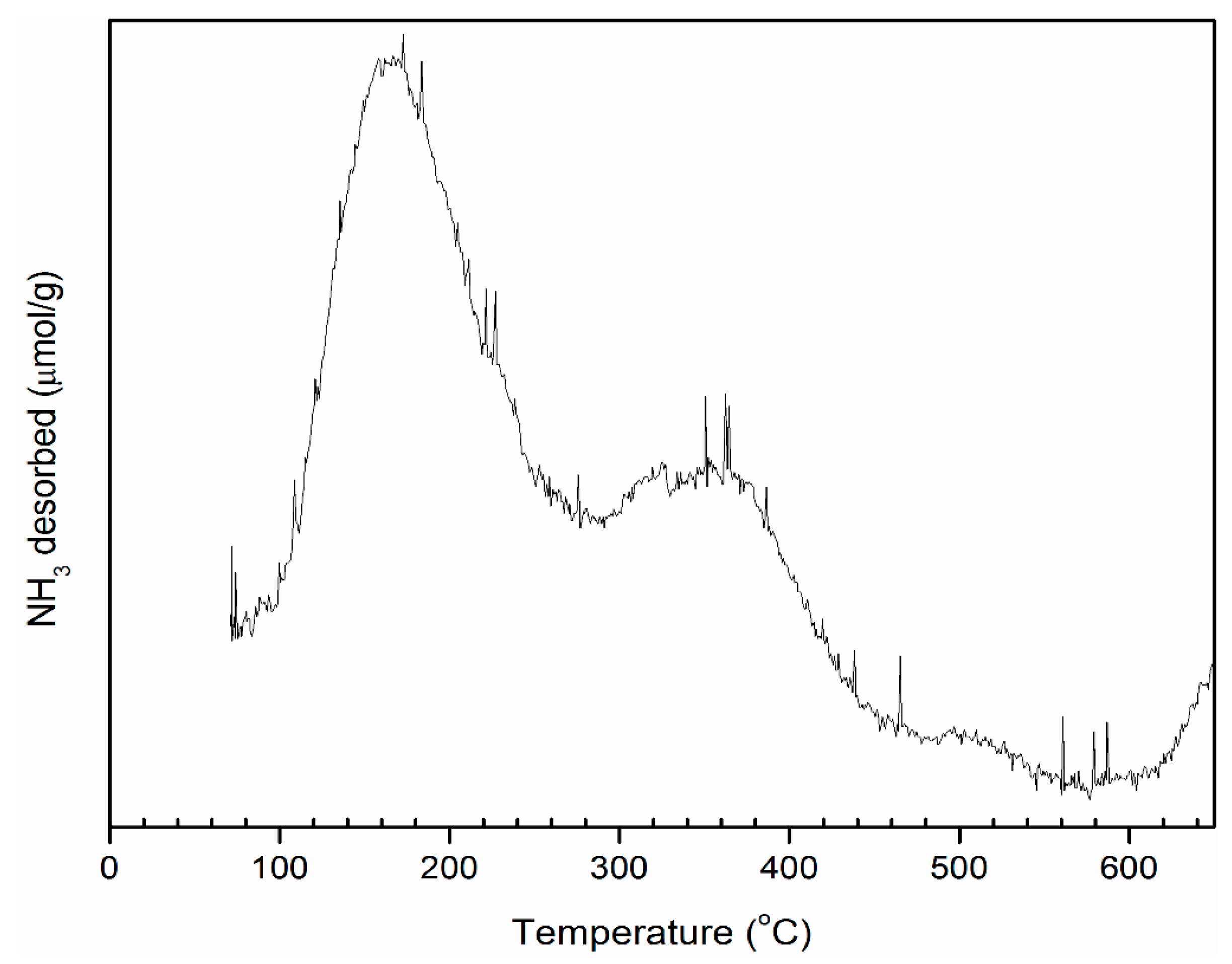
3.1. Catalyst Characterization
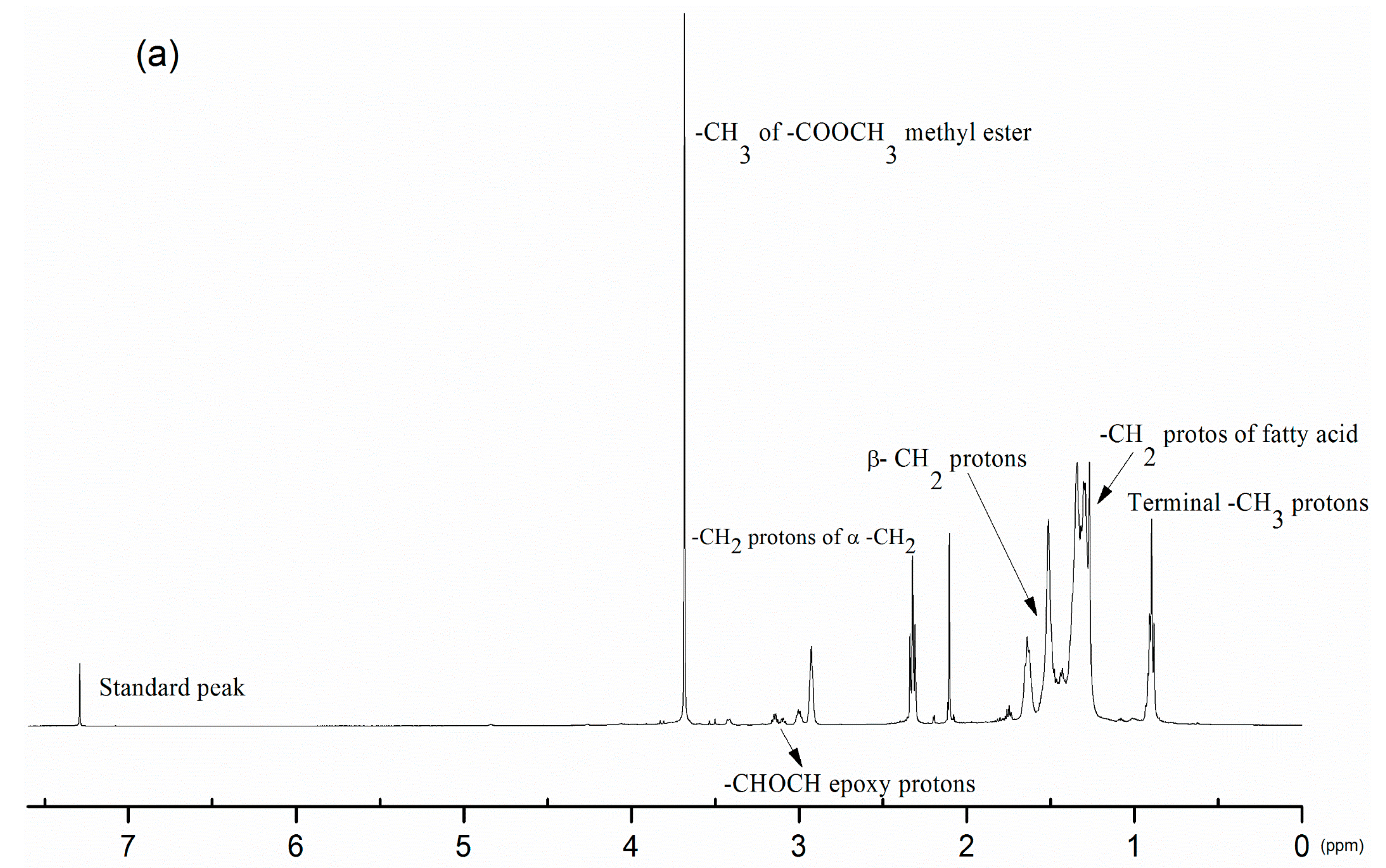
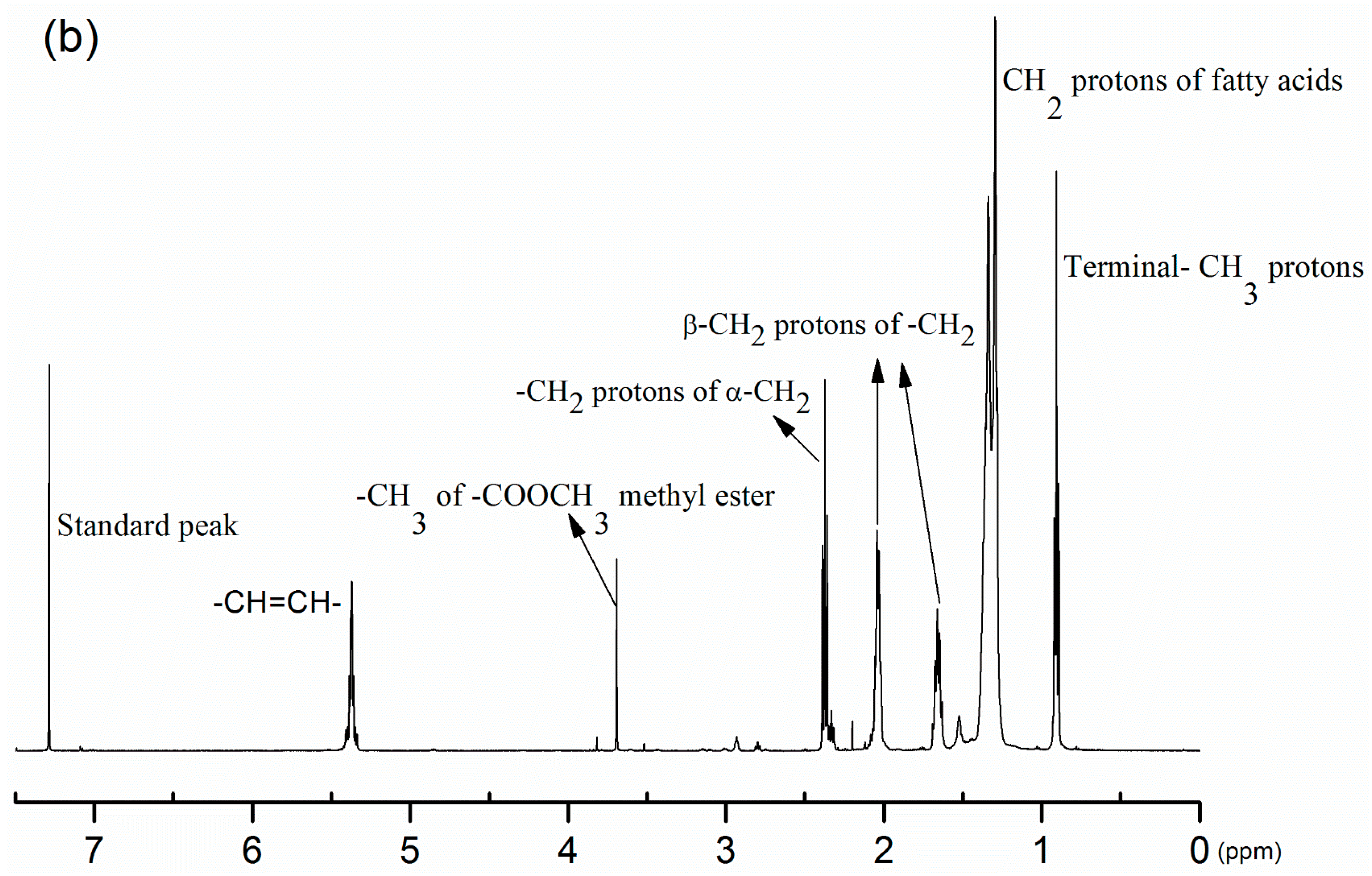
3.2. Product Confirmation and Characterization of Estolides by 1H-NMR
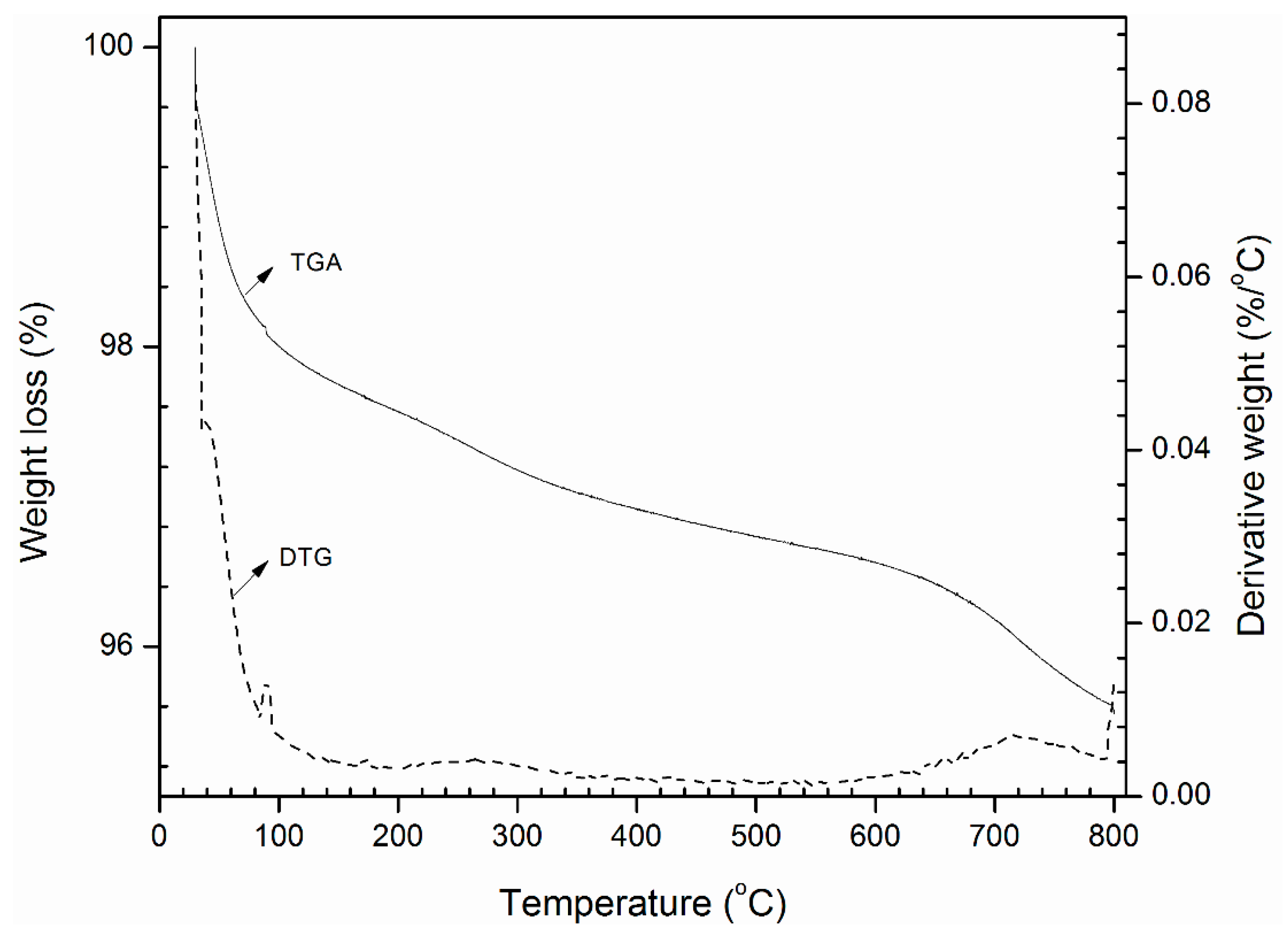
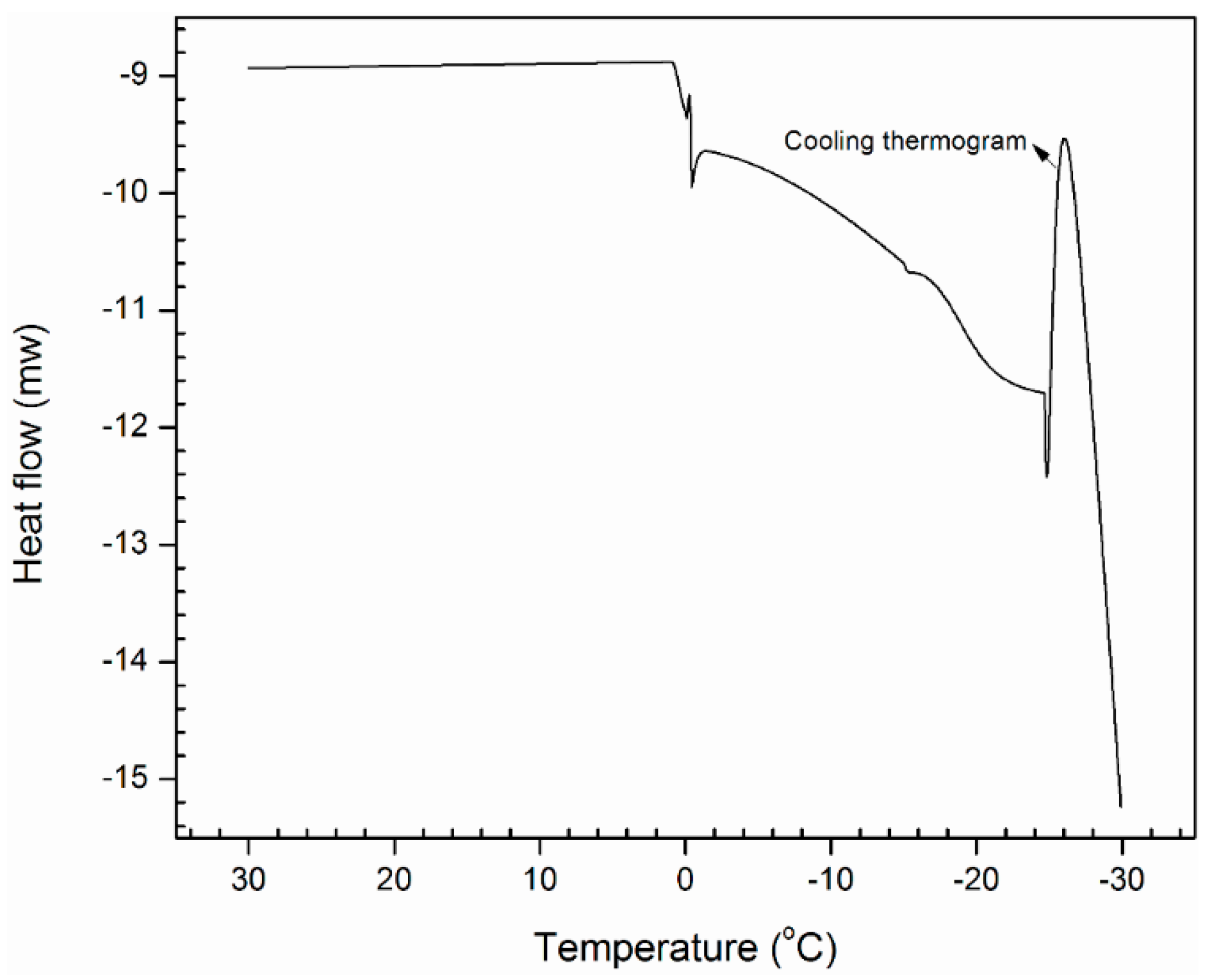
3.3. Detailed Physicochemical Characterization of Formulated Estolides
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yunus, R.; Luo, X. Thermochemical conversion of plant oils and derivatives to lubricants. Adv. Bioenergy 2017, 2, 183–231. [Google Scholar]
- Borugadda, V.B.; Goud, V.V. Improved low-temperature properties of chemically modified high free fatty acid castor oil–methyl esters: Blending and optimization study. J. Energy Eng. 2015. [Google Scholar] [CrossRef]
- Borugadda, V.B.; Goud, V.V. Comparative studies of thermal, oxidative and low-temperature properties of waste cooking oil and castor oil. J. Renew. Sustain. Energy 2013, 5. [Google Scholar] [CrossRef]
- Cermak, S.C.; Isbell, T.A. Improved oxidative stability of estolide esters. Ind Crops Prod. 2003, 18, 223–230. [Google Scholar] [CrossRef]
- Cermak, S.C.; Isbell, T.A. Physical properties of saturated estolides and their 2-Ethylhexyl esters. Ind. Crops Prod. 2002, 16, 119–127. [Google Scholar] [CrossRef]
- Kiatsimkul, P.P.; Suppes, G.J.; Hsieh, F.H.; Lozada, Z.; Tu, Y.C. Preparation of high hydroxyl equivalent weight polyols from vegetable oils. Ind. Crops Prod. 2008, 27, 257–264. [Google Scholar] [CrossRef]
- Ashby, R.D.; Solaiman, D.K.Y.; Liu, C.K.; Strahan, G.; Latona, N. Sophorolipid-derived unsaturated and epoxy fatty acid estolides as plasticizers for poly (3-Hydroxybutyrate). J. Am. Oil Chem. Soc. 2016, 93, 347–358. [Google Scholar] [CrossRef]
- Doll, K.M.; Cermak, S.C.; Kenar, J.A.; Isbell, T.A. Synthesis and characterization of estolide esters containing epoxy and cyclic carbonate groups. J. Am. Oil Chem. Soc. 2016, 93, 1149–1155. [Google Scholar] [CrossRef]
- Doll, K.M.; Cermak, S.C.; Kenar, J.A.; Walter, E.L.; Isbell, T.A. Derivatization of castor oil based estolide esters: Preparation of epoxides and cyclic carbonates. Ind. Crops Prod. 2017, 104, 269–277. [Google Scholar] [CrossRef]
- Borugadda, V.B.; Somidi, A.K.R.; Dalai, A.K. Chemical/structural modification of canola oil and canola biodiesel: Kinetic studies and biodegradability of the alkoxides. Lubricants 2017, 5, 1–14. [Google Scholar] [CrossRef]
- Bai, P.; Wu, P.; Xing, W.; Liu, D.; Zhao, L.; Wang, Y.; Xu, B.; Yan, Z.; Zhao, X.S. Synthesis and catalytic properties of ZSM-5 zeolite with hierarchical pores prepared in the presence of n-hexyltrimethylammonium bromide. J. Mater. Chem. 2015, 3, 18586–18597. [Google Scholar] [CrossRef]
- Feng, H.; Li, C.; Shan, H. In-situ synthesis and catalytic activity of ZSM-5 zeolite. App. Clay Sci. 2009, 42, 439–445. [Google Scholar] [CrossRef]
- Barot, S.; Bandyopadhyay, R.; Joshi, S.S. Catalytic conversion of jatropha oil to biofuel over titania, zirconia and ceria loaded amorphous alumino-silicate catalysts. Environ. Progres. Sustain. Energy 2017, 36, 749–757. [Google Scholar] [CrossRef]
- Kirschhock, C.E.A.; Ravishankar, R.; Verspeurt, F.; Grobet, P.J.; Jacobs, P.A.; Martens, J.A. Identification of precursor spices in the formation of MFI zeolite in the TPAOH-TEOS-H2O system. J. Phys. Chem. B 1999, 111, 4965–4971. [Google Scholar] [CrossRef]
- Choi, M.; Cho, H.S.; Srivastava, R.; Venkatesan, C.; Choi, D.H.; Ryoo, R. Amphiphilic organosilane-directed synthesis of crystalline zeolite with tunable mesoporosity. Nat. Mater. 2006, 5, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.; Na, K.; Kim, J.; Sakamoto, Y.; Terasaki, O.; Ryoo, R. Stable single-unit-cell nanosheets of zeolite MFI as active and long-lived catalysts. Nature 2009, 461, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Hassanpour, S.; Taghizadeh, M. Preparation, characterization, and activity evaluation of H-ZSM-5 catalysts in vapor-phase methanol dehydration to dimethyl ether. Ind. Eng. Chem. Res. 2010, 49, 4063–4069. [Google Scholar] [CrossRef]
- Barr, T.L.; Lishka, M.A. ESCA studies of the surface chemistry of zeolites. J. Am. Chem. Soc. 1986, 108, 3178. [Google Scholar] [CrossRef]
- Auroux, A.; Bolis, V.; Wierzchowski, P.; Gravelle, P.C.; Vedrine, J.C. Study of the acidity of ZSM-5 zeolite by microcalorimetry and infrared spectroscopy. J. Chem. Soc. 1979, 59, 2544–2555. [Google Scholar] [CrossRef]
- Badrinarayana, S.; Hegde, R.I.; Balakrishnan, L.; Kulkarni, S.B.; Ratnasamy, P. XPS study of nickel in NiHZSM5 catalysts. J. Catal. 1981, 71, 439–442. [Google Scholar] [CrossRef]
- Cermak, S.C.; Isbell, T.A. Synthesis of estolides from oleic and saturated fatty acids. J. Am. Chem. Soc. 2001, 78, 557–565. [Google Scholar] [CrossRef]
- Sharma, R.V.; Somidi, A.K.R.; Dalai, A.K. Preparation and property evaluation of biolubricants derived from canola oil and canola biodiesel. J. Agric. Food Chem. 2015, 63, 3235–3242. [Google Scholar] [CrossRef] [PubMed]
- Borugadda, V.B.; Goud, V.V. Synthesis of waste cooking oil epoxide as a bio-lubricant base stock: characterization and optimization study. J. Bioprocess Eng. Biorefin. 2014, 3, 57–72. [Google Scholar] [CrossRef]
- McNutt, J.; He, Q.S. Development of biolubricants from vegetable oils via chemical modification. J. Ind. Eng. Chem. 2016, 36, 1–12. [Google Scholar] [CrossRef]
- Adhvaryu, A.; Erhan, S.Z.; Perez, J.M. Tribological studies of thermally and chemically modified vegetable oils for use as environmentally friendly lubricants. Wear 2004, 257, 359–367. [Google Scholar] [CrossRef]
- Strotman, U.; Reuschenbach, P.; Schwarz, H.; Pagga, U. Development and evaluation of an online CO2 evolution test and multicomponent biodegradation test system. Appl. Environ. Microbiol. 2004, 70, 4621–4628. [Google Scholar] [CrossRef] [PubMed]
- Pakou, C.; Stamatelatou, K.; Kornaros, M.; Lyberatos, G. On the complete aerobic microbial mineralization of linear alkylbenzene sulfonate. Desalination 2007, 215, 198–208. [Google Scholar] [CrossRef]


| Process Parameter | Range |
|---|---|
| Oleic acid (mol) | 6–12 |
| Reaction time (h) | 0–9 |
| Reaction temperature (°C) | 30–120 |
| Catalyst loading (wt %) | 5–20 |
| Name of the Catalyst | Maximum Desorption Temperature (°C) | Amount of Ammonia (µL/g cat) | Acid Strength (mmol/g cat) |
|---|---|---|---|
| Mesoporous aluminosilicates | 167 | 49.91 | 0.484 |
| 401 | 53.67 | 0.520 | |
| 548 | 2.25 | 0.021 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borugadda, V.B.; Dalai, A.K. In-Situ Synthesis and Characterization of Biodegradable Estolides via Epoxidation from Canola Biodiesel. Lubricants 2018, 6, 94. https://doi.org/10.3390/lubricants6040094
Borugadda VB, Dalai AK. In-Situ Synthesis and Characterization of Biodegradable Estolides via Epoxidation from Canola Biodiesel. Lubricants. 2018; 6(4):94. https://doi.org/10.3390/lubricants6040094
Chicago/Turabian StyleBorugadda, Venu Babu, and Ajay K. Dalai. 2018. "In-Situ Synthesis and Characterization of Biodegradable Estolides via Epoxidation from Canola Biodiesel" Lubricants 6, no. 4: 94. https://doi.org/10.3390/lubricants6040094
APA StyleBorugadda, V. B., & Dalai, A. K. (2018). In-Situ Synthesis and Characterization of Biodegradable Estolides via Epoxidation from Canola Biodiesel. Lubricants, 6(4), 94. https://doi.org/10.3390/lubricants6040094







