Barriers and Facilitators to Genetic Testing for Familial Hypercholesterolemia in the United States: A Review
Abstract
1. Introduction
2. Methods
2.1. Search Strategy, Literature Selection, and Inclusion Criteria
2.2. Choice of Implementation Framework
2.3. Information Extraction
2.4. Analysis of Barriers and Facilitators
3. Results
3.1. CFIR Domain #1: Characteristics of Intervention
3.2. CFIR Domain #2: Outer Setting
3.3. CFIR Domain #3: Inner Setting
3.4. CFIR Domain #4: Characteristics of Individuals
3.5. CFIR Domain #5: Process
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Knowles, J.W.; Rader, D.J.; Khoury, M.J. Cascade Screening for Familial Hypercholesterolemia and the Use of Genetic Testing. JAMA 2017, 318, 381–382. [Google Scholar] [CrossRef] [PubMed]
- Knowles, J.W.; Stone, N.J.; Ballantyne, C.M. Familial Hypercholesterolemia and the 2013 American College of Cardiology/American Heart Association Guidelines: Myths, Oversimplification, and Misinterpretation Versus Facts. Am. J. Cardiol. 2015, 116, 481–484. [Google Scholar] [CrossRef] [PubMed]
- De Ferranti, S.D.; Rodday, A.M.; Mendelson, M.M.; Wong, J.B.; Leslie, L.K.; Sheldrick, R.C. Prevalence of Familial Hypercholesterolemia in the 1999 to 2012 United States National Health and Nutrition Examination Surveys (NHANES)CLINICAL PERSPECTIVE. Circulation 2016, 133, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, A.C.; Hopkins, P.N.; Toth, P.P.; Ballantyne, C.M.; Rader, D.J.; Robinson, J.G.; Daniels, S.R.; Gidding, S.S.; de Ferranti, S.D.; Ito, M.K.; et al. Familial Hypercholesterolemia: Screening, diagnosis and management of pediatric and adult patients: Clinical guidance from the National Lipid Association Expert Panel on Familial Hypercholesterolemia. J. Clin. Lipidol. 2011, 5, S1–S8. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.; Wang, J.; Hegele, R.A. Heterozygous familial hypercholesterolemia: An underrecognized cause of early cardiovascular disease. CMAJ 2006, 174, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Heart Disease Facts & Statistics. cdc.gov. Available online: https://www.cdc.gov/heartdisease/facts.htm (accessed on 31 May 2019).
- Akioyamen, L.E.; Genest, J.; Shan, S.D.; Reel, R.L.; Albaum, J.M.; Chu, A.; Tu, J.V. Estimating the prevalence of heterozygous familial hypercholesterolaemia: A systematic review and meta-analysis. BMJ Open 2017, 7, e016461. [Google Scholar] [CrossRef] [PubMed]
- Abul-Husn, N.S.; Manickam, K.; Jones, L.K.; Wright, E.A.; Hartzel, D.N.; Gonzaga-Jauregui, C.; O’Dushlaine, C.; Leader, J.B.; Kirchner, H.L.; Lindbuchler, D.M.; et al. Genetic identification of familial hypercholesterolemia within a single U.S. health care system. Science 2016, 354, aaf7000. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A.C.; Knowles, J.W.; Gidding, S.S.; Ahmad, Z.S.; Ahmed, C.D.; Ballantyne, C.M.; Baum, S.J.; Bourbon, M.; Carrié, A.; Cuchel, M.; et al. Clinical Genetic Testing for Familial Hypercholesterolemia: JACC Scientific Expert Panel. J. Am. Coll. Cardiol. 2018, 72, 662–680. [Google Scholar] [CrossRef]
- Cirino, A.L.; Harris, S.; Lakdawala, N.K.; Michels, M.; Olivotto, I.; Day, S.M.; Abrams, D.J.; Charron, P.; Caleshu, C.; Semsarian, C.; et al. Role of Genetic Testing in Inherited Cardiovascular Disease: A Review. JAMA Cardiol. 2017, 2, 1153–1160. [Google Scholar] [CrossRef]
- Minicocci, I.; Pozzessere, S.; Prisco, C.; Montali, A.; di Costanzo, A.; Martino, E.; Martino, F.; Arca, M. Analysis of Children and Adolescents with Familial Hypercholesterolemia. J. Pediatrics 2017, 183, 100–107.e3. [Google Scholar] [CrossRef]
- Setia, N.; Saxena, R.; Sawhney, J.P.S.; Verma, I.C. Familial Hypercholesterolemia: Cascade Screening in Children and Relatives of the Affected. Indian J. Pediatr. 2018, 85, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Séguro, F.; Rabès, J.-P.; Taraszkiewicz, D.; Ruidavets, J.-B.; Bongard, V.; Ferrières, J. Genetic diagnosis of familial hypercholesterolemia is associated with a premature and high coronary heart disease risk. Clin. Cardiol. 2018, 41, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Vohnout, B.; Gabcova, D.; Huckova, M.; Klimes, I.; Gasperikova, D.; Raslova, K. Genetic testing of familial hypercholesterolemia in a real clinical setting. Wien Klin. Wochenschr. 2016, 128, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Amor-Salamanca, A.; Castillo, S.; Gonzalez-Vioque, E.; Dominguez, F.; Quintana, L.; Lluís-Ganella, C.; Escudier, J.M.; Ortega, J.; Lara-Pezzi, E.; Alonso-Pulpon, L.; et al. Genetically Confirmed Familial Hypercholesterolemia in Patients with Acute Coronary Syndrome. J. Am. Coll. Cardiol. 2017, 70, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Sperlongano, S.; Gragnano, F.; Natale, F.; D’Erasmo, L.; Concilio, C.; Cesaro, A.; Golia, E.; Crisci, M.; Sperlongano, R.; Fimiani, F.; et al. Lomitapide in homozygous familial hypercholesterolemia: Cardiology perspective from a single-center experience. J. Cardiovasc. Med. 2018, 19, 83. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Martin, A.C.; Bates, T.R.; Hooper, A.J.; Bell, D.A.; Burnett, J.R.; Norman, R.; Watts, G.F. Parent–child genetic testing for familial hypercholesterolaemia in an Australian context. J. Paediatr. Child Health 2018, 54, 741–747. [Google Scholar] [CrossRef]
- Wu, X.; Pang, J.; Wang, X.; Peng, J.; Chen, Y.; Wang, S.; Watts, G.F.; Lin, J. Reverse cascade screening for familial hypercholesterolemia in high-risk Chinese families. Clin. Cardiol. 2017, 40, 1169–1173. [Google Scholar] [CrossRef]
- Averna, M.; Cefalù, A.B.; Casula, M.; Noto, D.; Arca, M.; Bertolini, S.; Calandra, S.; Catapano, A.L.; Tarugi, P. Familial hypercholesterolemia: The Italian Atherosclerosis Society Network (LIPIGEN). Atheroscler. Suppl. 2017, 29, 11–16. [Google Scholar] [CrossRef]
- Jones, L.K.; Rahm, A.K.; Manickam, K.; Butry, L.; Lazzeri, A.; Corcoran, T.; Komar, D.; Josyula, N.S.; Pendergrass, S.A.; Sturm, A.C.; et al. Healthcare Utilization and Patients’ Perspectives After Receiving a Positive Genetic Test for Familial Hypercholesterolemia. Circ. Genom. Precis. Med. 2018, 11, e002146. [Google Scholar] [CrossRef]
- Stimulating Implementation Science in Genomics and Precision Medicine for Heart, Lung, Blood and Sleep Diseases: The Case of Familial Hypercholesterolemia. Blogs|CDC. Available online: https://blogs.cdc.gov/genomics/2018/06/05/stimulating-implementation/ (accessed on 28 September 2018).
- Roberts, M.C.; Mensah, G.A.; Khoury, M.J. Leveraging Implementation Science to Address Health Disparities in Genomic Medicine: Examples from the Field. Ethn. Dis. 2019, 29, 187–192. [Google Scholar] [CrossRef]
- FH 1|Tier 1 Genomic Applications Toolkit for Public Health Departments. Genomics|CDC. Available online: https://www.cdc.gov/genomics/implementation/toolkit/fh_1.htm (accessed on 21 October 2018).
- Tier1|Tier 1 Genomic Applications Toolkit for Public Health Departments. Genomics|CDC. Available online: https://www.cdc.gov/genomics/implementation/toolkit/tier1.htm (accessed on 21 October 2018).
- Hendricks-Sturrup, R.M.; Lu, C.Y. Understanding Implementation Challenges to Genetic Testing for Familial Hypercholesterolemia in the United States. J. Pers. Med. 2019, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Feature|Familial Hypercholesterolemia: Clinician and Patient Insights. Available online: https://www.acc.org/latest-in-cardiology/articles/2018/10/14/12/42/feature-familial-hypercholesterolemia-clinician-and-patient-insights (accessed on 30 November 2018).
- Delikurt, T.; Williamson, G.R.; Anastasiadou, V.; Skirton, H. A systematic review of factors that act as barriers to patient referral to genetic services. Eur. J. Hum. Genet. 2015, 23, 739–745. [Google Scholar] [CrossRef] [PubMed]
- George, R.; Kovak, K.; Cox, S.L. Aligning policy to promote cascade genetic screening for prevention and early diagnosis of heritable diseases. J. Genet. Couns. 2015, 24, 388–399. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A.C. Cardiovascular Cascade Genetic Testing: Exploring the Role of Direct Contact and Technology. Front. Cardiovasc. Med. 2016, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.Y.; Loomer, S.; Ceccarelli, R.; Mazor, K.M.; Sabin, J.; Clayton, E.W.; Ginsburg, G.S.; Wu, A.C. Insurance Coverage Policies for Pharmacogenomic and Multi-Gene Testing for Cancer. J. Pers. Med. 2018, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Prince, A.E.R. Prevention for those who can pay: Insurance reimbursement of genetic-based preventive interventions in the liminal state between health and disease. J. Law Biosci. 2015, 2, 365–395. [Google Scholar] [CrossRef] [PubMed]
- Literature Search: Databases and Gray Literature. NIH Library. Available online: https://www.nihlibrary.nih.gov/services/systematic-review-service/literature-search-databases-and-gray-literature (accessed on 31 May 2019).
- Rahm, A.K.; Cragun, D.; Hunter, J.E.; Epstein, M.M.; Lowery, J.; Lu, C.Y.; Pawloski, P.A.; Sharaf, R.N.; Liang, S.-Y.; Burnett-Hartman, A.N.; et al. Implementing universal Lynch syndrome screening (IMPULSS): Protocol for a multi-site study to identify strategies to implement, adapt, and sustain genomic medicine programs in different organizational contexts. BMC Health Serv. Res. 2018, 18, 824. [Google Scholar] [CrossRef] [PubMed]
- Orlando, L.A.; Sperber, N.R.; Voils, C.; Nichols, M.; Myers, R.A.; Wu, R.R.; Rakhra-Burris, T.; Levy, K.D.; Levy, M.; Pollin, T.I.; et al. Developing a Common Framework for Evaluating the Implementation of Genomic Medicine Interventions in Clinical Care: The IGNITE Network’s Common Measures Working Group. Genet. Med. 2018, 20, 655–663. [Google Scholar] [CrossRef]
- Sperber, N.R.; Carpenter, J.S.; Cavallari, L.H.; Damschroder, L.J.; Cooper-DeHoff, R.M.; Denny, J.C.; Ginsburg, G.S.; Guan, Y.; Horowitz, C.R.; Levy, K.D.; et al. Challenges and strategies for implementing genomic services in diverse settings: Experiences from the Implementing GeNomics In pracTicE (IGNITE) network. BMC Med. Genom. 2017, 10, 35. [Google Scholar] [CrossRef]
- Levy, K.D.; Blake, K.; Fletcher-Hoppe, C.; Franciosi, J.; Goto, D.; Hicks, J.K.; Holmes, A.M.; Kanuri, S.H.; Madden, E.B.; Musty, M.D.; et al. Opportunities to implement a sustainable genomic medicine program: Lessons learned from the IGNITE Network. Genet. Med. 2019, 21, 743. [Google Scholar] [CrossRef]
- Birken, S.A.; Powell, B.J.; Shea, C.M.; Haines, E.R.; Alexis Kirk, M.; Leeman, J.; Rohweder, C.; Damschroder, L.; Presseau, J. Criteria for selecting implementation science theories and frameworks: Results from an international survey. Implement. Sci. 2017, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Kirk, M.A.; Kelley, C.; Yankey, N.; Birken, S.A.; Abadie, B.; Damschroder, L. A systematic review of the use of the Consolidated Framework for Implementation Research. Implement. Sci. 2016, 11, 72. [Google Scholar] [CrossRef] [PubMed]
- Giudicessi, J.R.; Kullo, I.J.; Ackerman, M.J. Precision Cardiovascular Medicine: State of Genetic Testing. Mayo Clin. Proc. 2017, 92, 642–662. [Google Scholar] [CrossRef] [PubMed]
- Sarraju, A.; Knowles, J.W. Genetic Testing and Risk Scores: Impact on Familial Hypercholesterolemia. Front. Cardiovasc. Med. 2019, 6. [Google Scholar] [CrossRef] [PubMed]
- Safarova, M.S.; Kullo, I.J. Lessening the Burden of Familial Hypercholesterolemia Using Health Information Technology. Circ. Res. 2018, 122, 26–27. [Google Scholar] [CrossRef] [PubMed]
- Migliara, G.; Baccolini, V.; Rosso, A.; D’Andrea, E.; Massimi, A.; Villari, P.; De Vito, C. Familial Hypercholesterolemia: A Systematic Review of Guidelines on Genetic Testing and Patient Management. Front. Public Health 2017, 5, 252. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.; Humanki, J.; Zierhut, H. A novel approach to screening for familial hypercholesterolemia in a large public venue. J. Community Genet. 2017, 8, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Khoury, M.J.; Bowen, M.S.; Clyne, M.; Dotson, W.D.; Gwinn, M.L.; Green, R.F.; Kolor, K.; Rodriguez, J.L.; Wulf, A.; Yu, W. From public health genomics to precision public health: A 20-year journey. Genet. Med. 2018, 20, 574–582. [Google Scholar] [CrossRef]
- Mues, K.E.; Bogdanov, A.N.; Monda, K.L.; Yedigarova, L.; Liede, A.; Kallenbach, L. How well can familial hypercholesterolemia be identified in an electronic health record database? Clin. Epidemiol. 2018, 10, 1667–1677. [Google Scholar] [CrossRef]
- Hasnie, A.A.; Kumbamu, A.; Safarova, M.S.; Caraballo, P.J.; Kullo, I.J. A Clinical Decision Support Tool for Familial Hypercholesterolemia Based on Physician Input. Mayo Clin. Proc. Innov. Qual. Outcomes 2018, 2, 103–112. [Google Scholar] [CrossRef]
- Knowles, J.W.; O’Brien, E.C.; Greendale, K.; Wilemon, K.; Genest, J.; Sperling, L.S.; Neal, W.A.; Rader, D.J.; Khoury, M.J. Reducing the burden of disease and death from familial hypercholesterolemia: A call to action. Am. Heart J. 2014, 168, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Welch, B.M.; Dere, W.; Schiffman, J.D. Family Health History: The Case for Better Tools. JAMA 2015, 313, 1711–1712. [Google Scholar] [CrossRef] [PubMed]
- Farwati, M.; Kumbamu, A.; Kochan, D.C.; Kullo, I.J. Patient and Provider Perspectives on a Decision Aid for Familial Hypercholesterolemia. J. Pers. Med. 2018, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, J.; Duprez, D.; Veach, P.M.; Zierhut, H.A. Barriers to the identification of familial hypercholesterolemia among primary care providers. J. Community Genet. 2018, 10, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Gleacher, A.A.; Olin, S.S.; Nadeem, E.; Pollock, M.; Ringle, V.; Bickman, L.; Douglas, S.; Hoagwood, K. Implementing a Measurement Feedback System in Community Mental Health Clinics: A Case Study of Multilevel Barriers and Facilitators. Adm. Policy Ment. Health 2016, 43, 426–440. [Google Scholar] [CrossRef]
- Lyon, A.R.; Lewis, C.C. Designing Health Information Technologies for Uptake: Development and Implementation of Measurement Feedback Systems in Mental Health Service Delivery. Adm. Policy Ment. Health 2016, 43, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Keith, R.E.; Crosson, J.C.; O’Malley, A.S.; Cromp, D.; Taylor, E.F. Using the Consolidated Framework for Implementation Research (CFIR) to produce actionable findings: A rapid-cycle evaluation approach to improving implementation. Implement. Sci. 2017, 12, 15. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, L.; Sun, L.-Y.; Wu, Y.; Wen, W.-H.; Wang, X.-F.; Liu, W.; Zhou, Y.-J.; Wang, L.-Y. Genetically confirmed familial hypercholesterolemia in outpatients with hypercholesterolemia. J. Geriatr. Cardiol. 2018, 15, 434–440. [Google Scholar]
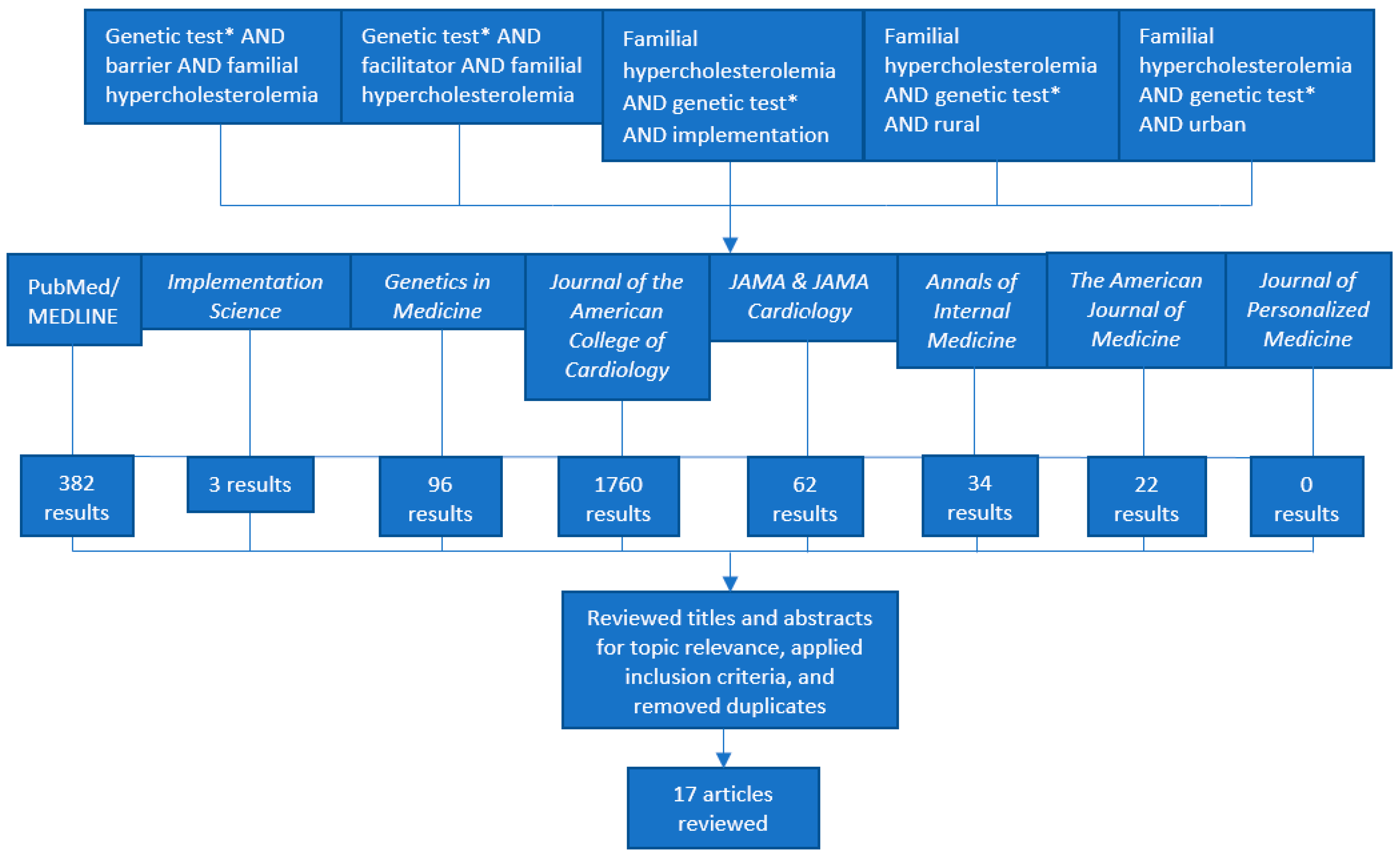
| Keywords/Strings Used for Literature Search | Databases and Journals | Inclusion Criteria |
|---|---|---|
|
|
|
| CFIR Domain | |
|---|---|
| 1. Characteristics of Intervention | Key attributes of interventions influence the success of implementation. Includes adaptability, complexity, cost, design quality and packaging, evidence strength and quality, intervention source, relative advantage, and trialability. |
| 2. Outer Setting | External influences including cosmopolitanism, external policies and incentives, patient needs and resources, and peer pressure. |
| 3. Inner Setting | Active interacting facets within a setting that include structural characteristics, networks and communications, culture, implementation climate, and readiness for implementation. |
| 4. Characteristics of Individuals | The actions and behaviors of individuals. Includes individual identification with an organization, individual stage of change, knowledge and beliefs about the intervention, other personal attributes, and self-efficacy. |
| 5. Process | The process of implementing an intervention. Includes or involves behaviors of engagement, execution, planning, reflecting, and evaluation. |
| Consolidated Framework for Implementation Research (CFIR) Domain #1, Characteristics of Intervention | |
| Facilitators | Barriers |
|
|
| CFIR Domain #2, Outer Setting | |
| Facilitators | Barriers |
| |
| CFIR Domain #3, Inner Setting | |
| Facilitators | Barriers |
|
|
| CFIR Domain #4, Characteristics of Individuals | |
| Facilitators | Barriers |
|
|
| CFIR Domain #5, Process | |
| Facilitators | Barriers |
|
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hendricks-Sturrup, R.M.; Mazor, K.M.; Sturm, A.C.; Lu, C.Y. Barriers and Facilitators to Genetic Testing for Familial Hypercholesterolemia in the United States: A Review. J. Pers. Med. 2019, 9, 32. https://doi.org/10.3390/jpm9030032
Hendricks-Sturrup RM, Mazor KM, Sturm AC, Lu CY. Barriers and Facilitators to Genetic Testing for Familial Hypercholesterolemia in the United States: A Review. Journal of Personalized Medicine. 2019; 9(3):32. https://doi.org/10.3390/jpm9030032
Chicago/Turabian StyleHendricks-Sturrup, Rachele M., Kathleen M. Mazor, Amy C. Sturm, and Christine Y. Lu. 2019. "Barriers and Facilitators to Genetic Testing for Familial Hypercholesterolemia in the United States: A Review" Journal of Personalized Medicine 9, no. 3: 32. https://doi.org/10.3390/jpm9030032
APA StyleHendricks-Sturrup, R. M., Mazor, K. M., Sturm, A. C., & Lu, C. Y. (2019). Barriers and Facilitators to Genetic Testing for Familial Hypercholesterolemia in the United States: A Review. Journal of Personalized Medicine, 9(3), 32. https://doi.org/10.3390/jpm9030032





