The Effect of a Personalized Approach to Patient Education on Heart Failure Self-Management
Abstract
1. Introduction
2. Methods
2.1. Design and Participants
2.2. Study Procedures
2.3. Outcome Measures
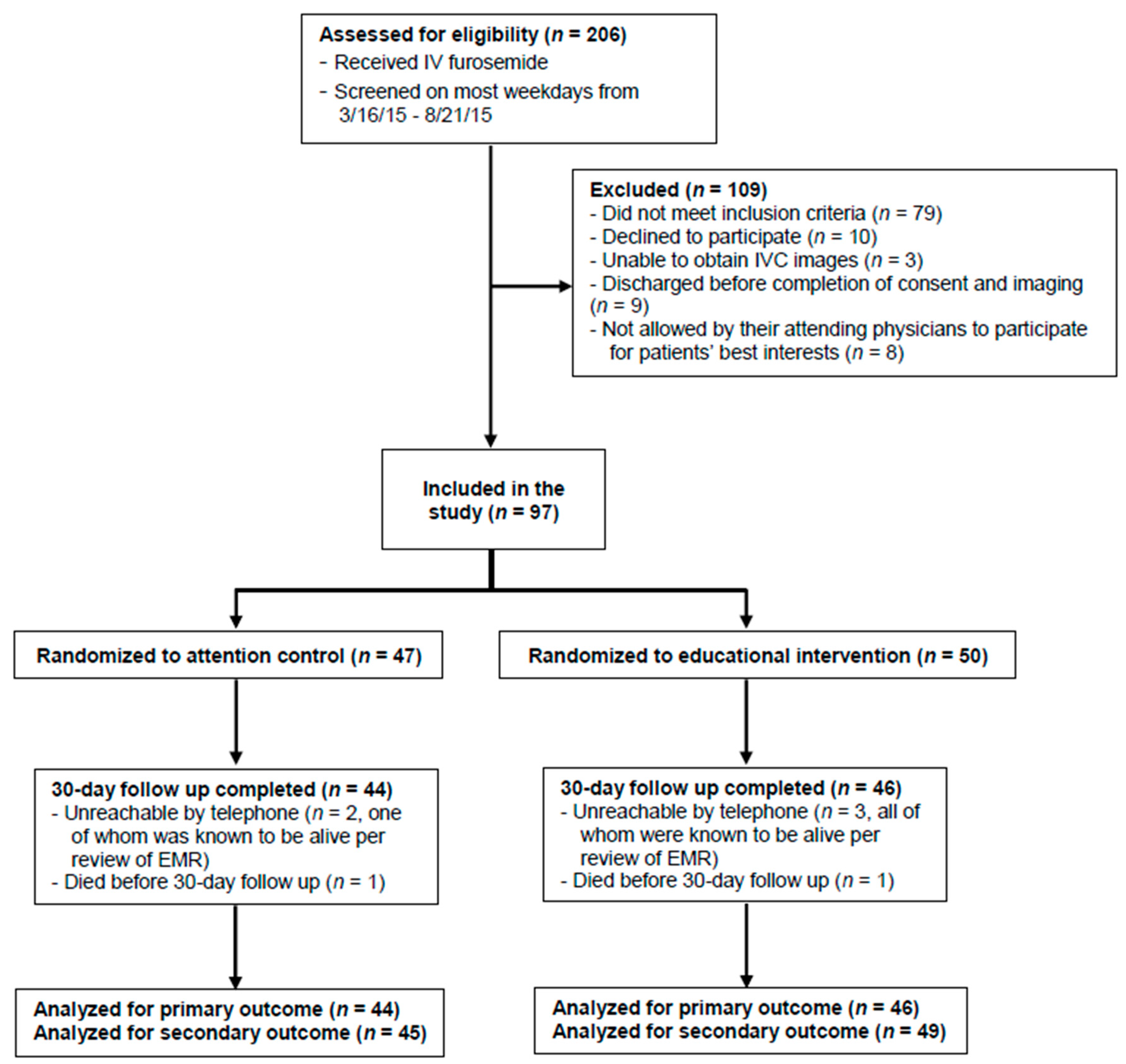
3. Results
3.1. Enrollment and Baseline Data
3.2. Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gheorghiade, M.; Vaduganathan, M.; Fonarow, G.C.; Bonow, R.O. Rehospitalization for heart failure: Problems and perspectives. J. Am. Coll. Cardiol. 2013, 61, 391–403. [Google Scholar] [CrossRef] [PubMed]
- Ambardekar, A.V.; Fonarow, G.C.; Hernandez, A.F.; Pan, W.; Yancy, C.W.; Krantz, M.J.; for the Get with the Guidelines Steering Committee and Hospitals. Characteristics and in-hospital outcomes for nonadherent patients with heart failure: Findings from Get with the Guidelines-Heart Failure (GWTG-HF). Am. Heart J. 2009, 158, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Zaya, M.; Phan, A.; Schwarz, E.R. The dilemma, causes and approaches to avoid recurrent hospital readmissions for patients with chronic heart failure. Heart Fail. Rev. 2012, 17, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chaiken, S. The heuristic-systematic model in its broader context. In Dual-Process Theories in Social Psychology, 1st ed.; Chaiken, S., Trope, Y., Eds.; Guilford: New York, NY, USA, 1999; pp. 73–96. ISBN 13 978-1572304215. [Google Scholar]
- Hollands, G.J.; Hankins, M.; Marteau, T.M. Visual feedback of individuals’ medical imaging results for changing health behaviour. Cochrane Database Syst. Rev. 2010, 1, CD007434. [Google Scholar] [CrossRef] [PubMed]
- Hollands, G.J.; Marteau, T.M. The impact of using visual images of the body within a personalized health risk assessment: An experimental study. Br. J. Health Psychol. 2013, 18, 263–278. [Google Scholar] [CrossRef] [PubMed]
- Bovet, P.; Perret, F.; Cornuz, J.; Quilindo, J.; Paccaud, F. Improved smoking cessation in smokers given ultrasound photographs of their own atherosclerotic plaques. Prev. Med. 2002, 34, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Hirschl, M.; Francesconi, C.; Chudik, M.; Katzenschlager, R.; Kundi, M. Degree of atherosclerosis predicts short-term commitment for smoking cessation therapy. Prev. Med. 2004, 39, 142–146. [Google Scholar] [CrossRef] [PubMed]
- DeWalt, D.A.; Malone, R.M.; Bryant, M.E.; Kosnar, M.C.; Corr, K.E.; Rothman, R.L.; Sueta, C.A.; Pignone, M.P. A heart failure self-management program for patients of all literacy levels: A randomized, controlled trial [ISRCTN11535170]. BMC Health Serv. Res. 2006, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Kalia, N.K.; Cespedes, L.; Youssef, G.; Li, D.; Budoff, M.J. Motivational effects of coronary artery calcium scores on statin adherence and weight loss. Coron. Artery Dis. 2015, 26, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Orakzai, R.H.; Nasir, K.; Orakzai, S.H.; Kalia, N.; Gopal, A.; Musunuru, K.; Blumenthal, R.S.; Budoff, M.J. Effect of patient visualization of coronary calcium by electron beam computed tomography on changes in beneficial lifestyle behaviors. Am. J. Cardiol. 2008, 101, 999–1002. [Google Scholar] [CrossRef] [PubMed]
- Goonewardena, S.N.; Gemignani, A.; Ronan, A.; Vasaiwala, S.; Blair, J.; Brennan, J.M.; Shah, D.P.; Spencer, K.T. Comparison of hand-carried ultrasound assessment of the inferior vena cava and N-terminal pro-brain natriuretic peptide for predicting readmission after hospitalization for acute decompensated heart failure. JACC Cardiovasc. Imaging 2008, 1, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Ziegelstein, R.C.; Fauerbach, J.A.; Stevens, S.S.; Romanelli, J.; Richter, D.P.; Bush, D.E. Patients with depression are less likely to follow recommendations to reduce cardiac risk during recovery from a myocardial infarction. Arch. Intern. Med. 2000, 160, 1818–1823. [Google Scholar] [CrossRef] [PubMed]
- Echocardiography for Emergency Physicians. Available online: https://www.acep.org/sonoguide/cardiac.html (accessed on 11 October 2018).
- Naylor, M.D.; Brooten, D.A.; Campbell, R.L.; Maislin, G.; McCauley, K.M.; Schwartz, J.S. Transitional care of older adults hospitalized with heart failure: A randomized, controlled trial. J. Am. Geriatr. Soc. 2004, 52, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Coleman, E.A.; Parry, C.; Chalmers, S.; Min, S.J. The care transitions intervention: Results of a randomized controlled trial. Arch. Intern. Med. 2006, 166, 1822–1828. [Google Scholar] [CrossRef] [PubMed]
- Ong, M.K.; Romano, P.S.; Edgington, S.; Aronow, H.U.; Auerbach, A.D.; Black, J.T.; De Marco, T.; Escarce, J.J.; Evangelista, L.S.; Hanna, B.; et al. Effectiveness of Remote Patient Monitoring after Discharge of Hospitalized Patients with Heart Failure: The Better Effectiveness after Transition—Heart Failure (BEAT-HF) Randomized Clinical Trial. JAMA Intern. Med. 2016, 176, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, B.D.; Moise, N.; Haerizadeh, M.; Ye, S.; Medina, V.; Kronish, I.M. Telemonitoring Adherence to Medications in Heart Failure Patients (TEAM-HF): A Pilot Randomized Clinical Trial. J. Card. Fail. 2017, 23, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Ziegelstein, R.C. Personomics: The Missing Link in the Evolution from Precision Medicine to Personalized Medicine. J. Pers. Med. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Control Group (CG) (n = 47) | Intervention Group (IG) (n = 50) | p Value |
|---|---|---|---|
| Female, n (%) | 28 (59.6) | 28 (56.0) | 0.72 |
| Age, years, mean (SD) | 72.4 (11.7) | 70.3 (13.7) | 0.43 |
| Comorbidities | |||
| Hypertension, n (%) | 41 (87.2) | 47 (94.0) | 0.31 |
| Diabetes mellitus, n (%) | 29 (61.7) | 18 (36.0) | 0.01 |
| Coronary artery disease, n (%) | 22 (46.8) | 28 (56.0) | 0.37 |
| Atrial fibrillation, n (%) | 19 (40.4) | 20 (40.0) | 0.97 |
| Chronic kidney disease (CrCl < 60 mL/min), n (%) | 22 (46.8) | 19 (38.0) | 0.38 |
| Left ventricular ejection fraction, a mean % (SD) | 46.5 (17.5) | 45.8 (16.1) | 0.84 |
| IVC Measurements | |||
| IVCmax (cm), mean (SD) | 2.43 (0.47) | 2.31 (0.46) | 0.21 |
| IVCmin (cm), mean (SD) | 1.66 (0.75) | 1.56 (0.70) | 0.52 |
| IVCCI (%), mean (SD) | 34.4 (20.1) | 34.5 (28.9) | 0.98 |
| Qualitative IVC Impression | |||
| Dilated and poorly collapsible (IVC > 2.5 cm diameter with poor phasicity; correlates with CVP > 15 mm Hg), n (%) | 22 (46.8) | 21 (42.0) | 0.88 |
| Normal diameter with <50% collapse (IVC 1.5–2.5 cm with poor collapsibility; correlates with CVP 10–15 mm Hg), n (%) | 7 (14.9) | 9 (18.0) | |
| Normal diameter with >50% collapse (IVC 1.5–2.5 cm with normal collapsibility; correlates with CVP 5–10 mm Hg), n (%) | 15 (31.9) | 18 (36.0) | |
| Small and very collapsible (IVC < 1.5 cm with ≥50% collapse); correlates with CVP 0–5 mm Hg), n (%) | 3 (6.4) | 2 (4.0) | |
| Control Group (CG) (n = 44) | Intervention Group (IG) (n = 46) | p Value for 30-Day Comparison | |||
| Baseline | 30 Days | Baseline | 30 Days | ||
| Total MOSSAS-3HFb, range 0–15, mean (SD) | 6.4 (3.7) | 11.7 (3.0) | 7.4 (3.4) | 11.8 (2.8) | 0.90 |
| Individual MOSSAS-3HF items | |||||
| Item 1: Took medication as prescribed (on time without skipping dose) (range 0–5), mean (SD) | 3.2 (1.6) | 4.7 (0.5) | 3.2 (1.8) | 4.4 (1.3) | 0.15 |
| Control Group (CG) (n = 44) | Intervention Group (IG) (n = 46) | p Value for 30-Day Comparison | |||
| Baseline | 30 Days | Baseline | 30 Days | ||
| Item 2: Followed a low-salt diet (range 0–5), mean (SD) | 1.9 (1.2) | 3.8 (1.0) | 2.4 (1.2) | 4.0 (1.0) | 0.16 |
| Item 3: Weighed yourself every day (range 0–5), mean (SD) | 1.3 (1.8) | 3.3 (2.0) | 1.8 (1.7) | 3.4 (1.7) | 0.85 |
| Survival without hospital readmission within 30 days, n (%) | 37 (84.1) | 39 (84.8) | 0.93 | ||
| Survival without readmission or ED visit within 30 days, n (%) | 37 (84.1) | 38 (82.6) | 0.85 | ||
| Time (days) from discharge to readmission or ED visit, mean (SD) | 9.7 (6.1) | 15.4 (9.3) | 0.20 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Athar, M.W.; Record, J.D.; Martire, C.; Hellmann, D.B.; Ziegelstein, R.C. The Effect of a Personalized Approach to Patient Education on Heart Failure Self-Management. J. Pers. Med. 2018, 8, 39. https://doi.org/10.3390/jpm8040039
Athar MW, Record JD, Martire C, Hellmann DB, Ziegelstein RC. The Effect of a Personalized Approach to Patient Education on Heart Failure Self-Management. Journal of Personalized Medicine. 2018; 8(4):39. https://doi.org/10.3390/jpm8040039
Chicago/Turabian StyleAthar, Muhammad W., Janet D. Record, Carol Martire, David B. Hellmann, and Roy C. Ziegelstein. 2018. "The Effect of a Personalized Approach to Patient Education on Heart Failure Self-Management" Journal of Personalized Medicine 8, no. 4: 39. https://doi.org/10.3390/jpm8040039
APA StyleAthar, M. W., Record, J. D., Martire, C., Hellmann, D. B., & Ziegelstein, R. C. (2018). The Effect of a Personalized Approach to Patient Education on Heart Failure Self-Management. Journal of Personalized Medicine, 8(4), 39. https://doi.org/10.3390/jpm8040039






