40 Years of Percutaneous Coronary Intervention: History and Future Directions
Abstract
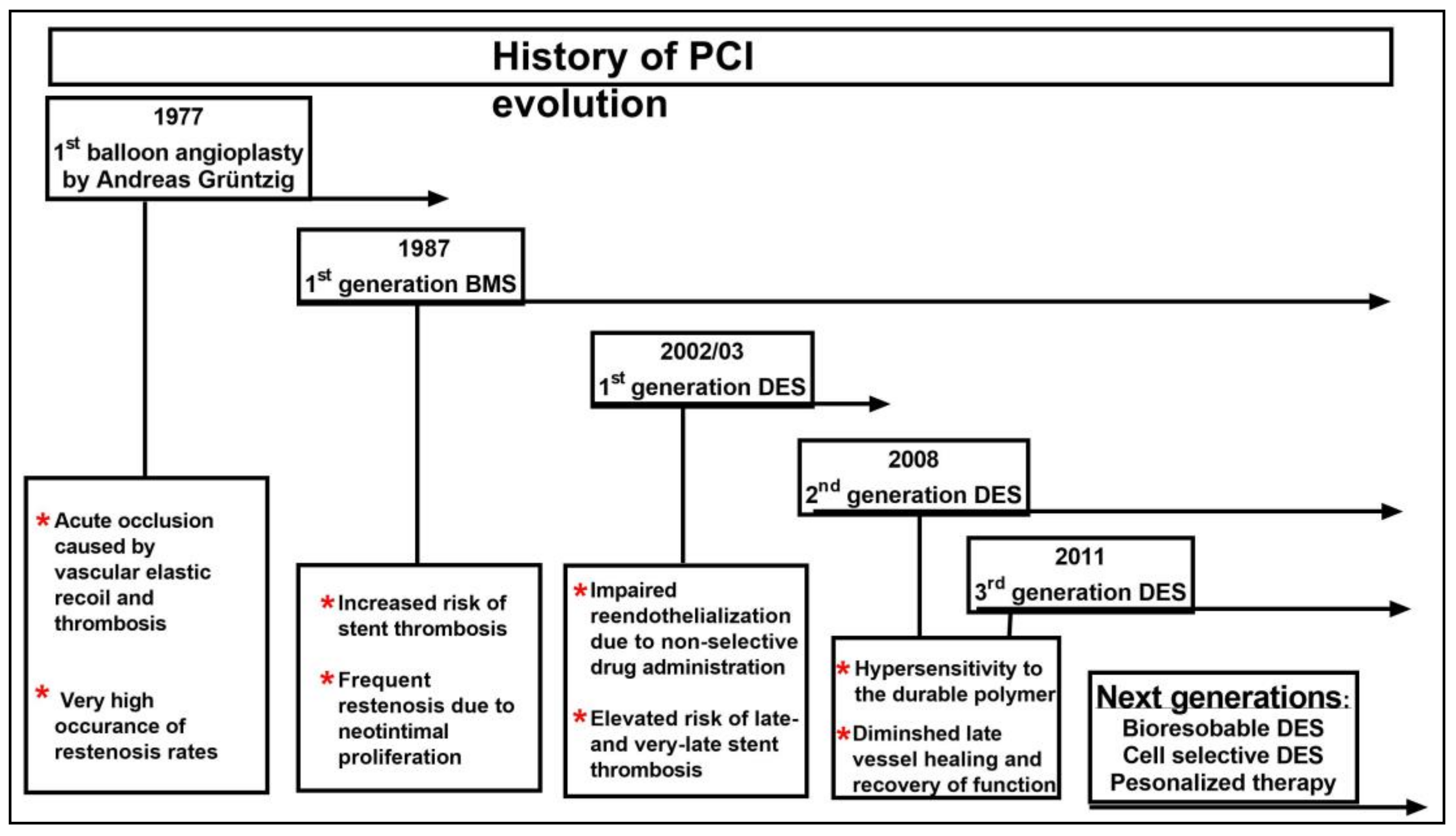
1. Introduction
2. Vascular Response to Stent Deployment
3. The Evolution of the First Generation of Drug-Eluting Stent
4. Vascular Response to Drug-Eluting Stent
5. New Generations of Drug-Eluting Stent
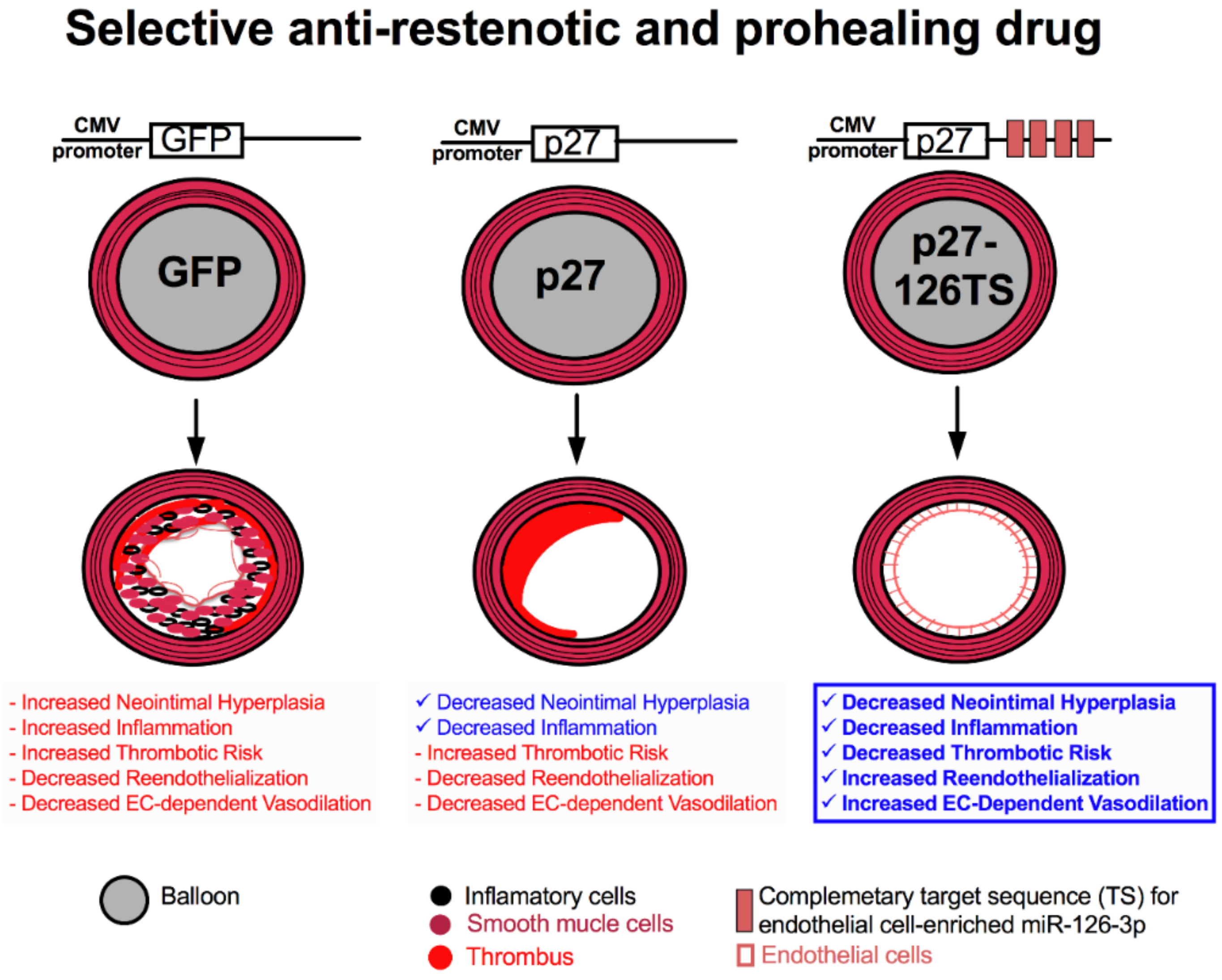
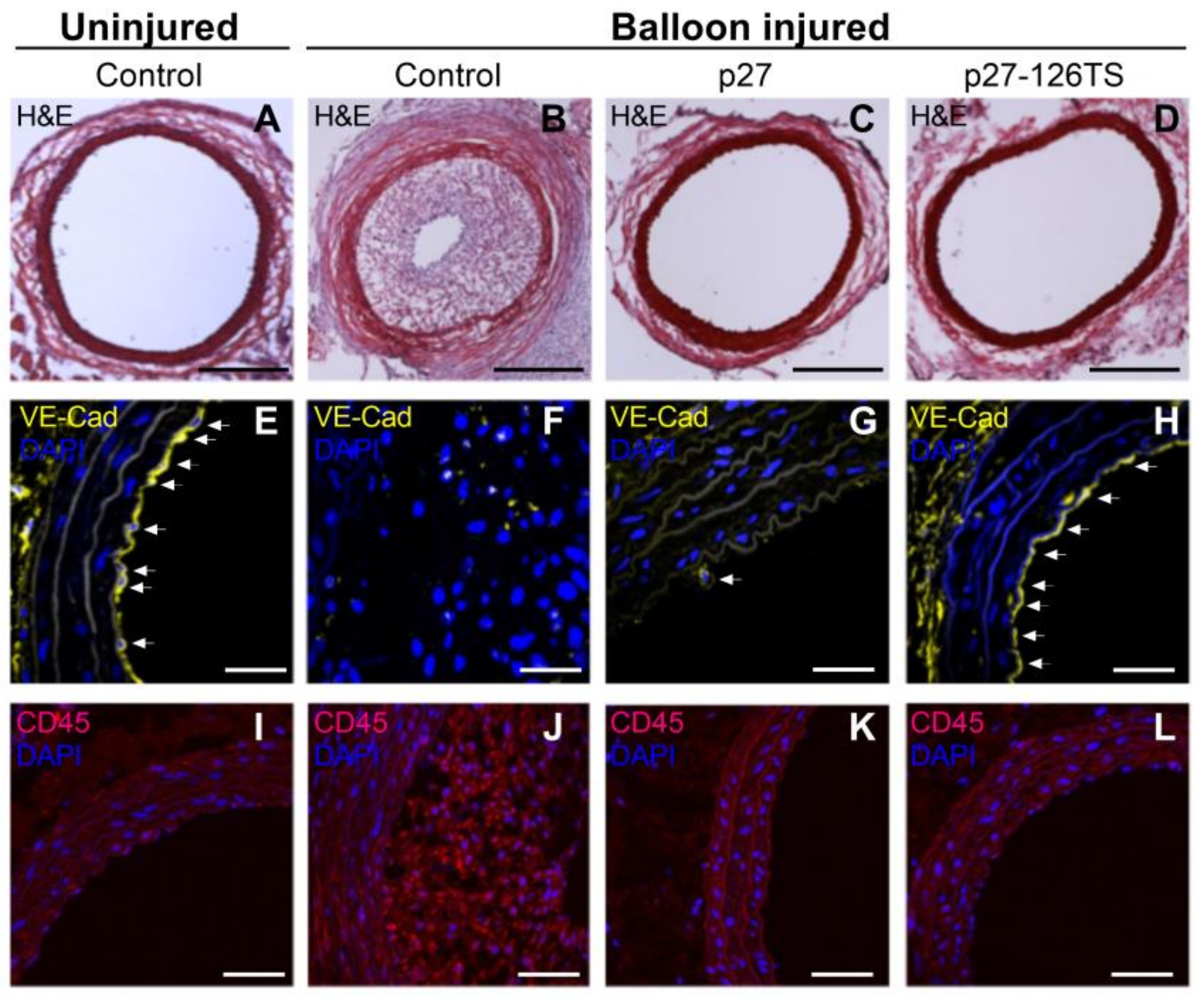
6. Future Directions for Percutaneous Coronary Intervention
6.1. Selective Anti-Restenotic and Prohealing Drug
6.2. Precision Medicine and Coronary Artery Disease
Author Contributions
Funding
Conflicts of Interest
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart disease and stroke statistics-2017 update: A report from the American heart association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- American Heart Association (AHA). Projections of Cardiovascular Disease Prevalence and Costs: 2015–2035; American Heart Association: Dallas, TX, USA, 2016; pp. 1–54. [Google Scholar]
- Laslett, L.J.; Alagona, P., Jr.; Clark, B.A., 3rd; Drozda, J.P., Jr.; Saldivar, F.; Wilson, S.R.; Poe, C.; Hart, M. The worldwide environment of cardiovascular disease: Prevalence, diagnosis, therapy, and policy issues: A report from the American college of cardiology. J. Am. Coll. Cardiol. 2012, 60, S1–S49. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, G.G.; Holmes, D.R., Jr. Drug-eluting coronary-artery stents. N. Engl. J. Med. 2013, 368, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Gruntzig, A. Transluminal dilatation of coronary-artery stenosis. Lancet 1978, 311, 1093. [Google Scholar] [CrossRef]
- Sigwart, U.; Puel, J.; Mirkovitch, V.; Joffre, F.; Kappenberger, L. Intravascular stents to prevent occlusion and restenosis after transluminal angioplasty. N. Engl. J. Med. 1987, 316, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Serruys, P.W.; Strauss, B.H.; Beatt, K.J.; Bertrand, M.E.; Puel, J.; Rickards, A.F.; Meier, B.; Goy, J.J.; Vogt, P.; Kappenberger, L.; et al. Angiographic follow-up after placement of a self-expanding coronary-artery stent. N. Engl. J. Med. 1991, 324, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Fischman, D.L.; Leon, M.B.; Baim, D.S.; Schatz, R.A.; Savage, M.P.; Penn, I.; Detre, K.; Veltri, L.; Ricci, D.; Nobuyoshi, M.; et al. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. Stent restenosis study investigators. N. Engl. J. Med. 1994, 331, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, R.; Mintz, G.S.; Dussaillant, G.R.; Popma, J.J.; Pichard, A.D.; Satler, L.F.; Kent, K.M.; Griffin, J.; Leon, M.B. Patterns and mechanisms of in-stent restenosis. A serial intravascular ultrasound study. Circulation 1996, 94, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Totary-Jain, H. Tailoring mTOR-based therapy: Molecular evidence and clinical challenges. Pharmacogenomics 2013, 14, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Poon, M.; Marx, S.O.; Gallo, R.; Badimon, J.J.; Taubman, M.B.; Marks, A.R. Rapamycin inhibits vascular smooth muscle cell migration. J. Clin. Investig. 1996, 98, 2277–2283. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.E.; Cao, W.; Huang, X.; Gregory, C.R.; Billingham, M.E.; Rowan, R.; Shorthouse, R.A. Rapamycin (Sirolimus) inhibits vascular smooth muscle DNA synthesis in vitro and suppresses narrowing in arterial allografts and in balloon-injured carotid arteries: Evidence that rapamycin antagonizes growth factor action on immune and nonimmune cells. Transpl. Proc. 1995, 27, 430–431. [Google Scholar]
- Gallo, R.; Padurean, A.; Jayaraman, T.; Marx, S.; Roque, M.; Adelman, S.; Chesebro, J.; Fallon, J.; Fuster, V.; Marks, A.; et al. Inhibition of intimal thickening after balloon angioplasty in porcine coronary arteries by targeting regulators of the cell cycle. Circulation 1999, 99, 2164–2170. [Google Scholar] [CrossRef] [PubMed]
- Moses, J.W.; Leon, M.B.; Popma, J.J.; Fitzgerald, P.J.; Holmes, D.R.; O’Shaughnessy, C.; Caputo, R.P.; Kereiakes, D.J.; Williams, D.O.; Teirstein, P.S.; et al. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N. Engl. J. Med. 2003, 349, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- Weisz, G.; Leon, M.B.; Holmes, D.R., Jr.; Kereiakes, D.J.; Clark, M.R.; Cohen, B.M.; Ellis, S.G.; Coleman, P.; Hill, C.; Shi, C.; et al. Two-year outcomes after sirolimus-eluting stent implantation: Results from the sirolimus-eluting stent in de novo native coronary lesions (sirius) trial. J. Am. Coll. Cardiol. 2006, 47, 1350–1355. [Google Scholar] [CrossRef] [PubMed]
- Weisz, G.; Leon, M.B.; Holmes, D.R., Jr.; Kereiakes, D.J.; Popma, J.J.; Teirstein, P.S.; Cohen, S.A.; Wang, H.; Cutlip, D.E.; Moses, J.W. Five-year follow-up after sirolimus-eluting stent implantation results of the sirius (sirolimus-eluting stent in de-novo native coronary lesions) trial. J. Am. Coll. Cardiol. 2009, 53, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Stone, G.W.; Ellis, S.G.; Cox, D.A.; Hermiller, J.; O’Shaughnessy, C.; Mann, J.T.; Turco, M.; Caputo, R.; Bergin, P.; Greenberg, J.; et al. A polymer-based, paclitaxel-eluting stent in patients with coronary artery disease. N. Engl. J. Med. 2004, 350, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Stone, G.W.; Moses, J.W.; Ellis, S.G.; Schofer, J.; Dawkins, K.D.; Morice, M.C.; Colombo, A.; Schampaert, E.; Grube, E.; Kirtane, A.J.; et al. Safety and efficacy of sirolimus- and paclitaxel-eluting coronary stents. N. Engl. J. Med. 2007, 356, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Stettler, C.; Wandel, S.; Allemann, S.; Kastrati, A.; Morice, M.C.; Schomig, A.; Pfisterer, M.E.; Stone, G.W.; Leon, M.B.; de Lezo, J.S.; et al. Outcomes associated with drug-eluting and bare-metal stents: A collaborative network meta-analysis. Lancet 2007, 370, 937–948. [Google Scholar] [CrossRef]
- Otsuka, F.; Finn, A.V.; Yazdani, S.K.; Nakano, M.; Kolodgie, F.D.; Virmani, R. The importance of the endothelium in atherothrombosis and coronary stenting. Nat. Rev. Cardiol. 2012, 9, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Tomey, M.; Mehran, R. Dual antiplatelet therapy dilemmas: Duration and choice of antiplatelets in acute coronary syndromes. Curr. Cardiol. Rep. 2013, 15, 405. [Google Scholar] [CrossRef] [PubMed]
- Levine, G.N.; Bates, E.R.; Bittl, J.A.; Brindis, R.G.; Fihn, S.D.; Fleisher, L.A.; Granger, C.B.; Lange, R.A.; Mack, M.J.; Mauri, L.; et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2016, 68, 1082–1115. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, F.; Byrne, R.A.; Yahagi, K.; Mori, H.; Ladich, E.; Fowler, D.R.; Kutys, R.; Xhepa, E.; Kastrati, A.; Virmani, R.; et al. Neoatherosclerosis: Overview of histopathologic findings and implications for intravascular imaging assessment. Eur. Heart J. 2015, 36, 2147–2159. [Google Scholar] [CrossRef] [PubMed]
- Toyota, T.; Shiomi, H.; Morimoto, T.; Kimura, T. Meta-analysis of long-term clinical outcomes of everolimus-eluting stents. Am. J. Cardiol. 2015, 116, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Palmerini, T.; Benedetto, U.; Biondi-Zoccai, G.; Della Riva, D.; Bacchi-Reggiani, L.; Smits, P.C.; Vlachojannis, G.J.; Jensen, L.O.; Christiansen, E.H.; Berencsi, K.; et al. Long-term safety of drug-eluting and bare-metal stents: Evidence from a comprehensive network meta-analysis. J. Am. Coll. Cardiol. 2015, 65, 2496–2507. [Google Scholar] [CrossRef] [PubMed]
- Bonaa, K.H.; Mannsverk, J.; Wiseth, R.; Aaberge, L.; Myreng, Y.; Nygard, O.; Nilsen, D.W.; Klow, N.E.; Uchto, M.; Trovik, T.; et al. Drug-eluting or bare-metal stents for coronary artery disease. N. Engl. J. Med. 2016, 375, 1242–1252. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.O.; Thayssen, P.; Christiansen, E.H.; Maeng, M.; Ravkilde, J.; Hansen, K.N.; Hansen, H.S.; Krusell, L.; Kaltoft, A.; Tilsted, H.H.; et al. Safety and efficacy of everolimus-versus sirolimus-eluting stents: 5-year results from SORT OUT IV. J. Am. Coll. Cardiol. 2016, 67, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, R.; Stefanini, G.G.; Franzone, A.; Spitzer, E.; Blochlinger, S.; Heg, D.; Juni, P.; Windecker, S. Safety and efficacy of resolute zotarolimus-eluting stents compared with everolimus-eluting stents: A meta-analysis. Circ. Cardiovasc. Interv. 2015, 8, e002223. [Google Scholar] [CrossRef] [PubMed]
- Ormiston, J.A.; Serruys, P.W.; Regar, E.; Dudek, D.; Thuesen, L.; Webster, M.W.; Onuma, Y.; Garcia-Garcia, H.M.; McGreevy, R.; Veldhof, S. A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): A prospective open-label trial. Lancet 2008, 371, 899–907. [Google Scholar] [CrossRef]
- Dudek, D.; Onuma, Y.; Ormiston, J.A.; Thuesen, L.; Miquel-Hebert, K.; Serruys, P.W. Four-year clinical follow-up of the absorb everolimus-eluting bioresorbable vascular scaffold in patients with de novo coronary artery disease: The absorb trial. EuroIntervention 2012, 7, 1060–1061. [Google Scholar] [CrossRef] [PubMed]
- Ellis, S.G.; Kereiakes, D.J.; Metzger, D.C.; Caputo, R.P.; Rizik, D.G.; Teirstein, P.S.; Litt, M.R.; Kini, A.; Kabour, A.; Marx, S.O.; et al. Everolimus-eluting bioresorbable scaffolds for coronary artery disease. N. Engl. J. Med. 2015, 373, 1905–1915. [Google Scholar] [CrossRef] [PubMed]
- Serruys, P.W.; Chevalier, B.; Sotomi, Y.; Cequier, A.; Carrie, D.; Piek, J.J.; Van Boven, A.J.; Dominici, M.; Dudek, D.; McClean, D.; et al. Comparison of an everolimus-eluting bioresorbable scaffold with an everolimus-eluting metallic stent for the treatment of coronary artery stenosis (ABSORB II): A 3 year, randomised, controlled, single-blind, multicentre clinical trial. Lancet 2016, 388, 2479–2491. [Google Scholar] [CrossRef]
- Toyota, T.; Morimoto, T.; Shiomi, H.; Yoshikawa, Y.; Yaku, H.; Yamashita, Y.; Kimura, T. Very late scaffold thrombosis of bioresorbable vascular scaffold: Systematic review and a meta-analysis. JACC Cardiovasc. Interv. 2017, 10, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Guagliumi, G.; Sirbu, V.; Musumeci, G.; Gerber, R.; Biondi-Zoccai, G.; Ikejima, H.; Ladich, E.; Lortkipanidze, N.; Matiashvili, A.; Valsecchi, O.; et al. Examination of the in vivo mechanisms of late drug-eluting stent thrombosis: Findings from optical coherence tomography and intravascular ultrasound imaging. JACC Cardiovasc. Interv. 2012, 5, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Vorpahl, M.; Yazdani, S.K.; Nakano, M.; Ladich, E.; Kolodgie, F.D.; Finn, A.V.; Virmani, R. Pathobiology of stent thrombosis after drug-eluting stent implantation. Curr. Pharm. Des. 2010, 16, 4064–4071. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, G.; Finn, A.V.; Joner, M.; Ladich, E.; Kutys, R.; Mont, E.K.; Gold, H.K.; Burke, A.P.; Kolodgie, F.D.; Virmani, R. Delayed arterial healing and increased late stent thrombosis at culprit sites after drug-eluting stent placement for acute myocardial infarction patients: An autopsy study. Circulation 2008, 118, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- Luscher, T.F.; Steffel, J.; Eberli, F.R.; Joner, M.; Nakazawa, G.; Tanner, F.C.; Virmani, R. Drug-eluting stent and coronary thrombosis: Biological mechanisms and clinical implications. Circulation 2007, 115, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Finn, A.V.; Joner, M.; Nakazawa, G.; Kolodgie, F.; Newell, J.; John, M.C.; Gold, H.K.; Virmani, R. Pathological correlates of late drug-eluting stent thrombosis: Strut coverage as a marker of endothelialization. Circulation 2007, 115, 2435–2441. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, G.; Nakano, M.; Otsuka, F.; Wilcox, J.N.; Melder, R.; Pruitt, S.; Kolodgie, F.D.; Virmani, R. Evaluation of polymer-based comparator drug-eluting stents using a rabbit model of iliac artery atherosclerosis. Circ. Cardiovasc. Interv. 2011, 4, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Bourantas, C.; Serruys, P.W. New concepts in the design of drug-eluting coronary stents. Nat. Rev. Cardiol. 2013, 10, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Alpert, J.S.; Kern, K.B.; Ewy, G.A. The risk of stent thrombosis after coronary arterial stent implantation. Am. J. Med. 123, 479–480. [CrossRef] [PubMed]
- Santulli, G.; Wronska, A.; Uryu, K.; Diacovo, T.G.; Gao, M.; Marx, S.O.; Kitajewski, J.; Chilton, J.M.; Akat, K.M.; Tuschl, T.; et al. A selective microrna-based strategy inhibits restenosis while preserving endothelial function. J. Clin. Investig. 2014, 124, 4102–4114. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Ingolia, N.T.; Weissman, J.S.; Bartel, D.P. Mammalian micrornas predominantly act to decrease target mRNA levels. Nature 2010, 466, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Ambros, V. The functions of animal micrornas. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.H.; Schmidt, M.; Jin, S.W.; Gray, A.M.; Beis, D.; Pham, T.; Frantz, G.; Palmieri, S.; Hillan, K.; Stainier, D.Y.; et al. The endothelial-cell-derived secreted factor Egfl7 regulates vascular tube formation. Nature 2004, 428, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Nichol, D.; Stuhlmann, H. EGFL7: A unique angiogenic signaling factor in vascular development and disease. Blood 2012, 119, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Campagnolo, L.; Leahy, A.; Chitnis, S.; Koschnick, S.; Fitch, M.J.; Fallon, J.T.; Loskutoff, D.; Taubman, M.B.; Stuhlmann, H. EGFL7 is a chemoattractant for endothelial cells and is up-regulated in angiogenesis and arterial injury. Am. J. Pathol. 2005, 167, 275–284. [Google Scholar] [CrossRef]
- Consortium, C.A.D.; Deloukas, P.; Kanoni, S.; Willenborg, C.; Farrall, M.; Assimes, T.L.; Thompson, J.R.; Ingelsson, E.; Saleheen, D.; Erdmann, J.; et al. Large-scale association analysis identifies new risk loci for coronary artery disease. Nat Genet. 2013, 45, 25–33. [Google Scholar]
- Lee, C.R.; Sriramoju, V.B.; Cervantes, A.; Howell, L.A.; Varunok, N.; Madan, S.; Hamrick, K.; Polasek, M.J.; Lee, J.A.; Clarke, M.; et al. Clinical outcomes and sustainability of using CYP2C19 genotype-guided antiplatelet therapy after percutaneous coronary intervention. Circ. Genom. Precis. Med. 2018, 11, e002069. [Google Scholar] [PubMed]
- Ladapo, J.A.; Budoff, M.J.; Azarmina, P.; Sharp, D.; Baker, A.; Maniet, B.; Herman, L.; Monane, M. Economic outcomes of a precision medicine blood test to assess obstructive coronary artery disease: Results from the preset registry. Manag. Care 2018, 27, 34–40. [Google Scholar] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canfield, J.; Totary-Jain, H. 40 Years of Percutaneous Coronary Intervention: History and Future Directions. J. Pers. Med. 2018, 8, 33. https://doi.org/10.3390/jpm8040033
Canfield J, Totary-Jain H. 40 Years of Percutaneous Coronary Intervention: History and Future Directions. Journal of Personalized Medicine. 2018; 8(4):33. https://doi.org/10.3390/jpm8040033
Chicago/Turabian StyleCanfield, John, and Hana Totary-Jain. 2018. "40 Years of Percutaneous Coronary Intervention: History and Future Directions" Journal of Personalized Medicine 8, no. 4: 33. https://doi.org/10.3390/jpm8040033
APA StyleCanfield, J., & Totary-Jain, H. (2018). 40 Years of Percutaneous Coronary Intervention: History and Future Directions. Journal of Personalized Medicine, 8(4), 33. https://doi.org/10.3390/jpm8040033



