Surveillance Recommendations in Reducing Risk of and Optimally Managing Breast Cancer-Related Lymphedema
Abstract
:1. Introduction
1.1. Definition
1.2. Incidence
1.3. Symptoms
| Clinical Stage | Description |
|---|---|
| 0 | A subclinical stage where swelling is not seen despite underlying changes in the lymphatic system |
| I | The initial stage of swelling which can be transient and where simple elevation can alleviate swelling |
| II | Swelling is constant and pitting without resolution using elevation |
| III | The tissue has become hard and fibrotic with associated skin changes |
| Severity | Based on volume differences between affected and contralateral limb in unilateral presentation |
| Mild ≤ 20% increase; Moderate = 20%–38% increase, Severe ≥ 38% increase |
2. Assessment and Management of LE
2.1. Assessment
2.2. Treatment
2.2.1. Non-Invasive Components of Care
2.2.2. Surgical Treatment
2.2.3. Nodal Status Determination
3. Adjunct Therapies
3.1. Pneumatic Compression Therapy
3.2. Aqua Lymphatic Therapy
3.3. Complementary and Alternative Medicine
3.4. Low-Level-Laser Therapy
4. Predisposing Factors
4.1. Exercise
4.2. Breast Cancer Surgery and Radiation Therapy
4.3. Comorbidities
4.4. Genetic Predisposition
5. Risk-Reduction Strategies and Preventive Interventions
5.1. Self-Management
Exercise and Weight-Lifting
5.2. Surgical Prevention
7. Supportive Care
7.1. Adherence to Self-Management of BCRL
7.2. Health Care Provider Knowledge
7.3. Palliative Care
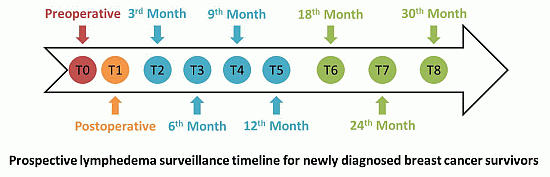
8. Interdisciplinary Surveillance
8.1. Measures for Surveillance
8.1.1. Lymphedema Breast Cancer Questionnaire (LBCQ)
8.1.2. Functional Assessment of Cancer Therapy (FACT-B+4)
8.2. Innovation in Qualitative Measures
9. Clinical Research
| Aims | (1) To determine if rigorous surveillance with web-based questionnaires in conjunction with arm volume measurements demonstrates earlier detection of BCRL compared to standard care. (2) To determine if earlier detection of BCRL using web-based HRQOL questionnaires in conjunction with arm volume measures improves QOL and functional outcomes. (3) To assess the cost and efficiency of nursing time in completing and evaluating questionnaires |
| Methods | Longitudinal, prospective, mixed-method study which will recruit women with newly-diagnosed breast cancer to complete a comprehensive symptom and quality of life assessment (LBCQ) and undergo arm volume measures pre-operatively, post-operatively, and quarterly. Preoperatively and at 12-month follow-up, patients will complete the FACT-B+4 questionnaire. Descriptive statistics and correlations will be used to examine relationships between actual measured arm volumes and subjective web-based questionnaire responses to determine if early symptoms correlate with limb volume changes. This study has been approved by the University of Missouri Health Sciences Institutional Review Board. |
| Results | Currently recruiting (n = 25 currently have given informed, written consent; up to 100 will be recruited) |
| Conclusion | Early diagnosis and intervention for BCRL is reported to improve the patients’ quality of life and functional outcomes. By utilizing a novel electronic tool developed in our institution (LBCQ) as a component of rigorous surveillance, we hope to show lymphedema is diagnosed earlier than with standard care, resulting in improved HRQOL and functional outcomes for our patients. |
| Clinical Trial Organization | Clinical Trial Number | Name of Trial | Principal Investigator(s) |
|---|---|---|---|
| Alliance (CALGB) | 70305 | A randomized education/exercise intervention study to reduce risk of lymphedema in women treated for breast cancer | Electra Paskett, Jane Armer, Lisa Yee, Michele Naughton, et al. |
| Alliance (ACOSOG) | Z1070 Successor Trial | Axillary management of T0-T3 node positive breast cancer receiving neoadjuvant chemotherapy | Judy C. Boughey MD, Tom Buchholz MD, Bruce Haffty MD, Vera Suman PhD, Janice Cormier, MD, MPH, Jane Armer, RN, PhD |
| Gynecologic Oncology Group (GOG) | The Lymphedema and Gynecologic Cancer (LEG) Study: Incidence, Risk Factors, and Impact | Richard Barakat, MD, MSKCC, NCI R01 |
10. Conclusions
10.1. Prospective Surveillance Model
10.2. Costs Associated with Traditional and Prospective Models
| Pre-op assessment with every woman who will undergo breast cancer surgery and/or RT. | H&P; height, weight, BMI, baseline bilateral limb (arm) volume measures with certified lymphedema therapist, functional assessment; provision of educational and resource information (predisposing factors, risk-reduction activities, signs and symptoms of early BCRL, support group information, team contact information; physical activity assessment and program information; nutritional information); introduction to team members and Q & A opportunities |
| Post-operative visit by team nurse and lymphedema therapist | Bilateral LV measure and assessment; supportive-educative visit; Ongoing: assess eligibility for clinical trials |
| Interval visits @ 1, 3, 6, 9, 12 months | Weight; BMI; bilateral LV measures; functional assessment; physical activity assessment; skin assessment. Ongoing: physical exercise program options, unless contraindicated; weight management program options; nutritional support and referral with dietician; support group information; virtual support group/blogs; contact with patient by team member monthly the first year post-breast cancer treatment; psychological/psychosocial assessment/Family assessment with a trained counselor. |
| Semi-Annual visits for 1–3 years | Same as above with team contact every 2 months to discuss status and evaluate need for resource referral. |
| Annual visits, if no BCRL is diagnosed | Team contact quarterly to discuss status and evaluate need for resource referral. |
| If BCRL is diagnosed: | Imaging: (lymphoscintigraphy/lymphangioscintigraphy, lymphography, MRI, ultrasound) to assess and/or rule out problems with lymphatic structures and flow and venous circulation, if indicated. Ongoing: Visits per treatment plan prescribed by lymphedema therapist for initial CDT. |
| Ongoing: support of self-management: Team contact every month or more often initially if necessary, treatment information, clinical trial information if available, support group information/other media information, new product information, Online LBCQ q 3 months or per institutional protocol, Fact-B+4 annually or per institutional protocol | |
| Ongoing throughout survivorship: | Celebration events at least annually |
10.3. Future Research
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Cancer Institute. Lymphedema (PDQ). Available online: www.cancer.gov/cancertopics/pdq/supportivecare/lymphedema/healthrofessional (accessed on 7 February 2014).
- Chang, C.J.; Cormier, J.N. Lymphedema intervetions: Exercise, surgery, and compression devices. Semin. Oncol. Nurs. 2013, 29, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Lasinski, B.; Thrift, K.; Squire, D.; Austin, M.K.; Smith, K.M.; Wanchai, A.; Green, J.M.; Stewart, B.R.; Cormier, J.N.; Armer, J.M. A systematic review of the evidence for complete decongestive therapy in the treatment of lymphedema from 2004 to 2011. Am. Acad. Phys. Med. Rehabil. 2012, 4, 580–601. [Google Scholar]
- Armer, J.M.; Fu, M.R.; Wainstock, J.M.; Zagar, E.; Jacobs, L.K. Lymphedema following breast cancer treatment, including sentinel lymph node biopsy. Lymphology 2004, 37, 73–91. [Google Scholar] [PubMed]
- Shah, C.; Arthur, D.; Riutta, J.; Whitworth, P.; Vicini, F. Breast cancer-related lymphedema: A review of procedure-specific incidence rates, clinical assessment aids, treatment paradigms, and risk reduction. Breast J. 2012, 18, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Textbook of Lymphology: For Physicians and Lymphedema Therapists, 3rd ed.; Fӧldi, M.; Fӧldi, E.; Strӧbenreuther, R.; Kubik, S. (Eds.) Elsevier Urban & Fischer: Munich, Germany, 2012.
- Armer, J.; Stewart, B. Post-breast cancer lymphedema: Incidence increases from 12 to 30 to 60 months. Lymphology 2010, 43, 118–122. [Google Scholar] [PubMed]
- Khan, S.A. Axillary reverse mapping to prevent lymphedema after breast cancer surgery: Defining the limits of the concept. J. Clin. Oncol. 2009, 27, 5494–5496. [Google Scholar] [CrossRef] [PubMed]
- Francis, W.P.; Abghari, P.; Du, W.; Rymal, C.; Suna, M.; Kosir, M.A. Improving surgical outcomes: Standardizing the reporting of incidence and severity of acute lymphedema after sentinel lymph node biopsy and axillary lymph node dissection. Am. J. Surg. 2006, 192, 636–663. [Google Scholar] [CrossRef] [PubMed]
- Wilke, L.G.; McCall, L.M.; Posther, K.E.; Whitworth, P.W.; Reintgen, D.S.; Leitch, A.M.; Gabram, S.G.; Lucci, A.; Cox, C.E.; Hunt, K.K.; et al. Surgical complications associated with sentinel lymph node biopsy: Results from a prospective international cooperative group trial. Ann. Surg. Oncol. 2006, 13, 491–500. [Google Scholar]
- Norman, S.A.; Localio, A.R.; Potashnik, S.L.; Torpey, H.A.S.; Kallan, M.J.; Weber, A.L.; Miller, L.T.; DeMichele, A.; Solin, L.J. Lymphedema in breast cancer survivors: Incidence, degree, time course, treatment, and symptoms. J. Clin. Oncol. 2009, 27, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Bernas, M. Assessment and risk reduction in lymphedema. Semin. Oncol. Nurs. 2013, 29, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Lacomba, M.; Sanchez, M.; Goni, A.; Merino, D.; del Moral, O.; Tellez, E.; Mogollon, E. Effectiveness of early physiotherapy to prevent lymphedema after surgery for breast cancer: Randomized, single blinded, clinical trial. Br. Med. J. 2010, 340. [Google Scholar] [CrossRef]
- Ridner, S.H.; Dietrich, M.S.; Kidd, N. Breast cancer treatment-related lymphedema self-care: Education, practices, symptoms, and quality of life. Support. Care Cancer 2011, 19, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Petrek, J.A.; Senie, R.T.; Peters, M.; Rosen, P.P. Lymphedema in a cohort of breast carcinoma survivors 20 years after diagnosis. Cancer 2001, 92, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Petrek, J.A.; Heelan, M.C. Incidence of breast carcinoma-related lymphedema. Cancer 1998, 83, A2776–A2781. [Google Scholar] [CrossRef]
- Radina, M.E.; Armer, J.M.; Stewart, B.R. Making self-care a priority for women at risk of breast cancer-related lymphedema. J. Fam. Nurs. 2014, 20, 226–249. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.L.; Specht, M.C.; Skolny, M.N.; Horick, N.; Jammallo, L.S.; O’Toole, J.; Shenouda, M.N.; Sadek, B.T.; Smith, B.L.; Taghian, A.G. Risk of lymphedema after mastectomy: Potential benefit of applying ACOSOG Z0011 protocol to mastectomy patients. Breast Cancer Res. Treat. 2014, 144, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Gergich, N.L.S.; Pfalzer, L.A.; McGarvey, C.; Springer, B.; Gerber, L.H.; Soballe, P. Preoperative assessment enables the early diagnosis and successful treatment of lymphedema. Cancer 2008, 112, 2809–2819. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.R.; Ridner, S.H.; Hu, S.H.; Stewart, B.R.; Cormier, J.N.; Armer, J.M. Psychosocial impact of lymphedema: A systematic review of literature from 2004 to 2011. Psycho-Oncology 2012, 7, 1466–1484. [Google Scholar]
- Armer, J.M.; Radina, M.E.; Porock, D.; Culberston, S. Predicting breast cancer-related lymphedema using self-reported symptoms. Nurs. Res. 2003, 52, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Armer, J.M.; Brooks, C.W.; Stewart, B.R. Limitations of self-care in reducing the risk of lymphedema: Supportive educative systems. Nurs. Sci. Quart. 2011, 24, 57–63. [Google Scholar] [CrossRef]
- Fu, M.R.; Rosedale, M. Breast Cancer survivors’ experiences of lymphedema-related symptoms. J. Pain Symptoms Manag. 2009, 38, 849–859. [Google Scholar] [CrossRef]
- International Society of Lymphology Executive Committee. The diagnosis and treatment of peripheral lymphedema: 2013 Consensus Document of the International Society of Lymphology. Lymphology 2013, 46, 1–11. [Google Scholar]
- Armer, J.M.; Shook, R.P.; Schneider, M.K.; Brooks, C.W.; Peterson, J.; Stewart, B.R. Enhancing supportive-educative nursing systems to reduce risk of post-breast cancer lymphedema. Self-Care Dependent-Care Nurs. 2009, 17, 6–15. [Google Scholar]
- Sherman, K.A.; Koelmeyer, L. Psychosocial predictors of adherence to lymphedema risk minimization guidelines among women with breast cancer. Psycho-Oncology 2013, 22, 1120–1126. [Google Scholar]
- Stout, N.L.; Pfalzer, L.A.; Springer, B.; Levy, E.; McGarvey, C.L.; Danoff, J.V.; Gerber, L.H.; Soballe, P.W. Breast cancer-related lymphedema: Comparing direct costs of a prospective surveillance model and a traditional model of care. J. Am. Phys. Ther. Assoc. 2012, 92, 152–163. [Google Scholar]
- Position statement of the National Lymphedema Network. Available online: http://www.lymphnet.org/ category/position-papers (accessed on 5 March 2014).
- Armer, J.M. The problem of post-breast cancer lymphedema: Impact and measurement issues. Cancer Investig. 2005, 23, 76–83. [Google Scholar] [CrossRef]
- Armer, J.M.; Stewart, B.R. Comparison of four diagnostic criteria for lymphedema in a post-breast cancer population. Lymphat. Res. Biol. 2005, 3, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Armer, J.M.; Hulett, J.M.; Bernas, M.; Ostby, P.; Stewart, B.R.; Cormier, J.N. Best-Practice guidelines in assessment, risk reduction, management, and surveillance for post-breast cancer lymphedema. Curr. Breast Cancer Rep. 2013, 5, 134–144. [Google Scholar] [CrossRef]
- Bernas, M.; Askew, R.; Armer, J.M.; Cormier, J.N. Lymphedema: How do we diagnose and reduce the risk of this dreaded complication of breast cancer treatment? Curr. Breast Cancer Rep. 2010, 2, 53–58. [Google Scholar]
- Armer, J.M.; Henggeler, M.H.; Brooks, C.W.; Zagar, E.A.; Homan, S.; Stewart, B.R. The health deviation of post-breast cancer lymphedema: Symptom assessment and impact on self-care agency. Self-Care Dependent-Care Nurs. 2008, 16, 14–21. [Google Scholar]
- Ward, L.C.; Czerniec, S.; Kilbreath, S.L. Quantitative bioimpedance spectroscopy for the assessment of lymphoedema. Breast Cancer Res. Treat. 2009, 117, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Mayrovitz, H.N.; Davey, S.; Shapiro, E. Local tissue water changes assessed by tissue dielectric constant: Single measurements versus averaging of multiple measurements. Lymphology 2008, 41, 186–188. [Google Scholar] [PubMed]
- Feldman, J.L.; Stout, N.L.; Wanchai, A.; Stewart, B.R.; Cormier, J.N.; Armer, J.M. Intermittent pneumatic compression therapy: A systematic review. Lymphology 2012, 45, 13–25. [Google Scholar] [PubMed]
- Cheifetz, O.; Haley, L. Management of secondary lymphedema related to breast cancer. Can. Fam. Phys. 2010, 56, 1277–1284. [Google Scholar]
- Lymphoedema Framework. In Best Practice for the Management of Lymphoedema: International Consensus; MEP Ltd: London, UK, 2006.
- Rockson, S.G. Lymphedema. Am. J. Med. 2001, 110, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Feldman, J.L.; Semel, J.D. The challenge of infection in lymphedema. LymphLink 2011, 23, 1–3. [Google Scholar]
- Cormier, J.N.; Askew, R.L.; Mungovan, K.S.; Xing, Y.; Ross, M.I.; Armer, J.M. Lymphedema beyond breast cancer. Cancer 2010, 116, 5138–5149. [Google Scholar] [CrossRef] [PubMed]
- Vignes, S.; Blanchard, M.; Yannoutsos, A.; Arrault, M. Complications of autologous lymph node transplantation for limb lymphoedema. Eur. J. Vasc. Endovasc. Surg. 2013, 45, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Cormier, J.N.; Rourke, L.; Crosby, M.; Chang, D.; Armer, J.M. The surgical treatment of lymphedema: A systematic review of the contemporary literature (2004–2010). Ann. Surg. Oncol. 2012, 19, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Damstra, R.J.; Voesten, H.G.J.; Schelven, W.D.; Lei, B. Lymphatic venous anastomosis (LVA) for treatment of secondary arm lymphedema. A prospective study of 11 LVA procedures in 10 patients with breast cancer-related lymphedema and a critical review of the literature. Breast Cancer Res. Treat. 2008, 113, 199–206. [Google Scholar]
- Granzow, J.W.; Soderberg, J.M.; Kaji, A.H.; Dauphine, C. Review of current surgical treatments for lymphedema. Ann. Surg. Oncol. 2014, 21, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.E.; Hunt, K.K.; Ballman, K.V.; Beitsch, P.D.; Whitworth, P.W.; Blumencranz, P.W.; Marilyn-Leitch, A.; Saha, S.; McCall, L.M.; Morrow, M. Axillary dissection vs. no axillary dissection in women with invasive breast cancer and sentinel node metastasis: A randomized clinical trial. JAMA 2011, 305, 569–575. [Google Scholar]
- Douek, M.; Klasse, J.; Monypenny, L.; Kothari, A.; Zechmeister, K.; Brown, D.; Pinder, S. Sentinel Node biopsy using a magnetic tracer versus standard technique: The SentiMAG multicentre trial. Ann. Surg. Oncol. 2014, 21, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Ridner, S.H.; McMahon, E.; Dietrich, M.S.; Hoy, S. Home-Based lymphedema treatment in patients with cancer-related lymphedema or noncancer-related lymphedema. Oncol. Nurs. Forum 2008, 35, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.E.; Rasmussen, J.C.; Darne, C.; Tan, I.; Aldrich, M.B.; Marshall, M.V.; Sevick-Muraca, E.M. Direct evidence of lymphatic function improvement after advanced pneumatic compression device treatment of lymphedema. Biomed. Opt. Express 2010, 1, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Tidhar, D.; Katz-Leurer, M. Aqua lymphatic therapy in women who suffer from breast cancer treatment-related lymphedema: A randomized controlled study. Support. Care Cancer 2010, 18, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Wanchai, A.; Armer, J.M.; Stewart, B.R. Complementary and alternative medicine and lymphedema. Semin. Oncol. Nurs. 2013, 29, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Kaviani, A.; Fateh, M.; Nooraie, R.Y.; Alinagi-Zadeh, M.; Ataie-Fashtami, L. Low-level laser therapy in management of postmastectomy lymphedema. Lasers Med. Sci. 2006, 21, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.T.B.R.M.; Lima, J.G.M.; Andrade, M.F.C.; Bergmann, A. Low-level laser therapy in secondary lymphedema after breast cancer: Systematic review. Lasers Med. Sci. 2012, 29, 1289–1295. [Google Scholar] [CrossRef] [PubMed]
- Ridner, S.; Poage-Hooper, E.; Kanar, C.; Doersam, J.; Bond, S.; Dietrich, M. A pilot randomized trial evaluating low-level laser therapy as an alternative treatment to manual lymphatic drainage for breast cancer-related lymphedema. Oncol. Nurs. Forum 2013, 40, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Dominick, S.A.; Madlensky, L.; Natarajan, L.; Pierce, J.P. Risk factors associated with breast cancer-related lymphedema in the WHEL study. J. Cancer Survivorship 2012, 7, 115–123. [Google Scholar] [CrossRef]
- Gho, S.A.; Steele, J.R.; Jones, S.C.; Munro, B.J. Self-Reported side effects of breast cancer treatment: A cross-sectional study of incidence, associations, and the influence of exercise. Cancer Causes Control. 2013, 24, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Ridner, S.H.; Dietrich, M.S.; Stewart, B.R.; Abbott, J.M. Body mass index and breast cancer-related lymphedema. Support. Care Cancer 2013, 19, 853–857. [Google Scholar] [CrossRef]
- Kwan, M.L.; Cohn, J.C.; Armer, J.M.; Stewart, B.R.; Cormier, J.N. Exercise in patients with lymphedema: A systematic review of the contemporary literature. J. Cancer Surv. 2011, 5, 320–336. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Ahmed, R.L.; Troxel, A.; Cheville, A.; Smith, R.; Lewis-Grant, L.; Greene, Q.P. Weight lifting in women with breast cancer-related lymphedema. N. Engl. J. Med. 2009, 261, 664–673. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Garshell, J.; Miller, D.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; et al. SEER Cancer Statistics Review, 1975–2011. Available online: http://seer.cancer.gov/csr/1975_2011/ (accessed on 1 March 2014).
- Bellury, L.; Ellington, L.; Beck, S.; Pett, M.; Clark, J.; Stein, K. Older breast cancer survivors: Can interaction analyses identify vulnerable subgroups? A report from the American Cancer Society Studies of cancer survivors. Oncol. Nurs. Forum 2013, 40, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Ridner, S.H.; Dietrich, M.S. Self-Reported comorbid conditions and medication usage in breast cancer survivors with and without lymphedema. Oncol. Nurs. Forum 2008, 35, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Keeley, V. Drugs that may exacerbate and those used to treat lymphoedema. J. Lymphoedema 2008, 3, 57–65. [Google Scholar]
- Newman, B.; Lose, F.; Kedda, M.; Francois, M.; Janda, M.; Hayes, S.C. Possible genetic predisposition to lymphedema after breast cancer. Lymphat. Res. Biol. 2012, 10, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Matchim, Y.; Armer, J.M.; Stewart, B.R. Effects of Mindfulness-Based Stress Reduction (MBSR) on health among breast cancer survivors. Western J. Nurs. Res. 2011, 33, 996–1016. [Google Scholar] [CrossRef]
- Ridner, S.H.; Bonner, C.M.; Deng, J.; Sinclair, V.G. Voices from the shadows: Living with lymphedema. Cancer Nurs. 2012, 35, E18–E26. [Google Scholar] [CrossRef] [PubMed]
- Boccardo, F.M.; Casabona, F.; Friedman, D.; Puglisi, M.; de Cain, F.; Ansaldi, F.; Campisi, C. Surgical prevention of arm lymphedema after breast cancer treatment. Ann. Surg. Oncol. 2011, 18, 2500–2505. [Google Scholar] [CrossRef] [PubMed]
- Rosedale, M.; Fu, M.R. Confronting the unexpected: Temporal, situational, and attributive dimensions of distressing symptom experience for breast cancer survivors. Oncol. Nurs. Forum 2010, 37, E28–E33. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.R.; Kang, Y. Psychosocial impact of living with cancer-related lymphedema. Semin. Oncol. Nurs. 2013, 29, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Radina, M.E. Breast cancer-related lymphedema: Implications for family leisure participation. Fam. Relat. 2009, 58, 445–459. [Google Scholar] [CrossRef]
- Radina, M.E.; Armer, J.M. Post-breast cancer lymphedema and the family: A qualitative investigation of families coping with chronic illness. J. Fam. Nurs. 2001, 7, 281–299. [Google Scholar] [CrossRef]
- Radina, M.E.; Armer, J.M. Surviving breast cancer and living with lymphedema: Resiliency among women in the context of their families. J. Fam. Nurs. 2004, 10, 484–505. [Google Scholar] [CrossRef]
- Kwan, M.L.; Shen, L.; Munneke, J.R.; Tam, E.K.; Partee, P.N.; André, M.; Thiadens, S.R.J. Patient awareness and knowledge of breast cancer-related lymphedema in a large, integrated health care delivery system. Breast Cancer Res. Treat. 2012, 135, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Khoury, M.; Sabb, B.R.; Haidar, L. Literature and the arts in medical education, autonomy and beneficence in an interactive theatre. Fam. Med. 2010, 42, 476–478. [Google Scholar] [PubMed]
- Pollack, L.A.; Adamache, W.; Ryerson, A.B.; Eheman, C.R.; Richardson, L.C. Care of long-term cancer survivors. Cancer 2009, 115, 5284–5295. [Google Scholar] [CrossRef] [PubMed]
- Tam, E.K.; Shen, L.; Munneke, J.R.; Ackerson, L.M.; Partee, P.N.; Somkin, C.P.; Kwan, M.L. Clinician awareness and knowledge of breast cancer-related lymphedema in a large, integrated health care delivery setting. Breast Cancer Res. Treat. 2012, 131, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Wanchai, A.; Stewart, B.R.; Armer, J.M. Experiences and management of breast cancer-related lymphoedema: A comparison between South Africa and the United States of America: LE experiences and management: SA and USA. Int. Nurs. Rev. 2012, 59, 117–124. [Google Scholar] [CrossRef]
- Wanchai, A.; Armer, J.M.; Stewart, B.R. Performance care practices in complementary and alternative medicine by Thai breast cancer survivors: An ethnonursing study. Nurs. Health Sci. 2012, 14, 339–344. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). WHO definition of palliative care. Available online: http://www.who.int/cancer/palliative/definition/en/ (accessed on 1 March 2014).
- Beck, M.; Wanchai, A.; Stewart, B.R.; Cormier, J.N.; Armer, J.M. Palliative care for cancer-related lymphedema: A systematic review. J. Palliat. Med. 2012, 15, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Cheville, A.L.; Andrews, K.; Kollasch, J.; Schmidt, K.; Basford, J. Adapting lymphedema treatment to the palliative setting. Am. J. Hosp. Palliat. Med. 2014, 31, 38–44. [Google Scholar] [CrossRef]
- National Accreditation Program for Breast Centers. Available online: https://www.facs.org/quality-programs/napbc/standards (accessed on 1 March 2014).
- American Cancer Society. What every woman with breast cancer should know. Available online: http://www.cancer.org/acs/groups/cid/documents/webcontent/002876-pdf.pdf (accessed on 1 March 2014).
- Armer, J.M.; Whitman, M. The problem of lymphedema following breast cancer treatment: Prevalence, symptoms, and self-management. Lymphology 2002, 35, 153–159. [Google Scholar] [PubMed]
- Brady, M.J.; Cella, D.F.; Mo, F.; Bonomi, A.E.; Tulsky, D.S.; Lloyd, S.R.; Deasy, S.; Cobleigh, M.; Shiomoto, G. Reliability and validity of the functional assessment of cancer therapy-breast quaility-of-life instrument. J. Clin. Oncol. 1997, 15, 974–986. [Google Scholar] [PubMed]
- Coster, S.; Poole, K.; Fallowfield, L.J. The validation of a quality of life scale to assess the impact of arm morbidity in breast cancer patients post-operatively. Breast Cancer Res. Treat. 2001, 68, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Schipper, H.; Clinch, J.; McMurray, A.; Levitt, M. Measuring the quality of life of cancer patients: The functional living index-cancer: Development and validation. J. Clin. Oncol. 1984, 2, 472–483. [Google Scholar] [PubMed]
- Keeley, V.; Crooks, S.; Locke, J.; Veigas, D.; Riches, K.; Hilliam, R. A quality of life measure for limb lymphoedema (LYMQOL). J. Lymphoedema 2010, 5, 26–37. [Google Scholar]
- Sherman, K.A.; Koelmeyer, L. The role of information sources and objective risk status on lymphedema risk-minimization behaviors in women recently diagnosed with breast cancer. Oncol. Nurs. Forum 2011, 38, E27–E36. [Google Scholar] [CrossRef] [PubMed]
- Birkballe, S.; Karlsmark, T.; Noerregaard, S.; Gottrup, F. A new concept of a multidisciplinary lymphoedema centre: Established in connection to a department of dermatology and the Copenhagen Wound Healing Center. Br. J. Dermatol. 2012, 167, 116–122. [Google Scholar] [CrossRef] [PubMed]
- European Wound Management Association (EWMA). Focus Document: Lymphoedema Bandaging in Practice; MEP Ltd.: London, UK, 2005. [Google Scholar]
- Reneker, J.; Armer, J.M.; Stewart, B.R.; Shyu, C.-R. Development of a minimum data set to assist in international collaborative lymphedema studies. In Lymphology: Progress in Lymphology XXIII, Presented at the 23rd International Congress of Lymphology; Malmӧ, Sweden, 19–23 September 2011, Brorson, H., Ed.; AM-TRYCK: Malmӧ, Sweden, 2011; pp. 230–233. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ostby, P.L.; Armer, J.M.; Dale, P.S.; Van Loo, M.J.; Wilbanks, C.L.; Stewart, B.R. Surveillance Recommendations in Reducing Risk of and Optimally Managing Breast Cancer-Related Lymphedema. J. Pers. Med. 2014, 4, 424-447. https://doi.org/10.3390/jpm4030424
Ostby PL, Armer JM, Dale PS, Van Loo MJ, Wilbanks CL, Stewart BR. Surveillance Recommendations in Reducing Risk of and Optimally Managing Breast Cancer-Related Lymphedema. Journal of Personalized Medicine. 2014; 4(3):424-447. https://doi.org/10.3390/jpm4030424
Chicago/Turabian StyleOstby, Pamela L., Jane M. Armer, Paul S. Dale, Margaret J. Van Loo, Cassie L. Wilbanks, and Bob R. Stewart. 2014. "Surveillance Recommendations in Reducing Risk of and Optimally Managing Breast Cancer-Related Lymphedema" Journal of Personalized Medicine 4, no. 3: 424-447. https://doi.org/10.3390/jpm4030424
APA StyleOstby, P. L., Armer, J. M., Dale, P. S., Van Loo, M. J., Wilbanks, C. L., & Stewart, B. R. (2014). Surveillance Recommendations in Reducing Risk of and Optimally Managing Breast Cancer-Related Lymphedema. Journal of Personalized Medicine, 4(3), 424-447. https://doi.org/10.3390/jpm4030424