Prognostic Value of Fibrosis 4 (FIB-4) Index in Sepsis Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection
2.3. Statistical Investigations
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.-L.; Jones, G.; David, S.; Olariu, E.; Cadwell, K.K. Frequency and mortality of septic shock in Europe and North America: A systematic review and meta-analysis. Crit. Care 2019, 23, 196. [Google Scholar] [CrossRef] [PubMed]
- Tsalik, E.L.; Woods, C.W. Sepsis redefined: The search for surrogate markers. Int. J. Antimicrob. Agents 2009, 34, S16–S20. [Google Scholar] [CrossRef] [PubMed]
- Gullo, A. Intensive and Critical Care Medicine Reflections, Recommendations and Perspectives; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Gunning, K.; Rowan, K. Outcome data and scoring systems. BMJ 1999, 319, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Gattani, S.C.; Baheti, A.H.; Dubey, A. Comparison of the performance of APACHE II, SOFA, and mNUTRIC scoring systems in critically ill patients: A 2-year cross-sectional study. Indian J. Crit. Care Med. 2020, 24, 1057. [Google Scholar] [PubMed]
- Xu, X.-L.; Jiang, L.-S.; Wu, C.-S.; Pan, L.-Y.; Lou, Z.-Q.; Peng, C.-T.; Dong, Y.; Ruan, B. The role of fibrosis index FIB-4 in predicting liver fibrosis stage and clinical prognosis: A diagnostic or screening tool? J. Formos. Med. Assoc. 2022, 121, 454–466. [Google Scholar] [CrossRef]
- Blanco-Grau, A.; Gabriel-Medina, P.; Rodriguez-Algarra, F.; Villena, Y.; Lopez-Martínez, R.; Augustín, S.; Pons, M.; Cruz, L.-M.; Rando-Segura, A.; Enfedaque, B.; et al. Assessing liver fibrosis using the FIB4 index in the community setting. Diagnostics 2021, 11, 2236. [Google Scholar] [CrossRef] [PubMed]
- Sterling, R.K.; Oakes, T.; Gal, T.S.; Stevens, M.P.; Dewit, M.; Sanyal, A.J. The fibrosis-4 index is associated with need for mechanical ventilation and 30-day mortality in patients admitted with coronavirus disease 2019. J. Infect. Dis. 2020, 222, 1794–1797. [Google Scholar] [CrossRef]
- Zhu, X.; Hu, X.; Qin, X.; Pan, J.; Zhou, W. An elevated Fibrosis-4 score is associated with poor clinical outcomes in patients with sepsis: An observational cohort study. Pol. Arch. Intern. Med. 2020, 130, 1064–1073. [Google Scholar] [CrossRef]
- Sumida, Y.; Yoneda, M.; Tokushige, K.; Kawanaka, M.; Fujii, H.; Yoneda, M.; Imajo, K.; Takahashi, H.; Eguchi, Y.; Ono, M.; et al. FIB-4 first in the diagnostic algorithm of metabolic-dysfunction-associated fatty liver disease in the era of the global metabodemic. Life 2021, 11, 143. [Google Scholar] [CrossRef]
- Houot, M.; Ngo, Y.; Munteanu, M.; Marque, S.; Poynard, T. Systematic review with meta-analysis: Direct comparisons of biomarkers for the diagnosis of fibrosis in chronic hepatitis C and B. Aliment. Pharmacol. Ther. 2016, 43, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, P.; Sabo, R.; Alsaadawi, R.; Robinson, A.; French, E.; Sterling, R.K. Fibrosis-4 (FIB-4) index as a predictor for mechanical ventilation and 30-day mortality across COVID-19 variants. J. Clin. Transl. Sci. 2023, 7, e213. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Kang, M.K.; Lee, Y.R.; Song, J.E.; Kim, N.Y.; Kweon, Y.O.; Tak, W.Y.; Jang, S.Y.; Lee, C.; Kim, B.S.; et al. Fibrosis-4 index as a predictor for mortality in hospitalised patients with COVID-19: A retrospective multicentre cohort study. BMJ Open 2020, 10, e041989. [Google Scholar] [CrossRef] [PubMed]
- Demir, N.; Yüzbasıoglu, B.; Calhan, T.; Ozturk, S. Prevalence and prognostic importance of high fibrosis-4 index in COVID-19 patients. Int. J. Clin. Pract. 2022, 2022, 1734896. [Google Scholar] [CrossRef]
- Shibata, N.; Kondo, T.; Kazama, S.; Kimura, Y.; Oishi, H.; Arao, Y.; Kato, H.; Yamaguchi, S.; Kuwayama, T.; Hiraiwa, H.; et al. Impact of predictive value of Fibrosis-4 index in patients hospitalized for acute heart failure. Int. J. Cardiol. 2021, 324, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Pranata, R.; Yonas, E.; Huang, I.; Lim, M.A.; Nasution, S.A.; Kuswardhani, R.A.T. Fibrosis-4 index and mortality in coronavirus disease 2019: A meta-analysis. Eur. J. Gastroenterol. Hepatol. 2021, 33, e368–e374. [Google Scholar] [CrossRef] [PubMed]
- Maeda, D.; Kanzaki, Y.; Sakane, K.; Tsuda, K.; Akamatsu, K.; Hourai, R.; Okuno, T.; Tokura, D.; Nakayama, S.; Hasegawa, H.; et al. Prognostic value of the liver fibrosis marker fibrosis-5 index in patients with acute heart failure. ESC Heart Fail. 2022, 9, 1380–1387. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, M.; Izumi, N. External validation of FIB-4: Diagnostic accuracy is limited in elderly populations. Hepatology 2008, 47, 352. [Google Scholar] [CrossRef]
- Ciardullo, S.; Monti, T.; Perseghin, G. High prevalence of advanced liver fibrosis assessed by transient elastography among US adults with type 2 diabetes. Diabetes Care 2021, 44, 519–525. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef]
- Guan, L.; Li, L.; Zou, Y.; Zhong, J.; Qiu, L. Association between FIB-4, all-cause mortality, cardiovascular mortality, and cardiovascular disease risk among diabetic individuals: NHANES 1999–2008. Front. Cardiovasc. Med. 2023, 10, 1172178. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-Y.; Adhikary, P.; Cheng, K. Cellular protein markers, therapeutics, and drug delivery strategies in the treatment of diabetes-associated liver fibrosis. Adv. Drug Deliv. Rev. 2021, 174, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Lomonaco, R.; Leiva, E.G.; Bril, F.; Shrestha, S.; Mansour, L.; Budd, J.; Romero, J.P.; Schmidt, S.; Chang, K.-L.; Samraj, G.; et al. Advanced liver fibrosis is common in patients with type 2 diabetes followed in the outpatient setting: The need for systematic screening. Diabetes Care 2021, 44, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Font, M.D.; Thyagarajan, B.; Khanna, A.K. Sepsis and Septic Shock-Basics of diagnosis, pathophysiology and clinical decision making. Med. Clin. 2020, 104, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Regan, J.; Fajnzylber, J.; Coxen, K.; Corry, H.; Wong, C.; Rosenthal, A.; Atyeo, C.; Fischinger, S.; Gillespie, E.; et al. Liver fibrosis index FIB-4 is associated with mortality in COVID-19. Hepatol. Commun. 2021, 5, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.V.; Milligan, S.; Frick, A.; Broestl, J.; Younossi, Z.; Afdhal, N.; Lai, M. Fibrosis-4 index can independently predict major adverse cardiovascular events in nonalcoholic fatty liver disease. Off. J. Am. Coll. Gastroenterol. ACG 2022, 117, 453–461. [Google Scholar] [CrossRef]
- Peleg, N.; Issachar, A.; Sneh-Arbib, O.; Shlomai, A. AST to Platelet Ratio Index and fibrosis 4 calculator scores for non-invasive assessment of hepatic fibrosis in patients with non-alcoholic fatty liver disease. Dig. Liver Dis. 2017, 49, 1133–1138. [Google Scholar] [CrossRef]
| n (%) | ||
|---|---|---|
| Gender | Female | 130 (42.1) |
| Male | 179 (57.9) | |
| Age | Mean ± Ss | 74.78 ± 13.92 |
| Median (Min–Max) | 77 (27–98) | |
| º Comorbidity | HT | 214 (69.3) |
| DM | 90 (29.1) | |
| CKD | 117 (37.9) | |
| CVD | 48 (15.5) | |
| CAD | 102 (33.0) | |
| Nasocomial infection | No | 84 (27.2) |
| Yes | 225 (72.8) | |
| Septic shock | No | 204 (66.0) |
| There is | 105 (34.0) | |
| º Breeding place | Blood | 124 (40.1) |
| Catheter | 21 (6.8) | |
| Tracheal aspirate culture/Phlegm | 112 (36.2) | |
| Urine | 93 (30.1) | |
| Wound site | 20 (6.5) | |
| Other (pleural fluid, peritoneum, etc.) | 4 (1.3) | |
| Effective | Acineto | 20 (6.5) |
| Klebsiella | 44 (14.2) | |
| Pseudomonas | 46 (14.9) | |
| E. coli | 96 (31.1) | |
| Enterococcus | 34 (11.0) | |
| MRSA | 10 (3.2) | |
| MSSA | 15 (4.9) | |
| Candida | 17 (5.5) | |
| Other | 33 (10.7) | |
| 28-day mortality | Survival | 167 (54.0) |
| Non-survival | 142 (46.0) |
| 28-Day Mortality | p | |||
|---|---|---|---|---|
| Survival (n = 167) | Non-Survival (n = 142) | |||
| Gender | Woman | 67 (40.1) | 63 (44.4) | a 0.451 |
| Male | 100 (59.9) | 79 (55.6) | ||
| Comorbidity | HT | 119 (71.3) | 95 (66.9) | a 0.408 |
| DM | 59 (35.3) | 31 (21.8) | a 0.009 ** | |
| CKD | 66 (39.5) | 51 (35.9) | a 0.515 | |
| CVD | 32 (19.2) | 16 (11.3) | a 0.056 | |
| CAD | 60 (35.9) | 42 (29.6) | a 0.237 | |
| Nasocomial | No | a 0.101 | ||
| 39 (23.4) | 45 (31.7) | |||
| Yes | 128 (76.6) | 97 (68.3) | ||
| Septic shock | No | 93 (55.7) | 111 (78.2) | a 0.001 ** |
| Yes | 74 (44.3) | 31 (21.8) | ||
| Place of reproduction Blood | a 0.639 | |||
| 0.65 (38.9) | 59 (41.5) | |||
| Catheter | 11 (6.6) | 10 (7.0) | a 0.874 | |
| TAC/Phlegm | 58 (34.7) | 54 (38.0) | a 0.548 | |
| Urine | 47 (28.1) | 46 (32.4) | a 0.417 | |
| Wound site | 17 (10.2) | 3 (2.1) | a 0.004 ** | |
| Other (pleural fluid, Peritoneum, etc.) | 2 (1.2) | 2 (1.4) | b 1.000 | |
| Agent | Acinetobacter | 10 (6) | 10 (7) | a 0.707 |
| Kleasiella | 22 (13.2) | 22 (15.5) | a 0.561 | |
| Pseudomonas | 25 (15.0) | 21 (14.8) | a 0.964 | |
| E. coli | 57 (34.1) | 39 (27.5) | a 0.207 | |
| Enterococcus | 17 (10.2) | 17 (12.0) | a 0.616 | |
| MRSA | 4 (2.4) | 6 (4.2) | b 0.522 | |
| MSSA | 8 (4.8) | 7 (4.9) | a 0.955 | |
| Candida | 8 (4,8) | 9 (6.3) | a 0.552 | |
| Other | 17 (10.2) | 16 (11.3) | a 0.758 |
| Total | 28-Day Mortality | p | |||
|---|---|---|---|---|---|
| Survival (n = 167) | Non-Survival (n = 142) | ||||
| Age | Mean ± Ss | 74.78 ± 13.92 | 73.85 ± 14.21 | 75.87 ± 13.54 | c 0.189 |
| Median (Min–Max) | 77 (27–98) | 77 (27–98) | 78 (32–98) | ||
| APAPCHE II | Mean ± Ss | 30.99 ± 6.43 | 28.75 ± 5.98 | 33.63 ± 5.94 | d 0.001 ** |
| Median (Min–Max) | 30 (15–51) | 28 (15–46) | 33 (18–51) | ||
| SOFA | Mean ± Ss | 9.17 ± 2.93 | 8.20 ± 2.70 | 10.32 ± 2.77 | d 0.001 ** |
| Median (Min–Max) | 9 (1–17) | 8 (2–17) | 10 (1–17) | ||
| Lactate | Mean ± Ss | 3.32 ± 2.54 | 2.89 ± 1.95 | 3.83 ± 3.01 | c 0.001 ** |
| Median (Min–Max) | 2.4 (1.4–24) | 2.3 (1.4–18) | 2.8 (1.6–24) | ||
| AST | Mean ± Ss | 96.57 ± 181.19 | 76.58 ± 154.54 | 120.08 ± 206.31 | c 0.011 * |
| Median (Min–Max) | 39 (6–1572) | 35 (8–1572) | 46.5 (6–1185) | ||
| ALT | Mean ± Ss | 55.36 ± 114.46 | 46.49 ± 96.48 | 65.8 ± 132.12 | c 0.252 |
| Median (Min–Max) | 19 (2–849) | 18 (2–753) | 20 (5–849) | ||
| Total bilirubin | Mean ± Ss | 1.60 ± 3.52 | 0.99 ± 1.53 | 2.30 ± 4.83 | c 0.001 ** |
| Median (Min–Max) | 0.7 (0.1–29.6) | 0.6 (0.1–14.4) | 0.8 (0.2–29.6) | ||
| Direct bilirubin | Mean ± Ss | 1.14 ± 2.92 | 0.63 ± 1.32 | 1.74 ± 3.98 | c 0.001 ** |
| Median (Min–Max) | 0.3 (0.1–22.4) | 0.3 (0.1–12.1) | 0.4 (0.1–22.4) | ||
| Urea | Mean ± Ss | 121.32 ± 76.9 | 110.57 ± 76.62 | 133.95 ± 75.55 | c 0.001 ** |
| Median (Min–Max) | 103 (14–494) | 90 (14–494) | 117.5 (19–404) | ||
| Creatinine | Mean ± Ss | 2.65 ± 1.86 | 2.59 ± 2.04 | 2.73 ± 1.63 | c 0.087 |
| Median (Min–Max) | 2.2 (0.3–10.8) | 2 (0.3–10.8) | 2.4 (0.3–7.7) | ||
| FIB-4 | Mean ± Ss | 6.49 ± 7.89 | 4.83 ± 6.37 | 8.44 ± 9.02 | c 0.001 ** |
| Median (Min–Max) | 3.8 (0.3–54.6) | 3.3 (0.3–54.6) | 5.3 (0.4–45) | ||
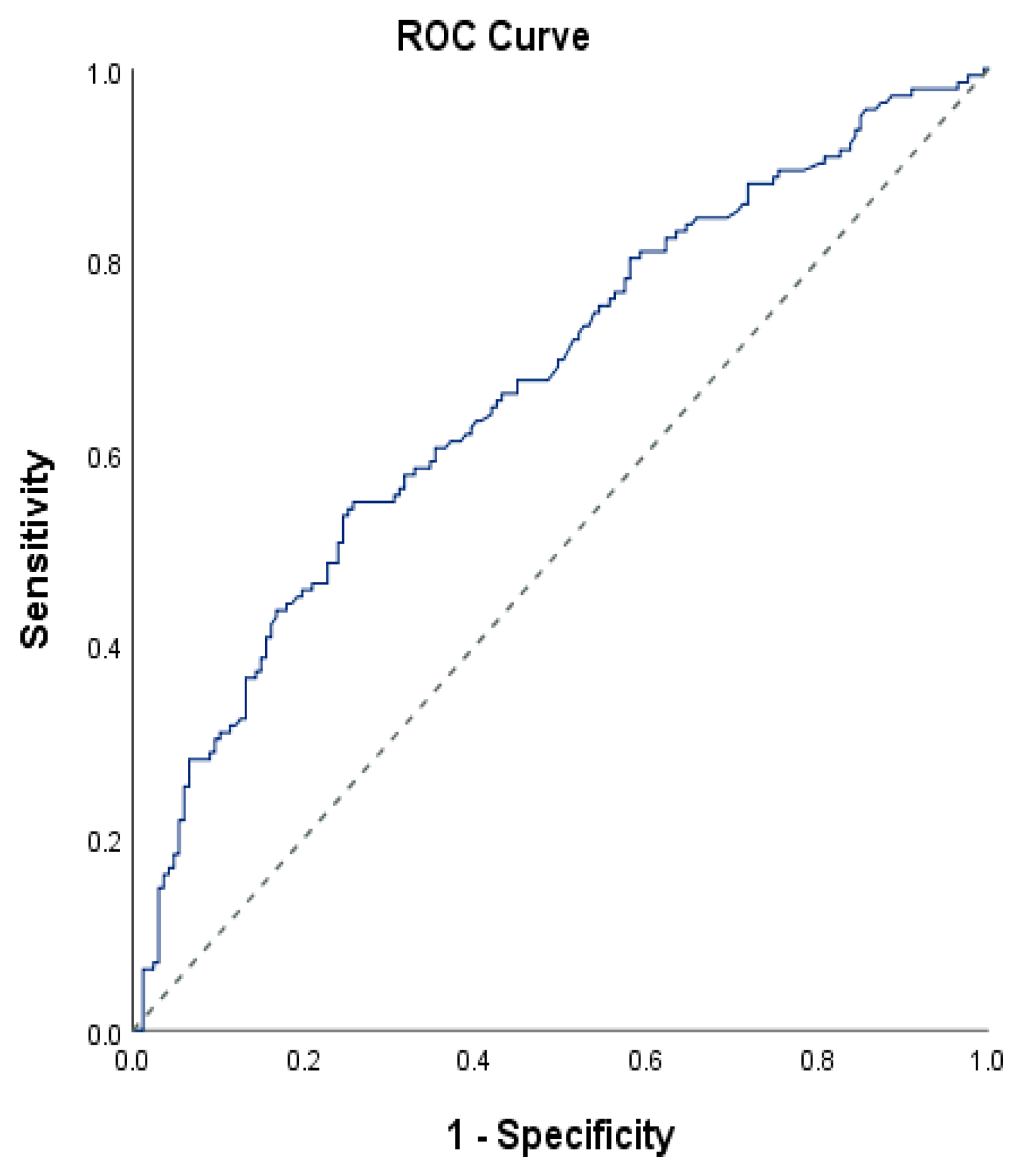
| Diagnostic Scan | ROC Curve | |||||||
|---|---|---|---|---|---|---|---|---|
| Cut-Off | Sensitivity | Specificity | Positive Predictive Value | Negative Predictive Value | Area | 95% Confidence Interval | p | |
| FIB-4 | 4.9 | 4.92 | 4.25 | 4.46 | 4.95 | 0.672 | 0.612–0.732 | 0.001 ** |
| 95% C.I.ODDS | ||||
|---|---|---|---|---|
| p | ODDS | Lower | Upper | |
| DM (+) | 0.105 | 0.606 | 0.331 | 1.110 |
| Septic shock (+) | 0.528 | 0.810 | 0.421 | 1.558 |
| Wound site infection | 0.058 | 0.258 | 0.064 | 1.045 |
| Lactate | 0.519 | 1.043 | 0.918 | 1.184 |
| AST | 0.775 | 1.000 | 0.998 | 1.001 |
| Total Bilirubin | 0.680 | 0.872 | 0.456 | 1.669 |
| Urea | 0.695 | 0.999 | 0.995 | 1.003 |
| APAPCHEII | 0.000 ** | 1.101 | 1.008 | 1.156 |
| SOFA | 0.037 * | 1.122 | 1.007 | 1.251 |
| Direct Bilirubin | 0.042 * | 1.228 | 1.080 | 1.497 |
| FIB-4 (≥4.9) | 0.006 ** | 2.127 | 1.237 | 3.659 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albayrak, T.; Yuksel, B. Prognostic Value of Fibrosis 4 (FIB-4) Index in Sepsis Patients. J. Pers. Med. 2024, 14, 531. https://doi.org/10.3390/jpm14050531
Albayrak T, Yuksel B. Prognostic Value of Fibrosis 4 (FIB-4) Index in Sepsis Patients. Journal of Personalized Medicine. 2024; 14(5):531. https://doi.org/10.3390/jpm14050531
Chicago/Turabian StyleAlbayrak, Tuna, and Beyza Yuksel. 2024. "Prognostic Value of Fibrosis 4 (FIB-4) Index in Sepsis Patients" Journal of Personalized Medicine 14, no. 5: 531. https://doi.org/10.3390/jpm14050531
APA StyleAlbayrak, T., & Yuksel, B. (2024). Prognostic Value of Fibrosis 4 (FIB-4) Index in Sepsis Patients. Journal of Personalized Medicine, 14(5), 531. https://doi.org/10.3390/jpm14050531






