Dendrimers, an Emerging Opportunity in Personalized Medicine?
Abstract
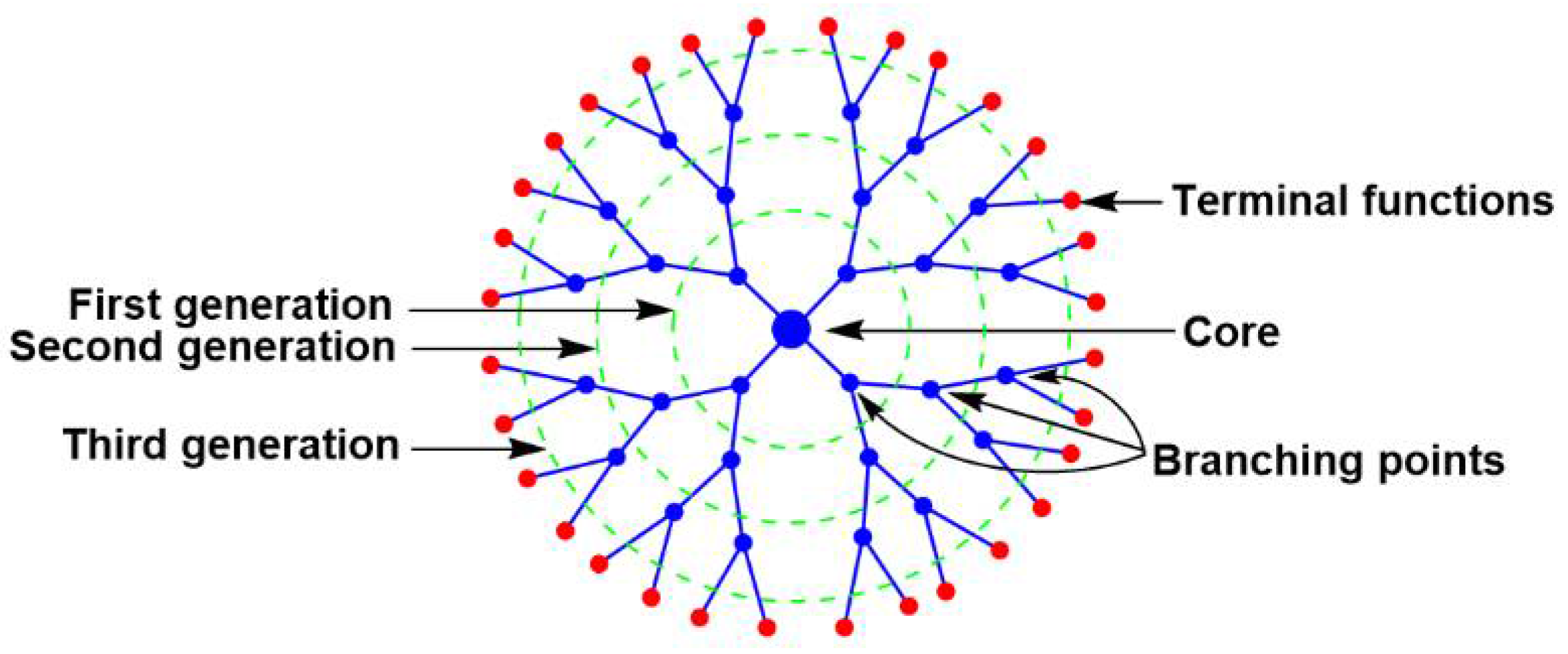
1. Introduction
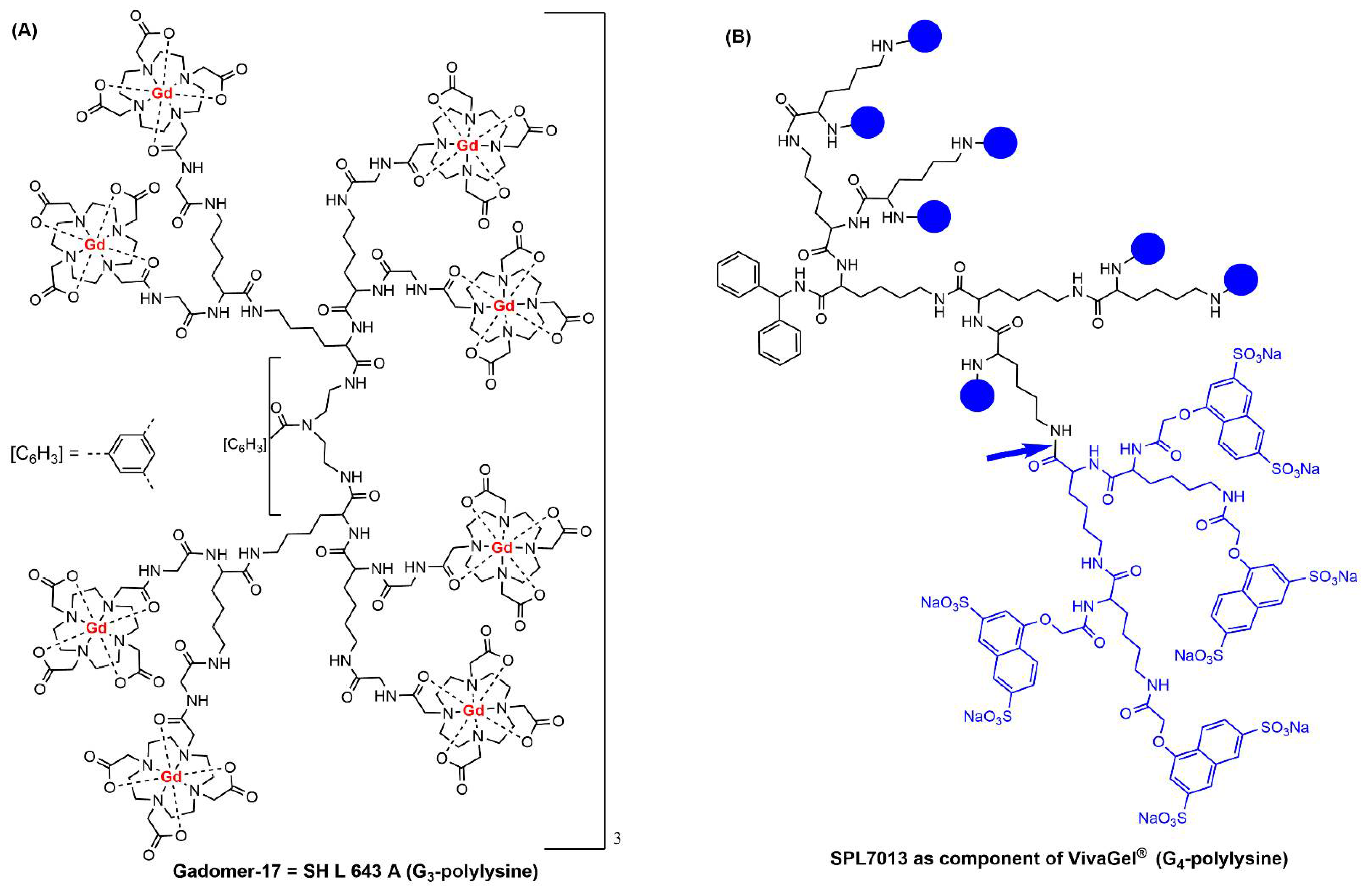
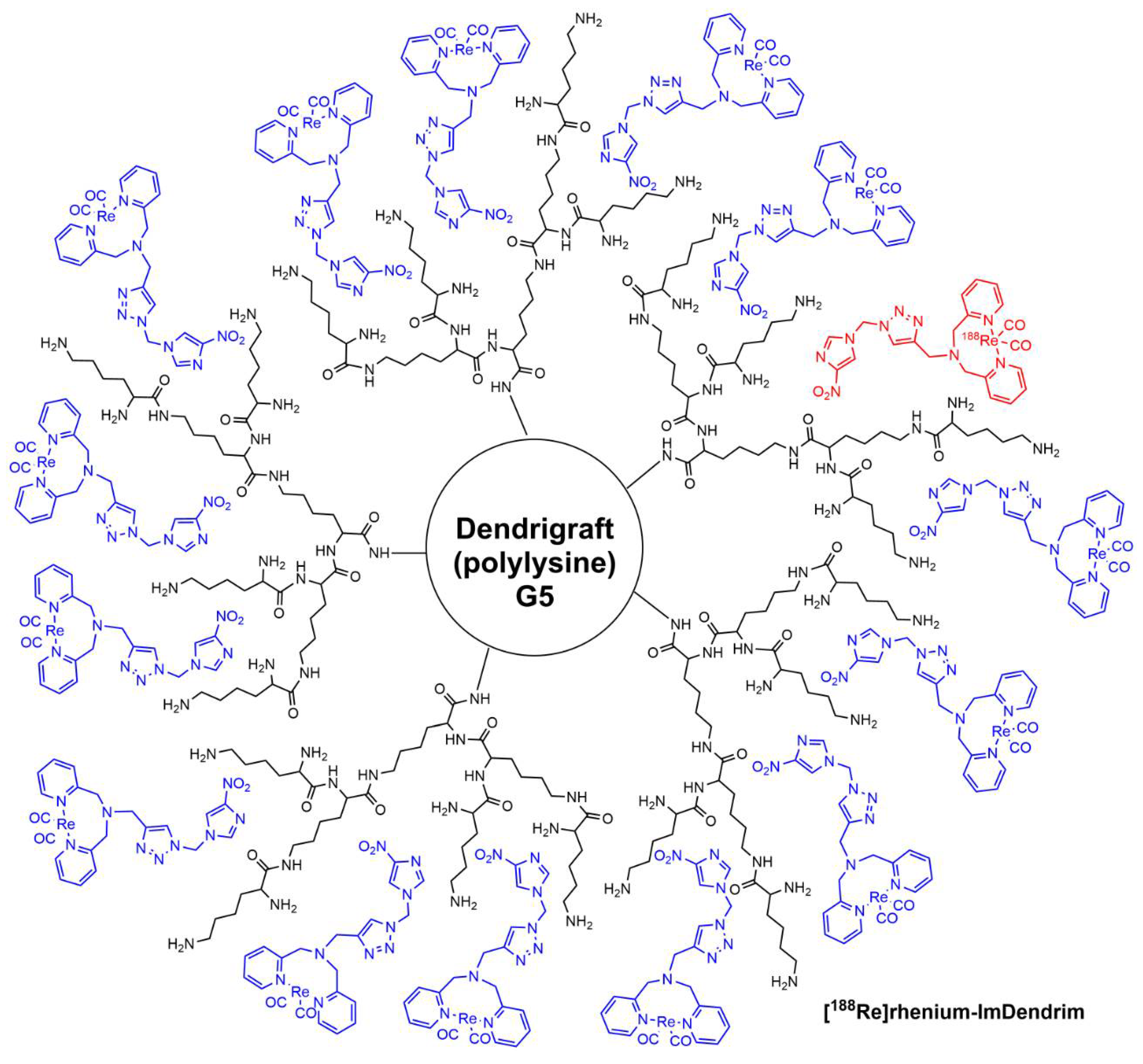
2. Clinical Trials with Dendrimers Based on Poly-L-Lysine
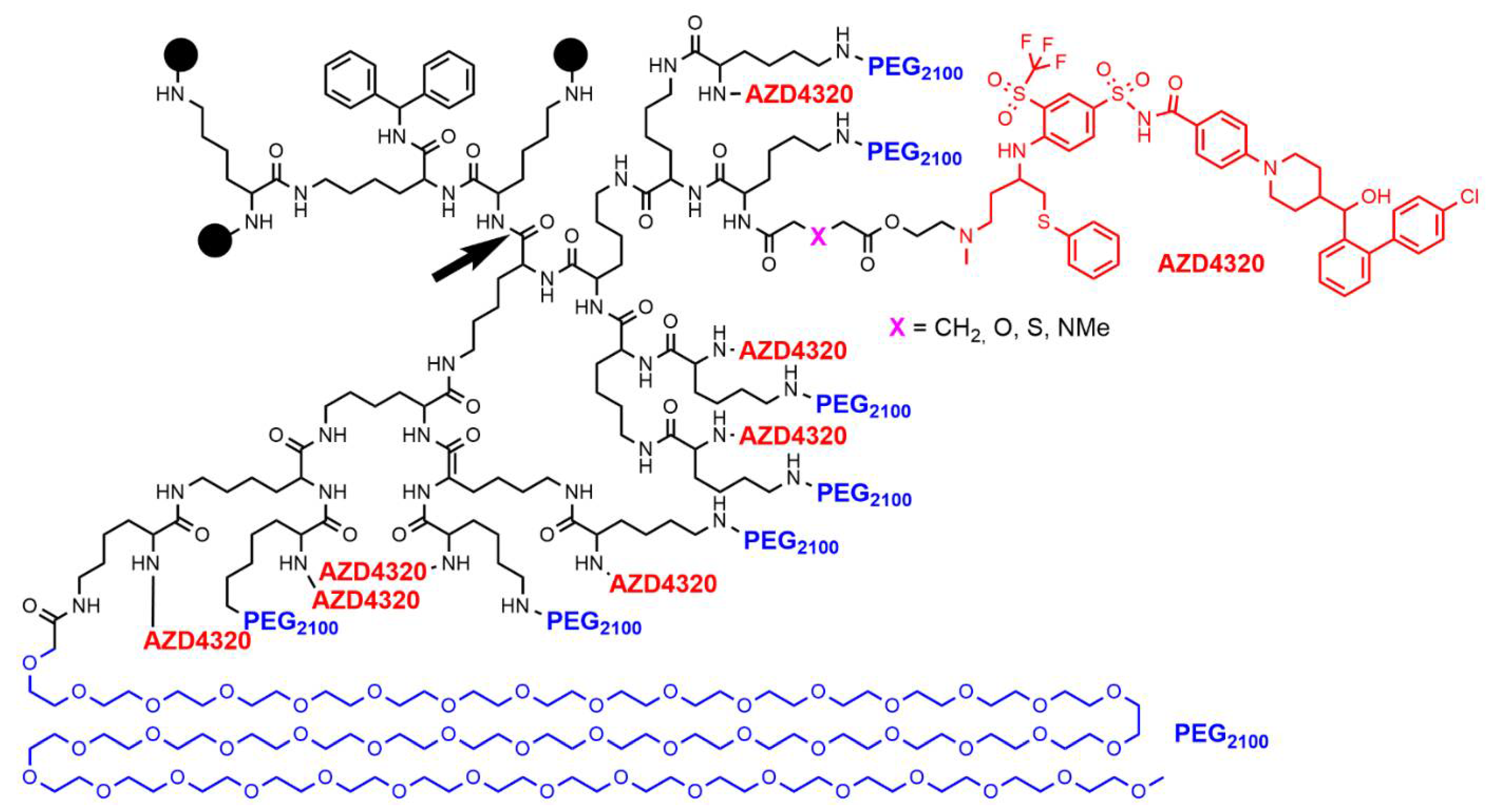
3. Clinical Trials with PAMAM Dendrimers
4. Clinical Trials with Other Types of Dendrimers
5. Discussion
6. Conclusions and Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Caminade, A.M.; Turrin, C.O.; Laurent, R.; Ouali, A.; DelavauxNicot, B. Dendrimers: Towards Catalytic, Material and Biomedical Uses; Wiley: Chichester, UK, 2011; pp. 1–538. [Google Scholar]
- Majoral, J.-P.; Zablocka, M.; Ciepluch, K.; Milowska, K.; Bryszewska, M.; Shcharbin, D.; Katir, N.; El Kadib, A.; Caminade, A.-M.; Mignani, S. Hybrid phosphorus-viologen dendrimers as new soft nanoparticles: Design and properties. Org. Chem. Front. 2021, 8, 4607–4622. [Google Scholar] [CrossRef]
- Roach, P.J.; Depaoli-Roach, A.A.; Hurley, T.D.; Tagliabracci, V.S. Glycogen and its metabolism: Some new developments and old themes. Biochem. J. 2012, 441, 763–787. [Google Scholar] [CrossRef]
- Balakshin, M.; Capanema, E.A.; Zhu, X.H.; Sulaeva, I.; Potthast, A.; Rosenau, T.; Rojas, O.J. Spruce milled wood lignin: Linear, branched or cross-linked? Green Chem. 2020, 22, 3985–4001. [Google Scholar] [CrossRef]
- Mekuria, S.L.; Ouyang, Z.; Song, C.; Rodrigues, J.; Shen, M.; Shi, X. Dendrimer-Based Nanogels for Cancer Nanomedicine Applications. Bioconjug. Chem. 2022, 33, 87–96. [Google Scholar] [CrossRef]
- Moorthy, H.; Govindaraju, T. Dendrimer Architectonics to Treat Cancer and Neurodegenerative Diseases with Implications in Theranostics and Personalized Medicine. ACS Appl. Bio Mater. 2021, 4, 1115–1139. [Google Scholar] [CrossRef]
- Mishra, V.; Yadav, N.; Saraogi, G.; Tambuwala, M.M.; Giri, N. Dendrimer based nanoarchitectures in diabetes management: An overview. Curr. Pharm. Des. 2019, 25, 2569–2583. [Google Scholar] [CrossRef]
- Patel, P.; Meghani, N.; Kansara, K.; Kumar, A. Nanotherapeutics for the Treatment of Cancer and Arthritis. Curr. Drug Metab. 2019, 20, 430–445. [Google Scholar] [CrossRef]
- Setrajcic-Tomic, A.J.; Popovic, J.K.; Vojnovic, M.; Dzambas, L.D.; Setrajcic, J.P. Review of core-multishell nanostructured models for nano-biomedical and nano-biopharmaceutical application. Bio-Med. Mater. Eng. 2018, 29, 451–471. [Google Scholar] [CrossRef]
- Xia, Y.; Matham, M.V.; Su, H.B.; Padmanabhan, P.; Gulyas, B. Nanoparticulate Contrast Agents for Multimodality Molecular Imaging. J. Biomed. Nanotechnol. 2016, 12, 1553–1584. [Google Scholar] [CrossRef]
- Cole, J.T.; Holland, N.B. Multifunctional nanoparticles for use in theranostic applications. Drug Deliv. Transl. Res. 2015, 5, 295–309. [Google Scholar] [CrossRef]
- Muthu, M.S.; Leong, D.T.; Mei, L.; Feng, S.S. Nanotheranostics—Application and Further Development of Nanomedicine Strategies for Advanced Theranostics. Theranostics 2014, 4, 660–677. [Google Scholar] [CrossRef] [PubMed]
- Svenson, S. The dendrimer paradox—High medical expectations but poor clinical translation. Chem. Soc. Rev. 2015, 44, 4131–4144. [Google Scholar] [CrossRef] [PubMed]
- Buhleier, E.; Wehner, F.; Vögtle, F. “Cascade-” and “Nonskid-chain-like” syntheses of molecular cavity topologies. Synthesis 1978, 78, 155–158. [Google Scholar] [CrossRef]
- Denkewalter, R.G.; Kolc, J.; Lukasavage, W.J. Macromolecular Highly Branched Homogeneous Compound Based on Lysine Units. US Patent 4,289,872, 15 September 1981. [Google Scholar]
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers—Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Newkome, G.R.; Yao, Z.Q.; Baker, G.R.; Gupta, V.K. Micelles. 1. Cascade molecules—A new approach to micelles—A [27]-arborol. J. Org. Chem. 1985, 50, 2003–2004. [Google Scholar] [CrossRef]
- Wooley, K.L.; Hawker, C.J.; Frechet JM, J. Hyperbranched macromolecules via a novel double-stage convergent growth approach. J. Am. Chem. Soc. 1991, 113, 4252–4261. [Google Scholar] [CrossRef]
- Zhou, L.L.; Roovers, J. Synthesis of novel carbosilane dendritic macromolecules. Macromolecules 1993, 26, 963–968. [Google Scholar] [CrossRef]
- de Brabander van den Berg, E.M.M.; Meijer, E.W. Poly(Propylene Imine) Dendrimers—Large-Scale Synthesis by Hetereogeneously Catalyzed Hydrogenations. Angew. Chem.-Int. Edit. Engl. 1993, 32, 1308–1311. [Google Scholar] [CrossRef]
- Launay, N.; Caminade, A.M.; Lahana, R.; Majoral, J.P. A general synthetic strategy for neutral phosphorus-containing dendrimers. Angew. Chem.-Int. Edit. Engl. 1994, 33, 1589–1592. [Google Scholar] [CrossRef]
- Clinical Trials USA. Available online: https://ClinicalTrials.gov (accessed on 2 August 2022).
- Clinical Trials EU. Available online: https://www.clinicaltrialsregister.eu (accessed on 2 August 2022).
- Runge, V.M.; Heverhagen, J.T. Advocating the Development of Next-Generation High-Relaxivity Gadolinium Chelates for Clinical Magnetic Resonance. Investig. Radiol. 2018, 53, 381–389. [Google Scholar] [CrossRef]
- Herborn, C.U.; Schmidt, M.; Bruder, O.; Nagel, E.; Shamsi, K.; Barkhausen, J. MR Coronary Angiography with SH L 643 A: Initial Experience in Patients with Coronary Artery Disease. Radiology 2004, 233, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Starpharma. Available online: https://starpharma.com (accessed on 2 August 2022).
- Holmes, W.R.; Maher, L.; Rosenthal, S.L. Attitudes of men in an Australian male tolerance study towards microbicide use. Sexual Health 2008, 5, 273–278. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, M.Y.; Millwood, I.Y.; Wand, H.; Poynten, M.; Law, M.; Kaldor, J.M.; Wesselingh, S.; Price, C.F.; Clark, L.J.; Paull, J.R.A.; et al. A Randomized Controlled Trial of the Safety of Candidate Microbicide SPL7013 Gel When Applied to the Penis. Jaids-J. Acquir. Immune Defic. Syndr. 2009, 50, 375–380. [Google Scholar] [CrossRef]
- O’Loughlin, J.; Millwood, I.Y.; McDonald, H.M.; Price, C.F.; Kaldor, J.M.; Paull, J.R.A. Safety, Tolerability, and Pharmacokinetics of SPL7013 Gel (VivaGel®): A Dose Ranging, Phase I Study. Sex. Transm. Dis. 2010, 37, 100–104. [Google Scholar] [CrossRef]
- Cohen, C.R.; Brown, J.; Moscicki, A.B.; Bukusi, E.A.; Paull, J.R.A.; Price, C.F.; Shiboski, S. A Phase I Randomized Placebo Controlled Trial of the Safety of 3% SPL7013 Gel (VivaGel®) in Healthy Young Women Administered Twice Daily for 14 Days. PLoS ONE 2011, 6, e16258. [Google Scholar] [CrossRef]
- Moscicki, A.B.; Kaul, R.; Ma, Y.; Scott, M.E.; Daud, I.I.; Bukusi, E.A.; Shiboski, S.; Rebbapragada, A.; Huibner, S.; Cohen, C.R. Measurement of Mucosal Biomarkers in a Phase 1 Trial of Intravaginal 3% StarPharma LTD 7013 Gel (VivaGel) to Assess Expanded Safety. J. Acquir. Immune Defic. Syndr. 2012, 59, 134–140. [Google Scholar] [CrossRef]
- McGowan, I.; Gomez, K.; Bruder, K.; Febo, I.; Chen, B.A.; Richardson, B.A.; Husnik, M.; Livant, E.; Price, C.; Jacobson, C.; et al. Phase 1 randomized trial of the vaginal safety and acceptability of SPL7013 gel (VivaGel) in sexually active young women (MTN-004). Aids 2011, 25, 1057–1064. [Google Scholar] [CrossRef]
- Carballo-Dieguez, A.; Giguere, R.; Dolezal, C.; Chen, B.A.; Kahn, J.; Zimet, G.; Mabragana, M.; Leu, C.S.; McGowan, I. ‘‘Tell Juliana’’: Acceptability of the Candidate Microbicide VivaGel® and Two Placebo Gels Among Ethnically Diverse, Sexually Active Young Women Participating in a Phase 1 Microbicide Study. AIDS Behav. 2012, 16, 1761–1774. [Google Scholar] [CrossRef]
- Price, C.F.; Tyssen, D.; Sonza, S.; Davie, A.; Evans, S.; Lewis, G.R.; Xia, S.; Spelman, T.; Hodsman, P.; Moench, T.R.; et al. SPL7013 Gel (VivaGel (R)) Retains Potent HIV-1 and HSV-2 Inhibitory Activity following Vaginal Administration in Humans. PLoS ONE 2011, 6, e24095. [Google Scholar] [CrossRef]
- Madan, R.P.; Dezzutti, C.S.; Rabe, L.; Hillier, S.L.; Marrazzo, J.; McGowan, I.; Richardson, B.A.; Herold, B.C.; Microbicide Trials Network, B.; Team, M.T.N.P. Soluble Immune Mediators and Vaginal Bacteria Impact Innate Genital Mucosal Antimicrobial Activity in Young Women. Am. J. Reprod. Immunol. 2015, 74, 323–332. [Google Scholar] [CrossRef]
- Waldbaum, A.S.; Schwebke, J.R.; Paull, J.R.A.; Price, C.F.; Edmondson, S.R.; Castellarnau, A.; McCloud, P.; Kinghorn, G.R. A phase 2, double-blind, multicenter, randomized, placebo-controlled, dose-ranging study of the efficacy and safety of Astodrimer Gel for the treatment of bacterial vaginosis. PLoS ONE 2020, 15, e0232394. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.D.; Karellas, P.; Henderson, S.A.; Giannis, M.; O’Keefe, D.F.; Heery, G.; Paull, J.R.A.; Matthews, B.R.; Holan, G. Dendrimers as drugs: Discovery and preclinical and clinical development of dendrimer-based microbicides for HIV and STI prevention. Mol. Pharm. 2005, 2, 312–318. [Google Scholar] [CrossRef]
- Chavoustie, S.E.; Carter, B.A.; Waldbaum, A.S.; Donders, G.G.G.; Peters, K.H.; Schwebke, J.R.; Paull, J.R.A.; Price, C.F.; Castellarnau, A.; McCloud, P.; et al. Two phase 3, double-blind, placebo-controlled studies of the efficacy and safety of Astodrimer 1% Gel for the treatment of bacterial vaginosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 245, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Schwebke, J.R.; Carter, B.A.; Waldbaum, A.S.; Agnew, K.J.; Paull, J.R.A.; Price, C.F.; Castellarnau, A.; McCloud, P.; Kinghorn, G.R. A phase 3, randomized, controlled trial of Astodrimer 1% Gel for preventing recurrent bacterial vaginosis. Eur. J. Obstetrics Gynecol. Reprod. Biol. X 2021, 10, 100121. [Google Scholar] [CrossRef] [PubMed]
- Mendling, W.; Holzgreve, W. Astodrimer sodium and bacterial vaginosis: A mini review. Arch. Gynecol. Obstet. 2022, 306, 101–108. [Google Scholar] [CrossRef]
- VIRALEZE. Available online: https://www.starpharma.com/viraleze/spl7013 (accessed on 2 August 2022).
- Castellarnau, A.; Heery, G.P.; Seta, A.; Luscombe, C.A.; Kinghorn, G.R.; Button, P.; McCloud, P.; Paull, J.R.A. Astodrimer sodium antiviral nasal spray for reducing respiratory infections is safe and well tolerated in a randomized controlled trial. Sci. Rep. 2022, 12, 10210. [Google Scholar] [CrossRef]
- Patterson, C.M.; Balachander, S.B.; Grant, I.; Pop-Damkov, P.; Kelly, B.; McCoull, W.; Parker, J.; Giannis, M.; Hill, K.J.; Gibbons, F.D.; et al. Design and optimisation of dendrimer-conjugated Bcl-2/xL inhibitor, AZD0466, with improved therapeutic index for cancer therapy. Commun. Biol. 2021, 4, 112. [Google Scholar] [CrossRef]
- Arulananda, S.; O’Brien, M.; Evangelista, M.; Jenkins, L.J.; Poh, A.R.; Walkiewicz, M.; Leong, T.; Mariadason, J.M.; Cebon, J.; Balachander, S.B.; et al. A novel BH3-mimetic, AZD0466, targeting BCL-XL and BCL-2 is effective in pre-clinical models of malignant pleural mesothelioma. Cell Death Discov. 2021, 7, 122. [Google Scholar] [CrossRef]
- Feeney, O.M.; Ardipradja, K.; Noi, K.F.; Mehta, D.; De Rose, R.; Yuen, D.; Johnston, A.P.R.; Kingston, L.; Ericsson, C.; Elmore, C.S.; et al. Subcutaneous delivery of a dendrimer-BH3 mimetic improves lymphatic uptake and survival in lymphoma. J. Control. Release 2022, 348, 420–430. [Google Scholar] [CrossRef]
- DEP® Docetaxel, DEP® Cabazitaxel, and DEP® Irinotecan. Available online: https://starpharma.com/drug_delivery (accessed on 1 August 2022).
- ColCom. Available online: https://www.colcom.eu/ (accessed on 2 August 2022).
- Yang, G.; Sadeg, N.; Belhadj-Tahar, H. New potential in situ anticancer agent derived from [188Re]rhenium nitro-imidazole ligand loaded 5th generation poly-L-lysine dendrimer for treatment of transplanted human liver carcinoma in nude mice. Drug Des. 2017, 6, 1000144. [Google Scholar] [CrossRef]
- Ashvattha Therapeutics. Available online: https://avttx.com/ (accessed on 2 August 2022).
- Sharma, R.; Sharma, A.; Kambhampati, S.P.; Reddy, R.R.; Zhang, Z.; Cleland, J.L.; Kannan, S.; Kannan, R.M. Scalable synthesis and validation of PAMAM dendrimer-N-acetyl cysteine conjugate for potential translation. Bioeng. Transl. Med. 2018, 3, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Gusdon, A.M.; Faraday, N.; Aita, J.S.; Kumar, S.; Mehta, I.; Choi, H.A.; Cleland, J.L.; Robinson, K.; McCullough, L.D.; Ng, D.K.; et al. Dendrimer nanotherapy for severe COVID-19 attenuates inflammation and neurological injury markers and improves outcomes in a phase2a clinical trial. Sci. Transl. Med. 2022, 14, eabo2652. [Google Scholar] [CrossRef] [PubMed]
- Kambhampati, S.P.; Bhutto, I.A.; Wu, T.; Ho, K.; McLeod, D.S.; Lutty, G.A.; Kannan, R.M. Systemic dendrimer nanotherapies for targeted suppression of choroidal inflammation and neovascularization in age-related macular degeneration. J. Control. Release 2021, 335, 527–540. [Google Scholar] [CrossRef]
- Cleland, J.; Sharma, R.; Appiani, S. Dendrimer Compositions and Methods for Drug Delivery to the Eye. WO 2021/113662, 10 June 2021. [Google Scholar]
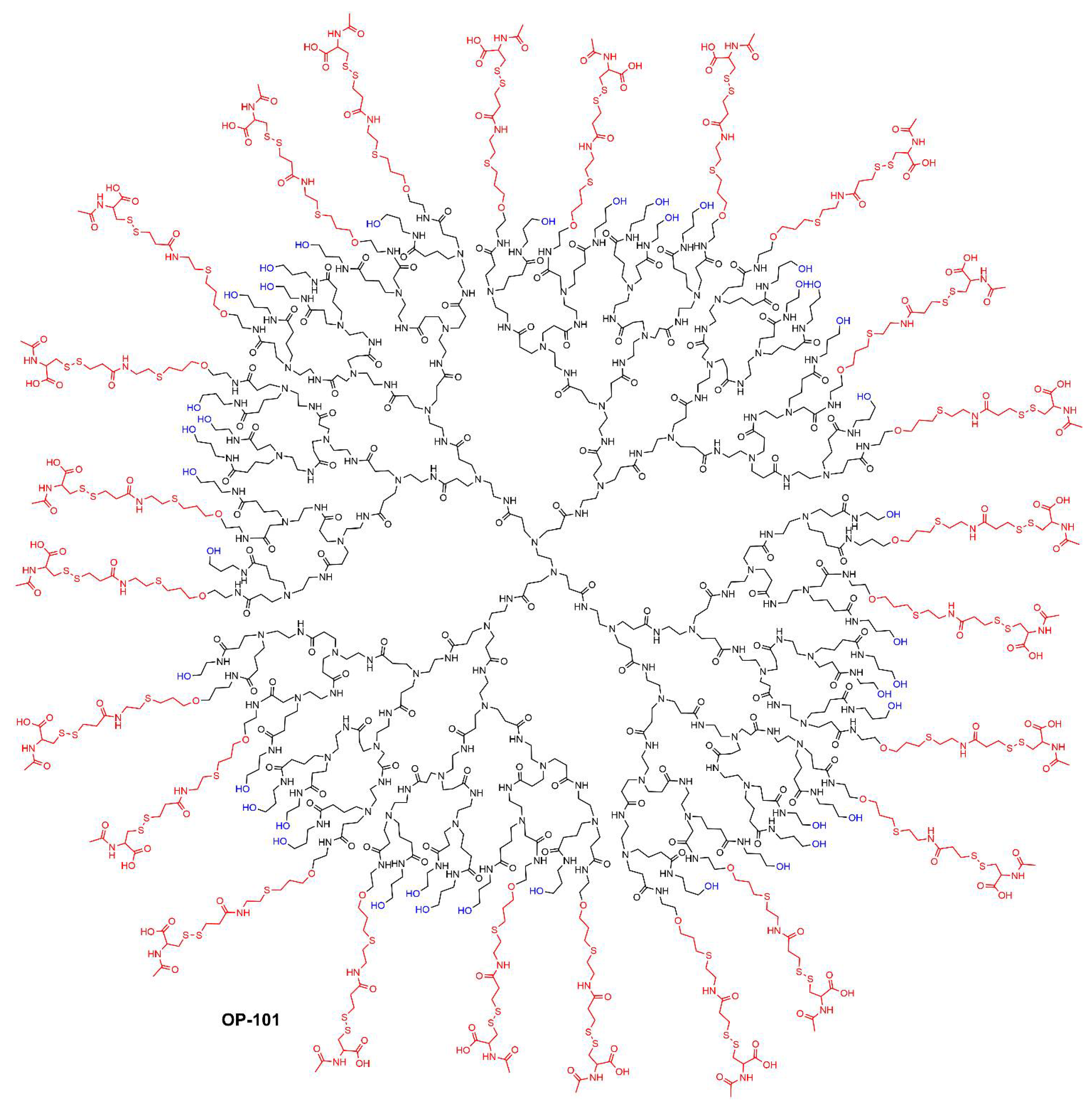
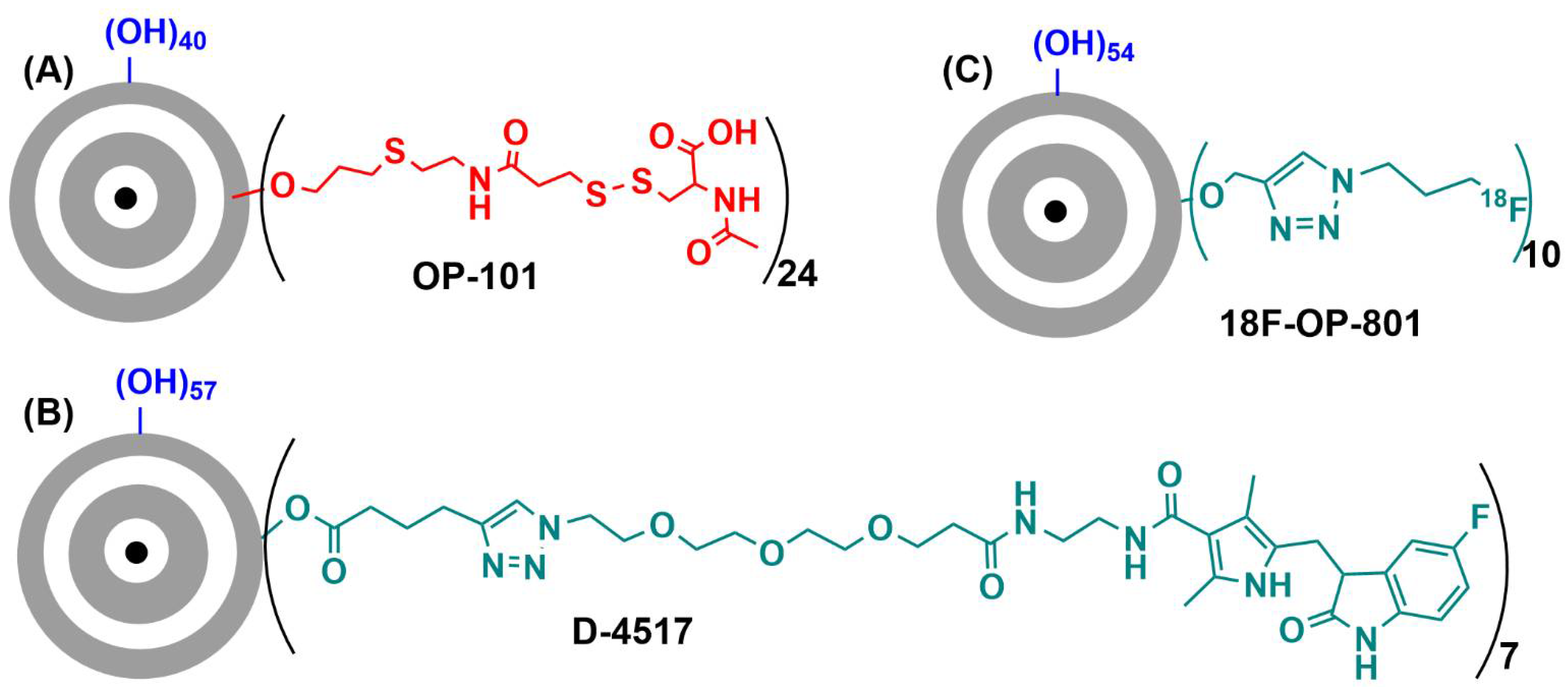
- Cleland, J.L.; Sharma, R.; Sun, M.; Appiani La Rosa, S. Radiolabeled ether dendrimer conjugates for PET imaging and radiotherapy. WO 2022/094327, 5 May 2022. [Google Scholar]
- Khaitov, M.R.; Shilovskii, I.P.; Kozhikhova, K.V.; Kofiadi, I.A.; Smirnov, V.V.; Koloskova, O.O.; Sergeev, I.V.; Trofimov, D.Y.; Trukhin, V.P.; Skvortsova, V.I. Combination Antiviral Formulation against SARS-CoV-2 Comprising SARS-CoV-2 Genome-Targeting siRNAs and Transfection-Enhancing Cationic Peptide Dendrimer. RU2746362 C1 2021-04-12, 11 March 2021. [Google Scholar]
- Khaitov, M.; Nikonova, A.; Shilovskiy, I.; Kozhikhova, K.; Kofiadi, I.; Vishnyakova, L.; Nikolskii, A.; Gattinger, P.; Kovchina, V.; Barvinskaia, E.; et al. Silencing of SARS-CoV-2 with modified siRNA-peptide dendrimer formulation. Allergy 2021, 76, 2840–2854. [Google Scholar] [CrossRef]
- Majoral, J.P.; Francois, J.M.; Fabre, R.; Senescau, A.; Mignani, S.; Caminade, A.M. Multiplexing technology for in vitro diagnosis of pathogens: The key contribution of phosphorus dendrimers. Sci. China-Mater. 2018, 61, 1454–1461. [Google Scholar] [CrossRef]
- Siemens-Healthineers. Available online: https://www.siemens-healthineers.com/cardiac/cardiac-systems/stratus-cs-acute-care (accessed on 2 August 2022).
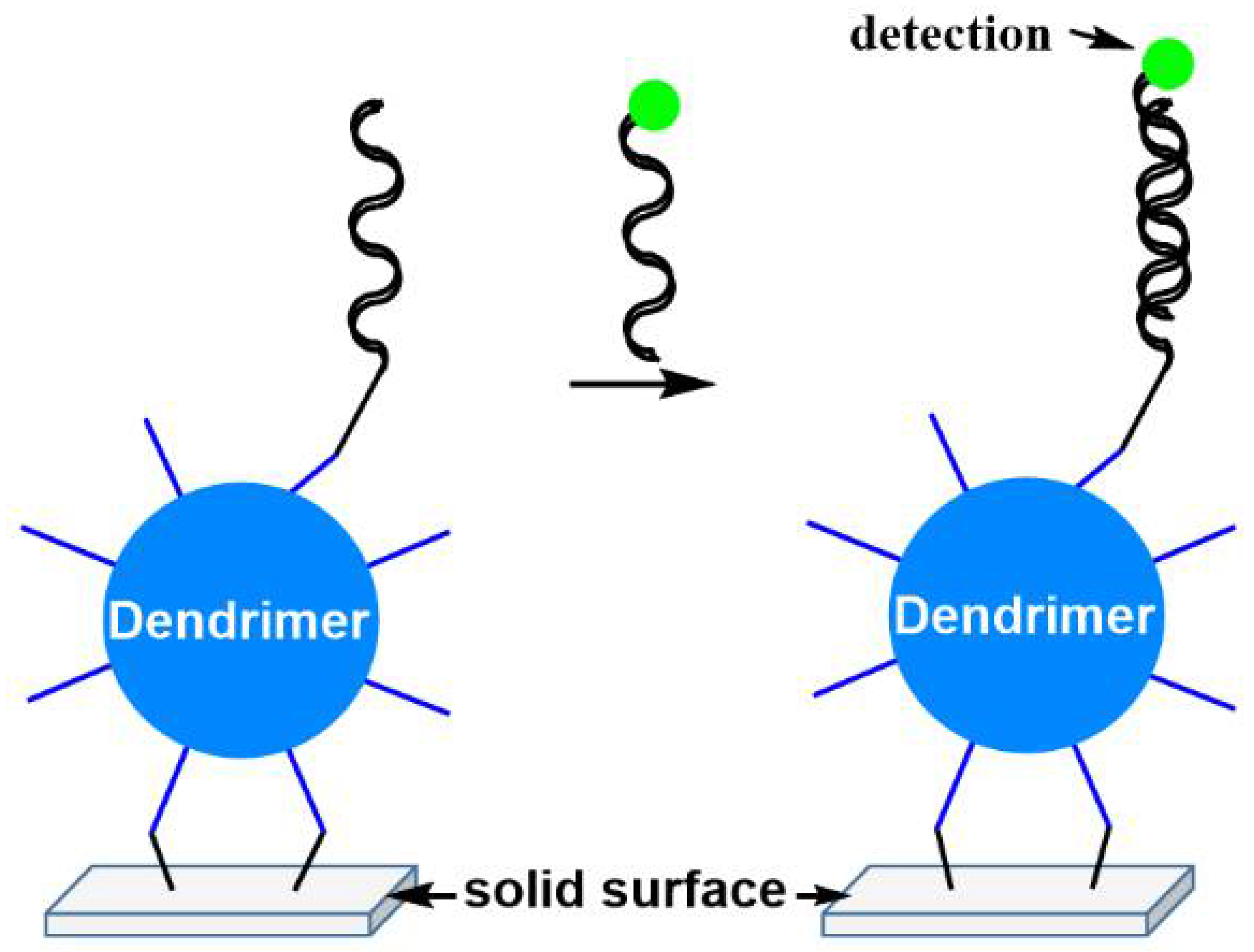
- Trevisiol, E.; Le Berre-Anton, V.; Leclaire, J.; Pratviel, G.; Caminade, A.M.; Majoral, J.P.; Francois, J.M.; Meunier, B. Dendrislides, dendrichips: A simple chemical functionalization of glass slides with phosphorus dendrimers as an effective means for the preparation of biochips. N. J. Chem. 2003, 27, 1713–1719. [Google Scholar] [CrossRef]
- Dendris. Available online: https://dendris.fr/medical-diagnostic/ (accessed on 2 August 2022).
- Bernard, E.; Peyret, T.; Plinet, M.; Contie, Y.; Cazaudarré, T.; Rouquet, Y.; Bernier, M.; Pesant, S.; Fabre, R.; Anton, A.; et al. The DendrisCHIP® Technology as a New, Rapid and Reliable Molecular Method for the Diagnosis of Osteoarticular Infections. Diagnostics 2022, 12, 1353. [Google Scholar] [CrossRef]
- Caminade, A.-M.; Turrin, C.-O.; Poupot, R. Curing inflammatory diseases using phosphorous dendrimers. Wiley Interdiscip. Rev.-Nanomed. Nanobiotechnol. 2022, e1783. [Google Scholar] [CrossRef]
- IMD-Pharma. Available online: http://www.imd-pharma.com/ (accessed on 2 August 2022).

| Study Number | Date Posted | Phase | Aims |
|---|---|---|---|
| NCT00331032 | 29 May 2006 | 1 | Safety and tolerability (female) |
| NCT00370357 | 31 August 2006 | 1 | Safety (male) |
| NCT00442910 | 5 March 2007 | 1 | Safety and acceptability (female) |
| NCT00490152 | 22 June 2007 | 1 | Adherence, acceptability (female) |
| NCT00740584 | 25 August 2008 | 1/2 | Retention and duration of activity (female) |
| NCT01201057 | 14 September 2010 | 2 | Efficacy against BV 1 (female) |
| NCT01437722 | 21 September 2011 | 2 | Prevention of recurrence of BV 1 (female) |
| NCT01577238 | 13 April 2012 | 3 | Treatment of BV 1 (female) |
| NCT01577537 | 16 April 2012 | 3 | Treatment of BV 1 (female) |
| 2012-000752-33 | 22 June 2012 | 3 | Treatment of BV 1 (female) |
| NCT02236156 | 10 September 2014 | 3 | Prevention of recurrence of BV 1 (female) |
| NCT02237950 | 12 September 2014 | 3 | Prevention of recurrence of BV 1 (female) |
| 2014-000694-39 | 21 October 2014 | 3 | Prevention of recurrence of BV 1 (female) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caminade, A.-M. Dendrimers, an Emerging Opportunity in Personalized Medicine? J. Pers. Med. 2022, 12, 1334. https://doi.org/10.3390/jpm12081334
Caminade A-M. Dendrimers, an Emerging Opportunity in Personalized Medicine? Journal of Personalized Medicine. 2022; 12(8):1334. https://doi.org/10.3390/jpm12081334
Chicago/Turabian StyleCaminade, Anne-Marie. 2022. "Dendrimers, an Emerging Opportunity in Personalized Medicine?" Journal of Personalized Medicine 12, no. 8: 1334. https://doi.org/10.3390/jpm12081334
APA StyleCaminade, A.-M. (2022). Dendrimers, an Emerging Opportunity in Personalized Medicine? Journal of Personalized Medicine, 12(8), 1334. https://doi.org/10.3390/jpm12081334






