Fetal Doppler Evaluation to Predict NEC Development
Abstract
:1. Introduction
2. Materials and Methods
- -
- -
- At delivery: GA; mode of delivery; reason for cesarean section; newborn’s sex and BW; Apgar score at 5 and 10 min; need for major or minor neonatal resuscitation;
- -
- On NICU admission: neonatal C-reactive protein, white blood cells (WBC) count, serum pH, hemoglobin, and platelet count.
3. Results
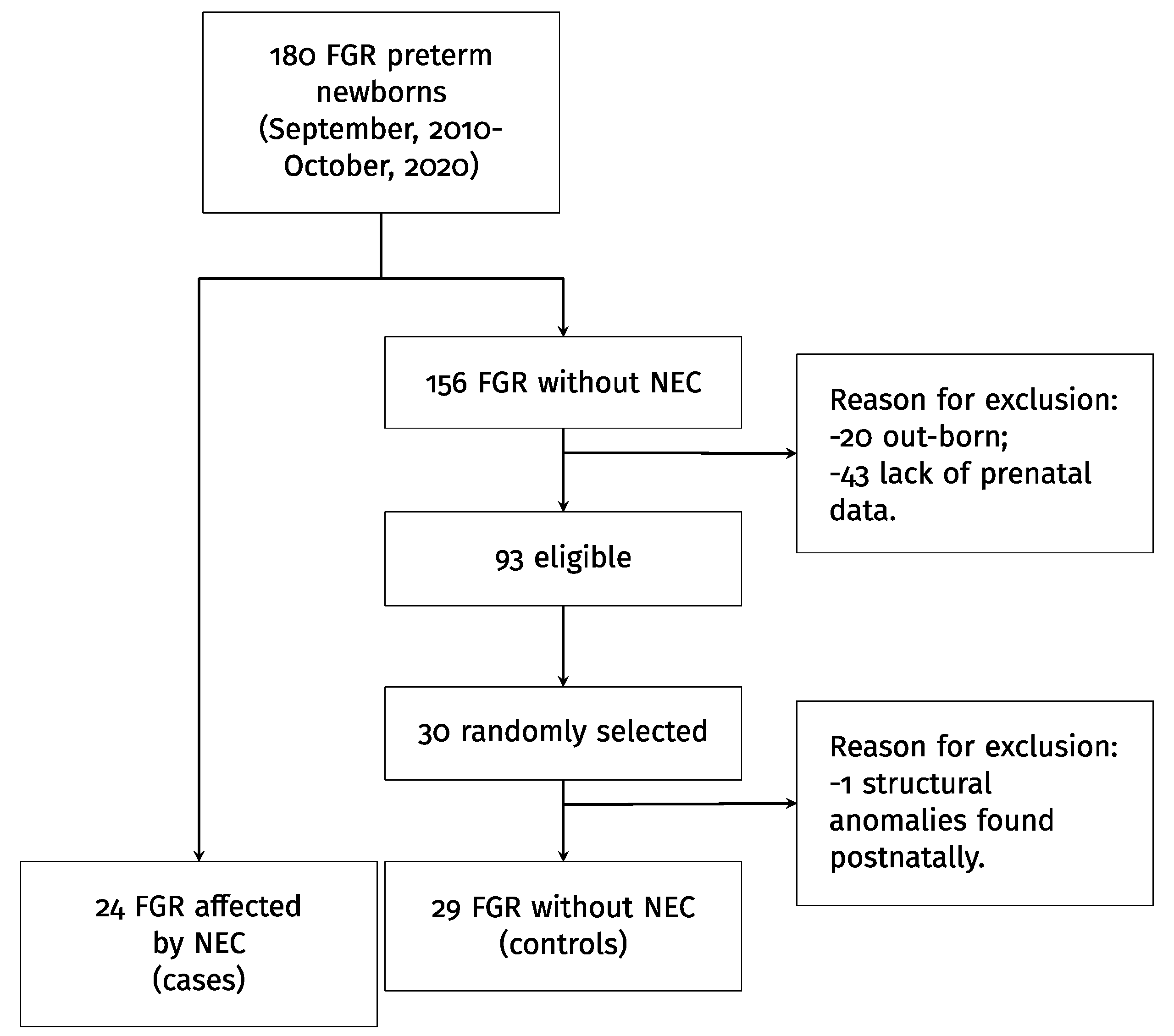
3.1. Population
3.2. Prenatal NEC Predictors
3.3. Postnatal NEC Predictors
4. Discussion
4.1. Principal Findings and Results
4.2. Clinical and Research Implications
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sacchi, C.; Marino, C.; Nosarti, C.; Vieno, A.; Visentin, S.; Simonelli, A. Association of intrauterine growth restriction and small for gestational age status with childhood cognitive outcomes: A systematic review and meta-analysis. JAMA Pediatr. 2020, 174, 772–781. [Google Scholar] [CrossRef] [PubMed]
- Lees, C.; Stampalija, T.; Baschat, A.; da Silva Costa, F.; Ferrazzi, E.; Figueras, F.; Hecher, K.; Kingdom, J.; Poon, L.C.; Salomon, L.J.; et al. ISUOG Practice guidelines: Diagnosis and management of small-for-gestational-age fetus and fetal growth restriction. Ultrasound Obstet. Gynecol. 2020, 56, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Lees, C.; Marlow, N.; Arabin, B.; Bilardo, C.M.; Brezinka, C.; Derks, J.B.; Duvekot, J.; Frusca, T.; Diemert, A.; TRUFFLE Group; et al. Perinatal morbidity and mortality in early-onset fetal growth restriction: Cohort outcomes of the trial of randomized umbilical and fetal flow in Europe (TRUFFLE). Ultrasound Obstet. Gynecol. 2013, 42, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Seravalli, V.; Miller, J.L.; Block-Abraham, D.; Baschat, A.A. Ductus venosus Doppler in the assessment of fetal cardiovascular health: An updated practical approach. Acta Obstet. Gynecol. Scand. 2016, 95, 635–644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanner, S.M.; Berryhill, T.F.; Ellenburg, J.L.; Jilling, T.; Cleveland, D.S.; Lorenz, R.G.; Martin, C.A. Pathogenesis of necrotizing enterocolitis: Modeling the innate immune response. Am. J. Pathol. 2015, 185, 4–16. [Google Scholar] [CrossRef] [Green Version]
- Bazacliu, C.; Neu, J. Necrotizing enterocolitis: Long term complications. Curr. Pediatr. Rev. 2019, 15, 115–124. [Google Scholar] [CrossRef]
- Manogura, A.C.; Turan, O.; Kush, M.L.; Berg, C.; Bhide, A.; Turan, S.; Moyano, D.; Bower, S.; Nicolaides, K.H.; Galan, H.L.; et al. Predictors of necrotizing enterocolitis in preterm growth-restricted neonates. Am. J. Obstet. Gynecol. 2008, 198, 638.e1–638.e6385. [Google Scholar] [CrossRef]
- Baschat, A.A.; Gembruch, U.; Reiss, I.; Gortner, L.; Weiner, C.P.; Harman, C.R. Relationship between arterial and venous Doppler and perinatal outcome in fetal growth restriction. Ultrasound Obstet. Gynecol. 2000, 16, 407–413. [Google Scholar] [CrossRef]
- Gordijn, S.J.; Beune, I.M.; Thilaganathan, B.; Papageorghiou, A.; Baschat, A.A.; Baker, P.N.; Silver, R.M.; Wynia, K.; Ganzevoort, W. Consensus definition of fetal growth restriction: A Delphi procedure. Ultrasound Obstet. Gynecol. 2016, 48, 333–339. [Google Scholar] [CrossRef]
- Hadlock, F.P.; Harrist, R.B.; Sharman, R.S.; Deter, R.L.; Park, S.K. Estimation of fetal weight with the use of head, body, and femur measurements—A prospective study. Am. J. Obstet. Gynecol. 1985, 151, 333–337. [Google Scholar] [CrossRef]
- Villar, J.; Giuliani, F.; Fenton, T.R.; Ohuma, E.O.; Cheikh Ismail, L.; Kennedy, S.H.; INTERGROWTH-21st Consortium. INTERGROWTH-21st very preterm size at birth reference charts. Lancet 2016, 387, 844–845, reprinted in Lancet 2016, 387, 944. [Google Scholar] [CrossRef] [Green Version]
- Fenton, T.R. A new growth chart for preterm babies: Babson and Benda’s chart updated with recent data and a new format. BMC Pediatr. 2003, 3, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American College of Obstetricians and Gynecologists. Gestational hypertension and preeclampsia: ACOG practice bulletin summary, number 222. Obstet. Gynecol. 2020, 135, 1492–1495. [Google Scholar] [CrossRef] [PubMed]
- Benhalima, K.; Mathieu, C.; van Assche, A.; Damm, P.; Devlieger, R.; Mahmood, T.; Dunne, F. Survey by the European Board and College of Obstetrics and Gynaecology on screening for gestational diabetes in Europe. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 201, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Tita, A.T.; Andrews, W.W. Diagnosis and management of clinical chorioamnionitis. Clin. Perinatol. 2010, 37, 339–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khong, T.Y.; Mooney, E.E.; Ariel, I.; Balmus, N.C.M.; Boyd, T.K.; Brundler, M.A.; Derricott, H.; Evans, M.J.; Faye-Petersen, O.M.; Gillan, J.E.; et al. Sampling and definitions of placental lesions: Amsterdam Placental Workshop Group Consensus Statement. Arch. Pathol. Lab. Med. 2016, 140, 698–713. [Google Scholar] [CrossRef] [Green Version]
- Siegler, Y.; Weiner, Z.; Solt, I. Prelabor Rupture of Membranes: ACOG Practice Bulletin, Number 217. Obstet Gynecol. 2020, 135, e80–e97. [Google Scholar] [CrossRef]
- Duci, M.; Fascetti-Leon, F.; Erculiani, M.; Priante, E.; Cavicchiolo, M.E.; Verlato, G.; Gamba, P. Neonatal independent predictors of severe NEC. Pediatr. Surg. Int. 2018, 34, 663–669. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; L. Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Hall, N.J.; Eaton, S.; Pierro, A. Royal Australasia of Surgeons Guest Lecture. Necrotizing enterocolitis: Prevention, treatment, and outcome. J. Pediatr. Surg. 2013, 48, 2359–2367. [Google Scholar] [CrossRef]
- Eaton, S.; Rees, C.M.; Hall, N.J. Current Research on the epidemiology, pathogenesis, and management of necrotizing enterocolitis. Neonatology 2017, 111, 423–430. [Google Scholar] [CrossRef]
- Duci, M.; Frigo, A.C.; Visentin, S.; Verlato, G.; Gamba, P.; Fascetti-Leon, F. Maternal and placental risk factors associated with the development of necrotizing enterocolitis (NEC) and its severity. J. Pediatr. Surg. 2019, 54, 2099–2102. [Google Scholar] [CrossRef] [PubMed]
- Westby Eger, S.H.; Kessler, J.; Kiserud, T.; Markestad, T.; Sommerfelt, K. Foetal Doppler abnormality is associated with increased risk of sepsis and necrotising enterocolitis in preterm infants. Acta Paediatr. 2015, 104, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Raboisson, M.-J.; Huissoud, C.; Lapointe, A.; Hugues, N.; Bigras, J.-L.; Brassard, M.; Lamoureux, J.; Fouron, J.-C. Assessment of uterine artery and aortic isthmus Doppler recordings as predictors of necrotizing enterocolitis. Am. J. Obstet. Gynecol. 2012, 206, 232.e1–232.e2326. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J.A.; Young, L.; Mc Guire, W. Pathogenesis and prevention of necrotizing enterocolitis. Curr. Opin. Infect. Dis. 2011, 24, 183–189. [Google Scholar] [CrossRef]
- Bhatt, A.B.; Tank, P.D.; Barmade, K.B.; Damania, K.R. Abnormal Doppler flow velocimetry in the growth restricted foetus as a predictor for necrotising enterocolitis. J. Postgrad. Med. 2002, 48, 182–185. [Google Scholar]
- Petrosyan, M.; Guner, Y.S.; Williams, M.; Grishin, A.; Ford, H.R. Current concepts regarding the pathogenesis of necrotizing enterocolitis. Pediatr. Surg. Int. 2009, 25, 309–318. [Google Scholar] [CrossRef]
- Downard, C.D.; Grant, S.N.; Matheson, P.J.; Guillaume, A.W.; Debski, R.; Fallat, M.E.; Garrison, R.N. Altered intestinal microcirculation is the critical event in the development of necrotizing enterocolitis. J. Pediatr. Surg. 2011, 46, 1023–1028. [Google Scholar] [CrossRef]
- Guang, Y.; Ying, D.; Sheng, Y.; Yiyong, F.; Jun, W.; Shuqiang, G.; Rong, J. Early Doppler ultrasound in the superior mesenteric artery and the prediction of necrotizing enterocolitis in preterm neonates. J. Ultrasound Med. 2019, 38, 3283–3289. [Google Scholar] [CrossRef]
- Christensen, R.D.; Yoder, B.A.; Baer, V.L.; Snow, G.L.; Butler, A. Early-onset neutropenia in small-for-gestational-age infants. Pediatrics 2015, 136, e1259–e1267. [Google Scholar] [CrossRef] [Green Version]
| Controls (29) | NEC (24) | p | |
|---|---|---|---|
| Maternal characteristics and pregnancy management | |||
| Maternal age (years) | 34.00 (32.00–36.00) | 36.50 (31.75–39.00) | 0.299 |
| Nulliparity | 41.38% (12/29) | 58.33% (14/24) | 0.219 |
| Medically assisted procreation | 13.79% (4/29) | 16.67% (4/24) | 0.771 |
| RDS prophylaxis | 62.07% (18/29) | 75.00% (18/24) | 0.315 |
| Delivery by CS | 96.55% (28/29) | 87.50% (21/24) | 0.214 |
| Pregnancy and fetal characteristics | |||
| Pre-eclampsia | 31.03% (9/29) | 41.67% (10/24) | 0.422 |
| Gestational diabetes | 3.45% (1/29) | 4.17% (1/24) | 0.891 |
| Premature preterm rupture of membranes | 13.79% (4/29) | 12.50% (3/24) | 0.890 |
| Chorioamnionitis | 6.90% (2/29) | 8.33% (2/24) | 0.844 |
| Maternal Doppler | |||
| Mean uterine arteries PI > 95th percentile | 41.38% (12/29) | 62.50% (15/24) | 0.126 |
| Bilateral uterine arteries notching | 24.14% (7/29) | 45.83% (11/24) | 0.097 |
| Fetal Doppler | |||
| MCA PI< 5th percentile | 41.38% (12/29) | 54.17% (13/24) | 0.353 |
| UA AREDF | 17.24% (5/29) | 41.67% (10/24) | <0.05 |
| DV PI > 95th percentile | 10.34% (3/29) | 41.67% (10/24) | <0.05 |
| Controls (29) | NEC (24) | p | |
|---|---|---|---|
| Neonatal characteristics | |||
| Neonatal male sex | 51.72% (15/29) | 66.67% (16/24) | 0.272 |
| Gestational age at delivery | |||
| Days | 213 (192–230) | 196 (185–211) | 0.100 |
| Weeks | 30.43 (27.43–32.86) | 27.93 (26.39–30.18) | 0.100 |
| Apgar score at 5 min | 8.00 (7.00–8.00) | 8.00 (7.00–8.00) | 0.857 |
| Apgar score at 10 min | 8.00 (8.00–9.00) | 8.00 (8.00–9.00) | 0.698 |
| Cord blood pH | 7.31 (7.28–7.34) | 7.32 (7.28–7.35) | 0.802 |
| Birthweight (grams) | 880.00 (650.00–1270.00) | 747.50 (558.75–943.75) | 0.186 |
| Birthweight (Fenton z-score) | −1.66 (−2.74–1.16) | −3.09 (−4.21–1.88) | <0.05 |
| Birthweight (Fenton MoM) | 0.70 (0.63–0.77) | 0.73 (0.64–0.78) | 0.681 |
| Birthweight (IG21 z-score) | −1.68 (−2.11–1.14) | −1.61 (−2.05–1.43) | 0.639 |
| Birthweight (IG21 MoM) | 0.75 (0.65–0.82) | 0.74 (0.65–0.90) | 0.754 |
| Fetal blood sample at birth | |||
| Hb (g/L) | 153.00 (145.00–170.00) | 159.00 (143.00–168.50) | 0.897 |
| Platelets (×109/L) | 183.00 (133.00–225.00) | 178.00 (107.50–224.00) | 0.587 |
| WBC (×109/L) | 6860.00 (5530.00–10,470.00) | 4290.00 (2535.00–6360.00) | <0.05 |
| Fetal cardio-pulmonary resuscitation | 44.83% (13/29) | 83.33% (20/24) | <0.05 |
| Fetal tracheal intubation at birth | 27.59% (8/29) | 37.50% (9/24) | 0.441 |
| Fetal tracheal intubation after birth | 3.45% (1/29) | 0.00% (0/24) | 0.358 |
| Neonatal ventilation within the first 24 h of life | |||
| 1 Spontaneous breathing | 24.14% (7/29) | 16.67% (4/24) | 0.504 |
| 2 Synchronized intermittent mandatory ventilation | 41.38% (12/29) | 70.83% (17/24) | <0.05 |
| 3 Nasal continuous positive airway pressure | 10.34% (3/29) | 8.33% (2/24) | 0.803 |
| 4 High-flow nasal cannula oxygen | 20.69% (6/29) | 4.17% (1/24) | 0.077 |
| 5 High-frequency oscillatory ventilation | 3.45% (1/29) | 0.00% (0/24) | 0.358 |
| Neonatal RDS | 48.28% (14/29) | 79.17% (19/24) | <0.05 |
| Surfactant use | 62.07% (18/29) | 79.17% (19/24) | 0.177 |
| Apnea of prematurity | 34.48% (10/29) | 45.83% (11/24) | 0.400 |
| IVH | 34.48% (10/29) | 25.00% (6/24) | 0.454 |
| Neonatal sepsis | 3.45% (1/29) | 0.00% (0/24) | 0.358 |
| Respiratory acidosis | 27.59% (8/29) | 41.67% (10/24) | 0.281 |
| PDA | 24.14% (7/29) | 54.17% (13/24) | <0.05 |
| PFO | 34.48% (10/29) | 45.83% (11/24) | 0.400 |
| Inotropic support | 6.90% (2/29) | 25.00% (6/24) | 0.067 |
| Enteral nutrition duration (days) | 1.00 (1.00–1.00) | 2.00 (1.00–3.00) | 0.188 |
| Type of Enteral nutrition HM (%) | 26/29 (89.65) | 20/24 (83.33%) | 0.6881 |
| OR (*) | p (ŧ) | AUC (*) | Specificity (*) (**) | Sensitivity (*) (**) | |
|---|---|---|---|---|---|
| Maternal and fetal Doppler (prenatal) | |||||
| Bilateral uterine artery notching | 2.6593 (0.8256–8.5657) | 0.101 | 61% (48–74%) | 76% (50–100%) | 46% (25–100%) |
| UA AREDF | 3.4286 (0.9728–12.084) | 0.055 | 62% (50–74%) | 83% (66–97%) | 42% (21–62%) |
| DV PI > 95th percentile | 6.1905 (1.46–26.2484) | 0.013 | 66% (54–77%) | 90% (79–100%) | 42% (21–62%) |
| Maternal and pregnancy characteristics | |||||
| Maternal age (years) | 1.0468 (0.935–1.172) | 0.427 | 58% (42–75%) | 86% (66–97%) | 46% (25–71%) |
| Nulliparity | 1.9833 (0.6618–5.9438) | 0.221 | 58% (45–72%) | 59% (0–100%) | 58% (0–100%) |
| Gestational age at delivery (days) | 0.9833 (0.961–1.0061) | 0.150 | 63% (48–79%) | 52% (31–97%) | 79% (29–96%) |
| Post-natal characteristics | |||||
| Fetal cardio-pulmonary resuscitation | 6.15385 (1.67835–22.56368) | 0.006 | 69% (57–81%) | 55% (38–72%) | 83% (67–96%) |
| PDA | 3.71429 (1.15316–11.96363) | 0.028 | 65% (52–78%) | 76% (59–90%) | 54% (33–75%) |
| Inotropic support | 4.5 (0.81564–24.827) | 0.084 | 59% (49–69%) | 93% (79–100%) | 25% (8–50%) |
| Birthweight (grams) | 0.9993 (0.9981–1.0006) | 0.289 | 61% (45–76%) | 38% (28–97%) | 88% (25–96%) |
| Birthweight (Fenton z-score) | 0.6682 (0.4713–0.9474) | 0.024 | 68% (54–83%) | 48% (24–93%) | 83% (42–100%) |
| Birthweight (Fenton z-score ≤ −1.62) | 4.6667 (1.2753–17.0772) | 0.020 | 66% (54–78%) | 48% (31–66%) | 83% (67–96%) |
| Newborn WBC (×109/L) | 0.9998 (0.9996–0.9999) | 0.015 | 74% (60–88%) | 79% (59–100%) | 71% (38–92%) |
| Newborn WBC ≤ 5255 × 109/L | 9.3095 (2.6465–32.7479) | 0.001 | 75% (63–87%) | 79% (62–93%) | 71% (50–88%) |
| OR (*) | p (ŧ) | AUC (*) | Specificity (*) (**) | Sensitivity (*) (**) | |
|---|---|---|---|---|---|
| Model 1 (p < 0.05) (ŧŧ) | 68% (55–81%) | 90% (69–100%) | 42% (21–71%) | ||
| UA AREDF | 2.0569 (0.5089–8.3144) | 0.312 | |||
| DV PI > 95th percentile | 4.6982 (1.0161–21.7226) | 0.048 | |||
| Model 2 (p < 0.05) (ŧŧ) | 68% (54–83%) | 41% (31–83%) | 96% (62–100%) | ||
| Birthweight (Fenton z-score ≤ −1.62) | 4.3918 (1.1861–16.2626) | 0.027 | |||
| Birthweight (grams) | 0.9995 (0.9982–1.0008) | 0.469 | |||
| Model 3 (p < 0.05) (ŧŧ) | 82% (71–94%) | 76% (59–90%) | 88% (71–100%) | ||
| Birthweight (Fenton z-score ≤ −1.62) | 3.5051 (0.7672–16.0132) | 0.106 | |||
| Newborn WBC ≤ 5255 × 109/L | 6.3635 (1.6871–24.0027) | 0.006 | |||
| PDA | 3.0366 (0.7747–11.9019) | 0.111 | |||
| Model 4 (p < 0.05) (ŧŧ) | 84% (72–95%) | 72% (55–93%) | 92% (71–100%) | ||
| Birthweight z-score Fenton ≤ −1.62 | 2.9388 (0.6127–14.0957) | 0.178 | |||
| Newborn WBC ≤ 5255 × 109/L | 5.362 (1.3667–21.0366) | 0.016 | |||
| PDA | 2.8815 (0.7208–11.5184) | 0.134 | |||
| DV PI > 95th percentile | 2.3867 (0.4509–12.633) | 0.306 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duci, M.; Cosmi, E.; Zorzato, P.; Londero, A.P.; Verlato, G.; Baraldi, E.; Ragazzi, E.; Fascetti Leon, F.; Visentin, S. Fetal Doppler Evaluation to Predict NEC Development. J. Pers. Med. 2022, 12, 1042. https://doi.org/10.3390/jpm12071042
Duci M, Cosmi E, Zorzato P, Londero AP, Verlato G, Baraldi E, Ragazzi E, Fascetti Leon F, Visentin S. Fetal Doppler Evaluation to Predict NEC Development. Journal of Personalized Medicine. 2022; 12(7):1042. https://doi.org/10.3390/jpm12071042
Chicago/Turabian StyleDuci, Miriam, Erich Cosmi, Pierpaolo Zorzato, Ambrogio Pietro Londero, Giovanna Verlato, Eugenio Baraldi, Eugenio Ragazzi, Francesco Fascetti Leon, and Silvia Visentin. 2022. "Fetal Doppler Evaluation to Predict NEC Development" Journal of Personalized Medicine 12, no. 7: 1042. https://doi.org/10.3390/jpm12071042
APA StyleDuci, M., Cosmi, E., Zorzato, P., Londero, A. P., Verlato, G., Baraldi, E., Ragazzi, E., Fascetti Leon, F., & Visentin, S. (2022). Fetal Doppler Evaluation to Predict NEC Development. Journal of Personalized Medicine, 12(7), 1042. https://doi.org/10.3390/jpm12071042









