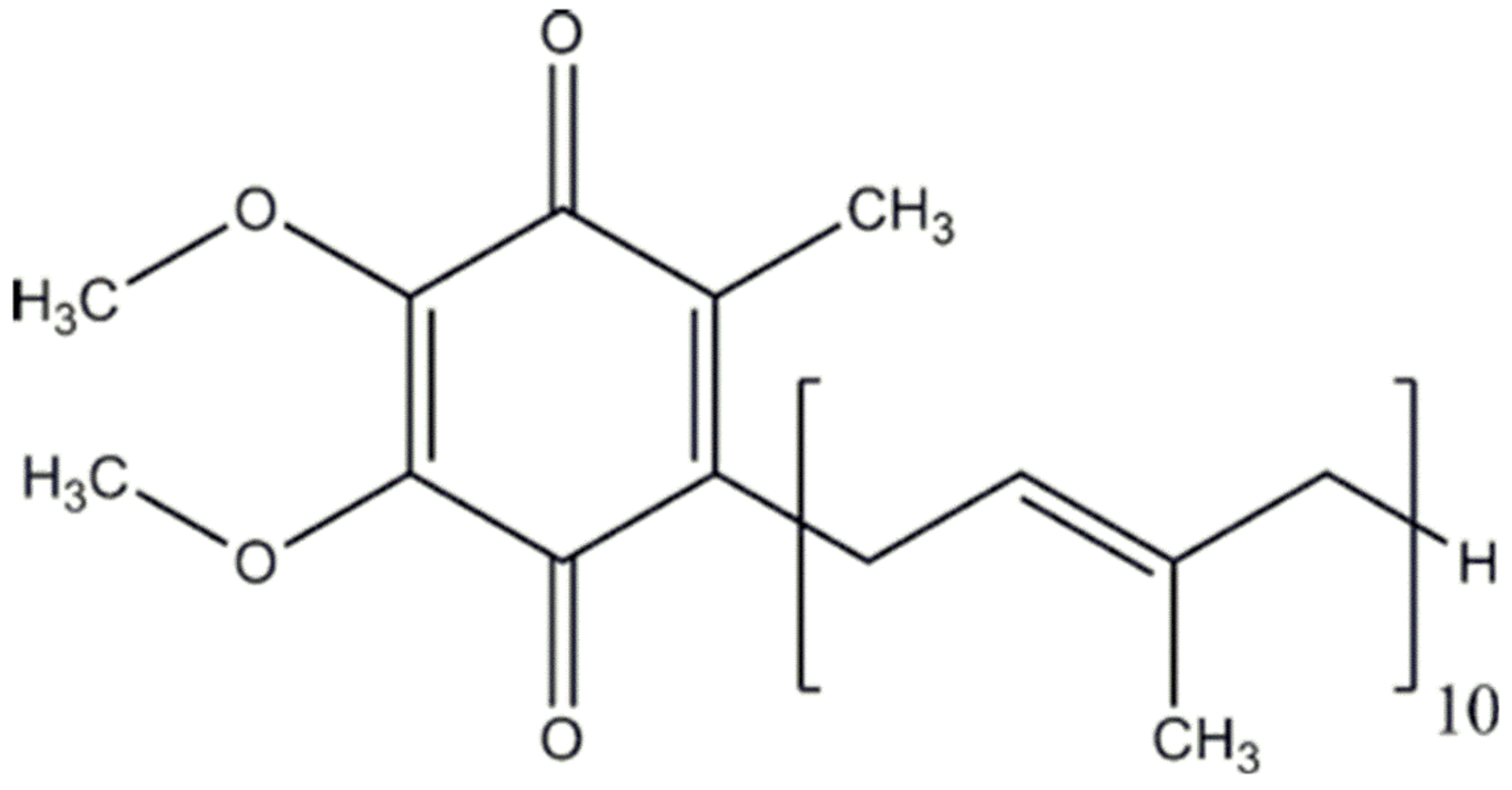
Coenzyme Q10 and Parkinsonian Syndromes: A Systematic Review
Abstract
:1. Introduction
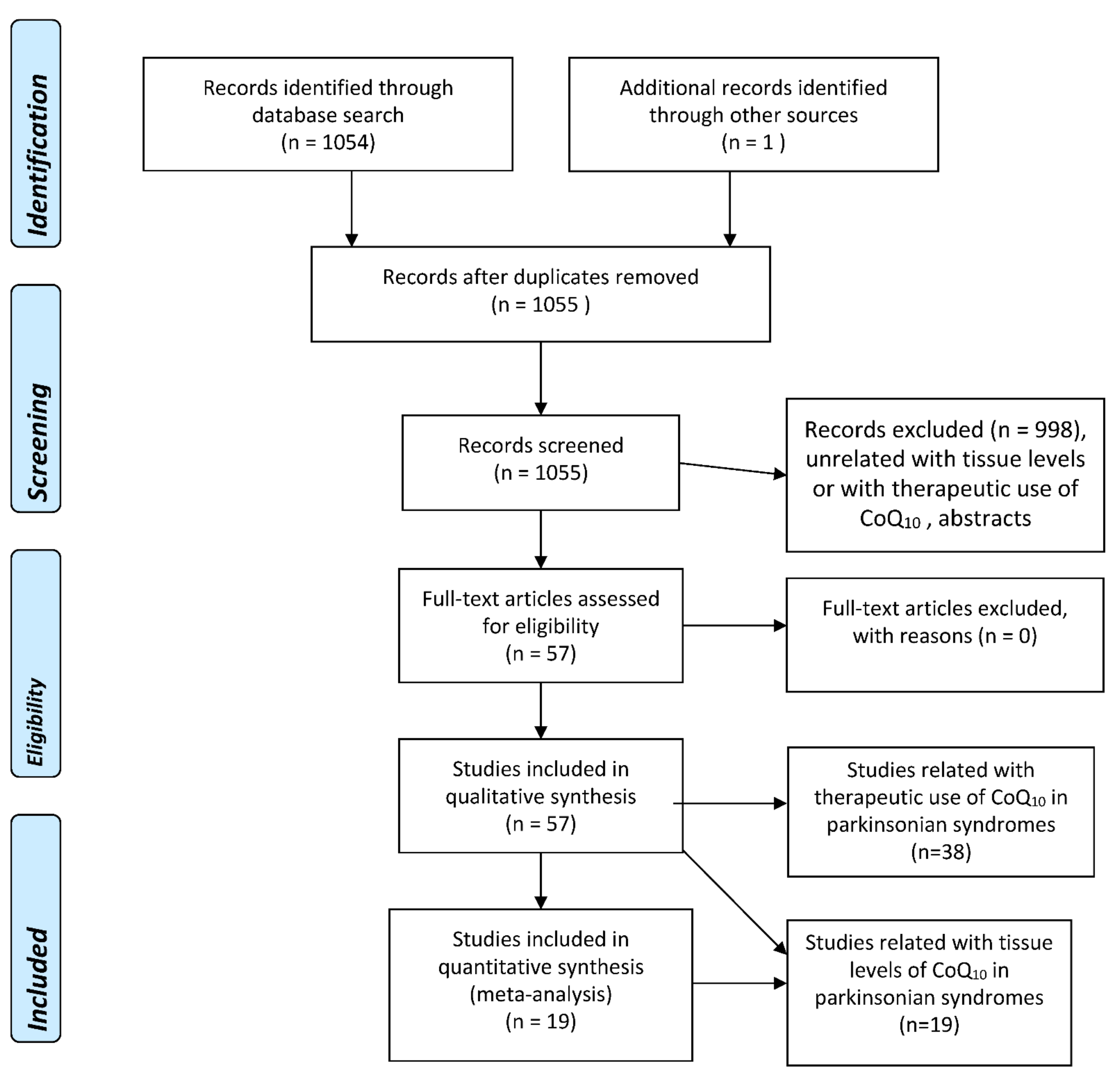
2. Methods
2.1. Search Strategy and Criteria for Eligibility of Studies
2.2. Selection of Studies and Methodology for the Meta-Analyses
3. Results
3.1. Studies Assessing Tissular CoQ10 Concentrations
3.1.1. Parkinson’s Disease
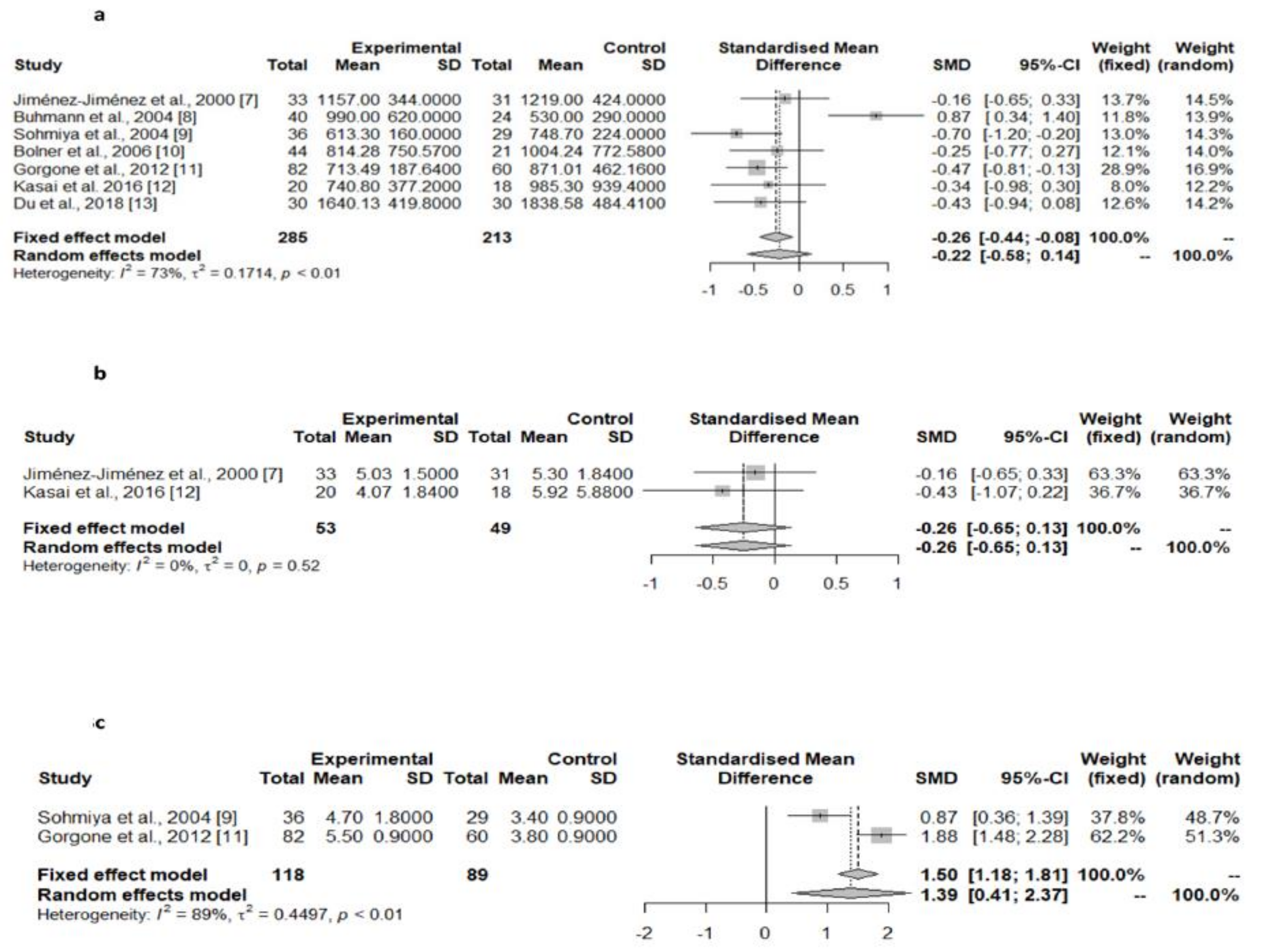
Serum/Plasma
Blood Cells
Cerebrospinal Fluid (CSF)
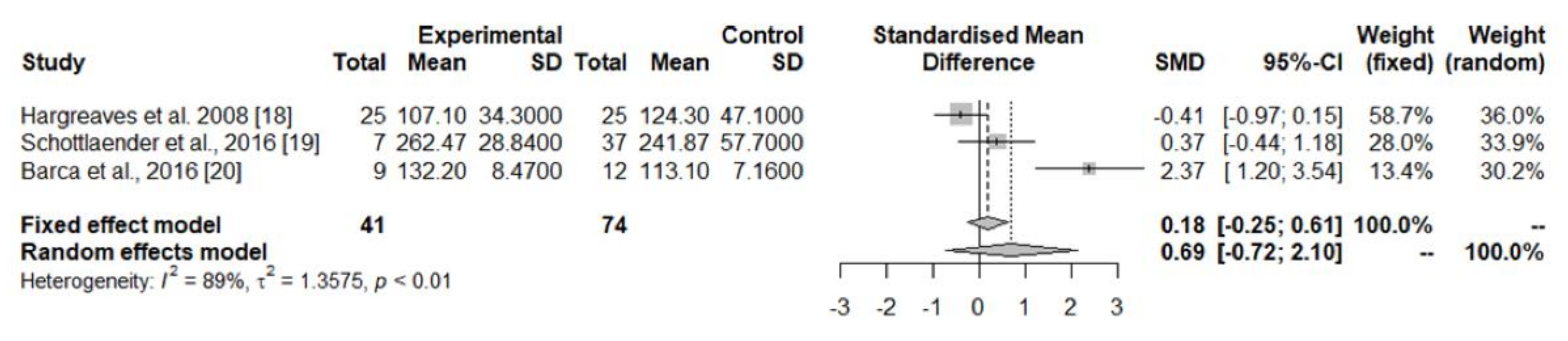
Brain
Skin Fibroblasts
3.1.2. Other Parkinsonian Syndromes
3.2. Studies Assessing Therapeutic Response to CoQ10 Administration
3.2.1. Parkinson’s Disease
3.2.2. Progressive Supranuclear Palsy
4. Discussion and Conclusions
- (a)
- (b)
- (c)
- (d)
- CoQ10 administration was also able to prevent iron-induced apoptosis in cultured human dopaminergic (SK-N-SH) neurons, in metallothionein gene-manipulated mice, and in alpha-synuclein knockout (alpha-synko) mice [56].
- (e)
- CoQ10 administration can prevent neurodegeneration and behavioral deterioration in rodents exposed to several toxins causing experimental parkinsonism, such as the pesticides paraquat [57,58], dichlorvos [59], and rotenone [60,61], and showed neuroprotective effects against rotenone in primary rat mesencephalic cultures [62] and human neuroblastoma cells [63]. Interestingly, the exposure of human neuroblastoma SH-SY5Y cells to commonly used organophosphate compounds, such as dichlorvos, methyl-parathion (parathion), and chlorpyrifos (CPF), induces an important decrease in CoQ10 levels and complex II + III activity—both related to a decrease in neuronal cell viability. In this model, CoQ10 supplementation can modestly although significantly increase complex II + III activity [64].
- (f)
- CoQ10 supplementation (with or without the concomitant treatment of levodopa) has shown a protective effect against chlorpromazine-induced parkinsonism in mice, including a reduction in mortality and catalepsy, an increase in dopamine levels, and a decrease in oxidative stress [65]. Similarly, CoQ10 improved the forced swimming test, locomotor activity test, catalepsy, muscle coordination, and akinesia test, and reduced the dopamine depletion in haloperidol-induced parkinsonism in rats [66].
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crane, F.L. Biochemical functions of coenzyme Q10. J. Am. Coll. Nutr. 2001, 20, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Heaton, R.A.; Hargreaves, I.P. Coenzyme Q10, Ageing and the Nervous System, An Overview. Antioxidants 2021, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Navarro, H.; Jiménez-Jiménez, F.J.; Pilo de la Fuente, B.; Plaza-Nieto, J.F. Mecanismos patogénicos de la enfermedad de Parkinson. In Tratado de los Trastornos del Movimiento, 2nd ed.; Jiménez-Jiménez, F.J., Luquin, M.R., Molina, J.A., Linazasoro, G., Eds.; Viguera Editores: Barcelona, Spain, 2008; Volume 1, pp. 425–485. [Google Scholar]
- Jurcau, A. Insights into the Pathogenesis of Neurodegenerative Diseases, Focus on Mitochondrial Dysfunction and Oxidative Stress. Int. J. Mol. Sci. 2021, 22, 11847. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses, the PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 10 May 2022).
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology, a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2009, 283, 2008–2012. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. Evid Based Ment Health. 2019, 22, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Jiménez, F.J.; Molina, J.A.; de Bustos, F.; García-Redondo, A.; Gómez-Escalonilla, C.; Martínez-Salio, A.; Berbel, A.; Camacho, A.; Zurdo, M.; Barcenilla, B.; et al. Serum levels of coenzyme Q10 in patients with Parkinson’s disease. J. Neural. Transm. 2000, 107, 177–181. [Google Scholar] [CrossRef]
- Buhmann, C.; Arlt, S.; Kontush, A.; Möller-Bertram, T.; Sperber, S.; Oechsner, M.; Stuerenburg, H.J.; Beisiegel, U. Plasma and CSF markers of oxidative stress are increased in Parkinson’s disease and influenced by antiparkinsonian medication. Neurobiol. Dis. 2004, 15, 160–170. [Google Scholar] [CrossRef]
- Sohmiya, M.; Tanaka, M.; Tak, N.W.; Yanagisawa, M.; Tanino, Y.; Suzuki, Y.; Okamoto, K.; Yamamoto, Y. Redox status of plasma coenzyme Q10 indicates elevated systemic oxidative stress in Parkinson’s disease. J. Neurol. Sci. 2004, 223, 161–166. [Google Scholar] [CrossRef]
- Bolner, A.; Micciolo, R.; Bosello, O.; Nordera, G.P. A Panel of Oxidative Stress Markers in Parkinson’s Disease. Clin. Lab. 2016, 62, 105–112. [Google Scholar] [CrossRef]
- Gorgone, G.; Currò, M.; Ferlazzo, N.; Parisi, G.; Parnetti, L.; Belcastro, V.; Tambasco, N.; Rossi, A.; Pisani, F.; Calabresi, P.; et al. Coenzyme Q10, hyperhomocysteinemia and MTHFR C677T polymorphism in levodopa-treated Parkinson’s disease patients. Neuromolecular Med. 2012, 14, 84–90. [Google Scholar] [CrossRef]
- Kasai, T.; Tokuda, T.; Ohmichi, T.; Ishii, R.; Tatebe, H.; Nakagawa, M.; Mizuno, T. Serum Levels of Coenzyme Q10 in Patients with Multiple System Atrophy. PLoS One 2016, 11, e0147574. [Google Scholar] [CrossRef]
- Du, J.; Wang, T.; Huang, P.; Cui, S.; Gao, C.; Lin, Y.; Fu, R.; Shen, J.; He, Y.; Tan, Y.; et al. Clinical correlates of decreased plasma coenzyme Q10 levels in patients with multiple system atrophy. Parkinsonism Relat. Disord. 2018, 57, 58–62. [Google Scholar] [CrossRef]
- Götz, M.E.; Gerstner, A.; Harth, R.; Dirr, A.; Janetzky, B.; Kuhn, W.; Riederer, P.; Gerlach, M. Altered redox state of platelet coenzyme Q10 in Parkinson’s disease. J. Neural. Transm. 2000, 107, 41–48. [Google Scholar]
- Mischley, L.K.; Allen, J.; Bradley, R. Coenzyme Q10 deficiency in patients with Parkinson’s disease. J. Neurol. Sci. 2012, 318, 72–75. [Google Scholar] [CrossRef] [Green Version]
- Isobe, C.; Murata, T.; Sato, C.; Terayama, Y. Increase of oxidized/total coenzyme Q-10 ratio in cerebrospinal fluid in patients with Parkinson’s disease. J. Clin. Neurosci. 2007, 14, 340–343. [Google Scholar] [CrossRef]
- Compta, Y.; Giraldo, D.M.; Muñoz, E.; Antonelli, F.; Fernández, M.; Bravo, P.; Soto, M.; Cámara, A.; Torres, F.; Martí, M.J.; et al. Cerebrospinal fluid levels of coenzyme Q10 are reduced in multiple system atrophy. Parkinsonism Relat. Disord. 2018, 46, 16–23. [Google Scholar] [CrossRef]
- Hargreaves, I.P.; Lane, A.; Sleiman, P.M. The coenzyme Q10 status of the brain regions of Parkinson’s disease patients. Neurosci Lett. 2008, 447, 17–19. [Google Scholar] [CrossRef]
- Schottlaender, L.V.; Bettencourt, C.; Kiely, A.P.; Chalasani, A.; Neergheen, V.; Holton, J.L.; Hargreaves, I.; Houlden, H. Coenzyme Q10 Levels Are Decreased in the Cerebellum of Multiple-System Atrophy Patients. PLoS One 2016, 11, e0149557. [Google Scholar] [CrossRef] [Green Version]
- Barca, E.; Kleiner, G.; Tang, G.; Ziosi, M.; Tadesse, S.; Masliah, E.; Louis, E.D.; Faust, P.; Kang, U.J.; Torres, J.; et al. Decreased Coenzyme Q10 Levels in Multiple System Atrophy Cerebellum. J. Neuropathol. Exp. Neurol. 2016, 75, 663–672. [Google Scholar] [CrossRef] [Green Version]
- Del Hoyo, P.; García-Redondo, A.; de Bustos, F.; Molina, J.A.; Sabed, Y.; Alonso-Navarro, H.; Caballero, L.; Arenas, J.; Agúndez, J.A.; Jiménez-Jiménez, F.J. Oxidative stress in skin fibroblasts cultures from patients with Parkinson’s disease. BMC Neurol. 2010, 10, 95. [Google Scholar] [CrossRef] [Green Version]
- Mitsui, J.; Matsukawa, T.; Yasuda, T.; Ishiura, H.; Tsuji, S. Plasma Coenzyme Q10 Levels in Patients With Multiple System Atrophy. JAMA Neurol. 2016, 73, 977–980. [Google Scholar] [CrossRef] [Green Version]
- Compagnoni, M.C.; Kleiner, G.; Bordoni, A.; Fortunato, F.; Ronchi, D.; Salani, S.; Guida, M.; Corti, C.; Pichler, I.; Bergamini, C.; et al. Mitochondrial dysfunction in fibroblasts of Multiple System Atrophy. Biochim. Biophys. Acta Mol. Basis. Dis. 2018, 1864, 3588–3597. [Google Scholar] [CrossRef]
- Molina, J.A.; de Bustos, F.; Ortiz, S.; Del Ser, T.; Seijo, M.; Benito-Léon, J.; Oliva, J.M.; Pérez, S.; Manzanares, J. Serum levels of coenzyme Q in patients with Lewy body disease. J. Neural. Transm. 2002, 109, 1195–1201. [Google Scholar] [CrossRef]
- Gironi, M.; Bianchi, A.; Russo, A.; Alberoni, M.; Ceresa, L.; Angelini, A.; Cursano, C.; Mariano, E.; Nemni, R.; Kullmann, C.; et al. Oxidative imbalance in different neurodegenerative diseases with memory impairment. Neurodegener. Dis. 2011, 8, 129–137. [Google Scholar] [CrossRef]
- Matsubara, T.; Azuma, T.; Yoshida, S.; Yamagami, T.; Okamoto, T.; Kishi, T. Serum coenzyme Q-10 level in Parkinson syndrome. In Biomedical and Clinical Aspects of Coenzyme Q; Folkers, K., Littarru, G.P., Yamagami, T., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1991; pp. 159–166. [Google Scholar]
- Strijks, E.; Kremer, H.P.; Horstink, M.W. Q10 therapy in patients with idiopathic Parkinson’s disease. Mol. Aspects Med. 1997, 18, S237–S240. [Google Scholar] [CrossRef]
- Shults, C.; Oakes, D.; Kieburtz, K.; Beal, M.F.; Haas, R.; Plumb, S.; Juncos, J.L.; Nutt, J.; Shoulson, I.; Carter, J.; et al. Effects of coenzyme Q10 in early Parkinson disease, evidence of slowing of the functional decline. Arch. Neurol. 2002, 59, 1541–1550. [Google Scholar] [CrossRef]
- Müller, T.; Büttner, T.; Gholipour, A.F.; Kuhn, W. Coenzyme Q10 supplementation provides mild symptomatic benefit in patients with Parkinson’s disease. Neurosci. Lett. 2003, 341, 201–204. [Google Scholar] [CrossRef]
- NINDS NET-PD Investigators. A randomized clinical trial of coenzyme Q10 and GPI-1485 in early Parkinson disease. Neurology 2007, 68, 20–28. [Google Scholar]
- Storch, A.; Jost, W.H.; Vieregge, P.; Spiegel, J.; Greulich, W.; Durner, J.; Müller, T.; Kupsch, A.; Henningsen, H.; Oertel, W.H.; et al. Randomized, double-blind, placebo-controlled trial on symptomatic effects of coenzyme Q(10) in Parkinson disease. Arch. Neurol. 2007, 64, 938–944. [Google Scholar] [CrossRef] [Green Version]
- Parkinson Study Group QE3 Investigators; Beal, M.F.; Oakes, D.; Shoulson, I.; Henchcliffe, C.; Galpern, W.R.; Haas, R.; Juncos, J.L.; Nutt, J.G.; Voss, T.S.; et al. A randomized clinical trial of high-dosage coenzyme Q10 in early Parkinson disease, no evidence of benefit. JAMA Neurol. 2014, 71, 543–552. [Google Scholar]
- Jie, Z. Clinical effects and safety of coenzyme Q10 in Parkinson disease. China Foreign Med. Treat. 2014, 23, 79–80. [Google Scholar]
- Wang, X.Y.; Yang, Z.M.; Zhang, X.J.; Xi, S.; Liu, C.; Li, J.; Wang, Q.; Wang, L. Clinical observation of coenzyme Q10 in Parkinson disease. HeBei J. TCM 2014, 36, 151–153. [Google Scholar]
- Li, Z.; Wang, P.; Yu, Z.; Cong, Y.; Sun, H.; Zhang, J.; Zhang, J.; Sun, C.; Zhang, Y.; Ju, X. The effect of creatine and coenzyme q10 combination therapy on mild cognitive impairment in Parkinson’s disease. Eur. Neurol. 2015, 73, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Yoritaka, A.; Kawajiri, S.; Yamamoto, Y.; Nakahara, T.; Ando, M.; Hashimoto, K.; Nagase, M.; Saito, Y.; Hattori, N. Randomized, double-blind, placebo-controlled pilot trial of reduced coenzyme Q10 for Parkinson’s disease. Parkinsonism Relat. Disord. 2015, 21, 911–916. [Google Scholar] [CrossRef]
- Liu, J.; Wang, L.; Zhan, S.Y.; Xia, Y. Coenzyme Q10 for Parkinson’s disease. Cochrane Database Syst. Rev. 2011, 12, CD008150, Update in Cochrane Database Syst. Rev. 2012, 5, CD008150. [Google Scholar]
- Negida, A.; Menshawy, A.; El Ashal, G.; Elfouly, Y.; Hani, Y.; Hegazy, Y.; El Ghonimy, S.; Fouda, S.; Rashad, Y. Coenzyme Q10 for Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. CNS Neurol. Disord. Drug Targets 2016, 15, 45–53. [Google Scholar] [CrossRef]
- Zhu, Z.G.; Sun, M.X.; Zhang, W.L.; Wang, W.W.; Jin, Y.M.; Xie, C.L. The efficacy and safety of coenzyme Q10 in Parkinson’s disease: A meta-analysis of randomized controlled trials. Neurol. Sci. 2017, 38, 215–224. [Google Scholar] [CrossRef]
- Mitsui, J.; Koguchi, K.; Momose, T.; Takahashi, M.; Matsukawa, T.; Yasuda, T.; Tokushige, S.I.; Ishiura, H.; Goto, J.; Nakazaki, S.; et al. Three-Year Follow-Up of High-Dose Ubiquinol Supplementation in a Case of Familial Multiple System Atrophy with Compound Heterozygous COQ2 Mutations. Cerebellum. 2017, 16, 664–672. [Google Scholar] [CrossRef] [Green Version]
- Stamelou, M.; Reuss, A.; Pilatus, U.; Magerkurth, J.; Niklowitz, P.; Eggert, K.M.; Krisp, A.; Menke, T.; Schade-Brittinger, C.; Oertel, W.H.; et al. Short-term effects of coenzyme Q10 in progressive supranuclear palsy: A randomized, placebo-controlled trial. Mov. Disord. 2008, 23, 942–949. [Google Scholar] [CrossRef]
- Apetauerova, D.; Scala, S.A.; Hamill, R.W.; Simon, D.K.; Pathak, S.; Ruthazer, R.; Standaert, D.G.; Yacoubian, T.A. CoQ10 in progressive supranuclear palsy: A randomized, placebo-controlled, double-blind trial. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e266. [Google Scholar] [CrossRef] [Green Version]
- Beal, M.F. Coenzyme Q10 administration and its potential for treatment of neurodegenerative diseases. Biofactors 1999, 9, 261–266. [Google Scholar] [CrossRef]
- Shults, C.W.; Haas, R.H.; Beal, M.F. A possible role of coenzyme Q10 in the etiology and treatment of Parkinson’s disease. Biofactors 1999, 9, 267–272. [Google Scholar] [CrossRef]
- Beal, M.F.; Matthews, R.T.; Tieleman, A.; Shults, C.W. Coenzyme Q10 attenuates the 1-methyl-4-phenyl-1.;2.;3.; Tetrahydropyridine (MPTP) induced loss of striatal dopamine and dopaminergic axons in aged mice. Brain Res. 1998, 783, 109–114. [Google Scholar] [CrossRef]
- Cleren, C.; Yang, L.; Lorenzo, B.; Calingasan, N.Y.; Schomer, A.; Sireci, A.; Wille, E.J.; Beal, M.F. Therapeutic effects of coenzyme Q10 (CoQ10) and reduced CoQ10 in the MPTP model of Parkinsonism. J. Neurochem. 2008, 104, 1613–1621. [Google Scholar] [CrossRef]
- Kobayashi, S.; Muroyama, A.; Matsushima, H.; Yoshimura, I.; Mitsumoto, Y. Oral administration of coenzyme Q10 reduces MPTP-induced loss of dopaminergic nerve terminals in the striatum in mice. Neurol. Sci. 2012, 33, 195–199. [Google Scholar] [CrossRef]
- Yan, A.; Liu, Z.; Song, L.; Wang, X.; Zhang, Y.; Wu, N.; Lin, J.; Liu, Y.; Liu, Z. Idebenone Alleviates Neuroinflammation and Modulates Microglial Polarization in LPS-Stimulated BV2 Cells and MPTP-Induced Parkinson’s Disease Mice. Front. Cell. Neurosci. 2019, 12, 529. [Google Scholar] [CrossRef]
- Horvath, T.L.; Diano, S.; Leranth, C.; Garcia-Segura, L.M.; Cowley, M.A.; Shanabrough, M.; Elsworth, J.D.; Sotonyi, P.; Roth, R.H.; Dietrich, E.H.; et al. Coenzyme Q induces nigral mitochondrial uncoupling and prevents dopamine cell loss in a primate model of Parkinson’s disease. Endocrinology 2003, 144, 2757–2760. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Calingasan, N.Y.; Wille, E.J.; Cormier, K.; Smith, K.; Ferrante, R.J.; Beal, M.F. Combination therapy with coenzyme Q10 and creatine produces additive neuroprotective effects in models of Parkinson’s and Huntington’s diseases. J. Neurochem. 2009, 109, 1427–1439. [Google Scholar] [CrossRef] [Green Version]
- Schulz, J.B.; Henshaw, D.R.; Matthews, R.T.; Beal, M.F. Coenzyme Q10 and nicotinamide and a free radical spin trap protect against MPTP neurotoxicity. Exp. Neurol. 1995, 132, 279–283. [Google Scholar] [CrossRef]
- Ghasemloo, E.; Mostafavi, H.; Hosseini, M.; Forouzandeh, M.; Eskandari, M.; Mousavi, S.S. Neuroprotective effects of coenzyme Q10 in Parkinson’s model via a novel Q10/miR-149-5p/MMPs pathway. Metab. Brain Dis. 2021, 36, 2089–2100. [Google Scholar] [CrossRef]
- Nezhadi, A.; Ghazi, F.; Rassoli, H.; Bakhtiari, M.; Ataiy, Z.; Soleimani, S.; Mehdizadeh, M. BMSC and CoQ10 improve behavioural recovery and histological outcome in rat model of Parkinson’s disease. Pathophysiology 2011, 18, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Kooncumchoo, P.; Sharma, S.; Porter, J.; Govitrapong, P.; Ebadi, M. Coenzyme Q(10) provides neuroprotection in iron-induced apoptosis in dopaminergic neurons. J. Mol. Neurosci. 2006, 28, 125–141. [Google Scholar] [CrossRef]
- Attia, H.N.; Maklad, Y.A. Neuroprotective effects of coenzyme Q10 on paraquat-induced Parkinson’s disease in experimental animals. Behav. Pharmacol. 2018, 29, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Somayajulu-Niţu, M.; Sandhu, J.K.; Cohen, J.; Sikorska, M.; Sridhar, T.S.; Matei, A.; Borowy-Borowski, H.; Pandey, S. Paraquat induces oxidative stress.; Neuronal loss in substantia nigra region and parkinsonism in adult rats, neuroprotection and amelioration of symptoms by water-soluble formulation of coenzyme Q10. BMC Neurosci. 2009, 10, 88. [Google Scholar] [CrossRef] [Green Version]
- Binukumar, B.K.; Gupta, N.; Bal, A.; Gill, K.D. Protection of dichlorvos induced oxidative stress and nigrostriatal neuronal death by chronic coenzyme Q10 pretreatment. Toxicol Appl. Pharmacol. 2011, 256, 73–82. [Google Scholar] [CrossRef]
- Abdin, A.A.; Hamouda, H.E. Mechanism of the neuroprotective role of coenzyme Q10 with or without L-dopa in rotenone-induced parkinsonism. Neuropharmacology. 2008, 55, 1340–1346. [Google Scholar] [CrossRef]
- Avcı, B.; Günaydın, C.; Güvenç, T.; Yavuz, C.K.; Kuruca, N.; Bilge, S.S. Idebenone Ameliorates Rotenone-Induced Parkinson’s Disease in Rats Through Decreasing Lipid Peroxidation. Neurochem. Res. 2021, 46, 513–522. [Google Scholar] [CrossRef]
- Moon, Y.; Lee, K.H.; Park, J.H.; Geum, D.; Kim, K. Mitochondrial membrane depolarization and the selective death of dopaminergic neurons by rotenone, protective effect of coenzyme Q10. J. Neurochem. 2005, 93, 1199–1208. [Google Scholar] [CrossRef]
- McCarthy, S.; Somayajulu, M.; Sikorska, M.; Borowy-Borowski, H.; Pandey, S. Paraquat induces oxidative stress and neuronal cell death; neuroprotection by water-soluble Coenzyme Q10. Toxicol. Appl. Pharmacol. 2004, 201, 21–31. [Google Scholar] [CrossRef]
- Turton, N.; Heaton, R.A.; Ismail, F.; Roberts, S.; Nelder, S.; Phillips, S.; Hargreaves, I.P. The Effect of Organophosphate Exposure on Neuronal Cell Coenzyme Q10 Status. Neurochem. Res. 2001, 46, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Onaolapo, O.J.; Odeniyi, A.O.; Jonathan, S.O.; Samuel, M.O.; Amadiegwu, D.; Olawale, A.; Tiamiyu, A.O.; Ojo, F.O.; Yahaya, H.A.; Ayeni, O.J.; et al. An Investigation of the Anti-Parkinsonism Potential of Co-enzyme Q10 and Co-enzyme Q10/Levodopa-carbidopa Combination in Mice. Curr. Aging Sci. 2021, 14, 62–75. [Google Scholar] [CrossRef]
- Gupta, B.K.; Kumar, S.; Kaur, H.; Ali, J.; Baboota, S. Attenuation of Oxidative Damage by Coenzyme Q10 Loaded Nanoemulsion Through Oral Route for the Management of Parkinson’s Disease. Rejuvenation Res. 2018, 21, 232–248. [Google Scholar] [CrossRef]
- Faust, K.; Gehrke, S.; Yang, Y.; Yang, L.; Beal, M.F.; Lu, B. Neuroprotective effects of compounds with antioxidant and anti-inflammatory properties in a Drosophila model of Parkinson’s disease. BMC Neurosci. 2009, 10, 109. [Google Scholar] [CrossRef] [Green Version]
- Tai, K.K.; Pham, L.; Truong, D.D. Idebenone induces apoptotic cell death in the human dopaminergic neuroblastoma SHSY-5Y cells. Neurotox. Res. 2011, 20, 321–328. [Google Scholar] [CrossRef]
- Shi, H.; Noguchi, N.; Xu, Y.; Niki, E. 1-Methyl-4-phenyl-2.;3-dihydropyridinium is transformed by ubiquinone to the selective nigrostriatal toxin 1-methyl-4-phenylpyridinium. FEBS Lett. 1999, 461, 196–200. [Google Scholar] [CrossRef] [Green Version]
- Lönnrot, K.; Metsä-Ketelä, T.; Molnár, G.; Ahonen, J.P.; Latvala, M.; Peltola, J.; Pietilä, T.; Alho, H. The effect of ascorbate and ubiquinone supplementation on plasma and CSF total antioxidant capacity. Free Radic. Biol. Med. 1996, 21, 211–217. [Google Scholar] [CrossRef]
- Shults, C.W.; Flint Beal, M.; Song, D.; Fontaine, D. Pilot trial of high dosages of coenzyme Q10 in patients with Parkinson’s disease. Exp. Neurol. 2004, 188, 491–494. [Google Scholar] [CrossRef]
- Nukui, K.; Yamagishi, T.; Miyawaki, H.; Kettawan, A.; Okamoto, T.; Belardinelli, R.; Tiano, L.; Littarru, G.P.; Sato, K. Blood CoQ10 levels and safety profile after single-dose or chronic administration of PureSorb-Q40, animal and human studies. Biofactors. 2008, 32, 209–219. [Google Scholar] [CrossRef]
- Seet, R.C.; Lim, E.C.; Tan, J.J.; Quek, A.M.; Chow, A.W.; Chong, W.L.; Ng, M.P.; Ong, C.N.; Halliwell, B. Does high-dose coenzyme Q10 improve oxidative damage and clinical outcomes in Parkinson’s disease? Antioxid. Redox Signal. 2014, 21, 211–217. [Google Scholar] [CrossRef]
- Prasuhn, J.; Brüggemann, N.; Hessler, N.; Berg, D.; Gasser, T.; Brockmann, K.; Olbrich, D.; Ziegler, A.; König, I.R.; Klein, C.; et al. An omics-based strategy using coenzyme Q10 in patients with Parkinson’s disease, concept evaluation in a double-blind randomized placebo-controlled parallel group trial. Neurol. Res. Pract. 2019, 1, 31. [Google Scholar] [CrossRef] [Green Version]
| Tissue | Author, Year [Ref] | Parameter | PD N | PD Mean ± SD (Except % in *) | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
|---|---|---|---|---|---|---|---|
| Serum/plasma | Jiménez-Jiménez et al., 2000 [9] | Total CoQ10 (nmol/L) | 33 | 1157 ± 344 | 31 | 1219 ± 424 | 62.00 (−130.39 to 254.39); 0.522 |
| Buhmann et al., 2004 [10] | Total CoQ10 (nmol/L) | 40 | 990 ± 620 | 24 | 530 ± 290 | −460.00 (−729.67 to −190.32); 0.001 | |
| Sohmiya et al., 2004 [11] | Total CoQ10 (nmol/L) | 36 | 613.3 ± 160 | 29 | 748.7 ± 224 | 135.40 (40.11 to 230.69); 0.006 | |
| Bolner et al., 2006 [12] | Total CoQ10 (nmol/L) | 44 | 814.28 ± 750.57 | 21 | 1004.24 ± 772.58 | 189.96 (−211.60 to 591.52); 0.348 | |
| Gorgone et al., 2012 [13] | Total CoQ10 (nmol/L) | 82 | 713.49 ± 187.64 | 60 | 871.01 ± 162.16 | 157.52 (97.95 to 217.09); <0.001 | |
| Kasai et al. 2016 [14] | Total CoQ10 (nmol/L) | 20 | 740.8 ± 377.2 | 18 | 985.3 ± 939.4 | 244.50 (−217.59 to 706.59); 0.290 | |
| Du et al., 2018 [15] | Total CoQ10 (nmol/L) | 30 | 1640.13 ± 419.80 | 30 | 1838.58 ± 481.41 | 198.45 (−34.98 to 431.88); 0.094 | |
| TOTAL SERIES | Total CoQ10 (nmol/L) | 285 | 906.01 ± 531.19 | 213 | 1025.65 ± 592.90 | Random-effects model p = 0.234 | |
| Jiménez-Jiménez et al., 2000 [9] | Total CoQ10/cholesterol | 33 | 5.03 ± 1.50 | 31 | 5.30 ± 1.84 | 02.7 (−0.57 to 1.11); 0.521 | |
| Kasai et al. 2016 [14] | Total CoQ10/cholesterol | 20 | 4.07 ± 1.84 | 18 | 5.92 ± 5.88 | 1.85 (−0.47 to 4.17); 0.115 | |
| TOTAL SERIES | Total CoQ10/cholesterol | 53 | 4.67 ± 1.69 | 49 | 5.53 ± 3.80 | Random-effects model p = 0.197 | |
| Sohmiya et al., 2004 [11] | % Oxidized/total CoQ10 | 36 | 4.7 ± 1.8 | 29 | 3.4 ± 0.9 | −1.30 (−2.03 to −0.57); <0.001 | |
| Gorgone et al., 2012 [13] | % Oxidized/total CoQ10 | 82 | 5.5 ± 0.9 | 60 | 3.8 ± 0.9 | −1.70 (−2.00 to −1.40); <0.001 | |
| TOTAL SERIES | % Oxidized/total CoQ10 | 118 | 5.26 ± 1.29 | 89 | 3.67 ± 0.9 | Random-effects model p = 0.006 | |
| Sohmiya et al., 2004 [11] | Oxidized CoQ10 (nmol/L) | 36 | 28.3 ± 10.5 | 29 | 24.7 ± 8.3 | −3.60 (−8.38 to 1.18); 0.137 | |
| Kasai et al. 2016 [14] | Oxidized CoQ10 (nmol/L) | 20 | 644.2 ± 382.4 | 18 | 900.2 ± 890.6 | 256.00 (−186.86 to 698.86); 0.249 | |
| Sohmiya et al., 2004 [11] | Reduced CoQ10 (nmol/L) | 36 | 585 ± 155 | 29 | 724 ± 219 | 139.00 (46.17 to 231.83); 0.004 | |
| Kasai et al. 2016 [14] | Reduced CoQ10 (nmol/L) | 20 | 96.6 ± 118.2 | 18 | 85.2 ± 66.6 | −11.40 (−75.52 to 52.72); 0.721 | |
| Platelets | Götz et al., 2000 [16] | Total CoQ10 (ng/109 platelets) | 20 | 80.6 ± 5.9 | 19 | 93.7 ± 5.1 | 13.10 (9.51 to 16.69); <0.001 |
| Götz et al., 2000 [16] | Reduced CoQ10 (ng/109 platelets) | 20 | 10.3 ± 2.4 | 19 | 20.3 ± 3.2 | 10.00 (8.17 to 11.83); <0.001 | |
| Götz et al., 2000 [16] | Oxidized CoQ10 (ng/109 platelets) | 20 | 70.3 ± 4.8 | 19 | 73.5 ± 4.7 | 3.20 (0.07 to 6.33); 0.045 | |
| Götz et al., 2000 [16] | Reduced/oxidizedCoQ10 | 20 | 0.15 ± 0.04 | 19 | 0.32 ± 0.07 | 0.17 (0.13 to 0.21); <0.001 | |
| Götz et al., 2000 [16] | Reduced/total CoQ10 | 20 | 0.11 ± 0.02 | 19 | 0.21 ± 0.03 | 0.10 (0.08 to 0.12); <0.001 | |
| Lymphocytes | Mischley et al., 2012 [17] * | % of patients with CoQ10 deficiency * | 22 | 32–36% | 88 | 8–9% | p = 0.0012–0.006 (according to authors data) |
| CSF | Isobe et al., 2007 [18] | Oxidized CoQ10 (nmol/L) | 20 | 5.2 ± 1.5 | 17 | 2.9 ± 1.3 | −2.30 (−3.25 to −1.35); <0.001 |
| Isobe et al., 2007 [18] | Reduced CoQ10 (nmol/L) | 20 | 0.7 ± 0.6 | 17 | 0.8 ± 0.7 | 0.10 (−0.33 to 0.53); 0.643 | |
| Isobe et al., 2007 [18] | Oxidized/total CoQ10 | 20 | 0.803 ± 0.179 | 17 | 0.682 ± 0.204 | −0.12 (−0.25 to 0.01); 0.063 | |
| Compta et al., 2018 [19] | Total CoQ10 (nmol/L) | 15 | 54.39 ± 7.16 | 15 | 36.02 ± 7.20 | −18.37 (−23.74 to −13.00); < 0.001 | |
| Brain | |||||||
| Striatum | Hargreaves et al. 2008 [20] | Total CoQ10 (pmol/mg protein) | 20 | 188.6 ± 51.4 | 20 | 214.3 ± 64.3 | 25.70 (−11.56 to 62.96); 0.171 |
| Substantia nigra | Hargreaves et al. 2008 [20] | Total CoQ10 (pmol/mg protein) | 8 | 102.9 ± 42.9 | 8 | 120.0 ± 4.3 | 17.10 (−15.59 to 49.79); 0.281 |
| Cerebellum cortex | Hargreaves et al. 2008 [20] | Total CoQ10 (pmol/mg protein) | 25 | 107.1 ± 34.3 | 25 | 124.3 ± 47.1 | 17.20 (−6.23 to 40.63); 0.147 |
| Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg protein) | 7 | 262.47 ± 28.84 | 37 | 241.87 ± 57.70 | −2.06 (−65.95 to 24.75); 0.365 | |
| Barca et al., 2016 [22] | Total CoQ10 (pmol/mg protein) | 9 | 132.2 ± 8.47 | 12 | 113.1 ± 7.16 | −19.10 (−26.24 to −11.96); <0.001 | |
| TOTAL SERIES | Total CoQ10 (pmol/mg protein) | 41 | 139.14 ± 64.49 | 74 | 181.27 ± 78.20 | Random-effects model p = 0.03358 | |
| Cerebral cortex | Hargreaves et al. 2008 [20] | Total CoQ10 (pmol/mg protein) | 13 | 128.6 ± 61.4 | 13 | 218.6 ± 55.7 | 90.00 (42.55 to 137.45); 0.0007 |
| Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 7 | 276.02 ± 71.37 | 37 | 259.39 ± 107.09 | −16.63 (−102.09 to 68.84); 0.697 | |
| TOTAL SERIES | Total CoQ10 (pmol/mg) | 20 | 180.20 ± 99.89 | 50 | 248.78 ± 97.53 | Random-effects model p = 0.143 | |
| Skin fibroblasts | Del Hoyo et al., 2010 [23] | Total CoQ10/CS | 20 | 1.16 ± 0.33 | 19 | 0.97 ± 0.25 | −0.19 (−0.38 to 0.00); 0.051 |
| Del Hoyo et al., 2010 [23] | Reduced CoQ10/CS | 20 | 0.41 ± 0.16 | 19 | 0.34 ± 0.11 | −0.07 (−0.16 to 0.02); 0.122 | |
| Del Hoyo et al., 2010 [23] | Oxidized CoQ10/CS | 20 | 0.75 ± 0.26 | 19 | 0.63 ± 0.23 | −0.12 (−0.28 to 0.04); 0.136 | |
| Del Hoyo et al., 2010 [23] | Total CoQ10/mg protein | 20 | 86.27 ± 29.07 | 19 | 71.86 ± 26.38 | −14.41 (−32.45 to 3.63); 0.114 | |
| Del Hoyo et al., 2010 [23] | Reduced CoQ10/mg protein | 20 | 24.50 ± 7.38 | 19 | 24.50 ± 7.38 | 0.00 (−4.79 to 4.79); 1.000 | |
| Del Hoyo et al., 2010 [23] | Oxidized CoQ10/mg protein | 20 | 56.49 ± 25.20 | 19 | 47.31 ± 23.50 | −9.18 (−25.01 to 6.65); 0.248 | |
| Del Hoyo et al., 2010 [23] | Oxidized CoQ10/Reduced CoQ10 | 20 | 0.60 ± 0.27 | 19 | 0.62 ± 0.27 | 0.02 (−0.16 to 0.20); 0.818 |
| Tissue | Author, Year [Ref] | Parameter | MSA N | MSA Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
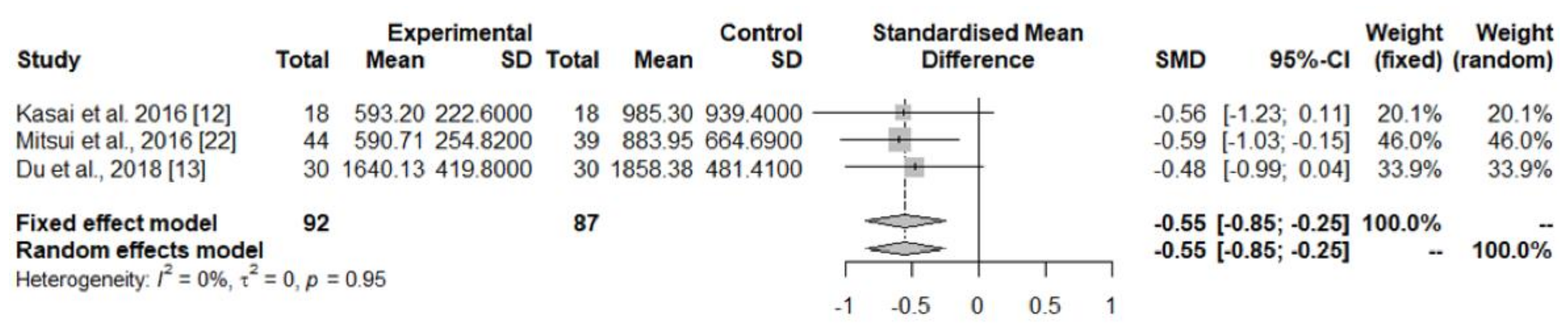
| Serum/plasma | Kasai et al. 2016 [14] | Total CoQ10 (nmol/L) | 18 | 593.2 ± 222.6 | 18 | 985.3 ± 939.4 | 392.10 (−70.34 to 854.54); 0.094 |
| Mitsui et al., 2016 [24] | Total CoQ10 (nmol/L) | 44 | 590.71 ± 254.82 | 39 | 833.95 ± 664.69 | 243.24 (28.09 to 458.39); 0.027 | |
| Du et al., 2018 [15] | Total CoQ10 (nmol/L) | 30 | 1640.13 ± 419.80 | 30 | 1858.38 ± 481.41 | 218.25 (−15.18 to 451.68); 0.066 | |
| TOTAL SERIES | Total CoQ10 (nmol/L) | 92 | 933.40 ± 583.47 | 87 | 1218.52 ± 817.98 | Random-effects model p = 0.001 | |
| Kasai et al. 2016 [14] | Total CoQ10/cholesterol | 18 | 3.04 ± 1.23 | 18 | 5.92 ± 5.88 | 2.88 (0.00 to 5.76); 0.050 | |
| Kasai et al. 2016 [14] | Oxidized CoQ10 (nmol/L) | 18 | 520.7 ± 202.8 | 18 | 900.2 ± 890.6 | 379.50 (−58.02 to 817.02); 0.087 | |
| Kasai et al. 2016 [14] | Reduced CoQ10 (nmol/L) | 18 | 72.4 ± 34.1 | 18 | 85.2 ± 66.6 | 12.80 (17.64 to 48.64); 0.473 | |
| CSF | Compta et al., 2018 [19] | Total CoQ10 (nmol/L) | 20 | 26.63 ± 3.70 | 15 | 36.02 ± 7.10 | 9.37 (5.61 to 13.13); <0.0001 |
| Brain | |||||||
| Cerebellum cortex | Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 20 | 169.30 ± 49.71 | 37 | 241.87 ± 57.70 | 72.57 (41.94 to 103.20); <0.001 |
| Barca et al., 2016 [22] | Total CoQ10 (pmol/mg) | 12 | 68.1 ± 10.03 | 12 | 113.1 ± 7.16 | 45.00 (37.62 to 52.38); <0.001 | |
| TOTAL SERIES | Total CoQ10 (pmol/mg) | 32 | 131.35 ± 63.47 | 49 | 210.33 ± 75.09 | Random-effects model p = 0.0977 | |
| Cerebral cortex frontal | Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 20 | 260.44 ± 70.22 | 37 | 259.39 ± 107.09 | −1.05 (−54.43 to 52.33); 0.969 |
| Cerebral cortex occipital | Barca et al., 2016 [22] | Total CoQ10 (nmol/mg protein) | 10 | 277.1 ± 29.73 | 9 | 267.3 ± 21.88 | −9.80 (−35.32 to 15.72); 0.429 |
| Striatum | Barca et al., 2016 [22] | Total CoQ10 (nmol/mg protein) | 7 | 244.2 ± 27.16 | 7 | 230.8 ± 28.62 | −13.40 (−45.89 to 10.09); 0.387 |
| Skin fibroblasts | Monzio Compagnoni et al., 2010 [25] | Total CoQ10 (pg/mg protein) | 14 | 27.83 ± 1.44 | 6 | 45.22 ± 3.48 | 17.39 (15.13 to 19.65); <0.001 |
| Lewy Body Dementia (LBD) | |||||||
|---|---|---|---|---|---|---|---|
| Tissue | Author, Year [Ref] | Parameter | LBD N | LBD Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Serum/plasma | Molina et al., 2002 [26] | Total CoQ10 (nmol/L) | 18 | 960.6 ± 359.1 | 20 | 1205.2 ± 362.2 | 244.60 (6.90 to 482.30); 0.044 |
| Gironi et al. 2011 [27] | Total CoQ10 (nmol/L) | 7 | 645.17 ± 290 | 66 | 622.12 ± 227.14 | −23.05 (−207.81 to 161.71); 0.804 | |
| TOTAL SERIES | Total CoQ10 (nmol/L) | 25 | 872.28 ± 365.05 | 86 | 757.72 ± 360.79 | Random-effects model: p = 0.409 | |
| Molina et al., 2002 [7] | Total CoQ10/cholesterol | 18 | 4.67 ± 1.75 | 20 | 5.05 ± 1.52 | 0.38 (−0.70 to 1.46); 0.478 | |
| Brain | |||||||
| Cerebellum cortex | Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 20 | 169.30 ± 49.71 | 37 | 241.87 ± 57.70 | 72.57 (41.94 to 103.20); <0.001 |
| Cerebral cortex frontal | Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 20 | 260.44 ± 70.22 | 37 | 259.39 ± 107.09 | −1.05 (−54.43 to 52.33); 0.969 |
| Progressive Supranuclear Palsy (PSP) | |||||||
| Tissue | Author, Year [Ref] | Parameter | PSP N | PSP Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| CSF | Compta et al., 2018 [19] | Total CoQ10 (nmol/L) | 10 | 47.67 ± 4.05 | 15 | 36.02 ± 7.10 | −11.65 (−16.79 to −6.51); 0.0001 |
| Cortical Basal Degeneration (CBD) | |||||||
| TISSUE | Author, Year [Ref] | Parameter | CBD N | CBD Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Cerebellum cortex | Schottlaender et al., 2016 [21] | Total CoQ10 (pmol/mg) | 15 | 271.18 ± 76.21 | 37 | 241.87 ± 57.70 | −29.31 (−68.31 to 9.69); 0.137 |
| Authors, Year [Ref] | Study Setting | Type of Study | Main Findings | Level of Evidence (Quality Score) |
|---|---|---|---|---|
| Strijks et al., 1997 [29] | 10 patients diagnosed with PD. Dosage of 200 mg/day. Assessment of motor performance with UPDRS and motor test. | 3 months open-label study |
| II (NA) |
| Shults et al., 2002 [30] | Eighty subjects with early PD not requiring treatment for their disability. Dosages of 300, 600, or 1200 mg/day Evaluation with the UPDRS at the screening, baseline, and 1-, 4-, 8-, 12-, and 16-month visits. Follow-up of 16 months or until disability requiring treatment with levodopa. | Multicenter, randomized, parallel-group, placebo-controlled, double-blind, dosage-ranging trial. |
| I (>50%) |
| Müller et al., 2003 [31] | Twenty-eight treated and stable PD patients. Dosage of 360 mg/day for 4 weeks. Scoring of PD symptoms, and visual function using the Farnsworth–Munsell 100 Hue test (FMT). | Monocenter, parallel-group, placebo-controlled, double-blind trial |
| I (>50%) |
| NINDS NET-PD Investigators 2007 [32] | Seventy-one untreated early PD patients assigned to CoQ10 therapy (2400 mg/day), 71 to GPI-1485, and 71 to placebo. Measurement of change in total UPDRS scores and subscores, Hoehn & Yahr staging, and Schwabb & England scale scores, either at the time requiring symptomatic therapy or at 12 months. | Randomized, double-blind, calibrated futility clinical trial |
| I (>50%) |
| Storch et al., 2007 [33] | One hundred thirty-one patients with PD without motor fluctuations and a stable antiparkinsonian treatment. Treatment with placebo or nanoparticular CoQ10 (100 mg 3 times a day, equivalent to 1200 mg/day of standard formulation) for 3 months. The stratification criterion was levodopa treatment. Evaluation with the UPDRS (sum score of parts II and III) at baseline, 1, 2, and 3 months at each visit monthly. | Multicenter, randomized, double-blind, placebo-controlled, stratified, parallel-group, single-dose trial. |
| I (>50%) |
| Parkinson Study Group QE3 Investigators [34] | Six hundred patients diagnosed with PD (from 67 hospitals in the USA) in the previous 5 years, free of dopaminergic therapy in the previous 3 months, with Hoehn & Yahr stage of 2.5 or less. Two hundred were assigned to CoQ10 1200 mg/day, 200 to CoQ10 2400 mg/day and 200 to placebo. All patients were taking vitamin E 1200 IU/day. Evaluation at 16 months from baseline or until a disability requiring dopaminergic treatment. The study was powered to detect a 3-point difference between active treatment and placebo. | Phase III randomized, placebo-controlled, double-blind clinical trial |
| I (>50%) |
| Jie et al., 2014 [35] | Eighty-eight patients diagnosed with PD and treated with levodopa. Forty-four were assigned to CoQ10 375–750 mg/day, and 44 to placebo Evaluation with the Webster Scale at baseline and 3 months | Monocenter, randomized, placebo-controlled, double-blind clinical trial |
| I (>50%) |
| Wang et al., 2014 [36] | Thirty-nine patients diagnosed with PD under conventional therapy. Twenty-one were assigned to CoQ10 450 or 1200 mg/day, and 18 to placebo as add-on therapy Evaluation with the UPDRS III and Webster Scale at baseline and 36 weeks | Monocenter, randomized, placebo-controlled, double-blind clinical trial |
| I (>50%) |
| Li et al., 2015 [37] | Seventy-five patients diagnosed with PD and MCI. Random assignation to treatment with CoQ10 100 mg b.i.d. and creatine 5 mg b.i.d. or to placebo. Evaluation with the UPDRS part III, and MoCa at 12 and 18 months. | Phase III randomized, placebo-controlled, double-blind clinical trial |
| I (>50%) |
| Yoritaka et al., 2015 [38] | Twenty-six patients with PD experiencing wearing off (group A) and 22 early PD patients without levodopa (with or without a dopamine agonist, group B). Treatment with 300 mg/day of ubiquinol-10 or placebo for 48 weeks (Group A, 14 ubiquinol-10, 12 placeboes) or 96 weeks (Group B, 14 ubiquinol-10, 8 placeboes). | Randomized, double-blind, placebo-controlled, parallel-group pilot trial |
| I (>50%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E.; Agúndez, J.A.G. Coenzyme Q10 and Parkinsonian Syndromes: A Systematic Review. J. Pers. Med. 2022, 12, 975. https://doi.org/10.3390/jpm12060975
Jiménez-Jiménez FJ, Alonso-Navarro H, García-Martín E, Agúndez JAG. Coenzyme Q10 and Parkinsonian Syndromes: A Systematic Review. Journal of Personalized Medicine. 2022; 12(6):975. https://doi.org/10.3390/jpm12060975
Chicago/Turabian StyleJiménez-Jiménez, Félix Javier, Hortensia Alonso-Navarro, Elena García-Martín, and José A. G. Agúndez. 2022. "Coenzyme Q10 and Parkinsonian Syndromes: A Systematic Review" Journal of Personalized Medicine 12, no. 6: 975. https://doi.org/10.3390/jpm12060975
APA StyleJiménez-Jiménez, F. J., Alonso-Navarro, H., García-Martín, E., & Agúndez, J. A. G. (2022). Coenzyme Q10 and Parkinsonian Syndromes: A Systematic Review. Journal of Personalized Medicine, 12(6), 975. https://doi.org/10.3390/jpm12060975








