Gastrointestinal Dysfunction in Parkinson’s Disease: Current and Potential Therapeutics
Abstract
:1. Introduction
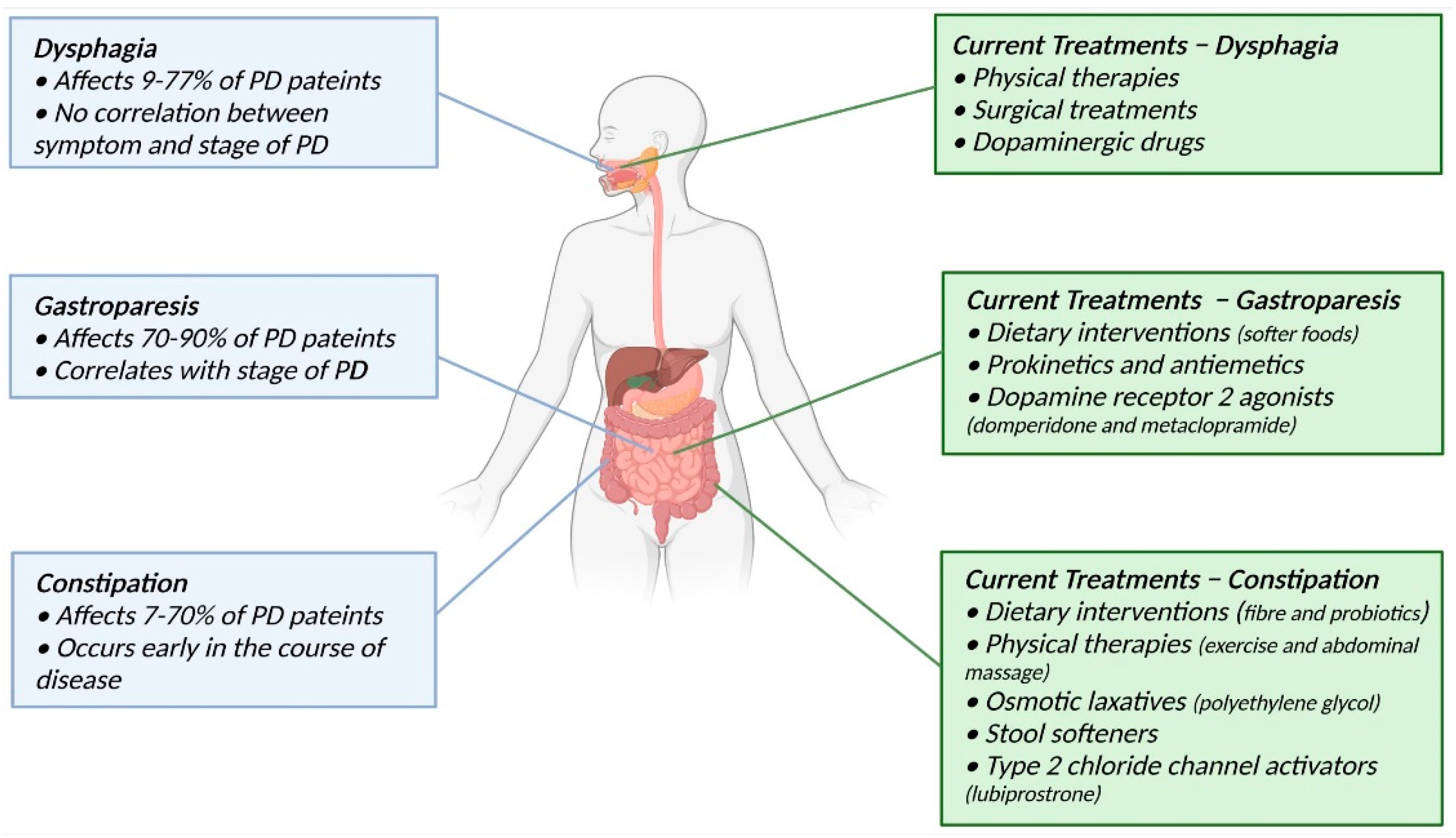
2. Gastrointestinal Dysfunction in Parkinson’s Disease
2.1. Dysphagia
2.2. Gastroparesis
2.3. Constipation
3. Current Treatments for Gastrointestinal Dysfunction in Parkinson’s Disease
3.1. Dysphagia
3.2. Gastroparesis
3.3. Constipation
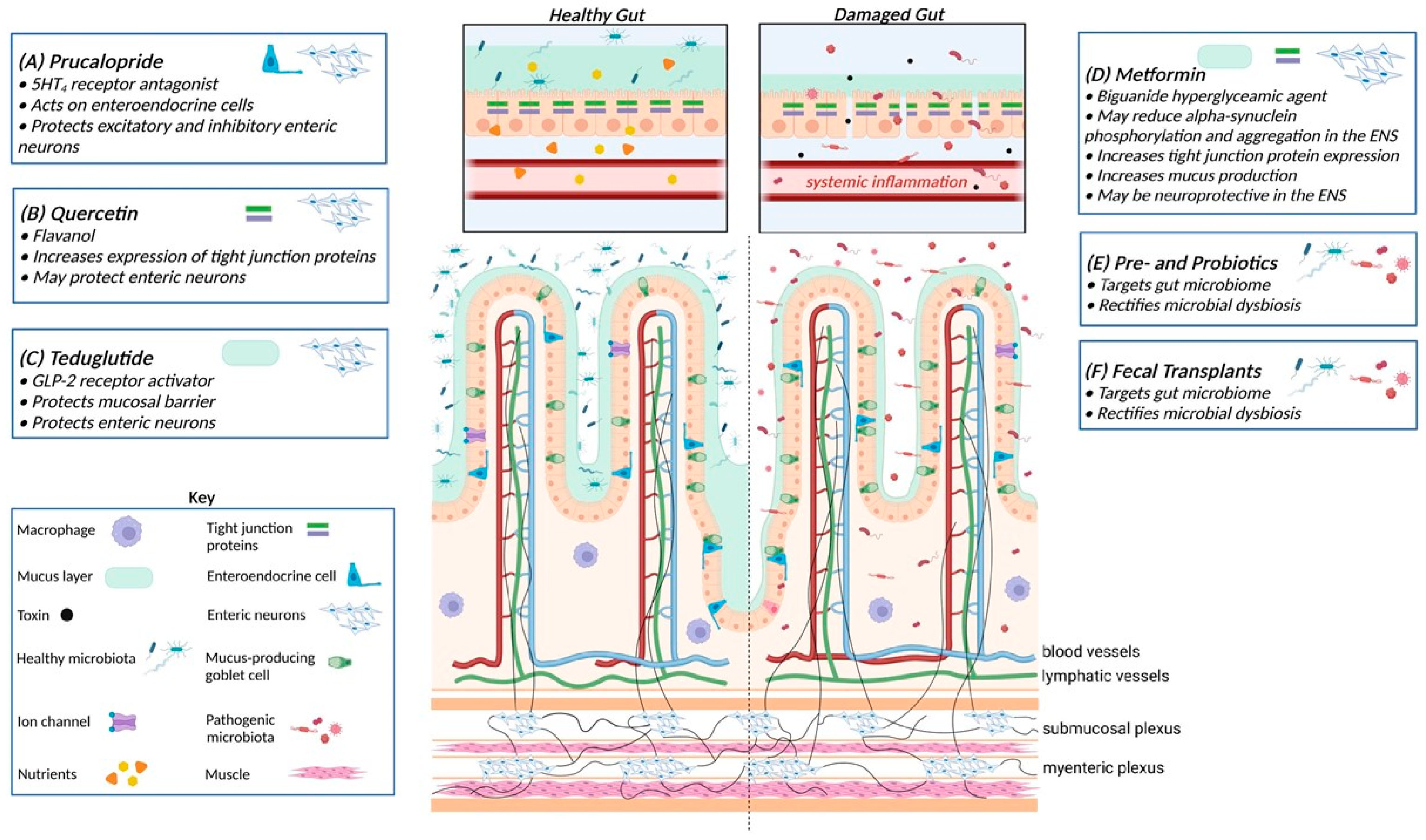
4. Potential Treatments for Gastrointestinal Dysfunction in Parkinson’s Disease
5. Targeting Enteric Neurons
5.1. 5-Hydroxytryptamine 4 (5-HT4) Receptor Agonists
5.2. Flavonoids
6. Targeting the Intestinal Barrier
6.1. Teduglutide
6.2. Metformin
7. Targeting the Gut Microbiota
Prebiotics, Probiotics, and Fecal Transplants
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 5-HT4 | 5-hydroxytryptamine/serotonin 4 |
| CNS | Central nervous system |
| DMV | Dorsal motor nucleus of the vagus |
| ENS | Enteric nervous system |
| FMT | Fecal microbiota transplant |
| FDA | Food and Drug Administration |
| GI | Gastrointestinal |
| GLP-2 | Glucagon-like peptide-2 |
| IBD | Inflammatory bowel disease |
| IBS | Inflammatory bowel syndrome |
| ITT | Intestinal transit time |
| LB | Lewy bodies |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| NMES | Neuromuscular electrical stimulation |
| PD | Parkinson’s disease |
| PEG | Polyethylene glycol |
| SNpc | Substantia nigra pars compacta |
| TJ | Tight junction |
| VIP | Vasoactive intestinal peptide |
References
- Nussbaum, R.L.; Ellis, C.E. Alzheimer’s disease and Parkinson’s disease. N. Engl. J. Med. 2003, 348, 1356–1364. [Google Scholar] [CrossRef] [Green Version]
- Fahn, S. Description of Parkinson’s disease as a clinical syndrome. Ann. N. Y. Acad. Sci. 2003, 991, 1–14. [Google Scholar] [CrossRef]
- Lees, A.J.; Hardy, J.; Revesz, T. Parkinson’s disease. Lancet 2009, 373, 2055–2066. [Google Scholar] [CrossRef]
- Shahed, J.; Jankovic, J. Motor symptoms in Parkinson’s disease. Handb. Clin. Neurol. 2007, 83, 329–342. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Takahashi, H.; Takeda, S.; Ohama, E.; Ikuta, F. Parkinson’s disease: The presence of Lewy bodies in Auerbach’s and Meissner’s plexuses. Acta Neuropathol. 1988, 76, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Del Tredici, K.; Duda, J.E. Peripheral Lewy body pathology in Parkinson’s disease and incidental Lewy body disease: Four cases. J. Neurol. Sci. 2011, 310, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, G.; Liu, J. Autonomic dysfunction in Parkinson’s disease: Implications for pathophysiology, diagnosis, and treatment. Neurobiol. Dis. 2020, 134, 104700. [Google Scholar] [CrossRef]
- Fasano, A.; Visanji, N.P.; Liu, L.W.; Lang, A.E.; Pfeiffer, R.F. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2015, 14, 625–639. [Google Scholar] [CrossRef]
- Lorefalt, B.; Ganowiak, W.; Palhagen, S.; Toss, G.; Unosson, M.; Granerus, A.K. Factors of importance for weight loss in elderly patients with Parkinson’s disease. Acta Neurol. Scand. 2004, 110, 180–187. [Google Scholar] [CrossRef]
- Park, K.; Oeda, T.; Kohsaka, M.; Tomita, S.; Umemura, A.; Sawada, H. Low body mass index and life prognosis in Parkinson’s disease. Parkinsonism Relat. Disord. 2018, 55, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Goldman, J.G.; Postuma, R. Premotor and nonmotor features of Parkinson’s disease. Curr. Opin. Neurol. 2014, 27, 434–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaudhuri, K.R.; Healy, D.G.; Schapira, A.H.; National Institute for Clinical Excellence. Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol. 2006, 5, 235–245. [Google Scholar] [CrossRef]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.J. The enteric nervous system and gastrointestinal innervation: Integrated local and central control. Adv. Exp. Med. Biol. 2014, 817, 39–71. [Google Scholar] [CrossRef]
- Annerino, D.M.; Arshad, S.; Taylor, G.M.; Adler, C.H.; Beach, T.G.; Greene, J.G. Parkinson’s disease is not associated with gastrointestinal myenteric ganglion neuron loss. Acta Neuropathol. 2012, 124, 665–680. [Google Scholar] [CrossRef] [Green Version]
- Anderson, G.; Noorian, A.R.; Taylor, G.; Anitha, M.; Bernhard, D.; Srinivasan, S.; Greene, J.G. Loss of enteric dopaminergic neurons and associated changes in colon motility in an MPTP mouse model of Parkinson’s disease. Exp. Neurol. 2007, 207, 4–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaumette, T.; Lebouvier, T.; Aubert, P.; Lardeux, B.; Qin, C.; Li, Q.; Accary, D.; Bezard, E.; Bruley des Varannes, S.; Derkinderen, P.; et al. Neurochemical plasticity in the enteric nervous system of a primate animal model of experimental Parkinsonism. Neurogastroenterol. Motil. 2009, 21, 215–222. [Google Scholar] [CrossRef]
- Lebouvier, T.; Neunlist, M.; Bruley des Varannes, S.; Coron, E.; Drouard, A.; N’Guyen, J.M.; Chaumette, T.; Tasselli, M.; Paillusson, S.; Flamand, M.; et al. Colonic biopsies to assess the neuropathology of Parkinson’s disease and its relationship with symptoms. PLoS ONE 2010, 5, e12728. [Google Scholar] [CrossRef] [Green Version]
- Zhu, H.C.; Zhao, J.; Luo, C.Y.; Li, Q.Q. Gastrointestinal dysfunction in a Parkinson’s disease rat model and the changes of dopaminergic, nitric oxidergic, and cholinergic neurotransmitters in myenteric plexus. J. Mol. Neurosci. 2012, 47, 15–25. [Google Scholar] [CrossRef] [Green Version]
- Forsyth, C.B.; Shannon, K.M.; Kordower, J.H.; Voigt, R.M.; Shaikh, M.; Jaglin, J.A.; Estes, J.D.; Dodiya, H.B.; Keshavarzian, A. Increased intestinal permeability correlates with sigmoid mucosa alpha-synuclein staining and endotoxin exposure markers in early Parkinson’s disease. PLoS ONE 2011, 6, e28032. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clairembault, T.; Leclair-Visonneau, L.; Coron, E.; Bourreille, A.; Le Dily, S.; Vavasseur, F.; Heymann, M.F.; Neunlist, M.; Derkinderen, P. Structural alterations of the intestinal epithelial barrier in Parkinson’s disease. Acta Neuropathol. Commun. 2015, 3, 12. [Google Scholar] [CrossRef] [Green Version]
- Romano, S.; Savva, G.M.; Bedarf, J.R.; Charles, I.G.; Hildebrand, F.; Narbad, A. Meta-analysis of the Parkinson’s disease gut microbiome suggests alterations linked to intestinal inflammation. NPJ Parkinsons Dis. 2021, 7, 27. [Google Scholar] [CrossRef]
- Muller, T.; Erdmann, C.; Bremen, D.; Schmidt, W.E.; Muhlack, S.; Woitalla, D.; Goetze, O. Impact of gastric emptying on levodopa pharmacokinetics in Parkinson disease patients. Clin. Neuropharmacol. 2006, 29, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Clifford, T.; Finnerty, J. The dental awareness and needs of a Parkinson’s disease population. Gerodontology 1995, 12, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Coates, C.; Bakheit, A.M. Dysphagia in Parkinson’s disease. Eur. Neurol. 1997, 38, 49–52. [Google Scholar] [CrossRef]
- Kaye, J.; Gage, H.; Kimber, A.; Storey, L.; Trend, P. Excess burden of constipation in Parkinson’s disease: A pilot study. J. Mov. Disord. 2006, 21, 1270–1273. [Google Scholar] [CrossRef] [PubMed]
- Proulx, M.; de Courval, F.P.; Wiseman, M.A.; Panisset, M. Salivary production in Parkinson’s disease. J. Mov. Disord. 2005, 20, 204–207. [Google Scholar] [CrossRef]
- Edwards, L.L.; Quigley, E.M.; Harned, R.K.; Hofman, R.; Pfeiffer, R.F. Characterization of swallowing and defecation in Parkinson’s disease. Am. J. Gastroenterol. 1994, 89, 15–25. [Google Scholar]
- Yang, T.; Zhan, Z.; Zhang, L.; Zhu, J.; Liu, Y.; Zhang, L.; Ge, J.; Zhao, Y.; Zhang, L.; Dong, J. Prevalence and Risk Factors for Malnutrition in Patients with Parkinson’s Disease. Front. Neurol. 2020, 11, 533731. [Google Scholar] [CrossRef]
- Dauer, W.; Przedborski, S. Parkinson’s disease: Mechanisms and models. Neuron 2003, 39, 889–909. [Google Scholar] [CrossRef] [Green Version]
- Lang, A.E.; Lozano, A.M. Parkinson’s disease. First of two parts. N. Engl. J. Med. 1998, 339, 1044–1053. [Google Scholar] [CrossRef]
- Marsden, C.D. Parkinson’s disease. Lancet 1990, 335, 948–952. [Google Scholar] [CrossRef]
- Ross, G.W.; Petrovitch, H.; Abbott, R.D.; Nelson, J.; Markesbery, W.; Davis, D.; Hardman, J.; Launer, L.; Masaki, K.; Tanner, C.M.; et al. Parkinsonian signs and substantia nigra neuron density in decendents elders without PD. Ann. Neurol. 2004, 56, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Barone, P.; Antonini, A.; Colosimo, C.; Marconi, R.; Morgante, L.; Avarello, T.P.; Bottacchi, E.; Cannas, A.; Ceravolo, G.; Ceravolo, R. The PRIAMO study: A multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov. Disord. Off. J. Mov. Disord. Soc. 2009, 24, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Bernal-Pacheco, O.; Limotai, N.; Go, C.L.; Fernandez, H.H. Nonmotor manifestations in Parkinson disease. Neurologist 2012, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Dubow, J.S. Autonomic dysfunction in Parkinson’s disease. Dis. Mon. 2007, 53, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Lyons, K.E.; Pahwa, R. The impact and management of nonmotor symptoms of Parkinson’s disease. Am. J. Manag. Care 2011, 17, S308. [Google Scholar] [PubMed]
- Knudsen, K.; Fedorova, T.D.; Bekker, A.C.; Iversen, P.; Østergaard, K.; Krogh, K.; Borghammer, P. Objective colonic dysfunction is far more prevalent than subjective constipation in Parkinson’s disease: A colon transit and volume study. J. Parkinson Dis. 2017, 7, 359–367. [Google Scholar] [CrossRef]
- Su, A.; Gandhy, R.; Barlow, C.; Triadafilopoulos, G. A practical review of gastrointestinal manifestations in Parkinson’s disease. Parkinsonism Relat. Disord. 2017, 39, 17–26. [Google Scholar] [CrossRef]
- Srivanitchapoom, P.; Pandey, S.; Hallett, M. Drooling in Parkinson’s disease: A review. Parkinsonism Relat. Disord. 2014, 20, 1109–1118. [Google Scholar] [CrossRef] [Green Version]
- Umemoto, G.; Tsuboi, Y.; Kitashima, A.; Furuya, H.; Kikuta, T. Impaired food transportation in Parkinson’s disease related to lingual bradykinesia. Dysphagia 2011, 26, 250–255. [Google Scholar] [CrossRef]
- Kikuchi, A.; Baba, T.; Hasegawa, T.; Kobayashi, M.; Sugeno, N.; Konno, M.; Miura, E.; Hosokai, Y.; Ishioka, T.; Nishio, Y.; et al. Hypometabolism in the supplementary and anterior cingulate cortices is related to dysphagia in Parkinson’s disease: A cross-sectional and 3-year longitudinal cohort study. BMJ Open 2013, 3, e002249. [Google Scholar] [CrossRef]
- Lee, K.D.; Koo, J.H.; Song, S.H.; Jo, K.D.; Lee, M.K.; Jang, W. Central cholinergic dysfunction could be associated with oropharyngeal dysphagia in early Parkinson’s disease. J. Neural. Transm. 2015, 122, 1553–1561. [Google Scholar] [CrossRef]
- Suntrup, S.; Teismann, I.; Bejer, J.; Suttrup, I.; Winkels, M.; Mehler, D.; Pantev, C.; Dziewas, R.; Warnecke, T. Evidence for adaptive cortical changes in swallowing in Parkinson’s disease. Brain 2013, 136, 726–738. [Google Scholar] [CrossRef]
- Picillo, M.; Amboni, M.; Erro, R.; Longo, K.; Vitale, C.; Moccia, M.; Pierro, A.; Santangelo, G.; De Rosa, A.; De Michele, G.; et al. Gender differences in non-motor symptoms in early, drug naive Parkinson’s disease. J. Neurol. 2013, 260, 2849–2855. [Google Scholar] [CrossRef] [PubMed]
- Johnston, B.T.; Li, Q.; Castell, J.A.; Castell, D.O. Swallowing and esophageal function in Parkinson’s disease. Am. J. Gastroenterol. 1995, 90, 1741–1746. [Google Scholar] [PubMed]
- Martinez-Ramirez, D.; Almeida, L.; Giugni, J.C.; Ahmed, B.; Higuchi, M.A.; Little, C.S.; Chapman, J.P.; Mignacca, C.; Wagle Shukla, A.; Hess, C.W.; et al. Rate of aspiration pneumonia in hospitalized Parkinson’s disease patients: A cross-sectional study. BMC Neurol. 2015, 15, 104. [Google Scholar] [CrossRef] [Green Version]
- Fuh, J.L.; Lee, R.C.; Wang, S.J.; Lin, C.H.; Wang, P.N.; Chiang, J.H.; Liu, H.C. Swallowing difficulty in Parkinson’s disease. Clin. Neurol. Neurosurg. 1997, 99, 106–112. [Google Scholar] [CrossRef]
- Potulska, A.; Friedman, A.; Krolicki, L.; Spychala, A. Swallowing disorders in Parkinson’s disease. Parkinsonism Relat. Disord. 2003, 9, 349–353. [Google Scholar] [CrossRef]
- Lin, C.W.; Chang, Y.C.; Chen, W.S.; Chang, K.; Chang, H.Y.; Wang, T.G. Prolonged swallowing time in dysphagic Parkinsonism patients with aspiration pneumonia. Arch. Phys. Med. Rehabil. 2012, 93, 2080–2084. [Google Scholar] [CrossRef]
- Heetun, Z.S.; Quigley, E.M. Gastroparesis and Parkinson’s disease: A systematic review. Parkinsonism Relat. Disord. 2012, 18, 433–440. [Google Scholar] [CrossRef]
- Tang, D.M.; Friedenberg, F.K. Gastroparesis: Approach, diagnostic evaluation, and management. Dis. Mon. 2011, 57, 74–101. [Google Scholar] [CrossRef]
- Goetze, O.; Nikodem, A.B.; Wiezcorek, J.; Banasch, M.; Przuntek, H.; Mueller, T.; Schmidt, W.E.; Woitalla, D. Predictors of gastric emptying in Parkinson’s disease. Neurogastroenterol. Motil. 2006, 18, 369–375. [Google Scholar] [CrossRef]
- Siddiqui, M.F.; Rast, S.; Lynn, M.J.; Auchus, A.P.; Pfeiffer, R.F. Autonomic dysfunction in Parkinson’s disease: A comprehensive symptom survey. Parkinsonism Relat. Disord. 2002, 8, 277–284. [Google Scholar] [CrossRef]
- Braak, H.; Rub, U.; Gai, W.P.; Del Tredici, K. Idiopathic Parkinson’s disease: Possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. J. Neural. Transm. 2003, 110, 517–536. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.; Coffin, B.; Gourcerol, G. Gastroparesis in Parkinson Disease: Pathophysiology, and Clinical Management. Brain Sci. 2021, 11, 831. [Google Scholar] [CrossRef] [PubMed]
- Doi, H.; Sakakibara, R.; Sato, M.; Masaka, T.; Kishi, M.; Tateno, A.; Tateno, F.; Tsuyusaki, Y.; Takahashi, O. Plasma levodopa peak delay and impaired gastric emptying in Parkinson’s disease. J. Neurol. Sci. 2012, 319, 86–88. [Google Scholar] [CrossRef] [PubMed]
- Mathers, S.E.; Kempster, P.A.; Swash, M.; Lees, A.J. Constipation and paradoxical puborectalis contraction in anismus and Parkinson’s disease: A dystonic phenomenon? J. Neurol. Neurosurg. Psychiatry 1988, 51, 1503–1507. [Google Scholar] [CrossRef] [Green Version]
- Jost, W.; Schimrigk, K.J.K.W. Constipation in Parkinson’s disease. Klin. Wochenschr. 1991, 69, 906–909. [Google Scholar] [CrossRef] [Green Version]
- Singer, C.; Weiner, W.J.; Sanchez-Ramos, J.R. Autonomic dysfunction in men with Parkinson’s disease. Eur. Neurol. 1992, 32, 134–140. [Google Scholar] [CrossRef]
- Yu, B.; Xiao, Z.Y.; Li, J.Z.; Yuan, J.; Liu, Y.M. Study of an integrated non-motor symptoms questionnaire for Parkinson’s disease. Chin. Med. J. 2010, 123, 1436–1440. [Google Scholar]
- Postuma, R.; Romenets, S.R.; Rakheja, R. Physician Guide: Non-Motor Symptoms of Parkinson’s Disease; McGill University Health Centre: Montreal, QC, Canada, 2012. [Google Scholar]
- Knudsen, K.; Krogh, K.; Ostergaard, K.; Borghammer, P. Constipation in parkinson’s disease: Subjective symptoms, objective markers, and new perspectives. Mov. Disord. 2017, 32, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ramallo, H.; Báez-Gutiérrez, N.; Prado-Mel, E.; Alfaro-Lara, E.R.; Santos-Ramos, B.; Sánchez-Fidalgo, S. Association between Anticholinergic Burden and Constipation: A Systematic Review. Healthcare 2021, 9, 581. [Google Scholar] [CrossRef] [PubMed]
- Andrews, C.N.; Storr, M. The pathophysiology of chronic constipation. Can. J. Gastroenterol. 2011, 25 (Suppl. B), 16B–21B. [Google Scholar] [CrossRef]
- Abbott, R.D.; Petrovitch, H.; White, L.R.; Masaki, K.H.; Tanner, C.M.; Curb, J.D.; Grandinetti, A.; Blanchette, P.L.; Popper, J.S.; Ross, G.W. Frequency of bowel movements and the future risk of Parkinson’s disease. Neurology 2001, 57, 456–462. [Google Scholar] [CrossRef]
- Ueki, A.; Otsuka, M. Life style risks of Parkinson’s disease: Association between decreased water intake and constipation. J. Neurol. 2004, 251 (Suppl. 7), vII18–vII23. [Google Scholar] [CrossRef] [PubMed]
- El Sharkawi, A.; Ramig, L.; Logemann, J.A.; Pauloski, B.R.; Rademaker, A.W.; Smith, C.H.; Pawlas, A.; Baum, S.; Werner, C. Swallowing and voice effects of Lee Silverman Voice Treatment (LSVT): A pilot study. J. Neurol. Neurosurg. Psychiatry 2002, 72, 31–36. [Google Scholar] [CrossRef]
- Troche, M.S.; Huebner, I.; Rosenbek, J.C.; Okun, M.S.; Sapienza, C.M. Respiratory-swallowing coordination and swallowing safety in patients with Parkinson’s disease. Dysphagia 2011, 26, 218–224. [Google Scholar] [CrossRef]
- Manor, Y.; Mootanah, R.; Freud, D.; Giladi, N.; Cohen, J.T. Video-assisted swallowing therapy for patients with Parkinson’s disease. Parkinsonism Relat. Disord. 2013, 19, 207–211. [Google Scholar] [CrossRef]
- Adamovich, S.V.; Berkinblit, M.B.; Hening, W.; Sage, J.; Poizner, H. The interaction of visual and proprioceptive inputs in pointing to actual and remembered targets in Parkinson’s disease. Neuroscience 2001, 104, 1027–1041. [Google Scholar] [CrossRef]
- Born, L.J.; Harned, R.H.; Rikkers, L.F.; Pfeiffer, R.F.; Quigley, E.M. Cricopharyngeal dysfunction in Parkinson’s disease: Role in dysphagia and response to myotomy. Mov. Disord. 1996, 11, 53–58. [Google Scholar] [CrossRef]
- Byrne, K.G.; Pfeiffer, R.; Quigley, E.M. Gastrointestinal dysfunction in Parkinson’s disease. A report of clinical experience at a single center. J. Clin. Gastroenterol. 1994, 19, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Heijnen, B.J.; Speyer, R.; Baijens, L.W.; Bogaardt, H.C. Neuromuscular electrical stimulation versus traditional therapy in patients with Parkinson’s disease and oropharyngeal dysphagia: Effects on quality of life. Dysphagia 2012, 27, 336–345. [Google Scholar] [CrossRef] [Green Version]
- Warnecke, T.; Suttrup, I.; Schroder, J.B.; Osada, N.; Oelenberg, S.; Hamacher, C.; Suntrup, S.; Dziewas, R. Levodopa responsiveness of dysphagia in advanced Parkinson’s disease and reliability testing of the FEES-Levodopa-test. Parkinsonism Relat. Disord. 2016, 28, 100–106. [Google Scholar] [CrossRef]
- Tison, F.; Wiart, L.; Guatterie, M.; Fouillet, N.; Lozano, V.; Henry, P.; Barat, M. Effects of central dopaminergic stimulation by apomorphine on swallowing disorders in Parkinson’s disease. Mov. Disord. 1996, 11, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Calne, D.; Shaw, D.; Spiers, A.; Stern, G. Swallowing in parkinsonism. Br. J. Radiol. 1970, 43, 456–457. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.; Leow, L.; Huckabee, M.-L.; Frampton, C.; Anderson, T. A pilot study of respiration and swallowing integration in Parkinson’s disease: “On” and “Off” levodopa. Dysphagia 2008, 23, 76–81. [Google Scholar] [CrossRef]
- Bushmann, M.; Dobmeyer, S.M.; Leeker, L.; Perlmutter, J.S. Swallowing abnormalities and their response to treatment in Parkinson’s disease. Neurology 1989, 39, 1309–1314. [Google Scholar] [CrossRef]
- Muller, B.; Assmus, J.; Larsen, J.P.; Haugarvoll, K.; Skeie, G.O.; Tysnes, O.B.; ParkWest Study Group. Autonomic symptoms and dopaminergic treatment in de novo Parkinson’s disease. Acta Neurol. Scand. 2013, 127, 290–294. [Google Scholar] [CrossRef]
- Hunter, P.C.; Crameri, J.; Austin, S.; Woodward, M.C.; Hughes, A.J. Response of parkinsonian swallowing dysfunction to dopaminergic stimulation. J. Neurol. Neurosurg. Psychiatry 1997, 63, 579–583. [Google Scholar] [CrossRef]
- Miller, N.; Allcock, L.; Hildreth, A.J.; Jones, D.; Noble, E.; Burn, D.J. Swallowing problems in Parkinson disease: Frequency and clinical correlates. J. Neurol. Neurosurg. Psychiatry 2009, 80, 1047–1049. [Google Scholar] [CrossRef]
- Noble, E.; Jones, D.; Miller, N. Perceived changes to swallowing in people with Parkinson’s disease. Int. J. Ther. Rehabil. 2015, 22, 573–581. [Google Scholar] [CrossRef]
- Camilleri, M.; Parkman, H.P.; Shafi, M.A.; Abell, T.L.; Gerson, L. Clinical guideline: Management of gastroparesis. Am. J. Gastroenterol. 2013, 108, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navas, C.M.; Patel, N.K.; Lacy, B.E. Symptomatic management of gastroparesis. Gastrointest. Endosc. Clin. 2019, 29, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Parkman, H.P.; Yates, K.; Hasler, W.L.; Nguyen, L.; Pasricha, P.J.; Snape, W.J.; Farrugia, G.; Koch, K.L.; Abell, T.L.; McCallum, R.W.; et al. Clinical features of idiopathic gastroparesis vary with sex, body mass, symptom onset, delay in gastric emptying, and gastroparesis severity. Gastroenterology 2011, 140, 101–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Natale, G.; Pasquali, L.; Ruggieri, S.; Paparelli, A.; Fornai, F. Parkinson’s disease and the gut: A well known clinical association in need of an effective cure and explanation. Neurogastroenterol. Motil. 2008, 20, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Brogden, R.N.; Carmine, A.A.; Heel, R.C.; Speight, T.M.; Avery, G.S. Domperidone. A review of its pharmacological activity, pharmacokinetics and therapeutic efficacy in the symptomatic treatment of chronic dyspepsia and as an antiemetic. Drugs 1982, 24, 360–400. [Google Scholar] [CrossRef] [PubMed]
- Van Nueten, J.M.; Schuurkes, J.A. Studies on the role of dopamine and dopamine blockers in gastroduodenal motility. Scand. J. Gastroenterol. Suppl. 1984, 96, 89–99. [Google Scholar]
- Nishikawa, N.; Nagai, M.; Tsujii, T.; Iwaki, H.; Yabe, H.; Nomoto, M. Coadministration of domperidone increases plasma levodopa concentration in patients with Parkinson disease. Clin. Neuropharmacol. 2012, 35, 182–184. [Google Scholar] [CrossRef]
- Soykan, I.; Sarosiek, I.; Shifflett, J.; Wooten, G.F.; McCallum, R.W. Effect of chronic oral domperidone therapy on gastrointestinal symptoms and gastric emptying in patients with Parkinson’s disease. Mov. Disord. 1997, 12, 952–957. [Google Scholar] [CrossRef]
- Johannes, C.B.; Varas-Lorenzo, C.; McQuay, L.J.; Midkiff, K.D.; Fife, D. Risk of serious ventricular arrhythmia and sudden cardiac death in a cohort of users of domperidone: A nested case-control study. Pharmacoepidemiol. Drug Saf. 2010, 19, 881–888. [Google Scholar] [CrossRef]
- van Noord, C.; Dieleman, J.P.; van Herpen, G.; Verhamme, K.; Sturkenboom, M.C. Domperidone and ventricular arrhythmia or sudden cardiac death: A population-based case-control study in the Netherlands. Drug Saf. 2010, 33, 1003–1014. [Google Scholar] [CrossRef]
- Ramprasad, C.; Douglas, J.Y.; Moshiree, B. Parkinson’s Disease and Current Treatments for Its Gastrointestinal Neurogastromotility Effects. Curr. Treat. Options Gastroenterol. 2018, 16, 489–510. [Google Scholar] [CrossRef]
- Avorn, J.; Gurwitz, J.H.; Bohn, R.L.; Mogun, H.; Monane, M.; Walker, A. Increased incidence of levodopa therapy following metoclopramide use. JAMA 1995, 274, 1780–1782. [Google Scholar] [CrossRef]
- Barone, J.A. Domperidone: A peripherally acting dopamine2-receptor antagonist. Ann. Pharmacother. 1999, 33, 429–440. [Google Scholar] [CrossRef]
- Hennessy, S.; Leonard, C.E.; Newcomb, C.; Kimmel, S.E.; Bilker, W.B. Cisapride and ventricular arrhythmia. Br. J. Clin. Pharmacol. 2008, 66, 375–385. [Google Scholar] [CrossRef] [Green Version]
- Loughlin, J.; Quinn, S.; Rivero, E.; Wong, J.; Huang, J.; Kralstein, J.; Earnest, D.L.; Seeger, J.D. Tegaserod and the risk of cardiovascular ischemic events: An observational cohort study. J. Cardiovasc. Pharmacol. Ther. 2010, 15, 151–157. [Google Scholar] [CrossRef]
- Ashraf, W.; Park, F.; Lof, J.; Quigley, E.M. Effects of psyllium therapy on stool characteristics, colon transit and anorectal function in chronic idiopathic constipation. Aliment. Pharmacol. Ther. 1995, 9, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Johanson, J.F. Review of the treatment options for chronic constipation. MedGenMed 2007, 9, 25. [Google Scholar] [PubMed]
- Fernandez-Martinez, M.N.; Hernandez-Echevarria, L.; Sierra-Vega, M.; Diez-Liebana, M.J.; Calle-Pardo, A.; Carriedo-Ule, D.; Sahagun-Prieto, A.M.; Anguera-Vila, A.; Garcia-Vieitez, J.J. A randomised clinical trial to evaluate the effects of Plantago ovata husk in Parkinson patients: Changes in levodopa pharmacokinetics and biochemical parameters. BMC Complement. Altern. Med. 2014, 14, 296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ernst, E. Abdominal massage therapy for chronic constipation: A systematic review of controlled clinical trials. Complement. Med. Res. 1999, 6, 149–151. [Google Scholar] [CrossRef]
- Sinclair, M. The use of abdominal massage to treat chronic constipation. J. Bodyw. Mov. Ther. 2011, 15, 436–445. [Google Scholar] [CrossRef] [PubMed]
- McClurg, D.; Hagen, S.; Dickinson, L. Abdominal massage for the treatment of constipation. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Mokhtare, M.; Karimi, S.; Bahardoust, M.; Sotoudeheian, M.; Ghazi, A.; Babaei-Ghazani, A. How adding the abdominal massage to polyethylene glycol can improve symptom and quality of life in patients with functional constipation in comparison with each one of the treatment modalities alone: A randomized clinical trial. Complement. Ther. Med. 2020, 52, 102495. [Google Scholar] [CrossRef] [PubMed]
- Seppi, K.; Ray Chaudhuri, K.; Coelho, M.; Fox, S.H.; Katzenschlager, R.; Perez Lloret, S.; Weintraub, D.; Sampaio, C.; Collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on Behalf of the Movement Disorders Society Evidence-Based Medicine Committee; Chahine, L. Update on treatments for nonmotor symptoms of Parkinson’s disease—An evidence-based medicine review. Mov. Disord. 2019, 34, 180–198. [Google Scholar] [CrossRef] [Green Version]
- Rossi, M.; Merello, M.; Perez-Lloret, S. Management of constipation in Parkinson’s disease. Expert Opin. Pharmacother. 2015, 16, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Zangaglia, R.; Martignoni, E.; Glorioso, M.; Ossola, M.; Riboldazzi, G.; Calandrella, D.; Brunetti, G.; Pacchetti, C. Macrogol for the treatment of constipation in Parkinson’s disease. A randomized placebo-controlled study. Mov. Disord. Off. J. Mov. Disord. Soc. 2007, 22, 1239–1244. [Google Scholar] [CrossRef]
- Eichhorn, T.E.; Oertel, W.H. Macrogol 3350/electrolyte improves constipation in Parkinson’s disease and multiple system atrophy. Mov. Disord. Off. J. Mov. Disord. Soc. 2001, 16, 1176–1177. [Google Scholar] [CrossRef]
- Coggrave, M.; Norton, C.; Cody, J.D. Management of faecal incontinence and constipation in adults with central neurological diseases. Cochrane Database Syst. Rev. 2014, 1, CD00211. [Google Scholar] [CrossRef] [Green Version]
- Di Palma, J.A.; Smith, J.R.; vB Cleveland, M. Overnight efficacy of polyethylene glycol laxative. Am. J. Gastroenterol. 2002, 97, 1776–1779. [Google Scholar] [CrossRef]
- DiPalma, J.A.; DeRidder, P.H.; Orlando, R.C.; Kolts, B.E.; vB Cleveland, M. A randomized, placebo-controlled, multicenter study of the safety and efficacy of a new polyethylene glycol laxative. Am. J. Gastroenterol. 2000, 95, 446–450. [Google Scholar] [CrossRef]
- Klaschik, E.; Nauck, F.; Ostgathe, C. Constipation—Modern laxative therapy. Support. Care Cancer 2003, 11, 679–685. [Google Scholar] [CrossRef]
- McKeage, K.; Plosker, G.L.; Siddiqui, M.A.A. Lubiprostone. Drugs 2006, 66, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Ondo, W.; Kenney, C.; Sullivan, K.; Davidson, A.; Hunter, C.; Jahan, I.; McCombs, A.; Miller, A.; Zesiewicz, T. Placebo-controlled trial of lubiprostone for constipation associated with Parkinson disease. Neurology 2012, 78, 1650–1654. [Google Scholar] [CrossRef] [PubMed]
- Cuppoletti, J.; Blikslager, A.T.; Chakrabarti, J.; Nighot, P.K.; Malinowska, D.H. Contrasting effects of linaclotide and lubiprostone on restitution of epithelial cell barrier properties and cellular homeostasis after exposure to cell stressors. BMC Pharmacol. 2012, 12, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Lisle, R.C. Lubiprostone stimulates small intestinal mucin release. BMC Gastroenterol. 2012, 12, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barboza, J.L.; Okun, M.S.; Moshiree, B. The treatment of gastroparesis, constipation and small intestinal bacterial overgrowth syndrome in patients with Parkinson’s disease. Expert Opin. Pharmacother. 2015, 16, 2449–2464. [Google Scholar] [CrossRef]
- Manfredsson, F.P.; Luk, K.C.; Benskey, M.J.; Gezer, A.; Garcia, J.; Kuhn, N.C.; Sandoval, I.M.; Patterson, J.R.; O’Mara, A.; Yonkers, R.; et al. Induction of alpha-synuclein pathology in the enteric nervous system of the rat and non-human primate results in gastrointestinal dysmotility and transient CNS pathology. Neurobiol. Dis. 2018, 112, 106–118. [Google Scholar] [CrossRef]
- Uemura, N.; Yagi, H.; Uemura, M.T.; Hatanaka, Y.; Yamakado, H.; Takahashi, R. Inoculation of alpha-synuclein preformed fibrils into the mouse gastrointestinal tract induces Lewy body-like aggregates in the brainstem via the vagus nerve. Mol. Neurodegener. 2018, 13, 21. [Google Scholar] [CrossRef] [Green Version]
- Holmqvist, S.; Chutna, O.; Bousset, L.; Aldrin-Kirk, P.; Li, W.; Bjorklund, T.; Wang, Z.Y.; Roybon, L.; Melki, R.; Li, J.Y. Direct evidence of Parkinson pathology spread from the gastrointestinal tract to the brain in rats. Acta Neuropathol. 2014, 128, 805–820. [Google Scholar] [CrossRef] [Green Version]
- Giancola, F.; Torresan, F.; Repossi, R.; Bianco, F.; Latorre, R.; Ioannou, A.; Guarino, M.; Volta, U.; Clavenzani, P.; Mazzoni, M.; et al. Downregulation of neuronal vasoactive intestinal polypeptide in Parkinson’s disease and chronic constipation. Neurogastroenterol. Motil. 2017, 29, e12995. [Google Scholar] [CrossRef] [Green Version]
- Chalazonitis, A.; Rao, M. Enteric nervous system manifestations of neurodegenerative disease. Brain Res. 2018, 1693, 207–213. [Google Scholar] [CrossRef]
- Braak, H.; de Vos, R.A.I.; Bohl, J.; Del Tredici, K. Gastric a-synuclein immunoreactive inclusions in Meissner’s and Auerbach’s plexuses in cases staged for Parkinson’s disease-related brain pathology. Neurosci. Lett. 2006, 396, 67–72. [Google Scholar] [CrossRef]
- Shannon, K.M.; Keshavarzian, A.; Mutlu, E.; Dodiya, H.B.; Daian, D.; Jaglin, J.A.; Kordower, J.H. Alpha-synuclein in colonic submucosa in early untreated Parkinson’s disease. Mov. Disord. 2012, 27, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Singaram, C.; Ashraf, W.; Gaumnitz, E.A.; Torbey, C.; Sengupta, A.; Pfeiffer, R.; Quigley, E.M. Dopaminergic defect of enteric nervous system in Parkinson’s disease patients with chronic constipation. Lancet 1995, 346, 861–864. [Google Scholar] [CrossRef]
- Bassotti, G.; Villanacci, V.; Nascimbeni, R.; Cadei, M.; Fisogni, S.; Antonelli, E.; Corazzi, N.; Salerni, B. Enteric neuroglial apoptosis in inflammatory bowel diseases. J. Crohn Colitis 2009, 3, 264–270. [Google Scholar] [CrossRef] [Green Version]
- McKeown, S.J.; Stamp, L.; Hao, M.M.; Young, H.M. Hirschsprung disease: A developmental disorder of the enteric nervous system. Wiley Interdiscip. Rev. Dev. Biol. 2013, 2, 113–129. [Google Scholar] [CrossRef] [PubMed]
- De Giorgio, R.; Cogliandro, R.F.; Barbara, G.; Corinaldesi, R.; Stanghellini, V. Chronic intestinal pseudo-obstruction: Clinical features, diagnosis, and therapy. Gastroenterol. Clin. N. Am. 2011, 40, 787–807. [Google Scholar] [CrossRef] [PubMed]
- Gershon, M.D.; Liu, M.T. Serotonin and neuroprotection in functional bowel disorders. Neurogastroenterol. Motil. 2007, 19 (Suppl. 2), 19–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mawe, G.M.; Hoffman, J.M. Serotonin signalling in the gut--functions, dysfunctions and therapeutic targets. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 473–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gershon, M.D.; Tack, J. The serotonin signaling system: From basic understanding to drug development for functional GI disorders. Gastroenterology 2007, 132, 397–414. [Google Scholar] [CrossRef]
- Poole, D.P.; Xu, B.; Koh, S.L.; Hunne, B.; Coupar, I.M.; Irving, H.R.; Shinjo, K.; Furness, J.B. Identification of neurons that express 5-hydroxytryptamine4 receptors in intestine. Cell Tissue Res. 2006, 325, 413–422. [Google Scholar] [CrossRef]
- Frampton, J.E. Prucalopride. Drugs 2009, 69, 2463–2476. [Google Scholar] [CrossRef]
- Müller-Lissner, S.; Rykx, A.; Kerstens, R.; Vandeplassche, L. A double-blind, placebo-controlled study of prucalopride in elderly patients with chronic constipation. Neurogastroenterol. Motil. 2010, 22, 991-e255. [Google Scholar] [CrossRef]
- Shin, A.; Camilleri, M.; Kolar, G.; Erwin, P.; West, C.; Murad, M. Systematic review with meta-analysis: Highly selective 5-HT4 agonists (prucalopride, velusetrag or naronapride) in chronic constipation. Aliment. Pharmacol. Ther. 2014, 39, 239–253. [Google Scholar] [CrossRef] [Green Version]
- Bouras, E.P.; Camilleri, M.; Burton, D.D.; McKinzie, S. Selective stimulation of colonic transit by the benzofuran 5HT4 agonist, prucalopride, in healthy humans. Gut 1999, 44, 682–686. [Google Scholar] [CrossRef] [Green Version]
- Bouras, E.P.; Camilleri, M.; Burton, D.D.; Thomforde, G.; McKinzie, S.; Zinsmeister, A.R. Prucalopride accelerates gastrointestinal and colonic transit in patients with constipation without a rectal evacuation disorder. Gastroenterology 2001, 120, 354–360. [Google Scholar] [CrossRef]
- Omer, A.; Quigley, E.M.M. An update on prucalopride in the treatment of chronic constipation. Ther. Adv. Gastroenterol. 2017, 10, 877–887. [Google Scholar] [CrossRef]
- Bianco, F.; Bonora, E.; Natarajan, D.; Vargiolu, M.; Thapar, N.; Torresan, F.; Giancola, F.; Boschetti, E.; Volta, U.; Bazzoli, F.; et al. Prucalopride exerts neuroprotection in human enteric neurons. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G768–G775. [Google Scholar] [CrossRef] [Green Version]
- Freitas, M.E.; Alqaraawi, A.; Lang, A.E.; Liu, L.W.C. Linaclotide and Prucalopride for Management of Constipation in Patients with Parkinsonism. Mov. Disord. Clin. Pract. 2018, 5, 218–220. [Google Scholar] [CrossRef] [Green Version]
- Pinyopornpanish, K.; Soontornpun, A.; Kijdamrongthum, P.; Teeyasoontranon, W.; Angkurawaranon, C.; Thongsawat, S. The effect of prucalopride on gastric emptying in Parkinson’s disease patients, a pilot randomized, open-label study. Dig. Syst. 2017, 1, 1–6. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, T.; Hara, H. Role of flavonoids in intestinal tight junction regulation. J. Nutr. Biochem. 2011, 22, 401–408. [Google Scholar] [CrossRef]
- El-Horany, H.E.; El-Latif, R.N.; ElBatsh, M.M.; Emam, M.N. Ameliorative Effect of Quercetin on Neurochemical and Behavioral Deficits in Rotenone Rat Model of Parkinson’s Disease: Modulating Autophagy (Quercetin on Experimental Parkinson’s Disease). J. Biochem. Mol. Toxicol. 2016, 30, 360–369. [Google Scholar] [CrossRef]
- Li, X.; Wang, H.; Gao, Y.; Li, L.; Tang, C.; Wen, G.; Zhou, Y.; Zhou, M.; Mao, L.; Fan, Y. Protective Effects of Quercetin on Mitochondrial Biogenesis in Experimental Traumatic Brain Injury via the Nrf2 Signaling Pathway. PLoS ONE 2016, 11, e0164237. [Google Scholar] [CrossRef] [Green Version]
- Sandhir, R.; Mehrotra, A. Quercetin supplementation is effective in improving mitochondrial dysfunctions induced by 3-nitropropionic acid: Implications in Huntington’s disease. Biochim. Biophys. Acta 2013, 1832, 421–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, C.; Hong, T.; Yang, Z.; Zhang, Y.; Wang, L.; Dong, M.; Zhao, J.; Mu, J.; Meng, Y. Effect of Quercetin in the 1-Methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine-Induced Mouse Model of Parkinson’s Disease. Evid. Based Complement. Altern. Med. 2012, 2012, 928643. [Google Scholar] [CrossRef] [PubMed]
- Haleagrahara, N.; Siew, C.J.; Mitra, N.K.; Kumari, M. Neuroprotective effect of bioflavonoid quercetin in 6-hydroxydopamine-induced oxidative stress biomarkers in the rat striatum. Neurosci. Lett. 2011, 500, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Figueira, I.; Menezes, R.; Macedo, D.; Costa, I.; Dos Santos, C.N. Polyphenols Beyond Barriers: A Glimpse into the Brain. Curr. Neuropharmacol. 2017, 15, 562–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, S.; Raj, K.; Singh, S. Neuroprotective Effect of Quercetin in Combination with Piperine against Rotenone- and Iron Supplement-Induced Parkinson’s Disease in Experimental Rats. Neurotox. Res. 2020, 37, 198–209. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Aschner, M.; Cheang, W.S.; Akkol, E.K. Neuroprotective Effects of Quercetin in Alzheimer’s Disease. Biomolecules 2019, 10, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, S.; Jamwal, S.; Kumar, P. Neuroprotective potential of Quercetin in combination with piperine against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neurotoxicity. Neural Regen. Res. 2017, 12, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E. Mucus layers in inflammatory bowel disease. Inflamm. Bowel Dis. 2014, 20, 2124–2131. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.; Sjovall, H.; Hansson, G.C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 352–361. [Google Scholar] [CrossRef] [Green Version]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Piche, T.; Barbara, G.; Aubert, P.; Bruley des Varannes, S.; Dainese, R.; Nano, J.L.; Cremon, C.; Stanghellini, V.; De Giorgio, R.; Galmiche, J.P.; et al. Impaired intestinal barrier integrity in the colon of patients with irritable bowel syndrome: Involvement of soluble mediators. Gut 2009, 58, 196–201. [Google Scholar] [CrossRef] [Green Version]
- Peeters, M.; Ghoos, Y.; Maes, B.; Hiele, M.; Geboes, K.; Vantrappen, G.; Rutgeerts, P. Increased permeability of macroscopically normal small bowel in Crohn’s disease. Dig. Dis. Sci. 1994, 39, 2170–2176. [Google Scholar] [CrossRef]
- Salat-Foix, D.; Tran, K.; Ranawaya, R.; Meddings, J.; Suchowersky, O. Increased intestinal permeability and Parkinson disease patients: Chicken or egg? Can. J. Neurol. Sci. 2012, 39, 185–188. [Google Scholar] [CrossRef] [Green Version]
- Kelly, L.P.; Carvey, P.M.; Keshavarzian, A.; Shannon, K.M.; Shaikh, M.; Bakay, R.A.; Kordower, J.H. Progression of intestinal permeability changes and alpha-synuclein expression in a mouse model of Parkinson’s disease. Mov. Disord. 2014, 29, 999–1009. [Google Scholar] [CrossRef]
- Thulesen, J. Glucagon-like peptide 2 (GLP-2), an intestinotrophic mediator. Curr. Protein Pept. Sci. 2004, 5, 51–65. [Google Scholar] [PubMed]
- Pini, A.; Garella, R.; Idrizaj, E.; Calosi, L.; Baccari, M.; Vannucchi, M. Glucagon-like peptide 2 counteracts the mucosal damage and the neuropathy induced by chronic treatment with cisplatin in the mouse gastric fundus. Neurogastroenterol. Motil. 2016, 28, 206–216. [Google Scholar] [CrossRef]
- Zhang, Z.; Hao, L.; Shi, M.; Yu, Z.; Shao, S.; Yuan, Y.; Zhang, Z.; Hölscher, C. Neuroprotective Effects of a GLP-2 Analogue in the MPTP Parkinson’s Disease Mouse Model. J. Parkinson Dis. 2021, 11, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Zhang, Z.; Li, H.; Ma, J.; Sun, L.; Shao, S.; Zhang, Z.; Hölscher, C. A GLP-2 Analogue Protects SH-SY5Y and Neuro-2a Cells Against Mitochondrial Damage, Autophagy Impairments and Apoptosis in a Parkinson Model. Drug Res. 2021, 71, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Yang, N.; Ji, C.; Zheng, J.; Wang, T.; Liu, Y.; Zuo, P. Neuroprotective effects of 5-(4-hydroxy-3-dimethoxybenzylidene)-thiazolidinone in MPTP induced Parkinsonism model in mice. Neuropharmacology 2015, 93, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Brakedal, B.; Flones, I.; Reiter, S.F.; Torkildsen, O.; Dolle, C.; Assmus, J.; Haugarvoll, K.; Tzoulis, C. Glitazone use associated with reduced risk of Parkinson’s disease. J. Mov. Disord. 2017, 32, 1594–1599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, L.; Li, D.; Feng, P.; Li, L.; Xue, G.F.; Li, G.; Holscher, C. A novel dual GLP-1 and GIP incretin receptor agonist is neuroprotective in a mouse model of Parkinson’s disease by reducing chronic inflammation in the brain. NeuroReport 2016, 27, 384–391. [Google Scholar] [CrossRef]
- Aviles-Olmos, I.; Dickson, J.; Kefalopoulou, Z.; Djamshidian, A.; Kahan, J.; Ell, P.; Whitton, P.; Wyse, R.; Isaacs, T.; Lees, A.; et al. Motor and cognitive advantages persist 12 months after exenatide exposure in Parkinson’s disease. J. Parkinsons Dis. 2014, 4, 337–344. [Google Scholar] [CrossRef] [Green Version]
- Labuzek, K.; Suchy, D.; Gabryel, B.; Bielecka, A.; Liber, S.; Okopien, B. Quantification of metformin by the HPLC method in brain regions, cerebrospinal fluid and plasma of rats treated with lipopolysaccharide. Pharmacol. Rep. 2010, 62, 956–965. [Google Scholar] [CrossRef]
- Campbell, J.M.; Stephenson, M.D.; de Courten, B.; Chapman, I.; Bellman, S.M.; Aromataris, E. Metformin Use Associated with Reduced Risk of Dementia in Patients with Diabetes: A Systematic Review and Meta-Analysis. J. Alzheimers Dis. 2018, 65, 1225–1236. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Yu, J.T.; Zhu, X.C.; Wang, H.F.; Tan, M.S.; Cao, L.; Zhang, Q.Q.; Gao, L.; Shi, J.Q.; Zhang, Y.D.; et al. Acute metformin preconditioning confers neuroprotection against focal cerebral ischaemia by pre-activation of AMPK-dependent autophagy. Br. J. Pharmacol. 2014, 171, 3146–3157. [Google Scholar] [CrossRef]
- Ng, T.P.; Feng, L.; Yap, K.B.; Lee, T.S.; Tan, C.H.; Winblad, B. Long-term metformin usage and cognitive function among older adults with diabetes. J. Alzheimers Dis. 2014, 41, 61–68. [Google Scholar] [CrossRef]
- Wahlqvist, M.L.; Lee, M.-S.; Hsu, C.-C.; Chuang, S.-Y.; Lee, J.-T.; Tsai, H.-N. Metformin-inclusive sulfonylurea therapy reduces the risk of Parkinson’s disease occurring with Type 2 diabetes in a Taiwanese population cohort. Parkinsonism Relat. Disord. 2012, 18, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Rotermund, C.; Machetanz, G.; Fitzgerald, J.C. The Therapeutic Potential of Metformin in Neurodegenerative Diseases. Front. Endocrinol. 2018, 9, 400. [Google Scholar] [CrossRef]
- Ahmadi, S.; Razazan, A.; Nagpal, R.; Jain, S.; Wang, B.; Mishra, S.P.; Wang, S.; Justice, J.; Ding, J.; McClain, D.A.; et al. Metformin Reduces Aging-Related Leaky Gut and Improves Cognitive Function by Beneficially Modulating Gut Microbiome/Goblet Cell/Mucin Axis. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, e9–e21. [Google Scholar] [CrossRef]
- Deng, J.; Zeng, L.; Lai, X.; Li, J.; Liu, L.; Lin, Q.; Chen, Y. Metformin protects against intestinal barrier dysfunction via AMPKalpha1-dependent inhibition of JNK signalling activation. J. Cell Mol. Med. 2018, 22, 546–557. [Google Scholar] [CrossRef] [Green Version]
- Scarpello, J.H.; Hodgson, E.; Howlett, H.C. Effect of metformin on bile salt circulation and intestinal motility in type 2 diabetes mellitus. Diabetes Med. 1998, 15, 651–656. [Google Scholar] [CrossRef]
- Dandona, P.; Fonseca, V.; Mier, A.; Beckett, A.G. Diarrhea and metformin in a diabetic clinic. Diabetes Care 1983, 6, 472–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menzies, D.G.; Campbell, I.W.; McBain, A.; Brown, I.R. Metformin efficacy and tolerance in obese non-insulin dependent diabetics: A comparison of two dosage schedules. Curr. Med. Res. Opin. 1989, 11, 273–278. [Google Scholar] [CrossRef]
- Minato, T.; Maeda, T.; Fujisawa, Y.; Tsuji, H.; Nomoto, K.; Ohno, K.; Hirayama, M. Progression of Parkinson’s disease is associated with gut dysbiosis: Two-year follow-up study. PLoS ONE 2017, 12, e0187307. [Google Scholar] [CrossRef] [Green Version]
- Mulak, A.; Bonaz, B. Brain-gut-microbiota axis in Parkinson’s disease. World J. Gastroenterol. 2015, 21, 10609–10620. [Google Scholar] [CrossRef]
- Bedarf, J.R.; Hildebrand, F.; Coelho, L.P.; Sunagawa, S.; Bahram, M.; Goeser, F.; Bork, P.; Wullner, U. Functional implications of microbial and viral gut metagenome changes in early stage L-DOPA-naive Parkinson’s disease patients. Genome Med. 2017, 9, 39. [Google Scholar] [CrossRef]
- Heintz-Buschart, A.; Pandey, U.; Wicke, T.; Sixel-Doring, F.; Janzen, A.; Sittig-Wiegand, E.; Trenkwalder, C.; Oertel, W.H.; Mollenhauer, B.; Wilmes, P. The nasal and gut microbiome in Parkinson’s disease and idiopathic rapid eye movement sleep behavior disorder. J. Mov. Disord. 2018, 33, 88–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill-Burns, E.M.; Debelius, J.W.; Morton, J.T.; Wissemann, W.T.; Lewis, M.R.; Wallen, Z.D.; Peddada, S.D.; Factor, S.A.; Molho, E.; Zabetian, C.P.; et al. Parkinson’s disease and Parkinson’s disease medications have distinct signatures of the gut microbiome. J. Mov. Disord. 2017, 32, 739–749. [Google Scholar] [CrossRef]
- Scheperjans, F.; Aho, V.; Pereira, P.A.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut microbiota are related to Parkinson’s disease and clinical phenotype. J. Mov. Disord. 2015, 30, 350–358. [Google Scholar] [CrossRef]
- Unger, M.M.; Spiegel, J.; Dillmann, K.U.; Grundmann, D.; Philippeit, H.; Burmann, J.; Fassbender, K.; Schwiertz, A.; Schafer, K.H. Short chain fatty acids and gut microbiota differ between patients with Parkinson’s disease and age-matched controls. Parkinsonism Relat. Disord. 2016, 32, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Barichella, M.; Cereda, E.; Cassani, E.; Pinelli, G.; Iorio, L.; Ferri, V.; Privitera, G.; Pasqua, M.; Valentino, A.; Monajemi, F.; et al. Dietary habits and neurological features of Parkinson’s disease patients: Implications for practice. Clin. Nutr. 2017, 36, 1054–1061. [Google Scholar] [CrossRef]
- Cassani, E.; Privitera, G.; Pezzoli, G.; Pusani, C.; Madio, C.; Iorio, L.; Barichella, M. Use of probiotics for the treatment of constipation in Parkinson’s disease patients. Minerva Gastroenterol. Dietol. 2011, 57, 117–121. [Google Scholar]
- Tan, A.H.; Lim, S.Y.; Chong, K.K.; MAA, A.M.; Hor, J.W.; Lim, J.L.; Low, S.C.; Chong, C.W.; Mahadeva, S.; Lang, A.E. Probiotics for Constipation in Parkinson Disease: A Randomized Placebo-Controlled Study. Neurology 2021, 96, e772–e782. [Google Scholar] [CrossRef]
- Miller, L.E.; Ouwehand, A.C. Probiotic supplementation decreases intestinal transit time: Meta-analysis of randomized controlled trials. World J. Gastroenterol. 2013, 19, 4718. [Google Scholar] [CrossRef]
- Huang, H.; Xu, H.; Luo, Q.; He, J.; Li, M.; Chen, H.; Tang, W.; Nie, Y.; Zhou, Y. Fecal microbiota transplantation to treat Parkinson’s disease with constipation: A case report. Medicine 2019, 98, e16163. [Google Scholar] [CrossRef]
- Kuai, X.Y.; Yao, X.H.; Xu, L.J.; Zhou, Y.Q.; Zhang, L.P.; Liu, Y.; Pei, S.F.; Zhou, C.L. Evaluation of fecal microbiota transplantation in Parkinson’s disease patients with constipation. Microb. Cell Fact. 2021, 20, 98. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C. The pros and cons of probiotics. Lancet Infect. Dis. 2013, 13, 571–572. [Google Scholar] [CrossRef]
- Brescia, P.; Rescigno, M. The gut vascular barrier: A new player in the gut–liver–brain axis. Trends Mol. Med. 2021, 27, 844–855. [Google Scholar] [CrossRef] [PubMed]
- Rocca, W.A. The burden of Parkinson’s disease: A worldwide perspective. Lancet Neurol. 2018, 17, 928–929. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, M.N.; Finkelstein, D.I.; McQuade, R.M.; Diwakarla, S. Gastrointestinal Dysfunction in Parkinson’s Disease: Current and Potential Therapeutics. J. Pers. Med. 2022, 12, 144. https://doi.org/10.3390/jpm12020144
Han MN, Finkelstein DI, McQuade RM, Diwakarla S. Gastrointestinal Dysfunction in Parkinson’s Disease: Current and Potential Therapeutics. Journal of Personalized Medicine. 2022; 12(2):144. https://doi.org/10.3390/jpm12020144
Chicago/Turabian StyleHan, Myat Noe, David I. Finkelstein, Rachel M. McQuade, and Shanti Diwakarla. 2022. "Gastrointestinal Dysfunction in Parkinson’s Disease: Current and Potential Therapeutics" Journal of Personalized Medicine 12, no. 2: 144. https://doi.org/10.3390/jpm12020144
APA StyleHan, M. N., Finkelstein, D. I., McQuade, R. M., & Diwakarla, S. (2022). Gastrointestinal Dysfunction in Parkinson’s Disease: Current and Potential Therapeutics. Journal of Personalized Medicine, 12(2), 144. https://doi.org/10.3390/jpm12020144






