Is It Time to Redefine Fetal Decelerations in Cardiotocography?
Abstract
1. Introduction
2. Materials and Methods
3. Synthesis
3.1. Early Decelerations
3.2. Variable Decelerations
3.3. Late Decelerations
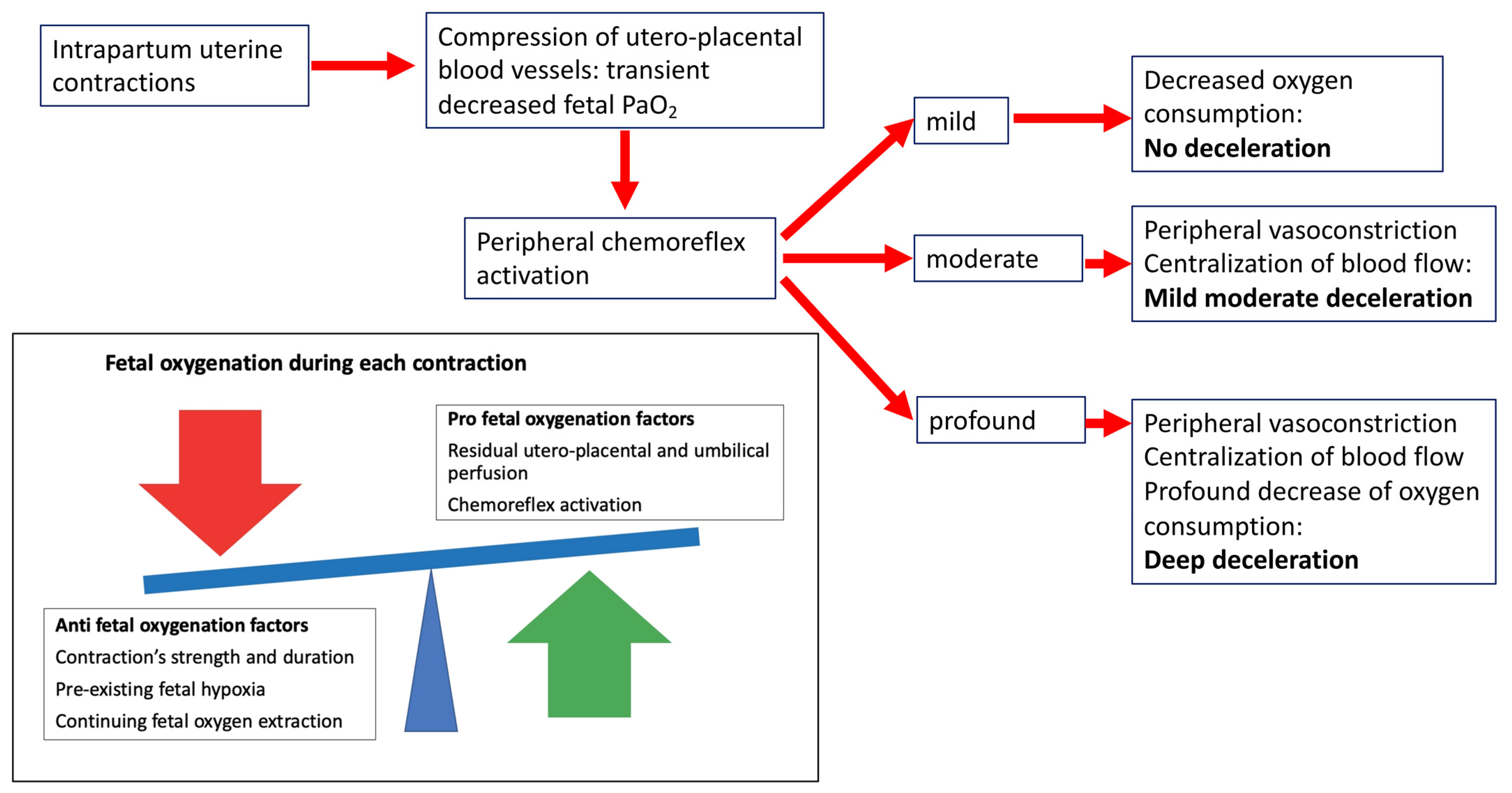
3.4. New Insights into the Pathophysiology of Fetal Intrapartum Decelerations
3.5. The State of the Art of Current Intrapartum Cardiotocography
4. Limitations to the Overview
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alfirevic, Z.; Devane, D.; Gyte, G.M.; Cuthbert, A. Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst. Rev. 2017, 2, CD006066. [Google Scholar] [CrossRef] [PubMed]
- ACOG Practice Bulletin No.106: Intrapartum fetal heart rate monitoring: Nomenclature, interpretation, and general management principles. Obstet Gynecol. 2009, 114, 192–202. [CrossRef] [PubMed]
- Ayres-de-Campos, D.; Spong, C.Y.; Chandraharan, E.; FIGO Intrapartum Fetal Monitoring Expert Consensus Panel. FIGO consensus guidelines on intrapartum fetal monitoring: Cardiotocography. Int. J. Gynaecol Obstet. 2015, 131, 13–24. [Google Scholar] [CrossRef] [PubMed]
- National Collaborating Centre for Women’s and Children’s Health (UK). Intrapartum Care: Care of Healthy Women and Their Babies during Childbirth; RCOG Press: London, UK, 2014. [Google Scholar]
- Mendez-Bauer, C.; Poseiro, J.J.; Arellano-Hernandez, G.; Zambrana, M.A.; Caldeyro-Barcia, R. Effects of atropine on the heart rate of the human fetus during labor. Am. J. Obstet. Gynecol. 1963, 85, 1033–1053. [Google Scholar] [CrossRef] [PubMed]
- Hon, E.H. The electronic evaluation of the fetal heart rate. Preliminary report. 1958. Am J Obstet Gynecol. 1996, 175 Pt 1, 747–748. [Google Scholar] [CrossRef] [PubMed]
- Lear, C.A.; Westgate, J.A.; Ugwumadu, A.; Nijhuis, J.G.; Stone, P.R.; Georgieva, A.; Ikeda, T.; Wassink, G.; Bennet, L.; Gunn, A.J. Understanding Fetal Heart Rate Patterns That May Predict Antenatal and Intrapartum Neural Injury. Semin. Pediatr. Neurol. 2018, 28, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Lear, C.A.; Wassink, G.; Westgate, J.A.; Nijhuis, J.G.; Ugwumadu, A.; Galinsky, R.; Bennet, L.; Gunn, A.J. The peripheral chemoreflex: Indefatigable guardian of fetal physiological adaptation to labour. J. Physiol. 2018, 596, 5611–5623. [Google Scholar] [CrossRef] [PubMed]
- Lear, C.A.; Kasai, M.; Booth, L.C.; Drury, P.P.; Davidson, J.O.; Maeda, Y.; Magawa, S.; Miyagi, E.; Ikeda, T.; Westgate, J.A.; et al. Peripheral chemoreflex control of fetal heart rate decelerations overwhelms the baroreflex during brief umbilical cord occlusions in fetal sheep. J. Physiol. 2020, 598, 4523–4536. [Google Scholar] [CrossRef] [PubMed]
- Lear, C.A.; Westgate, J.A.; Bennet, L.; Ugwumadu, A.; Stone, P.R.; Tournier, A.; Gunn, A.J. Fetal defenses against intrapartum head compression-implications for intrapartum decelerations and hypoxic-ischemic injury. Am. J. Obstet. Gynecol. 2021, 18, S0002-9378(21)02581-3. [Google Scholar] [CrossRef] [PubMed]
- Pinas, A.; Chandraharan, E. Continuous cardiotocography during labour: Analysis, classification and management. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 30, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Chung, F.; Hon, E.H. The electronic evaluation of fetal heart rate. I. With pressure on the fetal skull. Obstet. Gynecol. 1959, 13, 633–640. [Google Scholar] [PubMed]
- Paul, W.M.; Quilligan, E.J.; Maclachlan, T. Cardiovascular phenomenon associated with fetal head compression. Am. J. Obstet. Gynecol. 1964, 90, 824–826. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.P.; Koehler, R.C.; Gleason, C.A.; Jones, M.D., Jr.; Traystman, R.J. Cerebral and peripheral circulatory responses to intracranial hypertension in fetal sheep. Circ. Res. 1989, 64, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.P.; Koehler, R.C.; Nishijima, M.K.; Traystman, R.J.; Jones, M.D., Jr. Circulatory dynamics during periodic intracranial hypertension in fetal sheep. Am. J. Physiol. 1992, 263 Pt 2, R95–R102. [Google Scholar] [CrossRef] [PubMed]
- Barcroft Sir, J. Research on Prenatal Life. Part I; Blackwell Scientific Publications: Oxford, UK, 1947. [Google Scholar]
- Itskovitz, J.; LaGamma, E.F.; Rudolph, A.M. Heart rate and blood pressure responses to umbilical cord compression in fetal lambs with special reference to the mechanism of variable deceleration. Am. J. Obstet. Gynecol. 1983, 147, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.T.; Hon, E.H. Fetal hemodynamic response to umbilical cord compression. Obstet. Gynecol. 1963, 22, 553–562. [Google Scholar] [PubMed]
- Lear, C.A.; Galinsky, R.; Wassink, G.; Yamaguchi, K.; Davidson, J.O.; Westgate, J.A.; Bennet, L.; Gunn, A.J. The myths and physiology surrounding intrapartum decelerations: The critical role of the peripheral chemoreflex. J Physiol. 2016, 594, 4711–4725. [Google Scholar] [CrossRef] [PubMed]
- Mark, A.L. The Bezold-Jarisch reflex revisited: Clinical implications of inhibitory reflexes originating in the heart. J. Am. Coll. Cardiol. 1983, 1, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Itskovitz, J.; LaGamma, E.F.; Rudolph, A.M. Effects of cord compression on fetal blood flow distribution and O2 delivery. Am. J. Physiol. 1987, 252, H100–H109. [Google Scholar] [CrossRef] [PubMed]
- Giussani, D.A.; Unno, N.; Jenkins, S.L.; Wentworth, R.A.; Derks, J.B.; Collins, J.H.; Nathanielsz, P.W. Dynamics of cardiovascular responses to repeated partial umbilical cord compression in late-gestation sheep fetus. Am. J. Physiol. 1997, 273, H2351–H2360. [Google Scholar] [CrossRef]
- Aldrich, C.J.; D’Antona, D.; Spencer, J.A.; Delpy, D.T.; Reynolds, E.O.; Wyatt, J.S. Fetal heart rate changes and cerebral oxygenation measured by near-infrared spectroscopy during the first stage of labour. Eur. J. Obstet. Gynecol. Reprod. Biol. 1996, 64, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Hon, E.H.; Quilligan, E.J. Electronic evaluation of fetal heart rate. IX. Further observations on “pathologic” fetal bradycardia. Clin. Obstet. Gynecol. 1968, 11, 145–167. [Google Scholar] [CrossRef] [PubMed]
- Bax, M.; Nelson, K.B. Birth asphyxia: A statement. World Federation of Neurology Group. Dev. Med. Child Neurol. 1993, 35, 1022–1024. [Google Scholar] [CrossRef] [PubMed]
- Westgate, J.A.; Wibbens, B.; Bennet, L.; Wassink, G.; Parer, J.T.; Gunn, A.J. The intrapartum deceleration in center stage: A physiological approach to interpretation of fetal heart rate changes in labor. Am. J. Obstet. Gynecol. 2007, 197, 236.e1–236.e11. [Google Scholar] [CrossRef] [PubMed]
- Cahill, A.G.; Roehl, K.A.; Odibo, A.O.; Macones, G.A. Association and prediction of neonatal acidemia. Am J Obstet Gynecol. 2012, 207, e1–e206. [Google Scholar] [CrossRef] [PubMed]
- Yli, B.M.; Kjellmer, I. Pathophysiology of foetal oxygenation and cell damage during labour. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 30, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Kiserud, T.; Acharya, G. The fetal circulation. Prenat Diagn. 2004, 24, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Giussani, D.A. The fetal brain sparing response to hypoxia: Physiological mechanisms. J. Physiol. 2016, 594, 1215–1230. [Google Scholar] [CrossRef] [PubMed]
- Parer, J.T.; Ugwumadu, A. Impediments to a unified international approach to the interpretation and management of intrapartum cardiotocographs. J. Matern. Fetal. Neonatal. Med. 2017, 30, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Ugwumadu, A. Are we (mis)guided by current guidelines on intrapartum fetal heart rate monitoring? Case for a more physiological approach to interpretation. BJOG 2014, 121, 1063–1070, Erratum in BJOG 2015, 122, 599. [Google Scholar] [CrossRef] [PubMed]
- Berglund, S.; Grunewald, C.; Pettersson, H.; Cnattingius, S. Severe asphyxia due to delivery-related malpractice in Sweden 1990–2005. BJOG 2008, 115, 316–323. [Google Scholar] [CrossRef] [PubMed]
| Decelerations | ACOG 2009 | FIGO 2015 | NICE 2014 |
|---|---|---|---|
| Early | Usually symmetrical gradual decrease and return of the FHR associated with a uterine contraction. A gradual FHR decrease is defined as from the onset to the FHR nadir of 30 s or more. The decrease in FHR is calculated from the onset to the nadir of the deceleration. The nadir of the deceleration occurs at the same time as the peak of the contraction. In most cases the onset, nadir, and recovery of the deceleration are coincident with the beginning, peak, and ending of the contraction, respectively. | shallow, short-lasting, with normal variability within the deceleration and are coincident with contractions. They are believed to be caused by fetal head compression and do not indicate fetal hypoxia/acidosis. | |
| Late | Usually symmetrical gradual decrease and return of the FHR associated with a uterine contraction. A gradual FHR decrease is defined as from the onset to the FHR nadir of 30 s or more. The decrease in FHR is calculated from the onset to the nadir of the deceleration. The deceleration is delayed in timing, with the nadir of the deceleration occurring after the peak of the contraction. In most cases, the onset, nadir, and recovery of the deceleration occur after the beginning, peak, and ending of the contraction, respectively. | U-shaped with a gradual onset and/or a gradual return to the baseline and/or reduced variability within the deceleration. Gradual onset and return occurs when more than 30 s elapses between the beginning/end of a deceleration and its nadir. When contractions are adequately monitored, late decelerations start more than 20 seconds after the onset of a contraction, have a nadir after the acme, and a return to the baseline after the end of the contraction. These decelerations are indicative of a chemoreceptor-mediated response to fetal hypoxemia. In the presence of a tracing with no accelerations and reduced variability, the definition of late decelerations also includes those with an amplitude of 10−15 bpm. | Present for over 30 min; they do not improve with conservative measures and occurring with over 50% of contractions |
| Variable | Visually apparent abrupt decrease in FHR An abrupt FHR decrease is defined as from the onset of the deceleration to the beginning of the FHR nadir of less than 30 s. The decrease in FHR is calculated from the onset to the nadir of the deceleration. The decrease in FHR is 15 beats per minute or greater, lasting 15 seconds or greater, and less than 2 min in duration. When variable decelerations are associated with uterine contractions, their onset, depth, and duration commonly vary with successive uterine contractions. | V-shaped and exhibit a rapid drop (onset to nadir in less than 30 s), good variability within the deceleration, rapid recovery to the baseline, varying size, shape, and relationship to uterine contractions. They constitute the majority of decelerations during labor, and translate a baroreceptor-mediated response to increased arterial pressure, as occurs with umbilical cord compression. They are seldom associated with an important degree of fetal hypoxia/acidosis, unless they evolve to exhibit a U-shaped component, reduced variability within the deceleration, and/or their individual duration exceeds 3 min (prolonged decelerations). | Dropping from baseline by 60 beats/minute or less and for 30–90 min taking 60 seconds or less to recover. They are present for over 90 min and occurring with over 50% of contractions. OR Dropping from baseline by more than 60 beats/minute or taking over 60 seconds to recover. They are present for up to 30 min and occurring with over 50% of contractions. |
| Deceleration Type | Historical Belief | Why Historical Belief Is Misleading | New Insights in Pathophysiology of Fetal Decelerations |
|---|---|---|---|
| Early decelerations | -Results from head compression during fetal head engagement in the birth canal; -Considered benign and not caused by hypoxia; | -Only head compression severe enough to determine profound cerebral hypoperfusion triggers FHR decelerations; -Periods of head compression such as during spontaneous delivery do not critically impair cerebral perfusion and are not associated with FHR decelerations; | In case of extreme increases in intracranial pressure decelerations are not benign, being the fetal response to severe cerebral hypoperfusion |
| Variable decelerations | -Results from cord compression, therefore, having a variable relationship with contractions; -Potentially eliminable by changing the maternal position; | -The baroreflex mediated mechanism implicates that hypertension is consistently observed with complete cord occlusion, which was not experimentally confirmed; -The Bezold-Jarish reflex implicates that hypovolemia due to umbilical cord compression leads to a decreased venous return to the fetal heart, which in turn leads to the activation of the vagal reflex, thus provoking deceleration. However, in fetal sheep this reflex was not activated by reduced cardiac pressures. Moreover, when activated, this reflex induced a delayed deceleration, which is not consistent with the typical pattern of the variable deceleration. | These decelerations originate from hypoxemia, which is caused by at least a 50% reduction in intervillous perfusion, and are mediated by the peripheral chemoreflex |
| Late decelerations | -Results from utero-placental insufficiency. | No experimental evidence supports the conclusion that these decelerations are associated with a greater physiological challenge than early or variable decelerations. | Rhese decelerations originate from hypoxemia, which is caused by at least a 50% reduction in intervillous perfusion, and are mediated by the peripheral chemoreflex. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xodo, S.; Londero, A.P. Is It Time to Redefine Fetal Decelerations in Cardiotocography? J. Pers. Med. 2022, 12, 1552. https://doi.org/10.3390/jpm12101552
Xodo S, Londero AP. Is It Time to Redefine Fetal Decelerations in Cardiotocography? Journal of Personalized Medicine. 2022; 12(10):1552. https://doi.org/10.3390/jpm12101552
Chicago/Turabian StyleXodo, Serena, and Ambrogio P. Londero. 2022. "Is It Time to Redefine Fetal Decelerations in Cardiotocography?" Journal of Personalized Medicine 12, no. 10: 1552. https://doi.org/10.3390/jpm12101552
APA StyleXodo, S., & Londero, A. P. (2022). Is It Time to Redefine Fetal Decelerations in Cardiotocography? Journal of Personalized Medicine, 12(10), 1552. https://doi.org/10.3390/jpm12101552







