Pathophysiology of Takotsubo Syndrome as A Bridge to Personalized Treatment
Abstract
1. Introduction
2. Clinical Findings
3. Pathophysiology
3.1. Decrease of Estrogen Level
3.2. Microcirculation Disturbances
3.3. Endothelium Dysfunction
4. Low-Grade Inflammatory Process
4.1. Brain-Heart Axis
4.2. Genetic Factors
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Sato, H. Takotsubo-type cardiomyopathy due to multivessel spasm. In Clinical Aspect of Myocardial Injury: From Ischemia to Heart Failure; Kagakuhyouronsya: Tokyo, Japan, 1990; pp. 56–64. [Google Scholar]
- Ghadri, J.R.; Kato, K.; Cammann, V.L.; Gili, S.; Jurisic, S.; Di Vece, D.; Candreva, A.; Ding, K.J.; Micek, J.; Szawan, K.A. Long-Term Prognosis of Patients with Takotsubo Syndrome. J. Am. Coll. Cardiol. 2018, 72, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Ghadri, J.R.; Wittstein, I.S.; Prasad, A.; Sharkey, S.; Dote, K.; Akashi, Y.J.; Cammann, V.L.; Crea, F.; Galiuto, L.; Desmet, W. International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. Eur. Heart J. 2018, 39, 2032–2046. [Google Scholar] [CrossRef] [PubMed]
- Tagawa, M.; Nakamura, Y.; Ishiguro, M.; Satoh, K.; Chinushi, M.; Kodama, M.; Aizawaet, Y. Transient left ventricular apical ballooning developing after the Central Niigata Prefecture Earthquake: Two case reports. J. Cardiol. 2006, 48, 153–158. [Google Scholar] [PubMed]
- Chan, C.; Elliott, J.; Troughton, R.; Frampton, C.; Smyth, D.; Crozier, I.; Bridgman, P. Acute myocardial infarction and stress cardiomyopathy following the Christchurch earthquakes. PLoS ONE 2013, 8, e68504. [Google Scholar] [CrossRef] [PubMed]
- Butterly, S.J.; Indrajith, M.; Garrahy, P.; Ng, A.C.T.; Gould, P.A.; Wang, W.Y.S. Stress- induced takotsubo cardiomyopathy in survivors of the 2011 Queensland floods. Med. J. Aust. 2013, 198, 109–110. [Google Scholar] [CrossRef]
- Brandspiegel, H.Z.; Marinchak, R.A.; Rials, S.J.; Kowey, P.R. A broken heart. Circulation 1998, 98, 1349. [Google Scholar] [CrossRef]
- Ghadri, J.R.; Sarcon, A.; Diekmann, J.; Bataiosu, D.R.; Cammann, V.L.; Jurisic, S.; Napp, L.C.; Jaguszewski, M.; Scherff, F.; Brugger, P. InterTAK Co-investigators. Happy heart syndrome: Role of positive emotional stress in takotsubo syndrome. Eur. Heart J. 2016, 37, 2823–2829. [Google Scholar] [CrossRef]
- McCraty, R.; Atkinson, M.; Tiller, W.A.; Rein, G.; Watkins, A.D. The effects of emotions on short-term power spectrum analysis of heart rate variability. Am. J. Cardiol. 1995, 76, 1089–1093. [Google Scholar] [CrossRef]
- Medina de Chazal, H.; Del Buono, M.G.; Keyser-Marcus, L.; Ma, L.; Gerard Moeller, F.; Berrocal, D.; Abbate, A. Stress Cardiomyopathy Diagnosis and Treatment: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2018, 72, 1955–1971. [Google Scholar] [CrossRef]
- Mitsuma, W.; Kodama, M.; Ito, M. Serial electrocardiographic findings in women with Takotsubo cardiomyopathy. Am. J. Cardiol. 2007, 100, 106–109. [Google Scholar] [CrossRef]
- Ogura, R.; Hiasa, Y.; Takahashi, T.; Yamaguchi, K.; Fujiwara, K.; Ohara, Y.; Nada, T.; Ogata, T.; Kusunoki, K.; Yuba, K. Specific findings of the standard 12-lead ECG in patients with takotsubo cardiomyopathy: Comparison with the findings of acute anterior myocardial infarction. Circ. J. 2003, 67, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Guerra, F.; Rapaj, E.; Pongetti, G.; Fabbrizioli, A.; Pelizzoni, V.; Giannini, I.; Aschieri, D.; Costantini, C.; Capucciet, A. Differences and similarities of repolarization patterns during hospitalization for Takotsubo cardiomyopathy and acute coronary syndrome. Am. J. Cardiol. 2013, 112, 1720–1724. [Google Scholar] [CrossRef] [PubMed]
- Bybee, K.A.; Motiei, A.; Syed, I.S.; Kara, T.; Prasad, A.; Lennon, R.J.; Murphy, J.G.; Hammill, S.C.; Rihal, C.S.; Wright, R.S. Electrocardiography cannot reliably differentiate transient left ventricular apical ballooning syndrome from anterior ST-segment elevation myocardial infarction. J. Electrocardiol. 2007, 40, 38.e1–38.e6. [Google Scholar] [CrossRef] [PubMed]
- Tamura, A.; Watanabe, T.; Ishihara, M.; Ando, S.; Naono, S.; Zaizen, H.; Abe, Y.; Yano, S.; Shinozaki, K.; Kotoku, M. A New Electrocardiographic Criterion to Differentiate Between Takotsubo Cardiomyopathy and Anterior Wall ST-Segment Elevation Acute Myocardial Infarction. Am. J. Cardiol. 2011, 108, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Jim, M.H.; Chan, A.O.; Tsui, P.T.; Lau, S.T.; Siu, C.W.; Chow, W.H.; Lau, C.P. A new ECG criterion to identify takotsubo cardiomyopathy from anterior myocardial infarction: Role of inferior leads. Heart Vessel. 2009, 24, 124–130. [Google Scholar] [CrossRef]
- Parkkonen, O.; Allonen, J.; Vaara, S.; Viitasalo, M.; Nieminen, M.S.; Sinisalo, J. Differences in ST-elevation and T-wave amplitudes do not reliably differentiate takotsubo cardiomyopathy from acute anterior myocardial infarction. J. Electrocardiol. 2014, 47, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Templin, C.; Ghadri, J.R.; Diekmann, J.; Napp, L.C.; Bataiosu, D.R.; Jaguszewski, M.; Cammann, V.L.; Sarcon, A.; Geyer, V.; Neumann, C.A.; et al. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N. Engl. J. Med. 2015, 373, 929–938. [Google Scholar] [CrossRef]
- Budnik, M.; Nowak, R.; Fijałkowski, M.; Kochanowski, J.; Nargiełło, E.; Piątkowski, R.; Peller, M.; Kucharz, J.; Jaguszewski, M.; Gruchała, M. Sex-dependent differences in clinical characteristics and in-hospital outcomes in patients with takotsubo syndrome. Pol. Arch. Intern. Med. 2020, 130, 25–30. [Google Scholar]
- Sasaki, O.; Nishioka, T.; Akima, T.; Tabata, H.; Okamoto, Y.; Akanuma, M.; Uehata, A.; Takase, B.; Katsushika, S.; Isojima, K.; et al. Association of Takotsubo Cardiomyopathy and Long QT Syndrome. Circ. J. 2006, 70, 1220–1222. [Google Scholar] [CrossRef][Green Version]
- Cakici, M.; Cetin, M.; Polat, M.; Neret, A.S. Long QT-induced ventricular tachycardia associated with Takotsubo cardiomyopathy. Turk. Kardiyol. Dern. Ars. 2014, 42, 71–75. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Syed, F.F.; Asirvatham, S.J.; Francis, J. Arrhythmia occurrence with takotsubo cardiomyopathy: A literature review. EP Eur. 2011, 13, 780–788. [Google Scholar] [CrossRef]
- Budnik, M.; Kochanowski, J.; Piatkowski, R.; Wojtetra, K.; Peller, M.; Gaska, M.; Glowacka, P.; Karolczak, P.; Ochijewicz, D.; Opolski, G. Simple markers can distinguish Takotsubo cardiomyopathy from ST segment elevation myocardial infarction. Int. J. Cardiol. 2016, 219, 417–420. [Google Scholar] [CrossRef]
- Haghi, D.; Athanasiadis, A.; Papavassiliu, T.; Suselbeck, T.; Fluechter, S.; Mahrholdt, H.; Borggrefe, M.; Sechtem, U. Right ventricular involvement in Takotsubo cardiomyopathy. Eur. Heart. J. 2006, 27, 2433–2439. [Google Scholar] [CrossRef]
- Liu, K.; Carhart, R. ‘‘Reverse McConnell’s sign?’’: A unique right ventricular feature of Takotsubo cardiomyopathy. Am. J. Cardiol. 2013, 111, 1232–1235. [Google Scholar] [CrossRef]
- Lyon, A.R.; Bossone, E.; Schneider, B.; Sechtem, U.; Citro, R.; Underwood, S.R.; Sheppard, M.N.; Figtree, G.A.; Parodi, G.; Akashi, Y.J.; et al. Current state of knowledge on Takotsubo syndrome: A Position Statement from the Taskforce on Takotsubo Syndrome of the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2016, 18, 8–27. [Google Scholar] [CrossRef]
- Paur, H.; Wright, P.T.; Sikkel, M.; Tranter, M.H.; Mansfield, C.; O’Gara, P.; Stuckey, D.J.; Nikolaev, V.O.; Diakonov, I.; Pannell, L. High level of circulating epinephrine trigger apical cardiodepression in a β2-adrenergic receptor/Gi dependent manner: A new model of Takotsubo cardiomyopathy. Circulation 2012, 126, 697–706. [Google Scholar] [CrossRef]
- Möller, C.; Stiermaier, T.; Brabant, G.; Graf, T.; Thiele, H. Comprehensive assessment of sex hormones in Takotsubo syndrome. Int. J. Cardiol. 2018, 250, 11–15. [Google Scholar] [CrossRef]
- Pizzino, G.; Bitto, A.; Crea, P.; Khandheria, B.; Vriz, O.; Carerj, S.; Squadrito, F.; Minisini, R.; Citro, R.; Cusmà-Piccione, M.; et al. Takotsubo syndrome and estrogen receptor genes: Partners in crime? J. Cardiovasc. Med. 2017, 18, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Emre, A.; Sahin, S.; Erzik, C.; Nurkalem, Z.; Oz, D.; Cirakoglu, B.; Yesilcimen, K.; Erseket, B. Effect of Hormone Replacement Therapy on Plasma Lipoproteins and Apolipoproteins, Endothelial Function and Myocardial Perfusion in Postmenopausal Women with Estrogen Receptor-α IVS1–397 C/C Genotype and Established Coronary Artery Disease. Cardiology 2006, 106, 44–50. [Google Scholar] [CrossRef]
- Kobylecka, M.; Budnik, M.; Kochanowski, J.; Piatkowski, R.; Chojnowski, M.; Fronczewska-Wieniawska, K.; Mazurek, T.; Maczewska, J.; Peller, M.; Opolski, G.; et al. Takotsubo cardiomyopathy: FDG myocardial uptake pattern in fasting patients. Comparison of PET/CT, SPECT, and ECHO results. J. Nucl. Cardiol. 2018, 25, 1260–1270. [Google Scholar] [CrossRef]
- Akashi, Y.; Lyon, A. Microcirculatory dysfunction and autonomic disturbance in Takotsubo syndrome. Nat. Rev. Cardiol. 2015, 12, 497. [Google Scholar] [CrossRef]
- Naegele, M.; Flammer, A.; Enseleit, F.; Enseleit, F.; Roas, S.; Frank, M.; Hirt, A.; Kaiser, P.; Cantatore, S.; Templin, C.; et al. Endothelial function and sympathetic nervous system activity in patients with Takotsubo syndrome. Int. J. Cardiol. 2016, 224, 226–230. [Google Scholar] [CrossRef]
- Carbonara, R.; Giardinelli, F.; Pepe, M.; Luzzi, G.; Panettieri, I.; Vulpis, V.; Bortone, A.S.; Ciccone, M.M. Correlation between endothelial dysfunction and myocardial damage in acute phase of Tako-Tsubo cardiomyopathy: Brachial flow mediated dilation as a potential marker for assessment of patient with Tako-Tsubo. Heart Vessel. 2018, 33, 291–298. [Google Scholar] [CrossRef]
- Wilson, H.M.; Cheyenne, L.; Brown, P.A.; Ketrr, K.; Hannah, A.; Srinivasan, J.; Duniak, N.; Horgan, G.; Dawson, D. Characterization of the myocardial inflammatory response in acute stress-induced (Takotsubo) cardiomyopathy. J. Am. Coll. Cardiol. Basic Trans. Sci. 2018, 3, 766–778. [Google Scholar] [CrossRef]
- Steptoe, A.; Kivimäki, M. Stress and cardiovascular disease. Nat. Rev. Cardiol. 2012, 9, 360–370. [Google Scholar] [CrossRef]
- Land, S.; Niederer, S.; Louch, W.; Røe, A.T.; Aronsen, J.M.; Stuckey, D.J.; Sikkel, M.B.; Tranter, M.H.; Lyon, A.R.; Harding, S.E.; et al. Computational modeling of Takotsubo cardiomyopathy: Effect of spatially varying beta-adrenergic stimulation in the rat left ventricle. Am. J. Physiol. Heart Circ. Physiol. 2014, 307, H1487–H1496. [Google Scholar] [CrossRef]
- Heubach, J.F.; Ravens, U.; Kaumann, A.J. Epinephrine activates both Gs and Gi pathways, but norepinephrine activates only the Gs pathway through human 2-adrenoceptors overexpressed in mouse heart. Mol. Pharmacol. 2004, 65, 1313–1322. [Google Scholar] [CrossRef]
- Wright, P.; Bhopal, N.; Diakonov, I.; Pannell, L.; Perera, R.; Bork, N.; Schobesberger, S.; Lucarelli, C.; Faggian, G.; Alvarez-Laviada, A.; et al. Cardiomyocyte membrane structure and cAMP compartmentation produce anatomical variation in beta2AR-cAMP responsiveness in murine hearts. Cell Rep. 2018, 23, 459–469. [Google Scholar] [CrossRef]
- Shao, Y.; Redfors, B.; Ståhlman, M.; Täng, M.S.; Miljanovic, A.; Möllmann, H.; Troidl, C.; Szardien, S.; Hamm, C.; Nef, H.; et al. A mouse model reveals an important role for catecholamine-induced lipotoxicity in the pathogenesis of stress-induced cardiomyopathy. Eur. J. Heart Fail. 2013, 15, 9–22. [Google Scholar] [CrossRef]
- Suzuki, H.; Matsumoto, Y.; Kaneta, T.; Sugimura, K.; Takahashi, J.; Fukumoto, Y.; Talkahashi, S.; Shimokawa, H. Evidence for brain activation in patients with Takotsubo cardiomyopathy. Circ. J. 2014, 78, 256–258. [Google Scholar] [CrossRef]
- Templin, C.; Hänggi, J.; Klein, C.; Topka, M.S.; Hiestand, T.; Levinson, R.A.; Jurisic, S.; Lüscher, T.F.; Ghadri, J.-R.; Jäncke, L. Altered limbic and autonomic processing supports brain-heart axis in Takotsubo syndrome. Eur. Heart J. 2019, 40, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Hiestand, T.; Hänggi, J.; Klein, C.; Topka, M.S.; Jaguszewski, M.; Ghadri, J.R.; Lüscher, T.F.; Jäncke, L.; Templin, C. Takotsubo syndrome associated with structural brain alterations of the limbic system. Am. Coll. Cardiol. 2018, 71, 809–811. [Google Scholar] [CrossRef] [PubMed]
- Drevets, W.C.; Savitz, J.; Trimble, M. The subgenual anterior cingulate cortex in mood disorders. CNS Spectr. 2008, 13, 663–681. [Google Scholar] [CrossRef] [PubMed]
- Stepniewska, A.; Budnik, M.; Krzemiński, K.; Niewiadomski, W.; Gąsiorowska, A.; Opolski, G.; Kochanowski, J.; Mieczkowska, K.; Żukowska, K.; Szepietowska, K.; et al. Impaired hemodynamic response to tilt, handgrip and Valsalva manoeuvre in patients with takotsubo syndrome. Auton. Neurosci. 2019, 220, 102555. [Google Scholar] [CrossRef]
- Cherian, J.; Angelis, D.; Filiberti, A.; Saperia, G. Can takotsubo cardiomyopathy be familial? Int. J. Cardiol. 2007, 121, 74–75. [Google Scholar] [CrossRef]
- Kumar, G.; Holmes, D.R., Jr.; Prasad, A. “Familial” apical ballooning syndrome (Takotsubo cardiomyopathy). Int. J. Cardiol. 2010, 144, 444–445. [Google Scholar] [CrossRef]
- Ikutomi, M.; Yamasaki, M.; Matsusita, M.; Watari, Y.; Arashi, H.; Endo, G.; Yamaguchi, J.I.; Ohnishi, S. Takotsubo cardiomyopathy in siblings. Heart Vessel. 2014, 29, 119–122. [Google Scholar] [CrossRef]
- Caretta, G.; Robba, D.; Vizzardi, E.; Bonadei, I.; Raddino, R.; Metra, M. Tako-tsubo cardiomyopathy in two sisters: A chance finding or familial predisposition? Clin. Res. Cardiol. 2015, 104, 614–616. [Google Scholar] [CrossRef]
- Vriz, O.; Minisini, R.; Citro, R.; Guerra, V.; Zito, C.; De Luca, G.; Pavan, D.; Pirisi, M.; Limongelli, G.; Bossone, E. Analysis of beta1 and beta2-adrenergic receptors polymorphism in patients with apical ballooning cardiomyopathy. Acta Cardiol. 2011, 66, 787–790. [Google Scholar] [CrossRef]
- Spinelli, L.; Trimarco, V.; Di Marino, S.; Marino, M.; Iaccarino, G.; Trimarco, B. L41Q polymorphism of the G protein coupled receptor kinase 5 is associated with left ventricular apical ballooning syndrome. Eur. J. Heart Fail. 2010, 12, 13–16. [Google Scholar] [CrossRef]
- Citro, R.; D’Avenia, M.; DeMarco, M.; Giudice, R.; Mirra, M.; Ravera, A.; Silverio, A.; Farina, R.; Silvestri, F.; Gravina, P.; et al. Polymorphisms of the antiapoptotic protein bag3 may play a role in the pathogenesis of tako-tsubo cardiomyopathy. Int. J. Cardiol. 2013, 168, 1663–1665. [Google Scholar] [CrossRef]
- Novo, G.; Giambanco, S.; Guglielmo, M.; Arvigo, L.; Sutera, M.R.; Giambanco, F.; Evola, S.; Vaccarino, L.; Bova, M.; Lio, D.; et al. G-protein-coupled receptor kinase 5 polymorphism and Takotsubo cardiomyopathy. J. Cardiovasc. Med. 2015, 16, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Borchert, T.; Hubscher, D.; Guessoum, C.I.; Lam, T.D.D.; Ghadri, G.R.; Schellinger, I.N.; Tiburcy, M.; Liaw, N.Y.; Li, Y.; Haas, J.; et al. Catecholamine-dependent beta-adrenergic signaling in a pluripotent stem cell model of Takotsubo cardiomyopathy. J. Am. Coll. Cardiol. 2017, 70, 975–991. [Google Scholar] [CrossRef]
- Goodloe, A.H.; Evans, J.M.; Middha, S.; Prasad, A.; Olson, T.M. Characterizing genetic variation of adrenergic signalling pathways in Takotsubo (stress) cardiomyopathy exomes. Eur. J. Heart Fail. 2014, 16, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Limongelli, G.; D’Alessandro, R.; Masarone, D.; Maddaloni, V.; Vriz, O.; Minisini, R.; Citro, R.; Calabrò, P.; Russo, M.G.; Calabrò, R.; et al. Takotsubo cardiomyopathy: Do the genetics matter? Heart Fail. Clin. 2013, 9, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Figtree, G.A.; Bagnall, R.D.; Abdulla, I.; Buchholz, S.; Galougahi, K.K.; Yan, W.; Tan, T.; Neil, C.; Horowitz, J.; Semsarian, C.; et al. No association of G-protein-coupled receptor kinase 5 or β-adrenergic receptor polymorphisms with Takotsubo cardiomyopathy in a large Australian cohort. Eur. J. Heart Fail. 2013, 15, 730–733. [Google Scholar] [CrossRef]
- Mattsson, E.; Saliba-Gustafsson, P.; Ehrenborg, E.; Tornvall, P. Lack of genetic susceptibility in takotsubo cardiomyopathy: A case-control study. BMC Med. Genet. 2018, 19, 391–397. [Google Scholar] [CrossRef]
- Ghadri, J.R.; Wittstein, I.S.; Prasad, A.; Sharkey, S.; Dote, K.; Akashi, Y.J.; Cammann, V.L.; Crea, F.; Galiuto, L.; Desmet, W.; et al. International Expert Consensus Document on Takotsubo Syndrome (Part II): Diagnostic Workup, Outcome, and Management. Eur. Heart J. 2018, 39, 2047–2062. [Google Scholar] [CrossRef]
- Singh, K.; Parsaik, A.; Singh, B. Recurrent takotsubo cardiomyopathy: Variable pattern of ventricular involvement. Herz 2013, 39, 963–967. [Google Scholar] [CrossRef]
- Opolski, G.; Kochanowski, J.; Torbicki, A.; Scisło, P.; Kowalik, R.; Piotrowska-Kownacka, D.; Zarebiński, M.; Pruszczyk, P.; Kalarus, Z. The recurrence after ten years—“Mother in-law variant” of tako-tsubo syndrome. Kardiol. Pol. 2010, 68, 557–561. [Google Scholar]
- Opolski, G.; Budnik, M.; Kochanowski, J.; Kowalik, R.; Piatkowski, R.; Kochman, J. Four episodes of takotsubo cardiomyopathy in one patient. Int. J. Cardiol. 2016, 203, 53–54. [Google Scholar] [CrossRef]
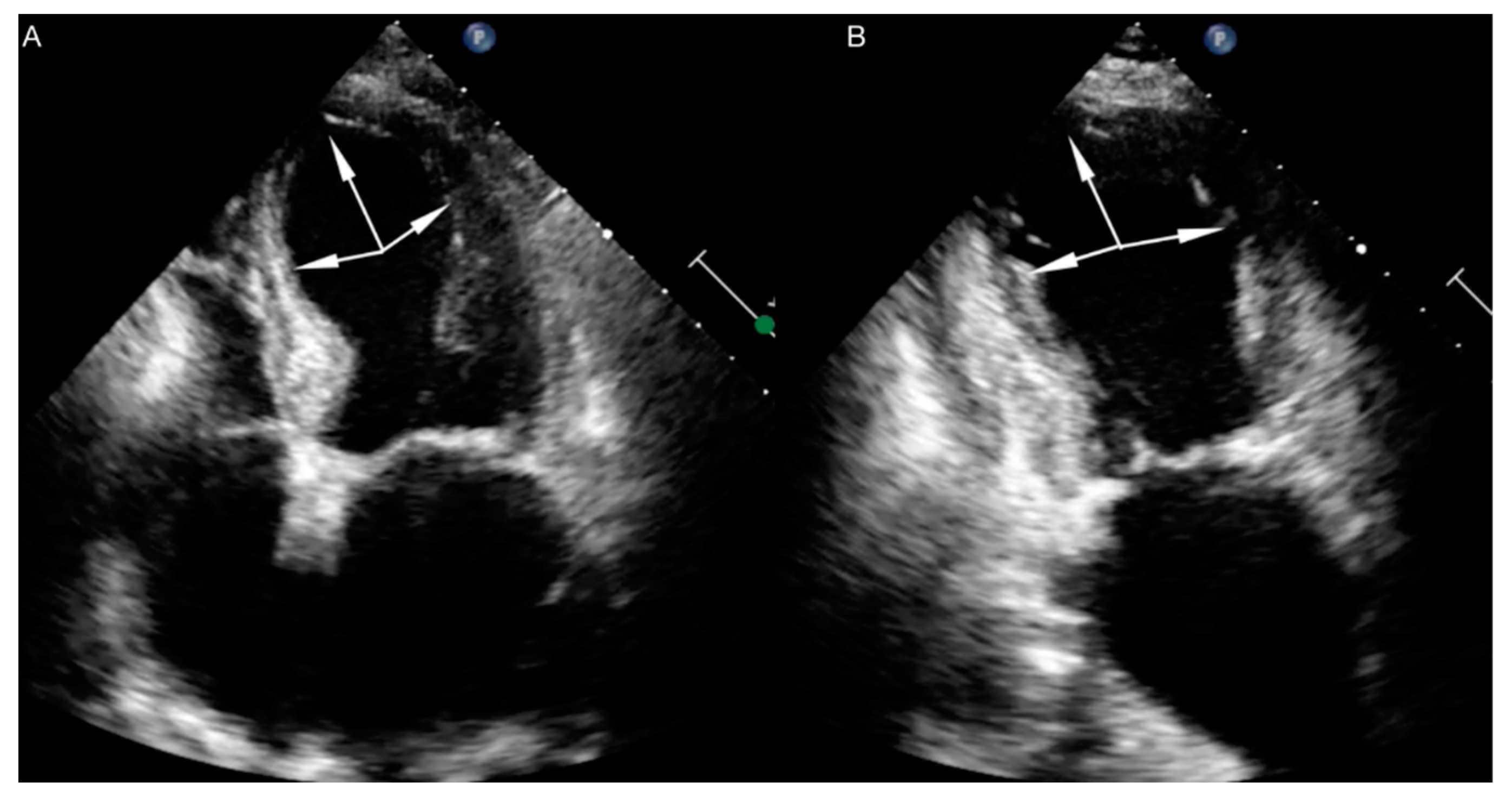
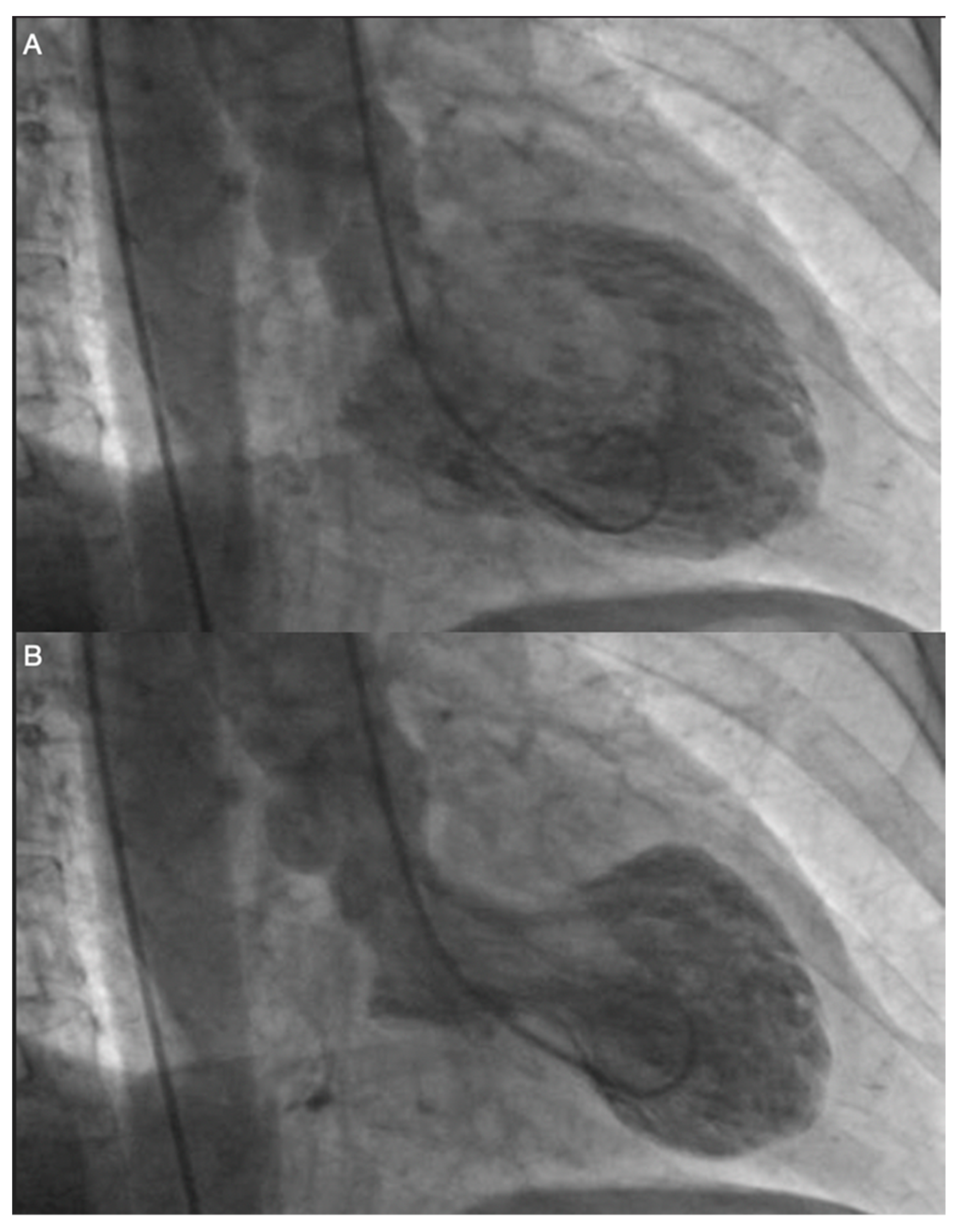
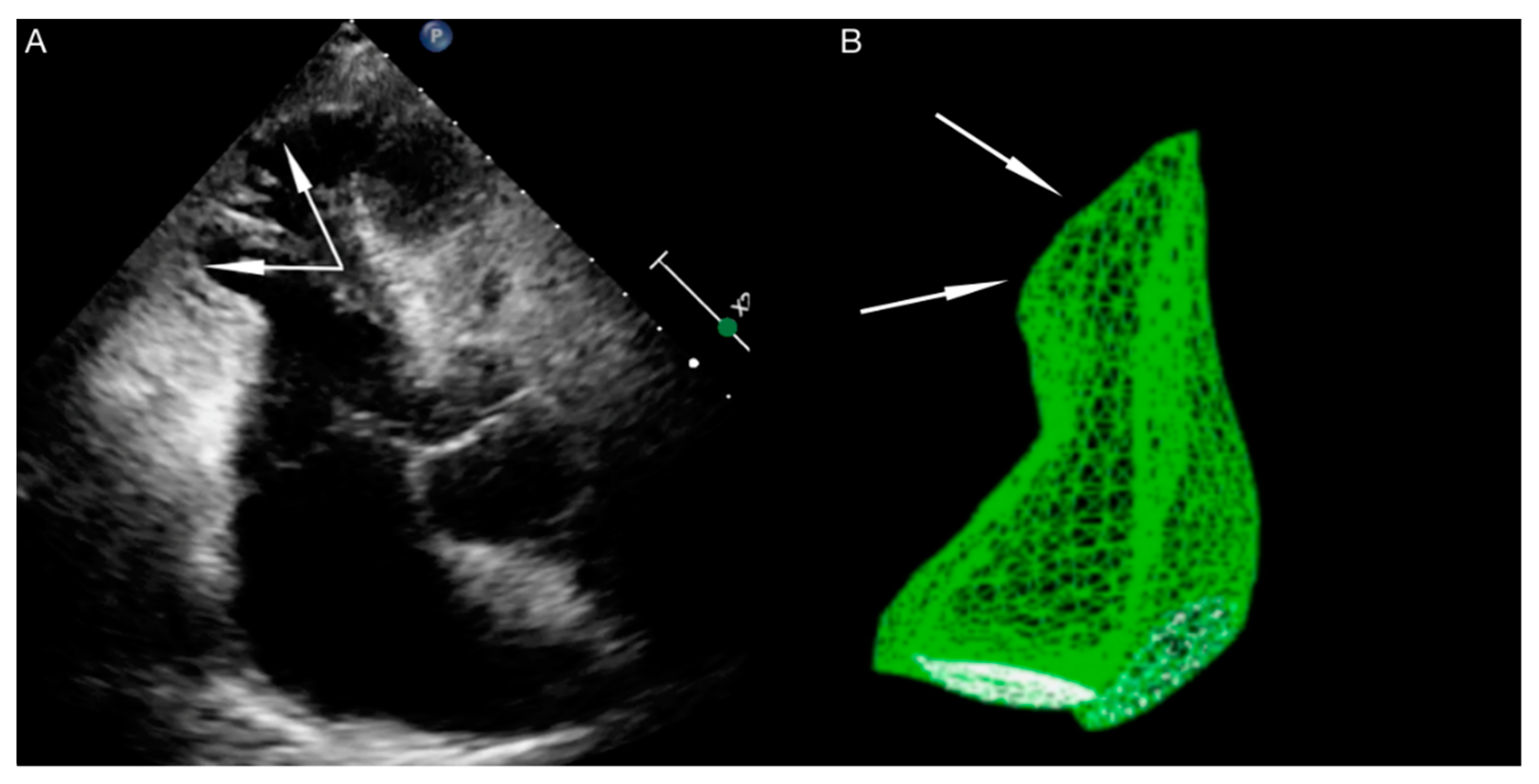
| International Takotsubo Diagnostic Criteria (InterTAK Diagnostic Criteria) a |
|---|
|
| InterTAK Classification |
|---|
| Class I: Takotsubo syndrome related to emotional stress Class II: Takotsubo syndrome related to physical stress Class IIa: Takotsubo syndrome secondary to physical activities, medical conditions, or procedures Class IIb: Takotsubo syndrome secondary to neurologic disorders Class III: Takotsubo syndrome without an identifiable triggering factor |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Budnik, M.; Piątkowski, R.; Ochijewicz, D.; Zaleska, M.; Grabowski, M.; Opolski, G. Pathophysiology of Takotsubo Syndrome as A Bridge to Personalized Treatment. J. Pers. Med. 2021, 11, 879. https://doi.org/10.3390/jpm11090879
Budnik M, Piątkowski R, Ochijewicz D, Zaleska M, Grabowski M, Opolski G. Pathophysiology of Takotsubo Syndrome as A Bridge to Personalized Treatment. Journal of Personalized Medicine. 2021; 11(9):879. https://doi.org/10.3390/jpm11090879
Chicago/Turabian StyleBudnik, Monika, Radosław Piątkowski, Dorota Ochijewicz, Martyna Zaleska, Marcin Grabowski, and Grzegorz Opolski. 2021. "Pathophysiology of Takotsubo Syndrome as A Bridge to Personalized Treatment" Journal of Personalized Medicine 11, no. 9: 879. https://doi.org/10.3390/jpm11090879
APA StyleBudnik, M., Piątkowski, R., Ochijewicz, D., Zaleska, M., Grabowski, M., & Opolski, G. (2021). Pathophysiology of Takotsubo Syndrome as A Bridge to Personalized Treatment. Journal of Personalized Medicine, 11(9), 879. https://doi.org/10.3390/jpm11090879






