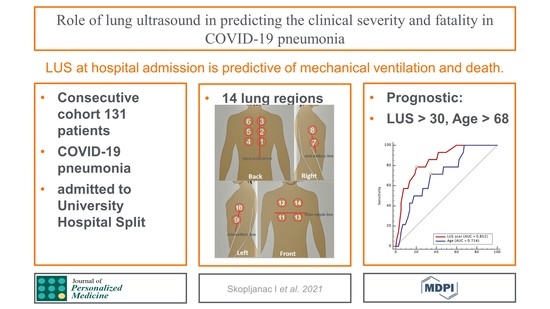
Role of Lung Ultrasound in Predicting Clinical Severity and Fatality in COVID-19 Pneumonia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion and Exclusion Criteria
Outcomes
2.3. Data Extraction
2.4. Acquisition Protocol
2.5. Bias
2.6. Study Size
2.7. Statistical Analysis
2.8. Reporting
3. Results
3.1. Patients and Characteristics
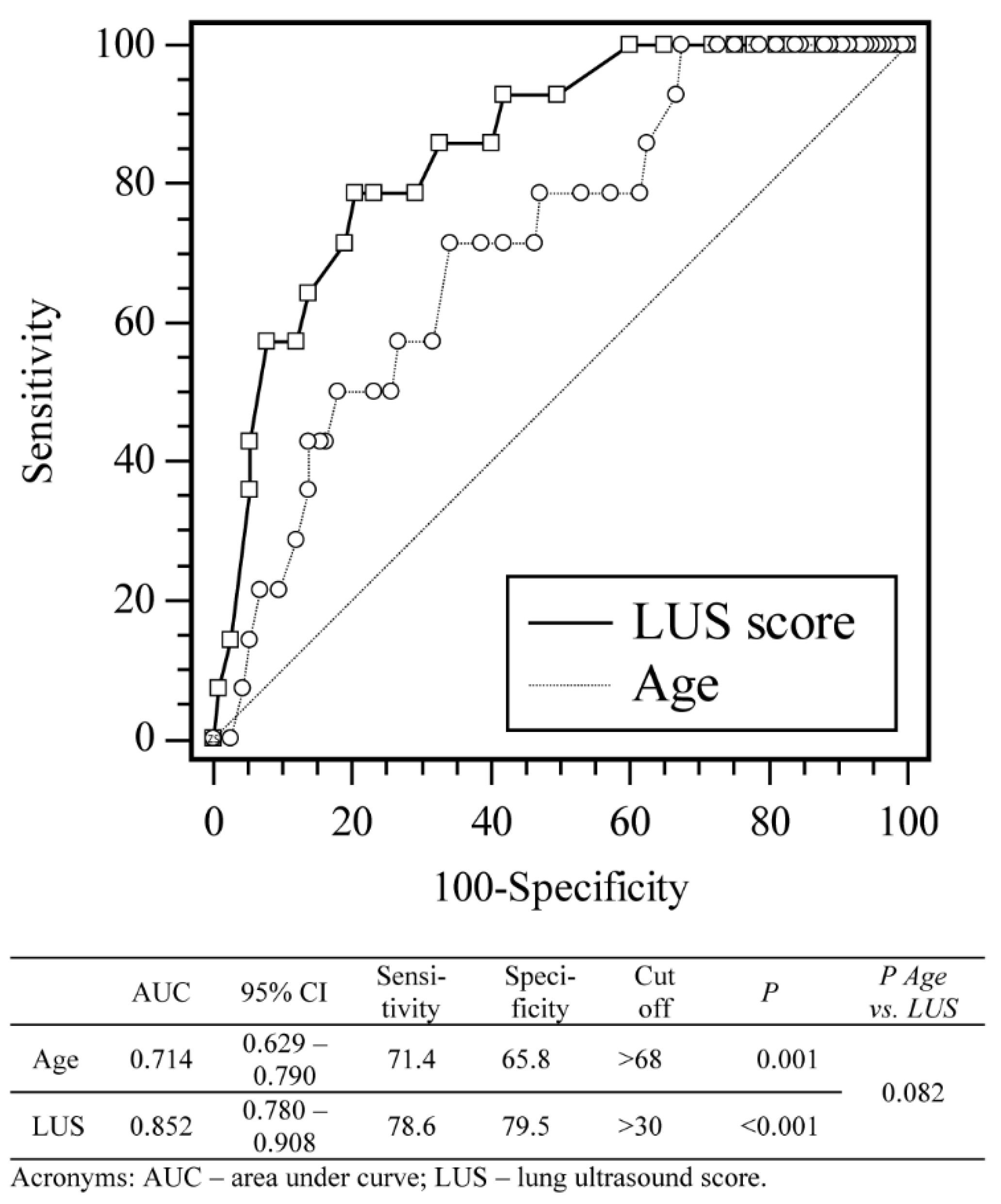
3.2. Impact of LUS Score on the Highest Respiratory Support Modality Used
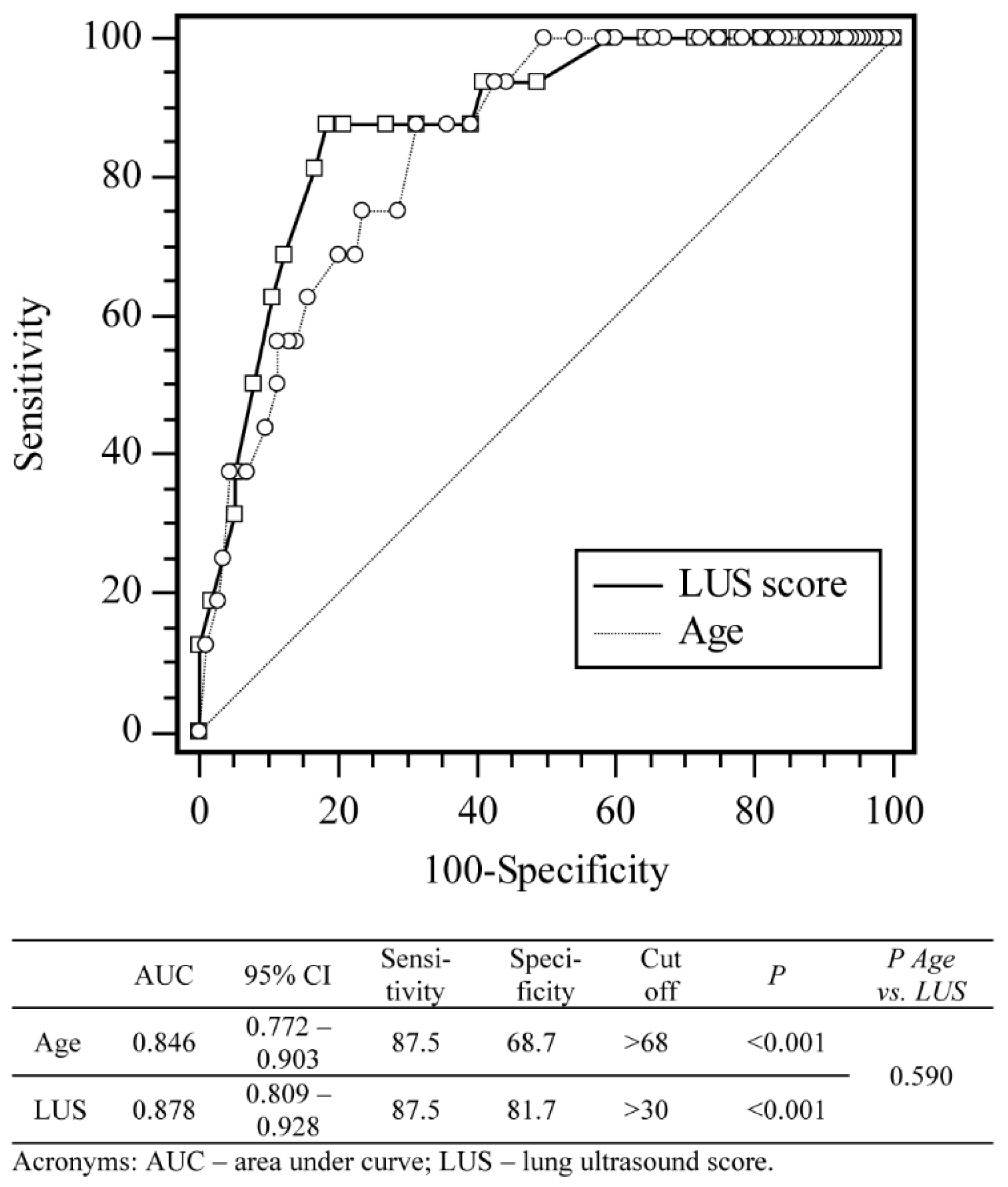
3.3. Impact of LUS Score and Risk Factors on the Patient Mortality
4. Discussion
4.1. Strengths
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, Evaluation, and Treatment of Coronavirus. In StatPearls; Uniformed Services University Of The Health Sciences: Treasure Island, FL, USA, 2020. [Google Scholar]
- Pascarella, G.; Strumia, A.; Piliego, C.; Bruno, F.; Del Buono, R.; Costa, F.; Scarlata, S.; Agro, F.E. COVID-19 diagnosis and management: A comprehensive review. J. Intern. Med. 2020, 288, 192–206. [Google Scholar] [CrossRef]
- Mohammad, S.; Aziz, R.; Al Mahri, S.; Malik, S.S.; Haji, E.; Khan, A.H.; Khatlani, T.S.; Bouchama, A. Obesity and COVID-19: What makes obese host so vulnerable? Immun. Ageing I A 2021, 18, 1. [Google Scholar] [CrossRef]
- Ji, D.; Zhang, D.; Xu, J.; Chen, Z.; Yang, T.; Zhao, P.; Chen, G.; Cheng, G.; Wang, Y.; Bi, J.; et al. Prediction for Progression Risk in Patients With COVID-19 Pneumonia: The CALL Score. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020, 71, 1393–1399. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Kovalic, A.J.; Graber, C.J. Prognostic Value of Leukocytosis and Lymphopenia for Coronavirus Disease Severity. Emerg. Infect. Dis. 2020, 26, 1839–1841. [Google Scholar] [CrossRef]
- Luo, X.; Zhou, W.; Yan, X.; Guo, T.; Wang, B.; Xia, H.; Ye, L.; Xiong, J.; Jiang, Z.; Liu, Y.; et al. Prognostic Value of C-Reactive Protein in Patients With Coronavirus 2019. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020, 71, 2174–2179. [Google Scholar] [CrossRef]
- Kaufman, A.E.; Naidu, S.; Ramachandran, S.; Kaufman, D.S.; Fayad, Z.A.; Mani, V. Review of radiographic findings in COVID-19. World J. Radiol. 2020, 12, 142–155. [Google Scholar] [CrossRef]
- Khosravi, B.; Aghaghazvini, L.; Sorouri, M.; Naybandi Atashi, S.; Abdollahi, M.; Mojtabavi, H.; Khodabakhshi, M.; Motamedi, F.; Azizi, F.; Rajabi, Z.; et al. Predictive value of initial CT scan for various adverse outcomes in patients with COVID-19 pneumonia. Heart Lung J. Crit. Care 2021, 50, 13–20. [Google Scholar] [CrossRef]
- Francone, M.; Iafrate, F.; Masci, G.M.; Coco, S.; Cilia, F.; Manganaro, L.; Panebianco, V.; Andreoli, C.; Colaiacomo, M.C.; Zingaropoli, M.A.; et al. Chest CT score in COVID-19 patients: Correlation with disease severity and short-term prognosis. Eur. Radiol. 2020, 30, 6808–6817. [Google Scholar] [CrossRef]
- Touw, H.R.; Tuinman, P.R.; Gelissen, H.P.; Lust, E.; Elbers, P.W. Lung ultrasound: Routine practice for the next generation of internists. Neth. J. Med. 2015, 73, 100–107. [Google Scholar]
- Jackson, K.; Butler, R.; Aujayeb, A. Lung ultrasound in the COVID-19 pandemic. Postgrad. Med. J. 2021, 97, 34–39. [Google Scholar] [CrossRef]
- Lieveld, A.W.E.; Kok, B.; Schuit, F.H.; Azijli, K.; Heijmans, J.; van Laarhoven, A.; Assman, N.L.; Kootte, R.S.; Olgers, T.J.; Nanayakkara, P.W.B.; et al. Diagnosing COVID-19 pneumonia in a pandemic setting: Lung Ultrasound versus CT (LUVCT)—A multicentre, prospective, observational study. ERJ Open Res. 2020, 6. [Google Scholar] [CrossRef]
- Ragnoli, B.; Malerba, M. Focus on the Potential Role of Lung Ultrasound in COVID-19 Pandemic: What More to Do? Int. J. Environ. Res. Public Health 2020, 17, 8398. [Google Scholar] [CrossRef]
- Ottaviani, S.; Franc, M.; Ebstein, E.; Demaria, L.; Lheure, C.; Debray, M.P.; Khalil, A.; Crestani, B.; Borie, R.; Dieude, P. Lung ultrasonography in patients with COVID-19: Comparison with CT. Clin. Radiol. 2020, 75, 877.e1–877.e6. [Google Scholar] [CrossRef]
- Lu, W.; Zhang, S.; Chen, B.; Chen, J.; Xian, J.; Lin, Y.; Shan, H.; Su, Z.Z. A Clinical Study of Noninvasive Assessment of Lung Lesions in Patients with Coronavirus Disease-19 (COVID-19) by Bedside Ultrasound. Ultraschall Der Med. 2020, 41, 300–307. [Google Scholar] [CrossRef]
- Nouvenne, A.; Zani, M.D.; Milanese, G.; Parise, A.; Baciarello, M.; Bignami, E.G.; Odone, A.; Sverzellati, N.; Meschi, T.; Ticinesi, A. Lung Ultrasound in COVID-19 Pneumonia: Correlations with Chest CT on Hospital admission. Respir. Int. Rev. Thorac. Dis. 2020, 99, 617–624. [Google Scholar] [CrossRef]
- Pivetta, E.; Goffi, A.; Tizzani, M.; Locatelli, S.M.; Porrino, G.; Losano, I.; Leone, D.; Calzolari, G.; Vesan, M.; Steri, F.; et al. Lung Ultrasonography for the Diagnosis of SARS-CoV-2 Pneumonia in the Emergency Department. Ann. Emerg. Med. 2021, 77, 385–394. [Google Scholar] [CrossRef]
- Gutsche, H.; Lesser, T.G.; Wolfram, F.; Doenst, T. Significance of Lung Ultrasound in Patients with Suspected COVID-19 Infection at Hospital Admission. Diagnostics 2021, 11, 921. [Google Scholar] [CrossRef]
- Ji, L.; Cao, C.; Gao, Y.; Zhang, W.; Xie, Y.; Duan, Y.; Kong, S.; You, M.; Ma, R.; Jiang, L.; et al. Prognostic value of bedside lung ultrasound score in patients with COVID-19. Crit. Care 2020, 24, 700. [Google Scholar] [CrossRef]
- Castelao, J.; Graziani, D.; Soriano, J.B.; Izquierdo, J.L. Findings and Prognostic Value of Lung Ultrasound in COVID-19 Pneumonia. J. Ultrasound Med. Off. J. Am. Inst. Ultrasound Med. 2020, 40, 1315–1324. [Google Scholar] [CrossRef]
- Lichter, Y.; Topilsky, Y.; Taieb, P.; Banai, A.; Hochstadt, A.; Merdler, I.; Gal Oz, A.; Vine, J.; Goren, O.; Cohen, B.; et al. Lung ultrasound predicts clinical course and outcomes in COVID-19 patients. Intensive Care Med. 2020, 46, 1873–1883. [Google Scholar] [CrossRef]
- Brahier, T.; Meuwly, J.Y.; Pantet, O.; Brochu Vez, M.J.; Gerhard Donnet, H.; Hartley, M.A.; Hugli, O.; Boillat-Blanco, N. Lung ultrasonography for risk stratification in patients with COVID-19: A prospective observational cohort study. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020. [Google Scholar] [CrossRef]
- de Alencar, J.C.G.; Marchini, J.F.M.; Marino, L.O.; da Costa Ribeiro, S.C.; Bueno, C.G.; da Cunha, V.P.; Lazar Neto, F.; Brandao Neto, R.A.; Souza, H.P.; Team, C.U.S.P.R. Lung ultrasound score predicts outcomes in COVID-19 patients admitted to the emergency department. Ann. Intensive Care 2021, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Ramos Hernandez, C.; Botana Rial, M.; Pazos Area, L.A.; Nunez Fernandez, M.; Perez Fernandez, S.; Rubianes Gonzalez, M.; Crespo Casal, M.; Fernandez Villar, A. Lung Ultrasound to Predict Unfavorable Progress in Patients Hospitalized for COVID-19. Arch. Bronconeumol. 2021, 57 (Suppl. S1), 47–54. [Google Scholar] [CrossRef]
- WHO. Diagnostic Testing for SARS-CoV-2. Available online: https://www.who.int/publications/i/item/diagnostic-testing-for-sars-cov-2 (accessed on 18 April 2021).
- Soldati, G.; Smargiassi, A.; Inchingolo, R.; Buonsenso, D.; Perrone, T.; Briganti, D.F.; Perlini, S.; Torri, E.; Mariani, A.; Mossolani, E.E.; et al. Proposal for International Standardization of the Use of Lung Ultrasound for Patients With COVID-19: A Simple, Quantitative, Reproducible Method. J. Ultrasound Med. Off. J. Am. Inst. Ultrasound Med. 2020, 39, 1413–1419. [Google Scholar] [CrossRef] [Green Version]
- Zanforlin, A.; Strapazzon, G.; Falk, M.; Gallina, V.; Viteritti, A.; Valzolgher, L.; La Guardia, M.; Ferro, F.; Pagani, L.; Vezzali, N. Lung Ultrasound in the Emergency Department for Early Identification of COVID-19 Pneumonia. Respir. Int. Rev. Thorac. Dis. 2021, 100, 145–153. [Google Scholar] [CrossRef]
- Smith, M.J.; Hayward, S.A.; Innes, S.M.; Miller, A.S.C. Point-of-care lung ultrasound in patients with COVID-19—A narrative review. Anaesthesia 2020, 75, 1096–1104. [Google Scholar] [CrossRef] [Green Version]
- Falgarone, G.; Pamoukdjian, F.; Cailhol, J.; Giocanti-Auregan, A.; Guis, S.; Bousquet, G.; Bouchaud, O.; Seror, O. Lung ultrasound is a reliable diagnostic technique to predict abnormal CT chest scan and to detect oxygen requirements in COVID-19 pneumonia. Aging 2020, 12, 19945–19953. [Google Scholar] [CrossRef]
- European Commission. Attitudes of Europeans towards Tobacco and Electronic cigarettes: Special Eurobarometer 458; European Commission: Brussels, Belgium, 2017. [Google Scholar] [CrossRef]
- Usman, M.S.; Siddiqi, T.J.; Khan, M.S.; Patel, U.K.; Shahid, I.; Ahmed, J.; Kalra, A.; Michos, E.D. Is there a smoker’s paradox in COVID-19? BMJ Evid. Based Med. 2020. [Google Scholar] [CrossRef]
- Gonzalez-Rubio, J.; Navarro-Lopez, C.; Lopez-Najera, E.; Lopez-Najera, A.; Jimenez-Diaz, L.; Navarro-Lopez, J.D.; Najera, A. A Systematic Review and Meta-Analysis of Hospitalised Current Smokers and COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 7394. [Google Scholar] [CrossRef]
- Farsalinos, K.; Bagos, P.G.; Giannouchos, T.; Niaura, R.; Barbouni, A.; Poulas, K. Smoking prevalence among hospitalized COVID-19 patients and its association with disease severity and mortality: An expanded re-analysis of a recent publication. Harm Reduct. J. 2021, 18, 9. [Google Scholar] [CrossRef]
- Strzelak, A.; Ratajczak, A.; Adamiec, A.; Feleszko, W. Tobacco Smoke Induces and Alters Immune Responses in the Lung Triggering Inflammation, Allergy, Asthma and Other Lung Diseases: A Mechanistic Review. Int. J. Environ. Res. Public Health 2018, 15, 1033. [Google Scholar] [CrossRef] [Green Version]
- Sopori, M. Effects of cigarette smoke on the immune system. Nat. Rev. Immunol. 2002, 2, 372–377. [Google Scholar] [CrossRef]
- Nizri, E.; Irony-Tur-Sinai, M.; Lory, O.; Orr-Urtreger, A.; Lavi, E.; Brenner, T. Activation of the cholinergic anti-inflammatory system by nicotine attenuates neuroinflammation via suppression of Th1 and Th17 responses. J. Immunol. 2009, 183, 6681–6688. [Google Scholar] [CrossRef] [Green Version]
- Arnson, Y.; Shoenfeld, Y.; Amital, H. Effects of tobacco smoke on immunity, inflammation and autoimmunity. J. Autoimmun. 2010, 34, J258–J265. [Google Scholar] [CrossRef]
- Margaritopoulos, G.A.; Vasarmidi, E.; Jacob, J.; Wells, A.U.; Antoniou, K.M. Smoking and interstitial lung diseases. Eur. Respir. Rev. Off. J. Eur. Respir. Soc. 2015, 24, 428–435. [Google Scholar] [CrossRef]
- Rojatti, M.; Regli, I.B.; Zanforlin, A.; Ferretti, E.; Falk, M.; Strapazzon, G.; Gamper, M.; Zanon, P.; Bock, M.; Rauch, S. Lung Ultrasound and Respiratory Pathophysiology in Mechanically Ventilated COVID-19 Patients-an Observational Trial. SN Compr. Clin. Med. 2020, 2, 1970–1977. [Google Scholar] [CrossRef] [PubMed]
| Score | Ultrasonic Observation |
|---|---|
| 0 | Regular finding: existence of a regular and not thickened pleural line, with a sliding sign, and the presence of A-lines; |
| 1 | Some loss of aeration: irregular pleural line with some B lines; |
| 2 | Severe loss of aeration: broken pleural line; small-to-large consolidated areas with associated areas of white below the consolidated area; |
| 3 | Complete loss of aeration: scanned area shows large, dense consolidations; “white lung”. |
| Comorbidity | Number (%) of Patients | p * | |||||
|---|---|---|---|---|---|---|---|
| 0 L | 1–10 L | 10–16 L or Venturi | HFNC | MV | Total | ||
| Arterial hypertension | 7 (50) | 46 (65.7) | 15 (75.0) | 11 (73.3) | 9 (69.2) | 88 (66.7) | 0.63 |
| Diabetes | 4 (28.6) | 16 (22.9) | 6 (30.0) | 4 (26.7) | 3 (23.1) | 33 (25) | 0.94 |
| Cardiovascular disease | 0 (0.0) | 19 (27.1) | 1 (5.0) | 4 (33.3) | 6 (46.2) | 30 (22.9) | 0.007 |
| Liver failure | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (6.7) | 0 (0.0) | 1 (0.8) | 0.32 |
| Lymphoma | 2 (14.3) | 2 (2.9) | 0 (0.0) | 1 (6.7) | 0 (0.0) | 5 (3.8) | 0.20 |
| Leukemia | 0 (0.0) | 0 (0.0) | 1 (5.0) | 0 (0.0) | 2 (15.4) | 3 (2.3) | 0.03 |
| Malignancy | 3 (21.4) | 7 (10) | 1 (5.0) | 3 (20) | 1 (8.3) | 15 (11.5) | 0.44 |
| Peripheral vascular disease | 0 (0.0) | 3 (4.3) | 2 (10.0) | 1 (6.7) | 1 (8.3) | 6 (4.6) | 0.59 |
| Dementia | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (6.7) | 0 (0.0) | 1 (0.8) | 0.32 |
| Myocardial infarction | 0 (0.0) | 6 (8.6) | 1 (5.0) | 1 (6.7) | 1 (8.3) | 9 (6.9) | 0.95 |
| CVI or TIA | 0 (0.0) | 1 (1.4) | 1 (5.0) | 0 (0.0) | 0 (0.0) | 2 (1.5) | 0.72 |
| COPD | 1 (7.1) | 3 (4.3) | 0 (0.0) | 1 (6.7) | 0 (0.0) | 4 (3.1) | 0.66 |
| Rheumatological disease | 1 (7.1) | 1 (1.4) | 1 (5.0) | 0 (0.0) | 0 (0.0) | 3 (2.3) | 0.39 |
| Hemiplegia | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (8.3) | 1 (0.8) | 0.09 |
| Renal failure | 0 (0.0) | 3 (4.3) | 1 (5.0) | 0 (0.0) | 0 (0.0) | 4 (3.1) | >0.99 |
| Mean ± SD or Median (IQR) According to the Highest Used Level of Respiratory Support Modality | p * | |||||
|---|---|---|---|---|---|---|
| 0 L (n = 14) | 1–10 L (n = 69) | 11–16 L or Venturi (n = 20) | HFNC (n = 14) | MV (n = 14) | ||
| LUS score | 10 (3–21) | 24 (19–26) | 32 (27–35) | 35 (32–38) | 36 (31–38) | <0.0001 † |
| Age (years) | 61.3 ± 16.0 | 63.8 ± 11.8 | 64.6 ± 10.4 | 64.6 ± 9.2 | 72.2 ± 8.7 | 0.115 |
| CRP (mg/L) | 118.8 | 74.5 | 85.6 | 121.0 | 125.6 | 0.340 |
| Leukocyte count (109/L) | 7.49 ± 3.66 | 8.53 ± 3.54 | 8.22 ± 4.39 | 9.84 ± 5.97 | 8.04 ± 3.78 | 0.614 |
| Neutrophils (%) | 74.7 ± 13.7 | 79.4 ± 8.5 | 76.5 ± 10.1 | 82.6 ± 8.5 | 78.7 ± 8.4 | 0.193 |
| Lymphocytes (%) | 17.4 ± 12.0 | 14.1 ± 6.0 | 17.7 ± 9.9 | 12.1 ± 7.8 | 15.0 ± 9.6 | 0.201 |
| D-dimer (µg/L) | 0.76 | 0.92 | 0.85 | 0.98 | 1.89 | 0.390 |
| LDH (U/L) | 302 | 342 | 405 | 365 | 439 | 0.166 |
| hs-Troponin (ng/L) | 21.7 | 10.3 | 10.2 | 11.0 | 11.7 | 0.623 |
| spO2 (%) | 96.0 | 91.0 | 87.5 | 89.0 | 90.0 | 0.0003 ‡ |
| pO2 (kPa) | 10.00 | 7.18 | 6.92 | 6.89 | 5.89 | 0.069 |
| Median (IQR) | Difference (95% CI) | p * | ||
|---|---|---|---|---|
| Survived (n = 115) | Died (n = 16) | |||
| Day of the illness | 10.0 (8.0–13.0) | 9.5 (6.0–12.0) | −1.0 (−4.0–1.0) | 0.306 |
| Presenting † respiratory support modality | 1.0 (0.3–1.0) | 1.0 (1.0–1.0) | 0.0 (0.0–0.0) | 0.375 |
| Highest ‡ respiratory support modality | 1.0 (1.0–2.0) | 4.0 (3.0–4.0) | 2.0 (2.0–3.0) | <0.001 |
| LUS score | 24.0 (19.5–29.0) | 35.5 (32.0–38.0) | 11.0 (7.0–14.0) | <0.001 |
| CRP (mg/L) | 82 (48–150) | 117 (74–147) | −17.2 (−51.5–21.6) | 0.372 |
| Leukocyte count (109/L) | 7.6 (5.9–10.6) | 7.7 (4.8–12.5) | 0.0 (−2.5–2.2) | 0.997 |
| Neutrophils (%) | 80.1 (74.6–85.3) | 82.2 (74.2–89.0) | 2.0 (−2.3–7.3) | 0.276 |
| Lymphocytes (%) | 13.7 (9.0–18.4) | 10.5 (6.3–18.5) | −2.9 (−6.9–0.9) | 0.149 |
| D-dimer (µg/L) | 0.87 (0.60–1.51) | 2.10 (1.53–3.37) | 1.12 (0.55–1.80) | 0.002 |
| LDH (U/L) | 361 (287–424) | 439 (284–463) | 40 (−33–118) | 0.330 |
| hs-Troponin (ng/L) | 10.2 (7.2–18.4) | 14.8 (9.8–50.9) | 4.5 (−1.9–36.2) | 0.171 |
| spO2 (%) | 91.0 (87.0–93.3) | 89.5 (81.0–93.0) | −3.0 (−6.0–0.0) | 0.093 |
| pO2 (kPa) | 7.14 (6.50–7.90) | 6.64 (5.56–8.09) | −0.42 (−1.38–0.57) | 0.419 |
| ß | Wald | p | OR | 95% CI | |
|---|---|---|---|---|---|
| Age | 0.081 | 5.051 | 0.025 | 1.08 | 1.01–1.16 |
| LUS score | 0.215 | 12.617 | <0.001 | 1.24 | 1.10–1.40 |
| Constant | −14.044 | 17.192 | <0.001 |
| ß | Wald | p | OR | 95% CI | |
|---|---|---|---|---|---|
| Age | 0.232 | 13.510 | <0.001 | 1.26 | 1.14–1.43 |
| LUS score | 0.344 | 14.589 | <0.001 | 1.41 | 1.18–1.68 |
| Constant | −28.864 | 18.882 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skopljanac, I.; Ivelja, M.P.; Barcot, O.; Brdar, I.; Dolic, K.; Polasek, O.; Radic, M. Role of Lung Ultrasound in Predicting Clinical Severity and Fatality in COVID-19 Pneumonia. J. Pers. Med. 2021, 11, 757. https://doi.org/10.3390/jpm11080757
Skopljanac I, Ivelja MP, Barcot O, Brdar I, Dolic K, Polasek O, Radic M. Role of Lung Ultrasound in Predicting Clinical Severity and Fatality in COVID-19 Pneumonia. Journal of Personalized Medicine. 2021; 11(8):757. https://doi.org/10.3390/jpm11080757
Chicago/Turabian StyleSkopljanac, Ivan, Mirela Pavicic Ivelja, Ognjen Barcot, Ivan Brdar, Kresimir Dolic, Ozren Polasek, and Mislav Radic. 2021. "Role of Lung Ultrasound in Predicting Clinical Severity and Fatality in COVID-19 Pneumonia" Journal of Personalized Medicine 11, no. 8: 757. https://doi.org/10.3390/jpm11080757
APA StyleSkopljanac, I., Ivelja, M. P., Barcot, O., Brdar, I., Dolic, K., Polasek, O., & Radic, M. (2021). Role of Lung Ultrasound in Predicting Clinical Severity and Fatality in COVID-19 Pneumonia. Journal of Personalized Medicine, 11(8), 757. https://doi.org/10.3390/jpm11080757