Ovarian Reserve after Chemotherapy in Breast Cancer: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Question
- Population: Fertile women who have been diagnosed with breast cancer and have gone through chemotherapy treatment
- Intervention: Chemotherapy treatment
- Comparison: Not Applicable
- Outcome: Ovarian reserve measured by AMH
- Setting/Time: All
2.2. Literature Search
2.3. Study Selection and Inclusion/Exclusion Criteria
2.4. Data Extraction
2.5. Data Synthesis and Statistical Analyses
2.6. Quality Assessment
3. Results
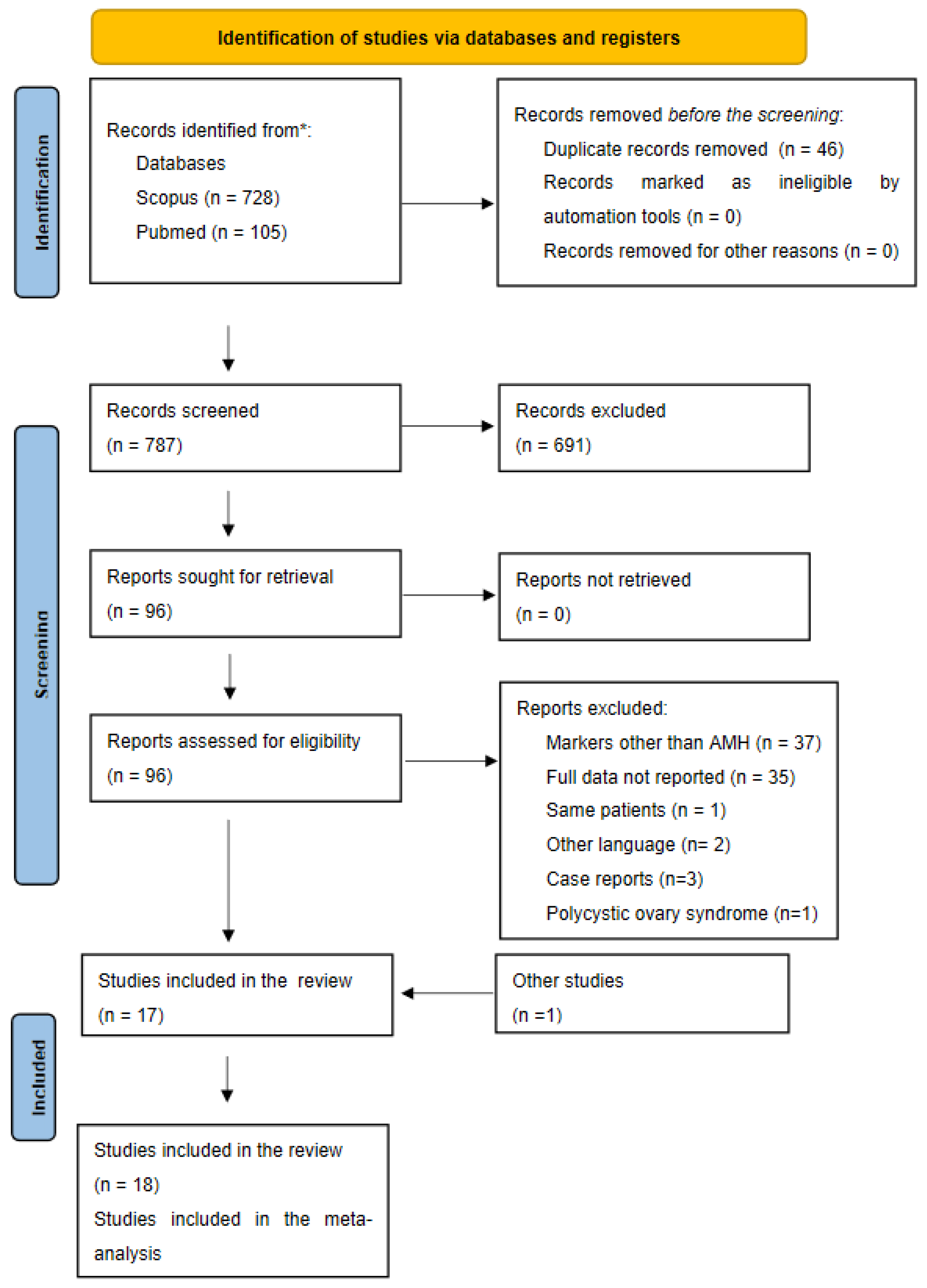
3.1. Bibliographical Search
3.2. Description of the Included Studies
3.3. Quality Assessment
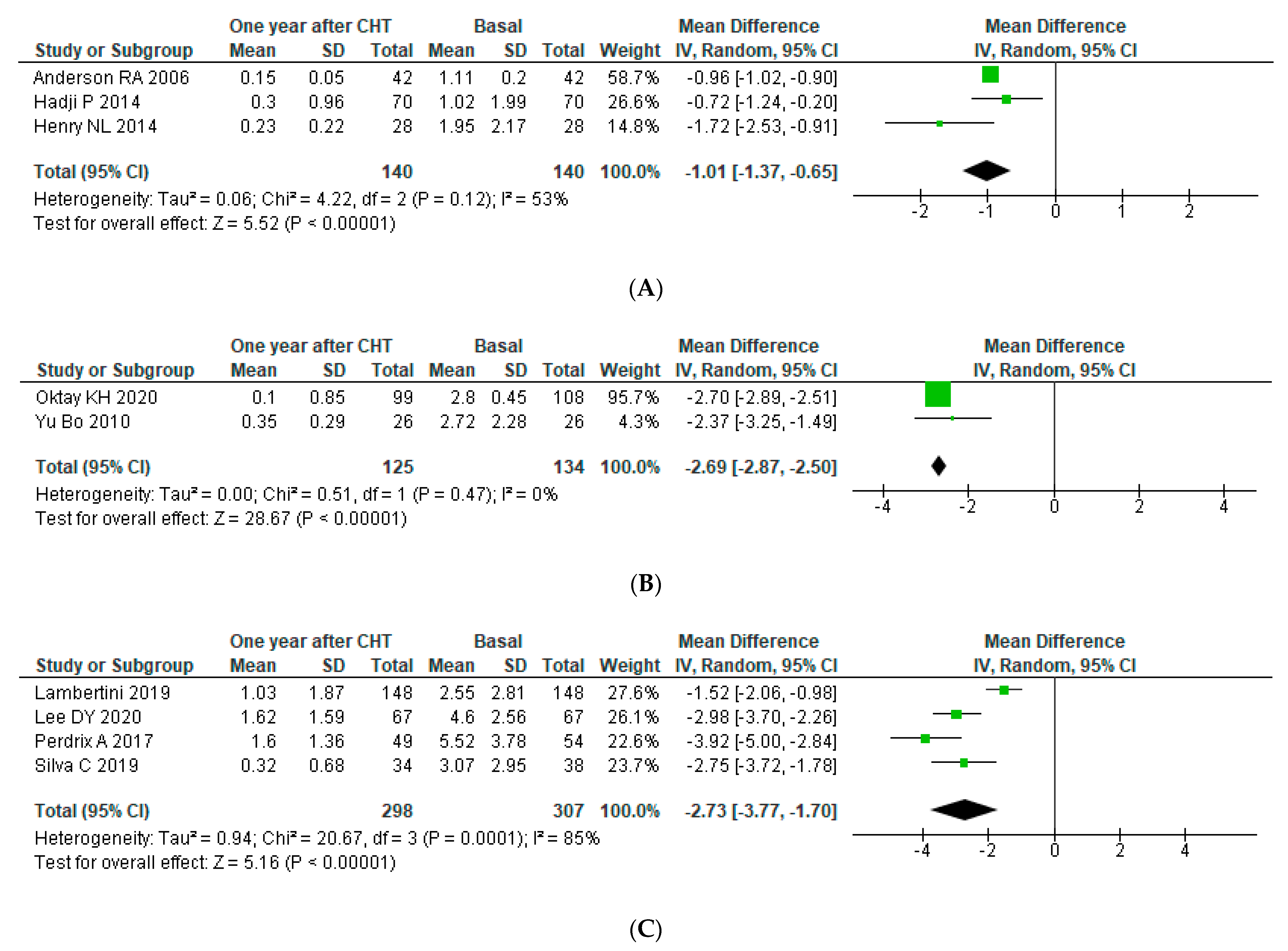
3.4. Meta-Analysis
3.4.1. Ovarian Reserve after Chemotherapy
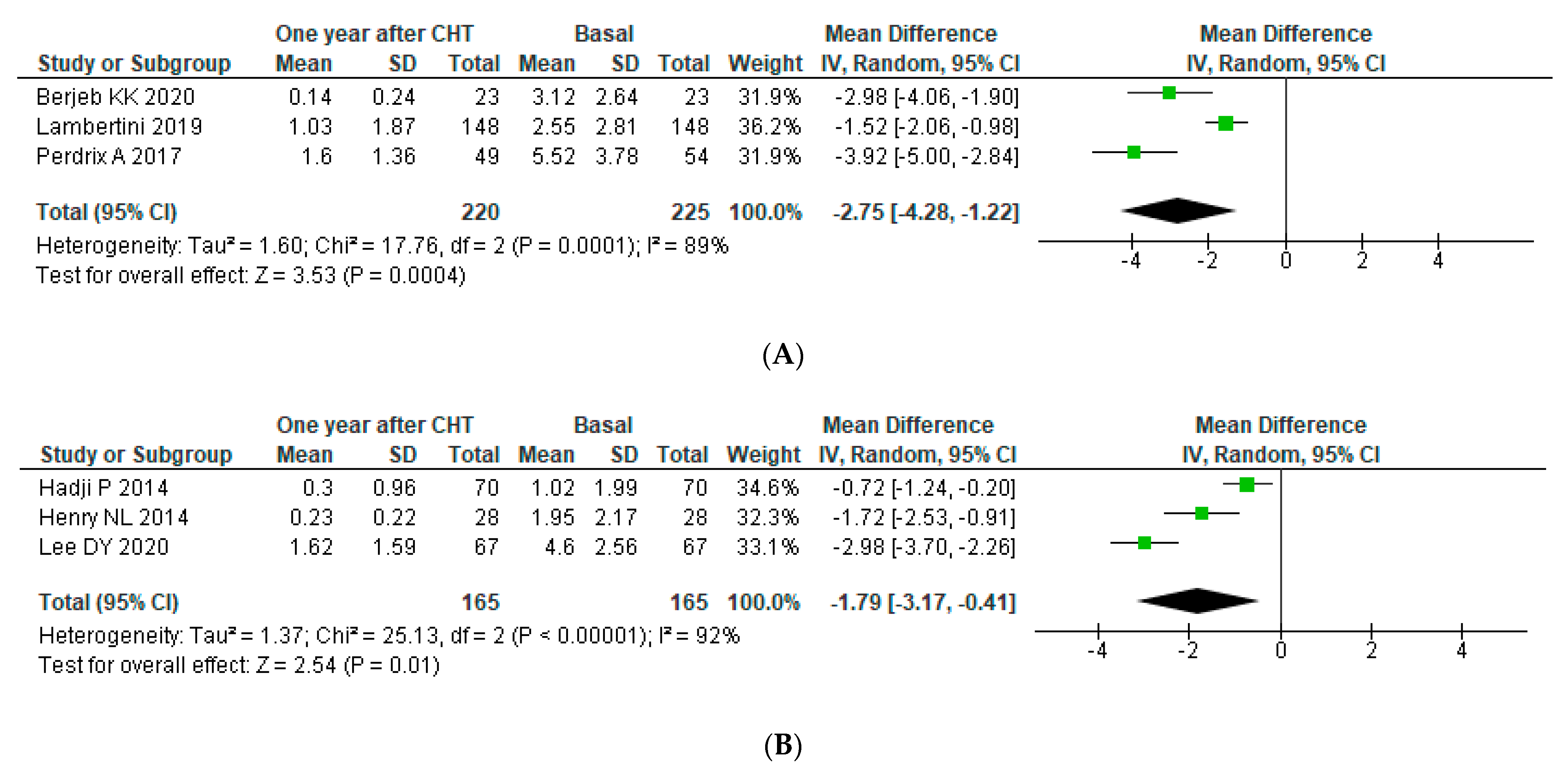
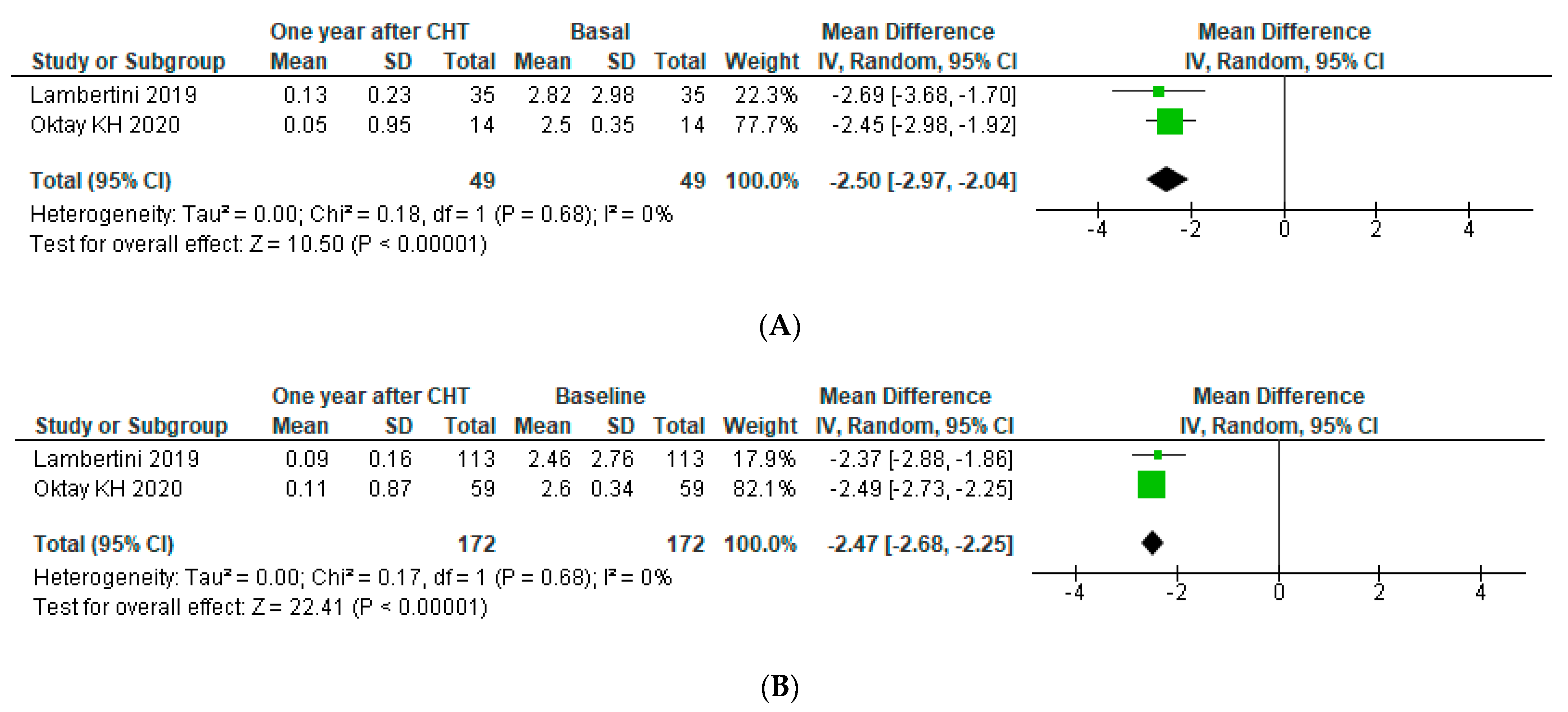
3.4.2. Subgroup Analysis
Subgroup Analysis: Age
Subgroup Analysis: AMH Assay
Subgroup Analysis: BRCA Subgroup Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Bardia, A.; Hurvitz, S. Targeted Therapy for Premenopausal Women with HR+, HER2− Advanced Breast Cancer: Focus on Special Considerations and Latest Advances. Clin. Cancer Res. 2018, 24, 5206–5218. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1194–1220. [Google Scholar] [CrossRef]
- Zavos, A.; Valachis, A. Risk of chemotherapy-induced amenorrhea in patients with breast cancer: A systematic review and meta-analysis. Acta Oncol. 2016, 55, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.R.; Hodnett, G.M.; Knowlton, N.; Craig, L.B. Correlation of ovarian reserve tests with histologically determined primordial follicle number. Fertil. Steril. 2011, 95, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Alipour, F.; Rasekhjahromi, A.; Maalhagh, M.; Sobhanian, S.; Hosseinpoor, M. Comparison of Specificity and Sensitivity of AMH and FSH in Diagnosis of Premature Ovarian Failure. Dis. Markers 2015, 2015, 1–4. [Google Scholar] [CrossRef]
- Broekmans, F.J.; Kwee, J.; Hendriks, D.J.; Mol, B.W.; Lambalk, C.B. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum. Reprod. Updat. 2006, 12, 685–718. [Google Scholar] [CrossRef]
- Eslami, B.; Jalaeefar, A.; Moini, A.; Omranipour, R.; Haghighi, M.; Alipour, S. Significant Post-Chemotherapy Decrease of Ovarian Reserve in Iranian Women With Breast Cancer. Acta Med. Iran. 2020, 58, 400–403. [Google Scholar] [CrossRef]
- Al-Rawi, S.A.; Saleh, B.O.; Al-Naqqash, M. Serum anti-müllerian hormone levels in evaluation of chemotherapy effect on ovarian reserve in women with breast cancer. Saudi Med. J. 2018, 39, 733–735. [Google Scholar] [CrossRef]
- Anderson, R.A.; Themmen, A.P.N.; Al-Qahtani, A.; Groome, N.P.; Cameron, D.A. The effects of chemotherapy and long-term gonadotrophin suppression on the ovarian reserve in premenopausal women with breast cancer. Hum. Reprod. 2006, 21, 2583–2592. [Google Scholar] [CrossRef]
- Anderson, R.A.; Cameron, D.A. Pretreatment Serum Anti-Müllerian Hormone Predicts Long-Term Ovarian Function and Bone Mass after Chemotherapy for Early Breast Cancer. J. Clin. Endocrinol. Metab. 2011, 96, 1336–1343. [Google Scholar] [CrossRef]
- Bala, J.; Seth, S.; Dhankhar, R.; Ghalaut, V.S. Chemotherapy: Impact on Anti-Müllerian Hormone Levels in Breast Carcinoma. J. Clin. Diagn. Res. 2016, 10, BC19–BC21. [Google Scholar] [CrossRef]
- Berjeb, K.K.; Debbabi, L.; Braham, M.; Zemni, Z.; Chtourou, S.; Hannachi, H.; Hamdoun, M.; Ayadi, M.; Kacem, K.; Zhioua, F.; et al. Evaluation of ovarian reserve before and after chemotherapy. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 102035. [Google Scholar] [CrossRef]
- D’Avila, Â.M.; Capp, E.; Corleta, H.V.E. Antral Follicles Count and Anti-Müllerian Hormone Levels after Gonadotoxic Chemotherapy in Patients with Breast Cancer: Cohort Study. Rev. Bras. Ginecol. Obs. RBGO Gynecol. Obstet. 2017, 39, 162–168. [Google Scholar] [CrossRef]
- Dezellus, A.; Barriere, P.; Campone, M.; Lemanski, C.; Vanlemmens, L.; Mignot, L.; Delozier, T.; Levy, C.; Bendavid, C.; Debled, M.; et al. Prospective evaluation of serum anti-Müllerian hormone dynamics in 250 women of reproductive age treated with chemotherapy for breast cancer. Eur. J. Cancer 2017, 79, 72–80. [Google Scholar] [CrossRef]
- Hadji, P.; Kauka, A.; Ziller, M.; Birkholz, K.; Baier, M.; Muth, M.; Kann, P. Effect of adjuvant endocrine therapy on hormonal levels in premenopausal women with breast cancer: The ProBONE II study. Breast Cancer Res. Treat. 2014, 144, 343–351. [Google Scholar] [CrossRef]
- Henry, N.L.; Xia, R.; Schott, A.F.; McConnell, D.; Banerjee, M.; Hayes, D.F. Prediction of Postchemotherapy Ovarian Function Using Markers of Ovarian Reserve. Oncology 2013, 19, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Lambertini, M.; Olympios, N.; LeQuesne, J.; Calbrix, C.; Fontanilles, M.; Loeb, A.; Leheurteur, M.; Demeestere, I.; Di Fiore, F.; Perdrix, A.; et al. Impact of Taxanes, Endocrine Therapy, and Deleterious Germline BRCA Mutations on Anti-müllerian Hormone Levels in Early Breast Cancer Patients Treated With Anthracycline- and Cyclophosphamide-Based Chemotherapy. Front. Oncol. 2019, 9, 575. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-Y.; Kim, J.-Y.; Yu, J.; Kim, S.W. Prediction of Successful Ovarian Protection Using Gonadotropin-Releasing Hormone Agonists During Chemotherapy in Young Estrogen Receptor-Negative Breast Cancer Patients. Front. Oncol. 2020, 10, 863. [Google Scholar] [CrossRef] [PubMed]
- Oktay, K.H.; Bedoschi, G.; Goldfarb, S.B.; Taylan, E.; Titus, S.; Palomaki, G.E.; Cigler, T.; Robson, M.; Dickler, M.N. Increased chemotherapy-induced ovarian reserve loss in women with germline BRCA mutations due to oocyte deoxyribonucleic acid double strand break repair deficiency. Fertil. Steril. 2020, 113, 1251–1260.e1. [Google Scholar] [CrossRef] [PubMed]
- Passildas, J.; Collard, O.; Savoye, A.-M.; Dohou, J.; Ginzac, A.; Thivat, E.; Durando, X.; Kwiatkowski, F.; Penault-Llorca, F.; Abrial, C.; et al. Impact of Chemotherapy-induced Menopause in Women of Childbearing Age With Non-metastatic Breast Cancer—Preliminary Results From the MENOCOR Study. Clin. Breast Cancer 2019, 19, e74–e84. [Google Scholar] [CrossRef]
- Perdrix, A.; Saint-Ghislain, M.; Degremont, M.; David, M.; Khaznadar, Z.; Loeb, A.; Leheurteur, M.; Di Fiore, F.; Clatot, F. Influence of adjuvant chemotherapy on anti-Müllerian hormone in women below 35 years treated for early breast cancer. Reprod. Biomed. Online 2017, 35, 468–474. [Google Scholar] [CrossRef]
- Silva, C.; Rama, A.C.R.; Soares, S.R.; Moura-Ramos, M.; Santos, T.A. Adverse reproductive health outcomes in a cohort of young women with breast cancer exposed to systemic treatments. J. Ovarian Res. 2019, 12, 1–10. [Google Scholar] [CrossRef]
- Trapp, E.; Steidl, J.; Rack, B.; Kupka, M.; Andergassen, U.; Jückstock, J.; Kurt, A.; Vilsmaier, T.; de Gregorio, A.; Tzschaschel, M.; et al. Anti-Müllerian hormone (AMH) levels in premenopausal breast cancer patients treated with taxane-based adjuvant chemotherapy—A translational research project of the SUCCESS A study. Breast 2017, 35, 130–135. [Google Scholar] [CrossRef]
- Yu, B.; Douglas, N.; Ferin, M.J.; Nakhuda, G.S.; Crew, K.; Lobo, R.A.; Hershman, D.L. Changes in markers of ovarian reserve and endocrine function in young women with breast cancer undergoing adjuvant chemotherapy. Cancer 2010, 116, 2099–2105. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 1–13. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating Characteristics of a Rank Correlation Test for Publication Bias. Biometrics 1994, 50, 1088–1099. [Google Scholar] [CrossRef]
- Galbraith, R.F. A note on graphical presentation of estimated odds ratios from several clinical trials. Stat. Med. 1988, 7, 889–894. [Google Scholar] [CrossRef]
- Anderson, R.; Mansi, J.; Coleman, R.; Adamson, D.; Leonard, R. The utility of anti-Müllerian hormone in the diagnosis and prediction of loss of ovarian function following chemotherapy for early breast cancer. Eur. J. Cancer 2017, 87, 58–64. [Google Scholar] [CrossRef]
- Wakimoto, Y.; Fukui, A.; Wakimoto, G.; Ikezawa, Y.; Matsuoka, M.; Omote, M.; Sugiyama, Y.; Ukita, Y.; Kato, T.; Shibahara, H. Association between spontaneous ovulation and serum anti-Müllerian hormone levels in a premature ovarian insufficiency patient after a multimodal treatment for breast cancer. J. Obstet. Gynaecol. Res. 2019, 45, 2297–2301. [Google Scholar] [CrossRef] [PubMed]
- Mahany, E.B.; Hershman, D.L.; Sauer, M.V.; Choi, J.M. Pregnancy despite ovarian insufficiency in a patient with breast cancer. Reprod. Med. Biol. 2013, 12, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Ohta, H.; Namba, N.; Tachibana, M.; Miyamura, T.; Miyashita, E.; Hashii, Y.; Oue, T.; Isobe, A.; Tsutsui, T.; et al. Low Serum Concentrations of Anti-Müllerian Hormone Are Common in 53 Female Childhood Cancer Survivors. Horm. Res. Paediatr. 2013, 79, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Andergassen, U.; Kasprowicz, N.S.; Hepp, P.; Schindlbeck, C.; Harbeck, N.; Kiechle, M.; Sommer, H.; Beckmann, M.W.; Friese, K.; Janni, W.; et al. Participation in the SUCCESS-A Trial Improves Intensity and Quality of Care for Patients with Primary Breast Cancer. Geburtshilfe Frauenheilkd. 2013, 73, 63–69. [Google Scholar] [CrossRef]
- Benedict, C.; Thom, B.; Friedman, D.N.; Pottenger, E.; Raghunathan, N.; Kelvin, J.F. Fertility information needs and concerns post-treatment contribute to lowered quality of life among young adult female cancer survivors. Support. Care Cancer 2018, 26, 2209–2215. [Google Scholar] [CrossRef] [PubMed]
- Gorman, J.R.; Su, H.I.; Mph, S.C.R.; Dominick, S.A.; Malcarne, V. Experiencing reproductive concerns as a female cancer survivor is associated with depression. Cancer 2015, 121, 935–942. [Google Scholar] [CrossRef]
- Oktay, K.; Harvey, B.; Partridge, A.H.; Quinn, G.; Reinecke, J.; Taylor, H.S.; Wallace, W.H.; Wang, E.T.; Loren, A.W. Fertility Preservation in Patients With Cancer: ASCO Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 1994–2001. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet 2005, 365, 1687–1717. [Google Scholar] [CrossRef]
- Smith, I.E.; Dowsett, M.; Yap, Y.-S.; Walsh, G.; Lonning, P.E.; Santen, R.J.; Hayes, D. Adjuvant Aromatase Inhibitors for Early Breast Cancer After Chemotherapy-Induced Amenorrhoea: Caution and Suggested Guidelines. J. Clin. Oncol. 2006, 24, 2444–2447. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.L.; Klitus, M.; Mintzer, D.M. Chemotherapy-Induced Amenorrhea from Adjuvant Breast Cancer Treatment: The Effect of the Addition of Taxanes. Clin. Breast Cancer 2005, 6, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Walshe, J.M.; Denduluri, N.; Swain, S.M. Amenorrhea in Premenopausal Women After Adjuvant Chemotherapy for Breast Cancer. J. Clin. Oncol. 2006, 24, 5769–5779. [Google Scholar] [CrossRef]
- Van Der Wijden, C.; Manion, C. Lactational amenorrhoea method for family planning. Cochrane Database Syst. Rev. 2015, 12, CD001329. [Google Scholar] [CrossRef]
- Polyzos, N.P.; Drakopoulos, P.; Parra, J.; Pellicer, A.; Santos-Ribeiro, S.; Tournaye, H.; Bosch, E.; Garcia-Velasco, J. Cumulative live birth rates according to the number of oocytes retrieved after the first ovarian stimulation for in vitro fertilization/intracytoplasmic sperm injection: A multicenter multinational analysis including ∼15,000 women. Fertil. Steril. 2018, 110, 661–670.e1. [Google Scholar] [CrossRef]
- Api, M. Is ovarian reserve diminished after laparoscopic ovarian drilling? Gynecol. Endocrinolol. 2009, 25, 159–165. [Google Scholar] [CrossRef]
- Decanter, C.; Morschhauser, F.; Pigny, P.; Lefebvre, C.; Gallo, C.; Dewailly, D. Anti-Müllerian hormone follow-up in young women treated by chemotherapy for lymphoma: Preliminary results. Reprod. Biomed. Online 2010, 20, 280–285. [Google Scholar] [CrossRef]
- Higgins, J.; Thompson, S.; Deeks, J.; Altman, D. Statistical heterogeneity in systematic reviews of clinical trials: A critical appraisal of guidelines and practice. J. Heal. Serv. Res. Policy 2002, 7, 51–61. [Google Scholar] [CrossRef]
- Su, H.I.; Sammel, M.D.; Homer, M.V.; Bui, K.; Haunschild, C.; Stanczyk, F.Z. Comparability of antimüllerian hormone levels among commercially available immunoassays. Fertil. Steril. 2014, 101, 1766–1772.e1. [Google Scholar] [CrossRef] [PubMed]
- Gassner, D.; Jung, R. First fully automated immunoassay for anti-Müllerian hormone. Clin. Chem. Lab. Med. 2014, 52, 1143–1152. [Google Scholar] [CrossRef]
- Venkitaraman, A.R. Cancer Susceptibility and the Functions of BRCA1 and BRCA2. Cell 2002, 108, 171–182. [Google Scholar] [CrossRef]
- Corrado, G.; Marchetti, C.; Trozzi, R.; Scambia, G.; Fagotti, A. Fertility preservation in patients with BRCA mutations or Lynch syndrome. Int. J. Gynecol. Cancer 2021, 31, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Francis, P.; Regan, M.M.; Fleming, G.F.; Láng, I.; Ciruelos, E.; Bellet, M.; Bonnefoi, H.R.; Climent, M.A.; Da Prada, G.A.; Burstein, H.J.; et al. Adjuvant Ovarian Suppression in Premenopausal Breast Cancer. N. Engl. J. Med. 2015, 372, 436–446. [Google Scholar] [CrossRef] [PubMed]
- Regan, M.M.; Pagani, O.; Fleming, G.F.; Walley, B.A.; Price, K.N.; Rabaglio, M.; Maibach, R.; Ruepp, B.; Coates, A.S.; Goldhirsch, A.; et al. Adjuvant treatment of premenopausal women with endocrine-responsive early breast cancer: Design of the TEXT and SOFT trials. Breast 2013, 22, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- Kennecke, H.; Yerushalmi, R.; Woods, R.; Cheang, M.C.U.; Voduc, D.; Speers, C.H.; Nielsen, T.O.; Gelmon, K. Metastatic Behavior of Breast Cancer Subtypes. J. Clin. Oncol. 2010, 28, 3271–3277. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romito, A.; Bove, S.; Romito, I.; Zace, D.; Raimondo, I.; Fragomeni, S.M.; Rinaldi, P.M.; Pagliara, D.; Lai, A.; Marazzi, F.; et al. Ovarian Reserve after Chemotherapy in Breast Cancer: A Systematic Review and Meta-Analysis. J. Pers. Med. 2021, 11, 704. https://doi.org/10.3390/jpm11080704
Romito A, Bove S, Romito I, Zace D, Raimondo I, Fragomeni SM, Rinaldi PM, Pagliara D, Lai A, Marazzi F, et al. Ovarian Reserve after Chemotherapy in Breast Cancer: A Systematic Review and Meta-Analysis. Journal of Personalized Medicine. 2021; 11(8):704. https://doi.org/10.3390/jpm11080704
Chicago/Turabian StyleRomito, Alessia, Sonia Bove, Ilaria Romito, Drieda Zace, Ivano Raimondo, Simona Maria Fragomeni, Pierluigi Maria Rinaldi, Domenico Pagliara, Antonella Lai, Fabio Marazzi, and et al. 2021. "Ovarian Reserve after Chemotherapy in Breast Cancer: A Systematic Review and Meta-Analysis" Journal of Personalized Medicine 11, no. 8: 704. https://doi.org/10.3390/jpm11080704
APA StyleRomito, A., Bove, S., Romito, I., Zace, D., Raimondo, I., Fragomeni, S. M., Rinaldi, P. M., Pagliara, D., Lai, A., Marazzi, F., Marchetti, C., Paris, I., Franceschini, G., Masetti, R., Scambia, G., Fabi, A., & Garganese, G. (2021). Ovarian Reserve after Chemotherapy in Breast Cancer: A Systematic Review and Meta-Analysis. Journal of Personalized Medicine, 11(8), 704. https://doi.org/10.3390/jpm11080704












