Glycosylation of IgG Associates with Hypertension and Type 2 Diabetes Mellitus Comorbidity in the Chinese Muslim Ethnic Minorities and the Han Chinese
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Diagnostic Criteria
2.3. Analysis of IgG N-glycosylation
2.4. Statistical Analysis
3. Results
3.1. Demographic and Biochemical Characteristics
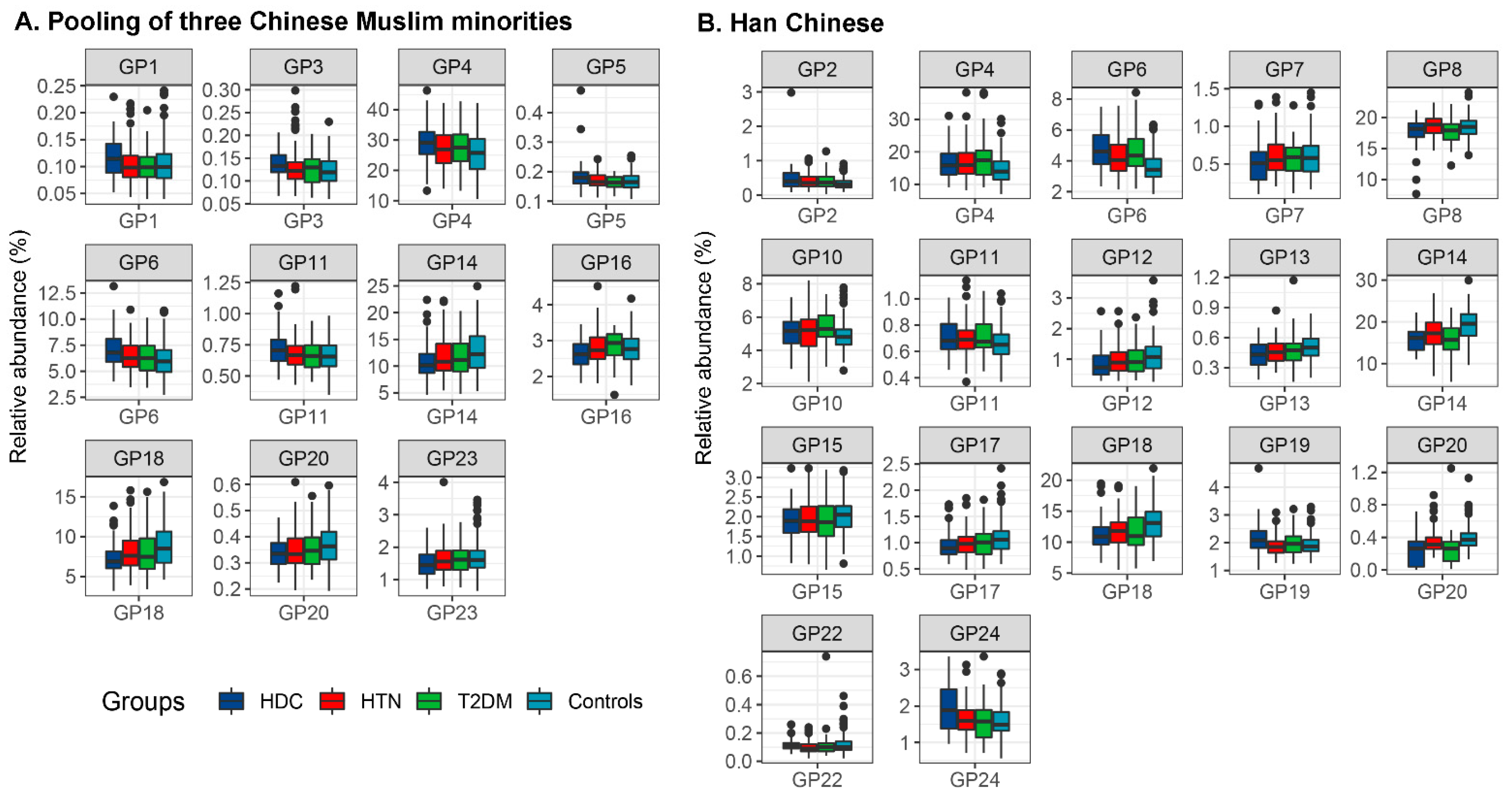
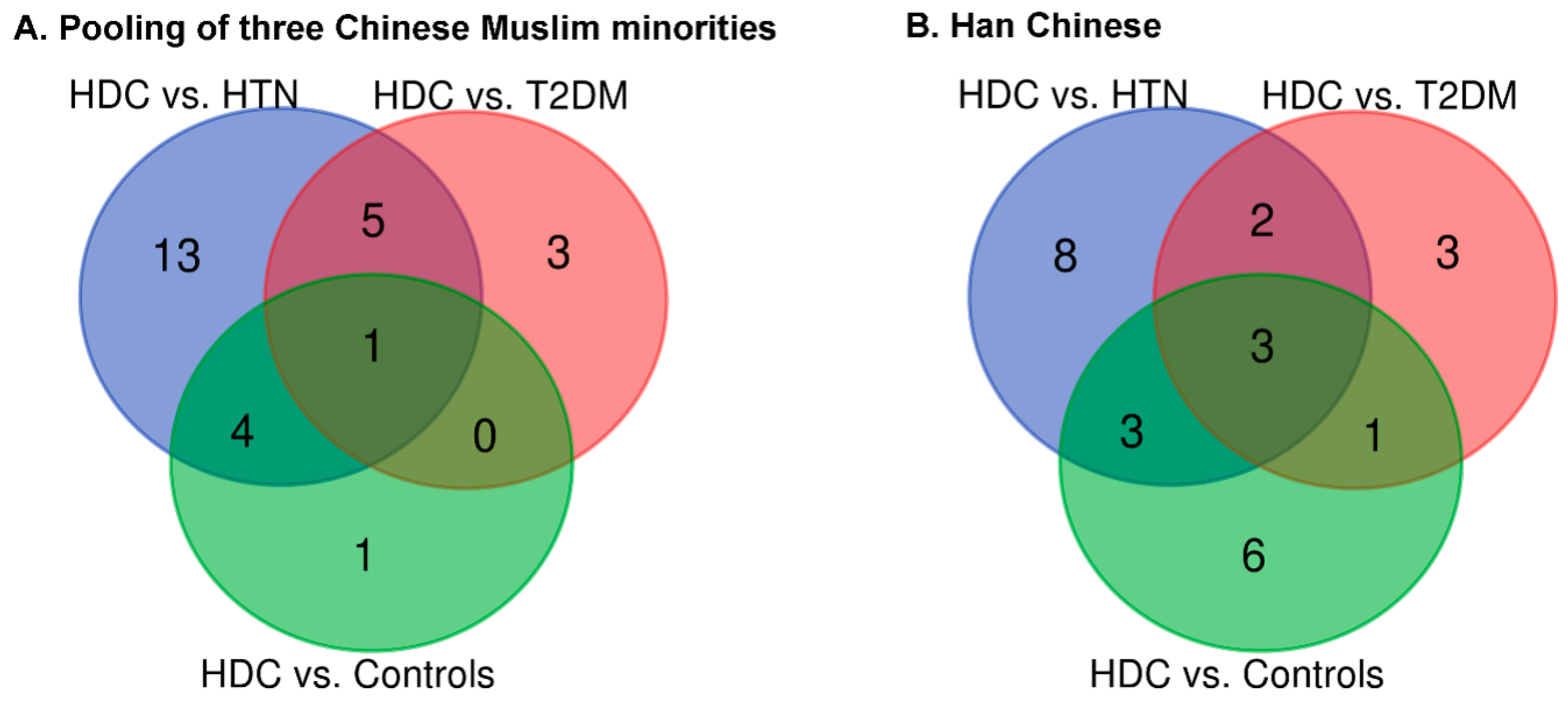
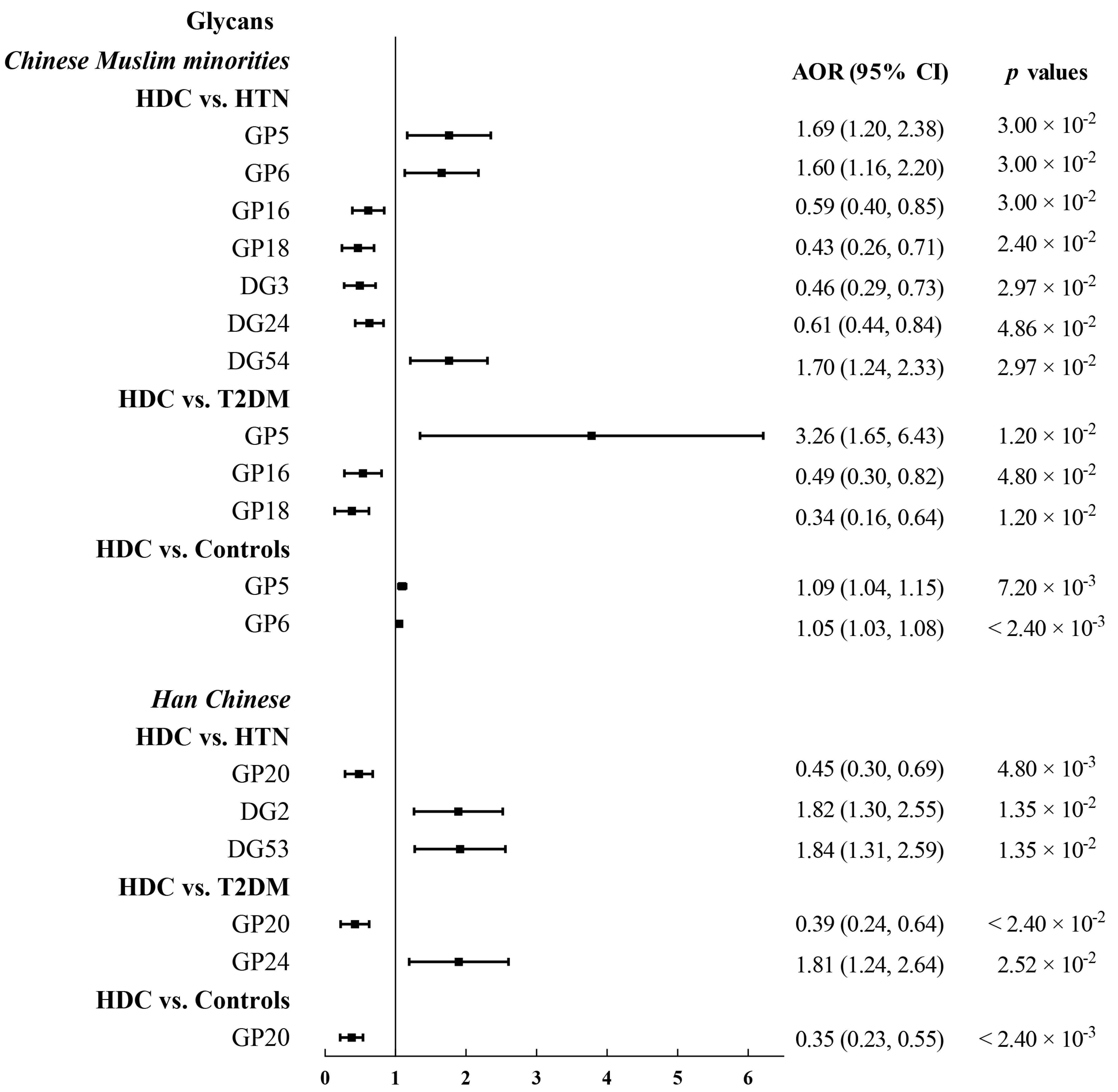
3.2. The Association of IgG N-Glycans with HDC
3.3. The Comparison of IgG N-Glycans between the Chinese Muslim Ethnic Minorities Pooled Samples and the Han Chinese Samples
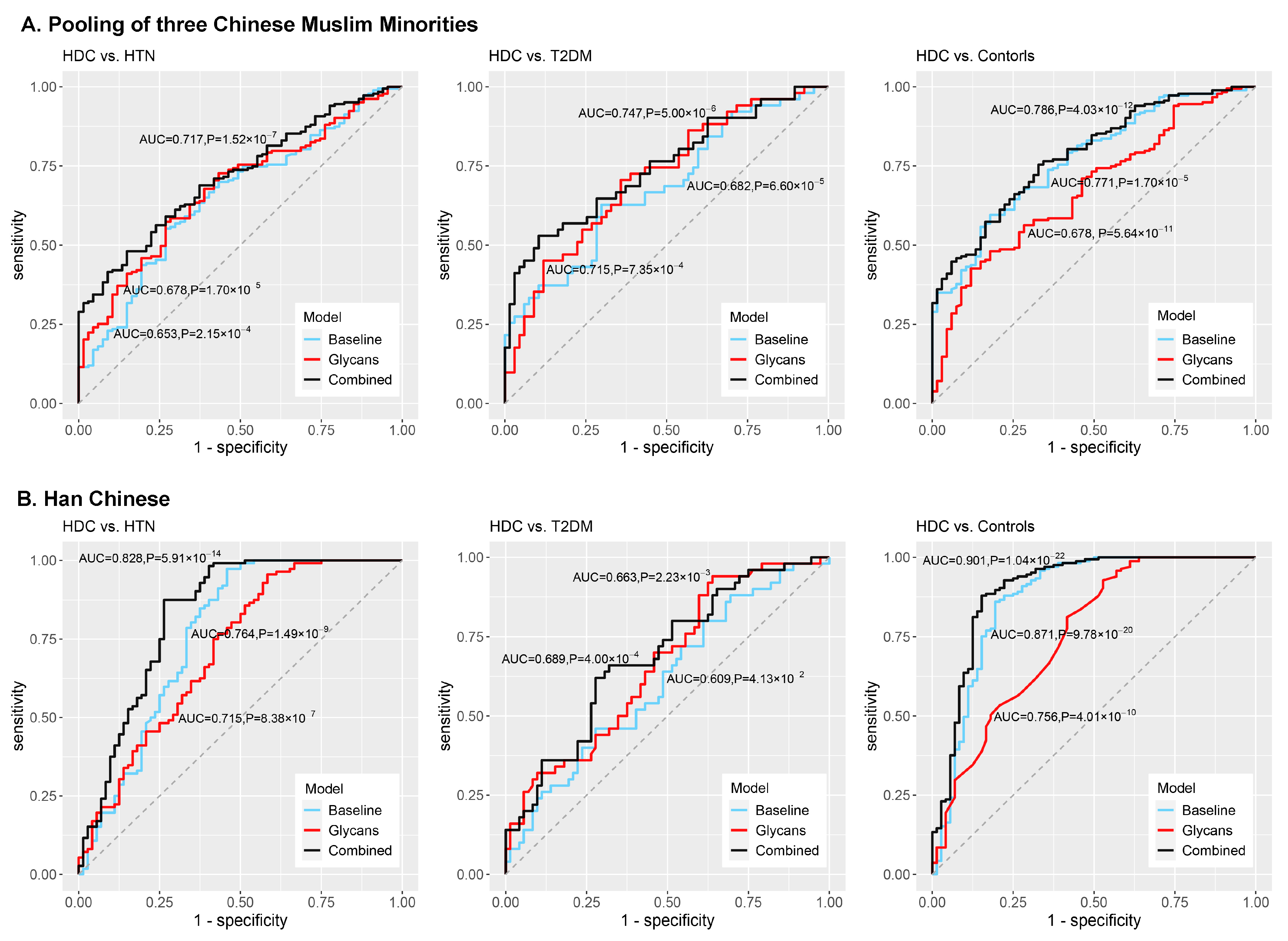
3.4. Development of the Classification Models and Discrimination of HDC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | area under the receiver operating characteristic curve |
| BMI | body mass index |
| CI | confidence interval |
| CVD | cardiovascular disease |
| DBP | diastolic blood pressure |
| DG | derived glycan |
| FBG | fasting blood glucose |
| GP | glycan peak |
| HDC | hypertension and type 2 diabetes mellitus comorbidity |
| HDL | high-density lipoprotein cholesterol |
| HILIC | hydrophilic interaction liquid chromatography |
| HTN | hypertension |
| IgG | immunoglobulin G |
| LDL | low-density lipoprotein cholesterol |
| OR | odds ratio |
| ROC | receiver operating characteristic |
| SBP | systolic blood pressure |
| T2DM | type 2 diabetes mellitus |
| TC | total cholesterol |
| TG | triglycerides |
| UPLC | ultra-performance liquid chromatography |
References
- Forouzanfar, M.H.; Alexander, L.; Anderson, H.R.; Bachman, V.F.; Biryukov, S.; Brauer, M.; Burnett, R.; Casey, D.; Coates, M.M.; Cohen, A.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 2287–2323. [Google Scholar] [CrossRef]
- Koye, D.N.; Magliano, D.J.; Nelson, R.G.; Pavkov, M.E. The Global Epidemiology of Diabetes and Kidney Disease. Adv. Chronic Kidney Dis. 2018, 25, 121–132. [Google Scholar] [CrossRef]
- Petrie, J.R.; Guzik, T.J.; Touyz, R.M. Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms. Can. J. Cardiol. 2018, 34, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Pavlou, D.I.; Paschou, S.A.; Anagnostis, P.; Spartalis, M.; Spartalis, E.; Vryonidou, A.; Tentolouris, N.; Siasos, G. Hypertension in patients with type 2 diabetes mellitus: Targets and management. Maturitas 2018, 112, 71–77. [Google Scholar] [CrossRef]
- Gillespie, C.D.; Hurvitz, K.A. Prevalence of hypertension and controlled hypertension–United States, 2007–2010. MMWR Suppl. 2013, 62, 144–148. [Google Scholar]
- Weir, M.R. Diabetes and hypertension: Blood pressure control and consequences. Am. J. Hypertens. 1999, 12, 170S–178S. [Google Scholar] [CrossRef]
- Hu, G.; Jousilahti, P.; Tuomilehto, J. Joint effects of history of hypertension at baseline and type 2 diabetes at baseline and during follow-up on the risk of coronary heart disease. Eur. Heart J. 2007, 28, 3059–3066. [Google Scholar] [CrossRef] [PubMed]
- Eaddy, M.T.; Shah, M.; Lunacsek, O.; Stanford, R.H. The burden of illness of hypertension and comorbid diabetes. Curr. Med. Res. Opin. 2008, 24, 2501–2507. [Google Scholar] [CrossRef]
- Kristic, J.; Lauc, G. Ubiquitous Importance of Protein Glycosylation. Methods Mol. Biol. 2017, 1503, 1–12. [Google Scholar] [PubMed]
- Gornik, O.; Wagner, J.; Pucic, M.; Knezevic, A.; Redzic, I.; Lauc, G. Stability of N-glycan profiles in human plasma. Glycobiology 2009, 19, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Zoldoš, V.; Horvat, T.; Lauc, G. Glycomics meets genomics, epigenomics and other high throughput omics for system biology studies. Curr. Opin. Chem. Biol. 2013, 17, 34–40. [Google Scholar] [CrossRef]
- Arnold, J.N.; Wormald, M.R.; Sim, R.B.; Rudd, P.M.; Dwek, R.A. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu. Rev. Immunol. 2007, 25, 21–50. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Y.; Kristic, J.; Dong, J.; Chu, X.; Ge, S.; Wang, H.; Fang, H.; Gao, Q.; Liu, D.; et al. Profiling IgG N-glycans as potential biomarker of chronological and biological ages: A community-based study in a Han Chinese population. Medicine 2016, 95, e4112. [Google Scholar] [CrossRef]
- Liu, J.N.; Dolikun, M.; Stambuk, J.; Trbojevic-Akmacic, I.; Zhang, J.; Wang, H.; Zheng, D.Q.; Zhang, X.Y.; Peng, H.L.; Zhao, Z.Y.; et al. The association between subclass-specific IgG Fc N-glycosylation profiles and hypertension in the Uygur, Kazak, Kirgiz, and Tajik populations. J. Hum. Hypertens. 2018, 32, 555–563. [Google Scholar] [CrossRef]
- Lemmers, R.F.H.; Vilaj, M.; Urda, D.; Agakov, F.; Šimurina, M.; Klaric, L.; Rudan, I.; Campbell, H.; Hayward, C.; Wilson, J.F.; et al. IgG glycan patterns are associated with type 2 diabetes in independent European populations. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2240–2249. [Google Scholar] [CrossRef] [PubMed]
- Reily, C.; Stewart, T.J.; Renfrow, M.B.; Novak, J. Glycosylation in health and disease. Nat. Rev. Nephrol. 2019, 15, 346–366. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Wang, Y.; Bili, K.; Guli, N.; Tian, Z.; Gan, X.; Tuerxun, G.; Liu, L.; Liu, C.; Bai, W.; et al. Population by Nationality in Main Years. In Xinjiang Statistic Yearbook-2019; Gao, W., Han, R., Eds.; China Statistics Press: Beijing, China, 2019; Volume 3, pp. 90–91. [Google Scholar]
- Yao, H.B.; Wang, C.C.; Tao, X.; Shang, L.; Wen, S.Q.; Zhu, B.; Kang, L.; Jin, L.; Li, H. Genetic evidence for an East Asian origin of Chinese Muslim populations Dongxiang and Hui. Sci. Rep. 2016, 6, 38656. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wise, C.; Baric, T.; Black, M.L.; Bittles, A.H. The origins and genetic structure of three co-resident Chinese Muslim populations: The Salar, Bo’an and Dongxiang. Hum. Genet. 2003, 113, 244–252. [Google Scholar] [CrossRef]
- Wang, L.; Li, N.; Heizhati, M.; Yao, X.; Duiyimuhan, G.; Zhou, K.; Cao, M.; Wang, M.; Hu, J.; Zhang, D. Prevalence, Awareness, Treatment, and Control and Related Factors of Hypertension in Multiethnic Agriculture, Stock-Raising, and Urban Xinjiang, Northwest China: A Cross-Sectional Screening for 47,000 Adults. Int. J. Hypertens. 2019, 2019, 3576853. [Google Scholar] [CrossRef]
- Wang, L.; Tao, Y.; Xie, Z.; Ran, X.; Zhang, M.; Wang, Y.; Luo, X.; Hu, M.; Gen, W.; Wufuer, H.; et al. Prevalence of metabolic syndrome, insulin resistance, impaired fasting blood glucose, and dyslipidemia in Uygur and Kazak populations. J. Clin. Hypertens. 2010, 12, 741–745. [Google Scholar] [CrossRef]
- Lu, J.; Lu, Y.; Wang, X.; Li, X.; Linderman, G.C.; Wu, C.; Cheng, X.; Mu, L.; Zhang, H.; Liu, J.; et al. Prevalence, awareness, treatment, and control of hypertension in China: Data from 1·7 million adults in a population-based screening study (China PEACE Million Persons Project). Lancet 2017, 390, 2549–2558. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Okosun, I.S.; Chandra, K.M.; Choi, S.; Christman, J.; Dever, G.E.; Prewitt, T.E. Hypertension and type 2 diabetes comorbidity in adults in the United States: Risk of overall and regional adiposity. Obes. Res. 2001, 9, 1–9. [Google Scholar] [CrossRef]
- Zhu, J.; Gao, R.; Zhao, S.; Lu, G.; Zhao, D.; Li, J. Guidelines for the prevention and treatment of dyslipidemia in Chinese adults (Revised Edition 2016). J. Chin. Circ. 2016, 31, 937–953. [Google Scholar]
- Trbojević-Akmačić, I.; Ugrina, I.; Lauc, G. Comparative Analysis and Validation of Different Steps in Glycomics Studies. Methods Enzymol. 2017, 586, 37–55. [Google Scholar] [PubMed]
- Pucic-Bakovic, M. High-Throughput Analysis of the IgG N-Glycome by UPLC-FLR. Methods Mol. Biol. 2017, 1503, 21–29. [Google Scholar]
- Pucić, M.; Knezević, A.; Vidic, J.; Adamczyk, B.; Novokmet, M.; Polasek, O.; Gornik, O.; Supraha-Goreta, S.; Wormald, M.R.; Redzić, I.; et al. High throughput isolation and glycosylation analysis of IgG-variability and heritability of the IgG glycome in three isolated human populations. Mol. Cell. Proteom. 2011, 10, M111.010090. [Google Scholar] [CrossRef] [PubMed]
- Vučković, F.; Krištić, J.; Gudelj, I.; Teruel, M.; Keser, T.; Pezer, M.; Pučić-Baković, M.; Štambuk, J.; Trbojević-Akmačić, I.; Barrios, C.; et al. Association of systemic lupus erythematosus with decreased immunosuppressive potential of the IgG glycome. Arthritis Rheumatol. 2015, 67, 2978–2989. [Google Scholar] [CrossRef]
- Gao, Q.; Dolikun, M.; Stambuk, J.; Wang, H.; Zhao, F.; Yiliham, N.; Wang, Y.; Trbojevic-Akmacic, I.; Zhang, J.; Fang, H.; et al. Immunoglobulin G N-Glycans as Potential Postgenomic Biomarkers for Hypertension in the Kazakh Population. Omics 2017, 21, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.P.; Knezevic, A.; Wang, Y.X.; Rudan, I.; Campbell, H.; Zou, Z.K.; Lan, J.; Lai, Q.X.; Wu, J.J.; He, Y.; et al. Screening novel biomarkers for metabolic syndrome by profiling human plasma N-glycans in Chinese Han and Croatian populations. J. Proteome Res. 2011, 10, 4959–4969. [Google Scholar] [CrossRef]
- Liu, J.; Dolikun, M.; Štambuk, J.; Trbojević-Akmačić, I.; Zhang, J.; Zhang, J.; Wang, H.; Meng, X.; Razdorov, G.; Menon, D.; et al. Glycomics for Type 2 Diabetes Biomarker Discovery: Promise of Immunoglobulin G Subclass-Specific Fragment Crystallizable N-glycosylation in the Uyghur Population. Omics 2019, 23, 640–648. [Google Scholar] [CrossRef]
- Glickman, M.E.; Rao, S.R.; Schultz, M.R. False discovery rate control is a recommended alternative to Bonferroni-type adjustments in health studies. J. Clin. Epidemiol. 2014, 67, 850–857. [Google Scholar] [CrossRef]
- Lastra, G.; Syed, S.; Kurukulasuriya, L.R.; Manrique, C.; Sowers, J.R. Type 2 diabetes mellitus and hypertension: An update. Endocrinol. Metab. Clin. N. Am. 2014, 43, 103–122. [Google Scholar] [CrossRef]
- Maverakis, E.; Kim, K.; Shimoda, M.; Gershwin, M.E.; Patel, F.; Wilken, R.; Raychaudhuri, S.; Ruhaak, L.R.; Lebrilla, C.B. Glycans in the immune system and The Altered Glycan Theory of Autoimmunity: A critical review. J. Autoimmun. 2015, 57, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Raju, T.S. Terminal sugars of Fc glycans influence antibody effector functions of IgGs. Curr. Opin. Immunol. 2008, 20, 471–478. [Google Scholar] [CrossRef]
- Shinkawa, T.; Nakamura, K.; Yamane, N.; Shoji-Hosaka, E.; Kanda, Y.; Sakurada, M.; Uchida, K.; Anazawa, H.; Satoh, M.; Yamasaki, M.; et al. The absence of fucose but not the presence of galactose or bisecting N-acetylglucosamine of human IgG1 complex-type oligosaccharides shows the critical role of enhancing antibody-dependent cellular cytotoxicity. J. Biol. Chem. 2003, 278, 3466–3473. [Google Scholar] [CrossRef]
- Liu, D.; Chu, X.; Wang, H.; Dong, J.; Ge, S.Q.; Zhao, Z.Y.; Peng, H.L.; Sun, M.; Wu, L.J.; Song, M.S.; et al. The changes of immunoglobulin G N-glycosylation in blood lipids and dyslipidaemia. J. Transl. Med. 2018, 16, 235. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Li, Q.; Dong, J.; Li, D.; Xu, X.; Xing, W.; Zhang, X.; Cao, W.; Hou, H.; Wang, H.; et al. The Association Between Normal BMI With Central Adiposity and Proinflammatory Potential Immunoglobulin G N-Glycosylation. Diabetes Metab. Syndr. Obes. 2019, 12, 2373–2385. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Klarić, L.; Yu, X.; Thaqi, K.; Dong, J.; Novokmet, M.; Wilson, J.; Polasek, O.; Liu, Y.; Krištić, J.; et al. The Association Between Glycosylation of Immunoglobulin G and Hypertension: A Multiple Ethnic Cross-Sectional Study. Medicine 2016, 95, e3379. [Google Scholar] [CrossRef]
- Vidarsson, G.; Dekkers, G.; Rispens, T. IgG subclasses and allotypes: From structure to effector functions. Front. Immunol. 2014, 5, 520. [Google Scholar] [CrossRef]
- Nakano, M.; Mishra, S.K.; Tokoro, Y.; Sato, K.; Nakajima, K.; Yamaguchi, Y.; Taniguchi, N.; Kizuka, Y. Bisecting GlcNAc Is a General Suppressor of Terminal Modification of N-glycan. Mol. Cell. Proteom. 2019, 18, 2044–2057. [Google Scholar] [CrossRef] [PubMed]
- Mihai, S.; Nimmerjahn, F. The role of Fc receptors and complement in autoimmunity. Autoimmun. Rev. 2013, 12, 657–660. [Google Scholar] [CrossRef] [PubMed]
- Karsten, C.M.; Pandey, M.K.; Figge, J.; Kilchenstein, R.; Taylor, P.R.; Rosas, M.; McDonald, J.U.; Orr, S.J.; Berger, M.; Petzold, D.; et al. Anti-inflammatory activity of IgG1 mediated by Fc galactosylation and association of FcγRIIB and dectin-1. Nat. Med. 2012, 18, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yuan, H.; Lyu, J.; Meng, X.; Tian, Q.; Li, Y.; Zhang, J.; Xu, X.; Su, J.; Hou, H.; et al. Association of dementia with immunoglobulin G N-glycans in a Chinese Han Population. NPJ Aging Mech. Dis. 2021, 7, 3. [Google Scholar] [CrossRef]
- Kemna, M.J.; Plomp, R.; van Paassen, P.; Koeleman, C.A.M.; Jansen, B.C.; Damoiseaux, J.; Cohen Tervaert, J.W.; Wuhrer, M. Galactosylation and Sialylation Levels of IgG Predict Relapse in Patients with PR3-ANCA Associated Vasculitis. EBioMedicine 2017, 17, 108–118. [Google Scholar] [CrossRef]
- Kaneko, Y.; Nimmerjahn, F.; Ravetch, J.V. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science 2006, 313, 670–673. [Google Scholar] [CrossRef]
- Hassinen, A.; Pujol, F.M.; Kokkonen, N.; Pieters, C.; Kihlström, M.; Korhonen, K.; Kellokumpu, S. Functional organization of Golgi N- and O-glycosylation pathways involves pH-dependent complex formation that is impaired in cancer cells. J. Biol. Chem. 2011, 286, 38329–38340. [Google Scholar] [CrossRef]
- Huffman, J.E.; Knezevic, A.; Vitart, V.; Kattla, J.; Adamczyk, B.; Novokmet, M.; Igl, W.; Pucic, M.; Zgaga, L.; Johannson, Å.; et al. Polymorphisms in B3GAT1, SLC9A9 and MGAT5 are associated with variation within the human plasma N-glycome of 3533 European adults. Hum. Mol. Genet. 2011, 20, 5000–5011. [Google Scholar] [CrossRef]
| Pooling of Chinese Muslim Ethnic Minorities | Han Chinese | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variables | HDC | HTN | T2DM | Controls | p * | HDC | HTN | T2DM | Controls | p * |
| (n = 67) | (n = 183) | (n = 51) | (n = 183) | (n = 72) | (n = 112) | (n = 50) | (n = 165) | |||
| Gender (male%) | 37 (55.22%) | 71 (38.80%) | 16 (31.37%) | 76 (41.53%) | 4.80 × 10−2 | 37 (51.39%) | 58 (50.43%) | 23 (46.00%) | 26 (15.76%) # | <1.00 × 10−3 |
| Age (years) | 63 (54, 69) | 59 (48, 67) | 54 (43, 68) | 51 (40, 61) $,# | <1.00 × 10−3 | 66 (49, 76) | 49 (43, 54) | 54 (46, 73) | 46 (42, 50) &,# | <1.00 × 10−3 |
| BMI (kg/m2) | 26.23 ± 4.52 | 27.04 ± 4.73 | 26.27 ± 4.39 | 25.85 ± 4.69 | 1.06 × 10−1 | 24.97 (23.03, 27.04) | 25.68 (23.84, 27.76) | 24.22 (21.73, 27.53) | 23.11 (21.57, 25.08) # | <1.00 × 10−3 |
| SBP (mmHg) | 151.03 ± 21.47 | 147.13 ± 21.72 | 116.27 ± 9.69 | 115.35 ± 10.62 $,# | <1.00 × 10−3 | 148.88 ± 15.77 | 132.77 ± 10.53 | 116.00 ± 16.16 | 104.87 ± 7.38 $,# | <1.00 × 10−3 |
| DBP (mmHg) | 93.78 ± 13.42 | 91.82 ± 14.06 | 72.84 ± 8.10 | 73.02 ± 8.93 $,# | <1.00 × 10−3 | 83.67 ± 10.31 | 93.13 ± 6.01 | 71.92 ± 10.14 | 68.57 ± 3.84 &,$,# | <1.00 × 10−3 |
| FBG (mmol/L) | 7.40 (6.70, 8.10) | 5.30 (5.00, 5.80) | 7.00 (6.70, 7.80) | 5.20 (4.90, 5.80) &,# | <1.00 × 10−3 | 7.37 (5.60, 8.49) | 5.32 (5.07, 5.78) | 7.58 (6.77, 7.87) | 5.03 (4.76, 5.36) &,# | <1.00 × 10−3 |
| TC (mmol/L) | 4.71 (3.75, 5.34) | 4.71 (4.06, 5.46) | 4.58 (3.94, 5.19) | 4.76 (4.18, 5.61) | 3.97 × 10−1 | 4.59 ± 1.47 | 5.19 ± 0.93 | 4.59 ± 1.15 | 5.06 ± 0.92 &,# | 7.00 × 10−3 |
| TG (mmol/L) | 3.10 (2.47, 3.70) | 3.07 (2.44, 3.70) | 3.10 (2.76, 3.68) | 3.19 (2.65, 3.78) | 3.48 × 10−1 | 1.45 (1.05, 2.17) | 1.52 (1.03, 2.38) | 1.10 (0.71, 1.97) | 0.98 (0.77, 1.44) $,# | <1.00 × 10−3 |
| HDL (mmol/L) | 1.83 (1.51, 2.10) | 1.64 (1.29, 2.04) | 1.71 (1.27, 1.98) | 1.78 (1.32, 2.13) | 3.06 × 10−1 | 1.20 (0.95, 1.50) | 1.49 (1.25, 1.70) | 1.41 (1.07, 1.66) | 1.66 (1.44, 1.89) &,# | <1.00 × 10−3 |
| LDL (mmol/L) | 2.60 (1.67, 3.47) | 2.29 (1.48, 3.17) | 2.50 (1.80, 3.43) | 2.17 (1.37, 3.28) | 1.04 × 10−1 | 2.39 (1.86, 3.02) | 2.76 (2.37, 3.26) | 2.35 (1.80, 2.96) | 2.65 (2.24, 3.19) &,# | <1.00 × 10−3 |
| Dyslipidemia (%) | 57 (85.07%) | 149 (81.42%) | 47 (92.16%) | 161 (87.98%) | 1.57 × 10−1 | 35 (48.61%) | 36 (32.14%) | 17 (34.00%) | 31 (18.79%) # | <1.00 × 10−3 |
| Type of Model | Pooling of Chinese Muslim Ethnic Minorities | Han Chinese | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Included Variables of Model | AUC (95%CI) | p * | Prediction Error | Included Variables of Model | AUC (95%CI) | p * | Prediction Error | ||
| HDC vs. HTN | baseline model | Age, gender, ethnicity, BMI, and dyslipidemia | 0.653 (0.578, 0.728) | 2.15 × 10−4 | 0.280 ± 0.010 | Age, gender, BMI, and dyslipidemia | 0.764 (0.686, 0.842) | 1.49 × 10−9 | 0.249 ± 0.003 |
| glycan-based model | GP5, GP6, GP16, DG24, and DG54 | 0.678 (0.607, 0.748) | 1.70 × 10−5 | 0.279 ± 0.001 | GP20, DG2, and DG53 | 0.715 (0.637, 0.794) | 8.38 × 10−7 | 0.281 ± 0.005 | |
| combined model | Age, gender, ethnicity, BMI, dyslipidemia, GP5, GP6, GP16, DG24, and DG54 | 0.717 (0.651, 0.782) | 1.52 × 10−7 | 0.291 ± 0.003 | Age, gender, BMI, dyslipidemia, GP20, DG2, and DG53 | 0.828 (0.761, 0.896) | 5.91 × 10−14 | 0.234 ± 0.008 | |
| HDC vs. T2DM | baseline model | Age, gender, ethnicity, BMI, and dyslipidemia | 0.682 (0.584, 0.779) | 6.60 × 10−5 | 0.439 ± 0.004 | Age, gender, BMI, and dyslipidemia | 0.609 (0.508, 0.710) | 4.13 × 10−2 | 0.452 ± 0.001 |
| glycan-based model | GP5, GP16, and GP18 | 0.715 (0.622, 0.808) | 7.35 × 10−4 | 0.420 ± 0.002 | GP20 and GP24 | 0.663 (0.567, 0.759) | 2.23 × 10−3 | 0.442 ± 0.009 | |
| combined model | Age, gender, ethnicity, BMI, dyslipidemia, GP5, GP16, and GP18 | 0.747 (0.656, 0.838) | 5.00 × 10−6 | 0.375 ± 0.010 | Age, gender, BMI, dyslipidemia, GP20, and GP24 | 0.689 (0.594, 0.783) | 4.00 × 10−4 | 0.426 ± 0.007 | |
| HDC vs. Controls | baseline model | Age, gender, ethnicity, BMI, and dyslipidemia | 0.771 (0.709, 0.846) | 1.70 × 10−5 | 0.251 ± 0.002 | Age, gender, BMI, and dyslipidemia | 0.871 (0.812, 0.931) | 9.78 × 10−20 | 0.143 ± 0.001 |
| glycan-based model | GP5 and GP6 | 0.678 (0.606, 0.749) | 5.64 × 10−11 | 0.269 ± 0.002 | GP20 | 0.756 (0.685, 0.826) | 4.01 × 10−10 | 0.203 ± 0.001 | |
| combined model | Age, gender, ethnicity, BMI, dyslipidemia, GP5, and GP6 | 0.786 (0.727, 0.846) | 4.03 × 10−12 | 0.238 ± 0.010 | Age, gender, BMI, dyslipidemia, and GP20 | 0.901 (0.850, 0.951) | 1.04 × 10−22 | 0.138 ± 0.001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, X.; Song, M.; Vilaj, M.; Štambuk, J.; Dolikun, M.; Zhang, J.; Liu, D.; Wang, H.; Zhang, X.; Zhang, J.; et al. Glycosylation of IgG Associates with Hypertension and Type 2 Diabetes Mellitus Comorbidity in the Chinese Muslim Ethnic Minorities and the Han Chinese. J. Pers. Med. 2021, 11, 614. https://doi.org/10.3390/jpm11070614
Meng X, Song M, Vilaj M, Štambuk J, Dolikun M, Zhang J, Liu D, Wang H, Zhang X, Zhang J, et al. Glycosylation of IgG Associates with Hypertension and Type 2 Diabetes Mellitus Comorbidity in the Chinese Muslim Ethnic Minorities and the Han Chinese. Journal of Personalized Medicine. 2021; 11(7):614. https://doi.org/10.3390/jpm11070614
Chicago/Turabian StyleMeng, Xiaoni, Manshu Song, Marija Vilaj, Jerko Štambuk, Mamatyusupu Dolikun, Jie Zhang, Di Liu, Hao Wang, Xiaoyu Zhang, Jinxia Zhang, and et al. 2021. "Glycosylation of IgG Associates with Hypertension and Type 2 Diabetes Mellitus Comorbidity in the Chinese Muslim Ethnic Minorities and the Han Chinese" Journal of Personalized Medicine 11, no. 7: 614. https://doi.org/10.3390/jpm11070614
APA StyleMeng, X., Song, M., Vilaj, M., Štambuk, J., Dolikun, M., Zhang, J., Liu, D., Wang, H., Zhang, X., Zhang, J., Cao, W., Momčilović, A., Trbojević-Akmačić, I., Li, X., Zheng, D., Wu, L., Guo, X., Wang, Y., Lauc, G., & Wang, W. (2021). Glycosylation of IgG Associates with Hypertension and Type 2 Diabetes Mellitus Comorbidity in the Chinese Muslim Ethnic Minorities and the Han Chinese. Journal of Personalized Medicine, 11(7), 614. https://doi.org/10.3390/jpm11070614









