Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Analysis
2.3. Ethical Approval
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease [COVID-19] Dashboard. 2021. Available online: https://covid19.who.int (accessed on 31 March 2021).
- Schwartz, D.A. An Analysis of 38 Pregnant Women with COVID-19, Their Newborn Infants, and Maternal-Fetal Transmission of SARS-CoV-2: Maternal Coronavirus Infections and Pregnancy Outcomes. Arch. Pathol. Lab. Med. 2020, 144, 799–805. [Google Scholar] [CrossRef] [Green Version]
- Di Mascio, D.; Khalil, A.; Saccone, G.; Rizzo, G.; Buca, D.; Liberati, M.; Vecchiet, J.; Nappi, L.; Scambia, G.; Berghella, V.; et al. Outcome of Coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: A systematic review and meta-analysis. Am. J. Obstet. Gynecol. MFM 2020, 2, 100107. [Google Scholar] [CrossRef]
- Dashraath, P.; Wong, J.L.J.; Lim, M.X.K.; Lim, L.M.; Li, S.; Biswas, A.; Choolani, M.; Mattar, C.; Su, L.L. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy. Am. J. Obstet. Gynecol. 2020, 222, 521–531. [Google Scholar] [CrossRef]
- Villalain, C.; Herraiz, I.; Luczkowiak, J.; Perez-Rivilla, A.; Folgueira, M.D.; Mejia, I.; Batllori, E.; Felipe, E.; Risco, B.; Galindo, A.; et al. Seroprevalence analysis of SARS-CoV-2 in pregnant women along the first pandemic outbreak and perinatal outcome. PLoS ONE 2020, 15, e0243029. [Google Scholar] [CrossRef]
- Martinez-Portilla, R.J.; Sotiriadis, A.; Chatzakis, C.; Torres-Torres, J.; Espino, Y.S.S.; Sandoval-Mandujano, K.; Castro-Bernabe, D.A.; Medina-Jimenez, V.; Monarrez-Martin, J.C.; Figueras, F.; et al. Pregnant women with SARS-CoV-2 infection are at higher risk of death and pneumonia: Propensity score matched analysis of a nationwide prospective cohort (COV19Mx). Ultrasound. Obstet. Gynecol. 2021, 57, 24–31. [Google Scholar] [CrossRef]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.T.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020, 20, 669–677. [Google Scholar] [CrossRef]
- Wong, S.F.; Chow, K.M.; Leung, T.N.; Ng, W.F.; Ng, T.K.; Shek, C.C.; Ng, P.C.; Lam, P.W.; Ho, L.C.; To, W.W.; et al. Pregnancy and perinatal outcomes of women with severe acute respiratory syndrome. Am. J. Obstet. Gynecol. 2004, 191, 292–297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centro de Coordinación de Alertas y Emergencias Sanitarias. Actualización n° 343. Enfermedad por el Coronavirus (COVID-19); Ministerio de Sanidad: Madrid, Spain, 2021. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/Actualizacion_242_COVID-19pdf (accessed on 31 March 2021).
- Cunarro-Lopez, Y.; Cano-Valderrama, O.; Pintado-Recarte, P.; Cueto-Hernandez, I.; Gonzalez-Garzon, B.; Garcia-Tizon, S.; Bujan, J.; Asúnsolo, Á.; Ortega, M.A.; De León-Luis, J.A. Maternal and Perinatal Outcomes in Patients with Suspected COVID-19 and Their Relationship with a Negative RT-PCR Result. J. Clin. Med. 2020, 9, 3552. [Google Scholar] [CrossRef] [PubMed]
- Pollan, M.; Perez-Gomez, B.; Pastor-Barriuso, R.; Oteo, J.; Hernan, M.A.; Perez-Olmeda, M.; Sanmartín, J.L.; Fernández-García, A.; Cruz, I.; de Larrea, N.F.; et al. Prevalence of SARS-CoV-2 in Spain (ENE-COVID): A nationwide, population-based seroepidemiological study. Lancet 2020, 396, 535–544. [Google Scholar] [CrossRef]
- Soriano, V.; Ganado-Pinilla, P.; Sanchez-Santos, M.; Gomez-Gallego, F.; Barreiro, P.; de Mendoza, C.; Corral, O. Main Differences Between the First and Second Waves of COVID-19 in Madrid, Spain. Int. J. Infect. Dis. 2021, 105, 374–376. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef] [Green Version]
- Encinas Pardilla, M.B.; Cano Aguilar, A.; Marcos Puig, B.; Sanz Lorenzana, A.; Rodriguez de la Torre, I.; Hernando Lopez de la Manzanara, P.; Fernandez Bernardo, A.; Martinez Perez, O. Spanish registry of Covid-19 screening in asymptomatic pregnants. Rev. Esp. Salud Publica 2020, 94, e202009092. [Google Scholar]
- Di Mascio, D. COVID Wwgo. Maternal and Perinatal Outcomes of Pregnant Women with SARS-COV-2 infection. Ultrasound. Obstet. Gynecol. 2020, 57, 232–241. [Google Scholar]
- Centro de coordinación de Alertas y Emergencias Sanitarias. Actualización n° 152. Enfermedad por el Coronavirus (COVID-19); Ministerio de Sanidad: Madrid, Spain, 2020. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/Actualizacion_152_COVID-19.pdf (accessed on 31 March 2021).
- Centro de Coordinación de Alertas y Emergencias Sanitarias. Actualización n° 242. Enfermedad por el Coronavirus (COVID-19); Ministerio de Sanidad: Madrid, Spain, 2020. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/Actualizacion_242_COVID-19.pdf (accessed on 31 March 2021).
- Say, L.; Souza, J.P.; Pattinson, R.C. Mortality WHOwgoM, Morbidity c. Maternal near miss—Towards a standard tool for monitoring quality of maternal health care. Best Pract. Res. Clin. Obstet. Gynaecol. 2009, 23, 287–296. [Google Scholar] [CrossRef]
- Mullins, E.; Hudak, M.L.; Banerjee, J.; Getzlaff, T.; Townson, J.; Barnette, K.; Playle, R.; Perry, A.; Bourne, T.; Lees, C.C.; et al. Pregnancy and neonatal outcomes of COVID-19: Co-reporting of common outcomes from PAN-COVID and AAP SONPM registries. Ultrasound Obstet. Gynecol. 2021, 57, 573–581. [Google Scholar] [CrossRef] [PubMed]
- D’Antonio, F.; Sen, C.; Mascio, D.D.; Galindo, A.; Villalain, C.; Herraiz, I.; Arisoy, R.; Ovayolu, A.; Eroğlu, H.; Canales, G.; et al. Maternal and perinatal outcomes in high vs low risk-pregnancies affected by SARS-COV-2 infection (Phase-2): The WAPM (World Association of Perinatal Medicine) working group on COVID-19. Am. J. Obstet. Gynecol. MFM 2021, 3, 100329. [Google Scholar]
- Instituto Nacional de Estadística. Estadísticas del Movimiento Natural de la Población (MNP)—Nacimientos, Defunciones y Matrimonios. Primer Semestre del 2020. Datos Provisionales, 2020. Available online: https://www.ine.es/prensa/mnp_1s2020_p.pdf (accessed on 31 March 2021).
- Cunarro-Lopez, Y.; Pintado-Recarte, P.; Cueto-Hernandez, I.; Hernandez-Martin, C.; Paya-Martinez, M.P.; Munoz-Chapuli, M.D.M.; Cano-Valderrama, Ó.; Bravo, C.; Bujan, J.; Álvarez-Mon, M.; et al. The Profile of the Obstetric Patient with SARS-CoV-2 Infection According to Country of Origin of the Publication: A Systematic Review of the Literature. J. Clin. Med. 2021, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Antoun, L.; Taweel, N.E.; Ahmed, I.; Patni, S.; Honest, H. Maternal COVID-19 infection, clinical characteristics, pregnancy, and neonatal outcome: A prospective cohort study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 252, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Coronado-Arroyo, J.C.; Concepcion-Zavaleta, M.J.; Zavaleta-Gutierrez, F.E.; Concepcion-Urteaga, L.A. Is COVID-19 a risk factor for severe preeclampsia? Hospital experience in a developing country. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 256, 502–503. [Google Scholar] [CrossRef] [PubMed]
- Tutiya, C.; Mello, F.; Chaccur, G.; Almeida, C.; Galvão, E.; de Souza, A.C.B.; Kondo, M.M.; Torloni, M.R.; Siaulys, M. Risk factors for severe and critical Covid-19 in pregnant women in a single center in Brazil. J. Matern. Fetal Neonatal Med. 2021, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Team CC-R. Severe Outcomes Among Patients with Coronavirus Disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb. Mortal Wkly. Rep. 2020, 69, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Kalafat, E.; Benlioglu, C.; O’Brien, P.; Morris, E.; Draycott, T.; Thangaratinam, S.; Le Doare, K.; Heath, P.; Ladhani, S.; et al. SARS-CoV-2 infection in pregnancy: A systematic review and meta-analysis of clinical features and pregnancy outcomes. EClinicalMedicine 2020, 25, 100446. [Google Scholar] [CrossRef] [PubMed]
- Guallar, M.P.; Meirino, R.; Donat-Vargas, C.; Corral, O.; Jouve, N.; Soriano, V. Inoculum at the time of SARS-CoV-2 exposure and risk of disease severity. Int. J. Infect. Dis. 2020, 97, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Narang, K.; Enninga, E.A.L.; Gunaratne, M.; Ibirogba, E.R.; Trad, A.T.A.; Elrefaei, A.; Theiler, R.N.; Ruano, R.; Szymanski, M.; Chakraborty, R.; et al. SARS-CoV-2 Infection and COVID-19 During Pregnancy: A Multidisciplinary Review. Mayo Clin. Proc. 2020, 95, 1750–1765. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, D.J.; Theiler, R.N.; Rasmussen, S.A. Emerging infections and pregnancy. Emerg. Infect. Dis. 2006, 12, 1638–1643. [Google Scholar] [CrossRef]
- Novoa, R.H.; Quintana, W.; Llancari, P.; Urbina-Quispe, K.; Guevara-Rios, E.; Ventura, W. Maternal clinical characteristics and perinatal outcomes among pregnant women with coronavirus disease 2019. A systematic review. Travel Med. Infect. Dis. 2021, 39, 101919. [Google Scholar] [CrossRef]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Bekkar, B.; Pacheco, S.; Basu, R.; DeNicola, N. Association of Air Pollution and Heat Exposure with Preterm Birth, Low Birth Weight, and Stillbirth in the US: A Systematic Review. JAMA Netw. Open 2020, 3, e208243. [Google Scholar] [CrossRef]
- Been, J.V.; Burgos Ochoa, L.; Bertens, L.C.M.; Schoenmakers, S.; Steegers, E.A.P.; Reiss, I.K.M. Impact of COVID-19 mitigation measures on the incidence of preterm birth: A national quasi-experimental study. Lancet Public Health 2020, 5, e604–e611. [Google Scholar] [CrossRef]
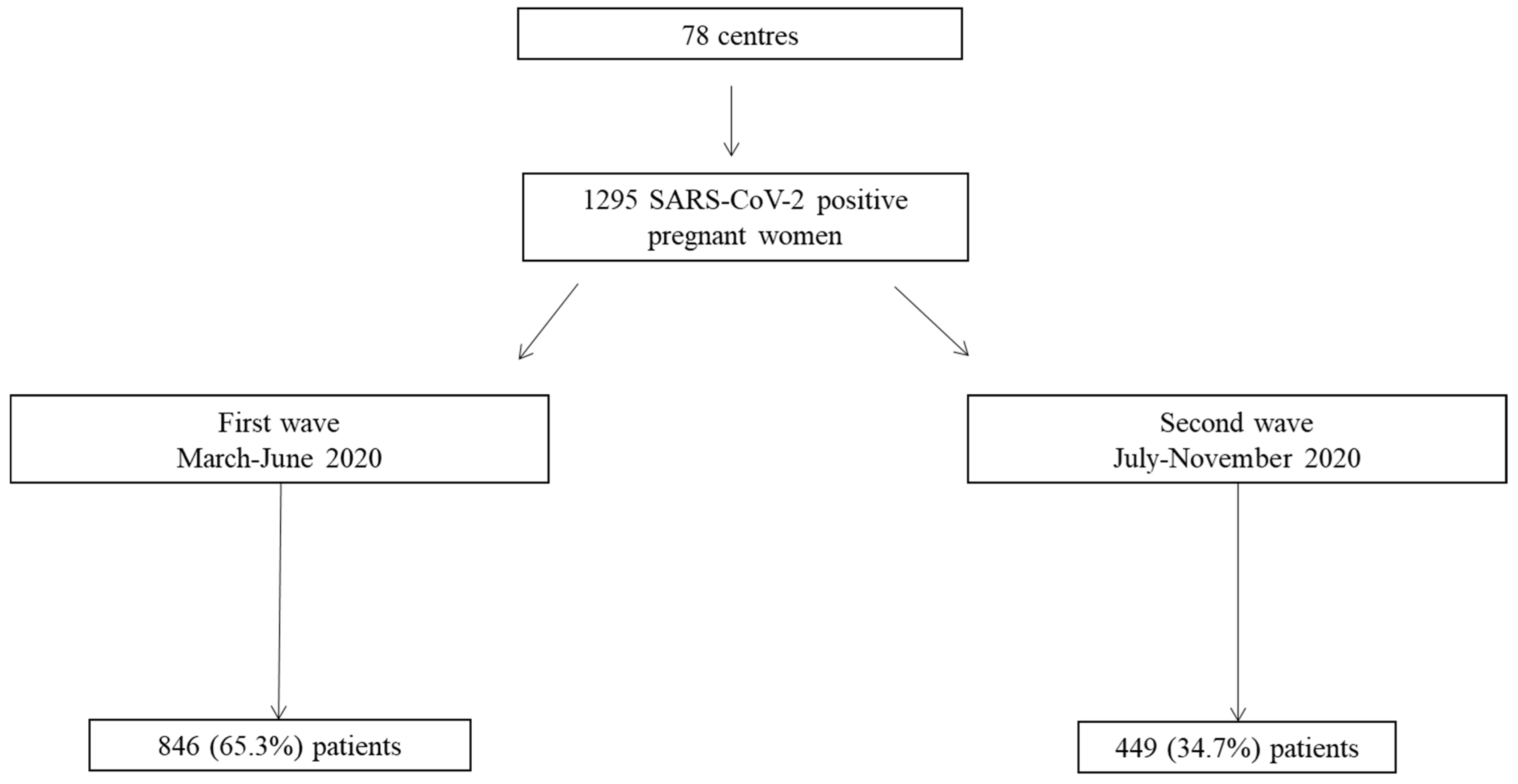
| Number (%) of Patients Reporting Results | First Wave | Second Wave | p-Value | |
|---|---|---|---|---|
| n, % | 1295 (100) | 846 (65.3) | 449 (34.7) | |
| Maternal characteristics | ||||
| Age, mean, 95% CI | 1284 (99.2) | 33.3 (32.8–33.7) | 31.7 (31.1–32.3) | <0.001 |
| Maternal age > 35 years, n, % | 1284 (99.2) | 358 (42.6) | 145 (32.7) | <0.001 |
| Tobacco use, n, % | 1239 (95.7) | 94 (11.6) | 35 (8.2) | 0.054 |
| BMI, mean, 95% CI | 1119 (86.4) | 26.4 (26.1–26.8) | 26.8 (26.3–27.4) | 0.193 |
| Morbidities, n, % | 1182 (91.3) | 310 (40.2) | 155 (37.8) | 0.431 |
| Nuliparous, n, % | 1281 (98.9) | 316 (37.9) | 175 (39.1) | 0.692 |
| Symptoms at triage, n, % | 1103 (85.2) | 457 (62.4) | 155 (41.8) | <0.001 |
| Pneumonia, n, % | 978 (75.5) | 104 (16.1) | 17 (5.1) | <0.001 |
| Treatment, n, % | 1295 (100) | 656 (77.5) | 354 (78.8) | 0.590 |
| Laboratory tests | ||||
| Haemoglobin count (x1ꝰ/L), mean, 95% CI | 892 (68.9) | 12.2 (11.1–13.4) | 12.4 (10.6–14.2) | 0.891 |
| Haematocrito count (x1ꝰ/L), mean, 95% CI | 875 (67.6) | 32.5 (31.7–33.3) | 31.7 (30.5–32.9) | 0.262 |
| Leukocyte count (x1ꝰ/L), mean, 95% CI | 902 (69.7) | 37.2 (24.7–49.7) | 10.9 (6.8–15.2) | 0.003 |
| Lymphocyte count (x1ꝰ/L), mean, 95% CI | 847 (65.4) | 3.6 (2.5–4.6) | 4.4 (3.3–5.6) | 0.319 |
| Lymphocytopenia, n, % | 847 (65.4) | 191 (35.6) | 84 (27.0) | 0.009 |
| Platelet count (x1ꝰ/L), mean, 95% CI | 899 (69.4) | 177.8 (169.8–185.7) | 186.9 (174.6–197.1) | 0.242 |
| Thrombocytopenia, n, % | 899 (69.4) | 184 (31.4) | 93 (29.8) | 0.634 |
| aPTT, mean, 95% CI | 641 (50.0) | 25.8 (24.1–27.5) | 26.4 (22.7–30.1) | 0.753 |
| Obstetric and perinatal characteristics | ||||
| Obstetric morbidities, n, % | 1057 (81.6) | 299 (43.5) | 150 (40.5) | 0.349 |
| Gestational age at triage, mean, 95% CI | 1295 (100) | 33.6 (33.0–34.1) | 37.3 (37.0–37.6) | <0.001 |
| Gestational age at delivery, mean, 95% CI | 1295 (100) | 38.5 (38.3–38.7) | 38.7 (38.4–38.9) | 0.185 |
| Birthweight, mean, 95% CI | 1275 (98.5) | 3357.2 (3115.6–3198.9) | 3232.4 (3176.9–3288.0) | 0.036 |
| Maternal and perinatal mortality | ||||
| Maternal mortality, n, % | 1295 (100) | 2 (0.2) | 0 (0.0) | 0.547 |
| Perinatal mortality, n, % | 1295 (100) | 9 (1.1) | 7 (1.6) | 0.439 |
| Overall maternal morbidity | 1020 (78.8) | 305 (42.4) | 122 (41.7) | 0.617 |
| COVID-19 maternal morbidity | 1295 (100) | 77 (9.1) | 16 (3.6) | <0.001 |
| Oxygen therapy, n, % | 1295 (100) | 60 (7.1) | 13 (2.9) | 0.001 |
| Mechanical ventilation, n, % | 1295 (100) | 15 (1.8) | 2 (0.5) | 0.029 |
| Admission to ICU, n, % | 1295 (100) | 25 (3.0) | 11 (2.5) | 0.595 |
| Non COVID-19 maternal morbidity | 1015 (78.4) | 276 (38.5) | 118 (39.6) | 0.743 |
| C-section, n, % | 1293 (99.8) | 254 (30.1) | 108 (24.1) | 0.022 |
| Haemorrhagic disorders, n, % | 938 (72.4) | 32 (4.7) | 11 (4.2) | 0.711 |
| Hypertensive disorders, n, % | 1071 (82.7) | 25 (3.4) | 12 83.6) | 0.878 |
| Perinatal morbidity | 1068 (82.5) | 144 (20.4) | 60 (16.6) | 0.137 |
| Preterm birth, n, % | 1295 (100) | 105 (12.4) | 39 (8.7) | 0.039 |
| Neonatal near miss, n, % | 1037 (80.1) | 41 (6.9) | 21 (5.9) | 0.964 |
| Gestational age < 33 weeks, n, % | 1295 (100) | 36 (4.3) | 17 (3.8) | 0.683 |
| Birthweight < 1750 g, n, % | 1275 (98.5) | 24 (2.9) | 10 (2.3) | 0.521 |
| Apgar 5 min < 7, n, % | 1277 (98.6) | 10 (1.2) | 2 (0.5) | 0.165 |
| Admission to NICU, n, % | 1295 (100) | 93 (11.0) | 41 (9.1) | 0.291 |
| Maternal Morbidity | Perinatal Morbidity | |||
|---|---|---|---|---|
| First Wave | Second Wave | First Wave | Second Wave | |
| OR/r | OR/r | OR/r | OR/r | |
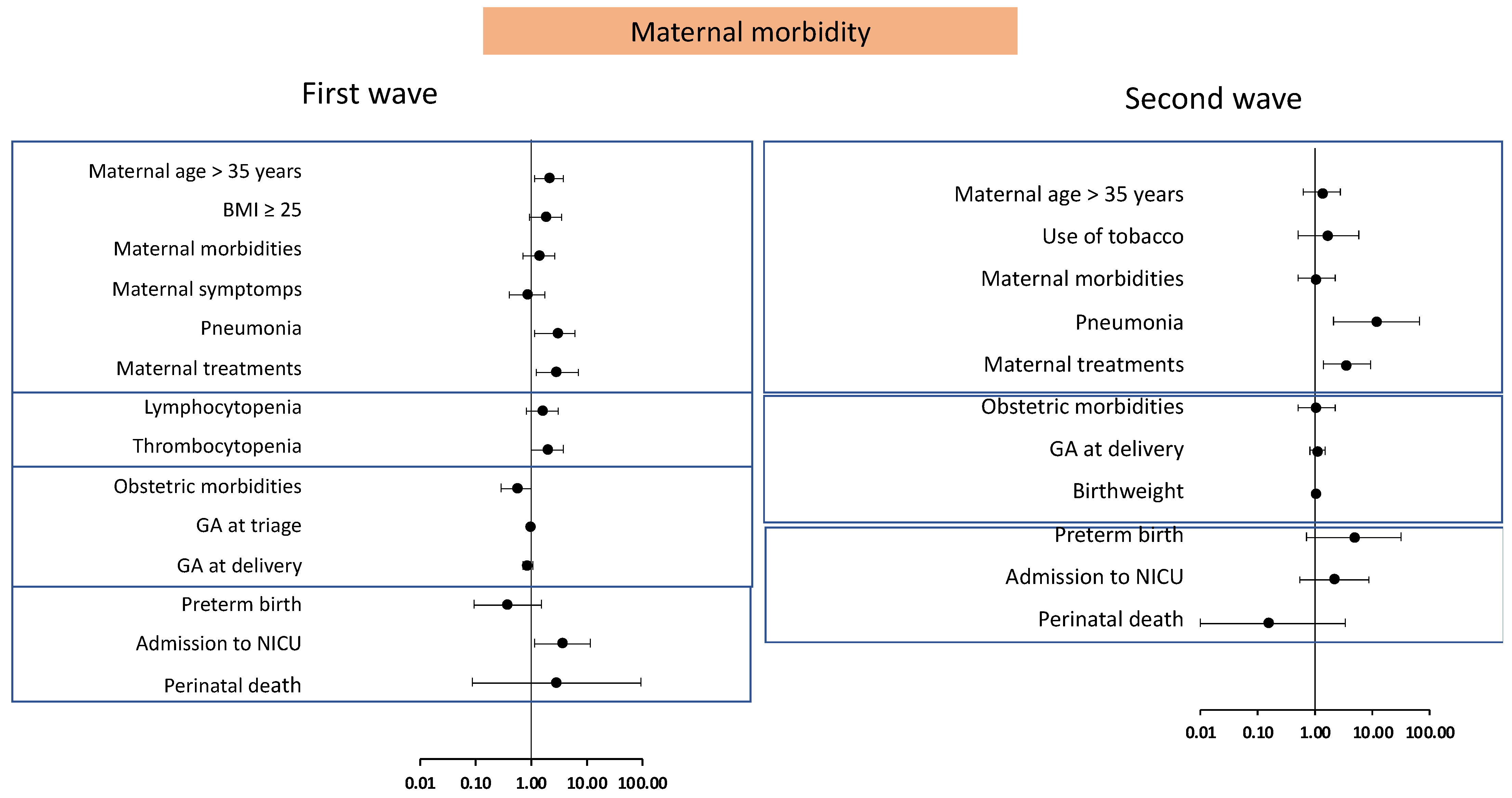
| Maternal characteristics | ||||
| Age, r | 0.1 (0–0.2) | 0.1 (0–0.3) | 0.01 (−0.1–0.1) | 0 (−0.1–0.1) |
| Maternal age > 35 years, OR | 1.5 (1.1–2.1) | 1.6 (1–2.6) | 1 (0.7–1.4) | 0.8 (0.4–1.5) |
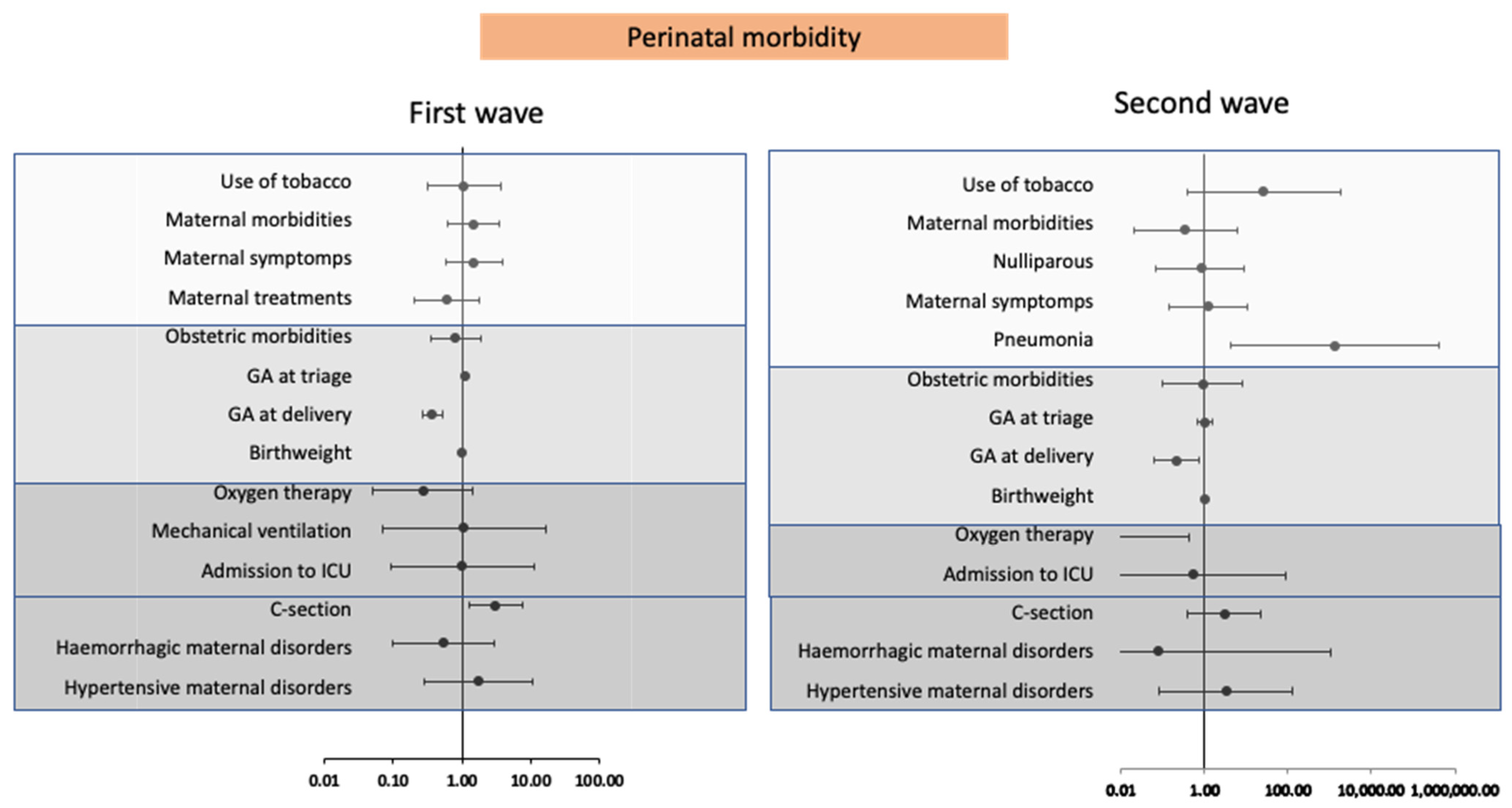
| Tobacco use, OR | 1 (0.6–1.6) | 1.7 (0.7–3.8) | 1.6 (0.9–2.8) | 1.6 (0.6–3.8) |
| BMI, r | 0.2 (0.1–0.2) | 0.1 (0–0.2) | 0 (0–0) | 0 (−0.1–0.1) |
| BMI ≧ 25, OR | 1.9 (1.3–2.6) | 1.3 (0.8–2.1) | 1.2 (0.7–1.6) | 1.1 (0.6–2.1) |
| Morbidities, OR | 2.2 (1.6–3) | 1.6 (1–2.6) | 1.7 (1.1–2.5) | 1.7 (0.9–3.1) |
| Nuliparous, OR | 1.2 (0.9–1.6) | 0.7 (0.4–1.1) | 1.4 (1–2) | 0.7 (0.4–1.2) |
| Symptoms at triage, OR | 2.1 (1.5–3) | 1.2 (0.7–2.1) | 1.9 (1.3–2.9) | 2 (1.1–3.7) |
| Pneumonia, OR | 2.9 (1.8–4.5) | 9.4 (2.1–43.3) | 1.1 (0.7–1.9) | 2.4 (0.8–7.4) |
| Treatment, OR | 2.1 (1.4–3.2) | 2.8 (1.6–5.2) | 1.6 (1–2.6) | 1.2 (0.6–2.6) |
| Laboratory tests | ||||
| Haemoglobin count (x1ꝰ/L), r | 0 (−0.1–0) | 0 (−0.1–0.2) | 0.1 (−0.1–0) | −0.1 (−0.2–0) |
| Haematocrito coun (x1ꝰ/L), r | 0 (−0.2–0) | 0 (−0.1–0.2) | 0.1 (−0.1–0) | 0 (−0.1–0.1) |
| Leukocyte count (x1ꝰ/L), r | 0 (−0.1–0.1) | 0 (−0.1–0.2) | 0.1 (0–0.2) | 0 (−0.2–0.1) |
| Lymphocite count (x1ꝰ/L), r | 0 (−0.1–0.1) | −0.1 (−0.2–0.1) | 0.1 (−0.1–0) | 0 (−0.12–0.1) |
| Lymphocytopenia, OR | 1.9 (1.3–2.8) | 1.4 (0.7–2.6) | 0.9 (0.6–1.4) | 0.8 (0.4–1.7) |
| Platelet count (x1ꝰ/L), r | −0.1 (−0.2–0) | −0.1 (−0.2–0.1) | 0 (−0.1–0.1) | 0 (−0.1–0.1) |
| Thrombocytopenia, OR | 1.8 (1.2–2.6) | 1.5 (0.8–2.7) | 1.1 (0.7–1.8) | 1.1 (0.5–2.2) |
| aPTT, r | 0 (−0.1–0.1) | −0.1 (−0.2–0.1) | 0 (−0.1–0.1) | 0 (−0.1–0.1) |
| Obstetric and perinatal characteristics | ||||
| Obstetric morbidities, OR | 1.4 (1–2) | 2 (1.2–3.3) | 2.1 (1.4–3) | 4.6 (2.3–9.1) |
| Gestational age at triage, r | −0.1 (−0.2–0) | −0.1 (−0.2–0.1) | −0.1 (−0.1–0) | −0.4 (−0.5–−0.4) |
| Gestational age at delivery, r | −0.2 (−0.3–−0.2) | −0.15 (−0.3–0) | −0.7 (−0.7–−0.6) | −0.6 (−0.7–−0.6) |
| Birthweight, r | −0.1 (−0.2–−0.1) | −0.2 (−0.3–0) | −0.5 (−0.6–−0.5) | −0.4 (−0.6–−0.3) |
| Maternal and perinatal mortality | ||||
| Maternal mortality, n, % | n.a. | n.a. | 1 (n.a.–n.a.) | 1 (n.a.–n.a.) |
| Perinatal mortality, OR | 8.3 (1–69.4) | 0.6 (0.1–3) | n.a. | n.a. |
| Overall Maternal Morbidity | n.a. | n.a. | 3.8 (2.5–5.8) | 3.3 (1.7–6.3) |
| COVID-19 Maternal Morbidity | n.a. | n.a. | 3 (1.8–5.1) | 10.4 (3.4–32.4) |
| Oxygen therapy, OR | n.a. | n.a. | 1.7 (0.9–3.2) | 6.6 (1.9–22.3) |
| Mechanical ventilation, OR | n.a. | n.a. | 10.4 (3.2–33.8) | 1 (n.a.–n.a.) |
| Admission to ICU, OR | n.a. | n.a. | 11.6 (4.5–30.2) | 52.9 (6.6–426.8) |
| Not COVID-19 Maternal Morbidity | n.a. | n.a. | 3.9 (2.6–5.8) | 3.4 (1.8–6.7) |
| C-section, OR | n.a. | n.a. | 3.8 (2.6–5.5) | 3.2 (1.8–5.6) |
| Haemorrhagic disorders, OR | n.a. | n.a. | 3.2 (1.5–6.8) | 2.6 (0.6–10.8) |
| Hypertensive disorders, OR | n.a. | n.a. | 4.3 (1.7–10.6) | 23.5 (4.9–112.7) |
| Perinatal morbidity | 3.84 (2.5–5.8) | 3.3 (1.7–6.3) | n.a. | n.a. |
| Preterm birth, OR | 3.8 (2.4–6) | 3.5 (1.6–7.7) | n.a. | n.a. |
| Apgar 5 min < 7, n, % | 1 (n.a.–n.a.) | 1 (n.a.–n.a.) | n.a. | n.a. |
| Admission to NICU, OR | 5.2 (3.1–8.7) | 3.9 (1.8–8.5) | n.a. | n.a. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuñarro-López, Y.; Pintado-Recarte, P.; Hernández-Martín, C.; Paya-Martínez, P.; López-Pérez, R.; Cueto-Hernández, I.; Ruiz-Labarta, J.; Cano-Valderrama, Ó.; Martínez-Pérez, Ó.; Bravo-Arribas, C.; et al. Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic. J. Pers. Med. 2021, 11, 599. https://doi.org/10.3390/jpm11070599
Cuñarro-López Y, Pintado-Recarte P, Hernández-Martín C, Paya-Martínez P, López-Pérez R, Cueto-Hernández I, Ruiz-Labarta J, Cano-Valderrama Ó, Martínez-Pérez Ó, Bravo-Arribas C, et al. Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic. Journal of Personalized Medicine. 2021; 11(7):599. https://doi.org/10.3390/jpm11070599
Chicago/Turabian StyleCuñarro-López, Yolanda, Pilar Pintado-Recarte, Concepción Hernández-Martín, Pilar Paya-Martínez, Rocío López-Pérez, Ignacio Cueto-Hernández, Javier Ruiz-Labarta, Óscar Cano-Valderrama, Óscar Martínez-Pérez, Coral Bravo-Arribas, and et al. 2021. "Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic" Journal of Personalized Medicine 11, no. 7: 599. https://doi.org/10.3390/jpm11070599
APA StyleCuñarro-López, Y., Pintado-Recarte, P., Hernández-Martín, C., Paya-Martínez, P., López-Pérez, R., Cueto-Hernández, I., Ruiz-Labarta, J., Cano-Valderrama, Ó., Martínez-Pérez, Ó., Bravo-Arribas, C., Ortega, M. A., & De León-Luis, J. A. (2021). Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic. Journal of Personalized Medicine, 11(7), 599. https://doi.org/10.3390/jpm11070599









