Conventional CT versus Dedicated CT Angiography in DIEP Flap Planning: A Feasibility Study
Abstract
:1. Introduction
2. Materials and Methods
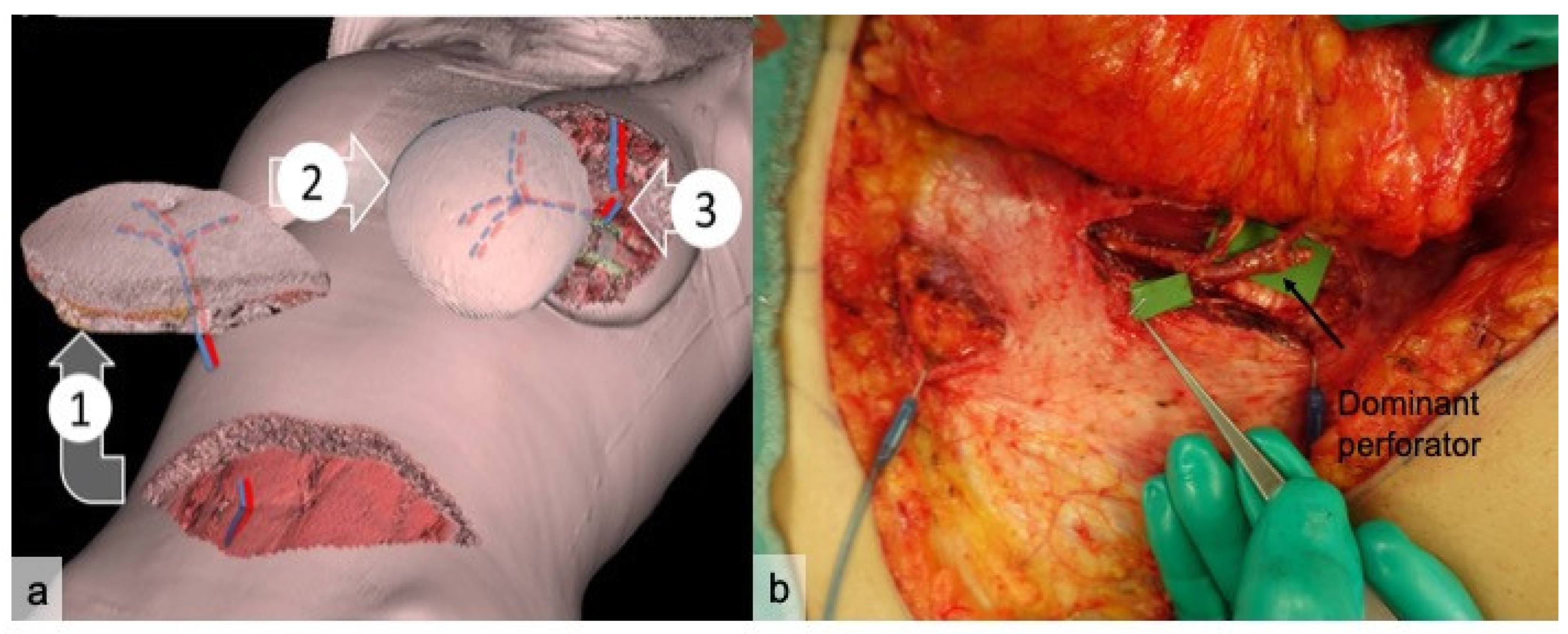
2.1. CTa and cCT Technique
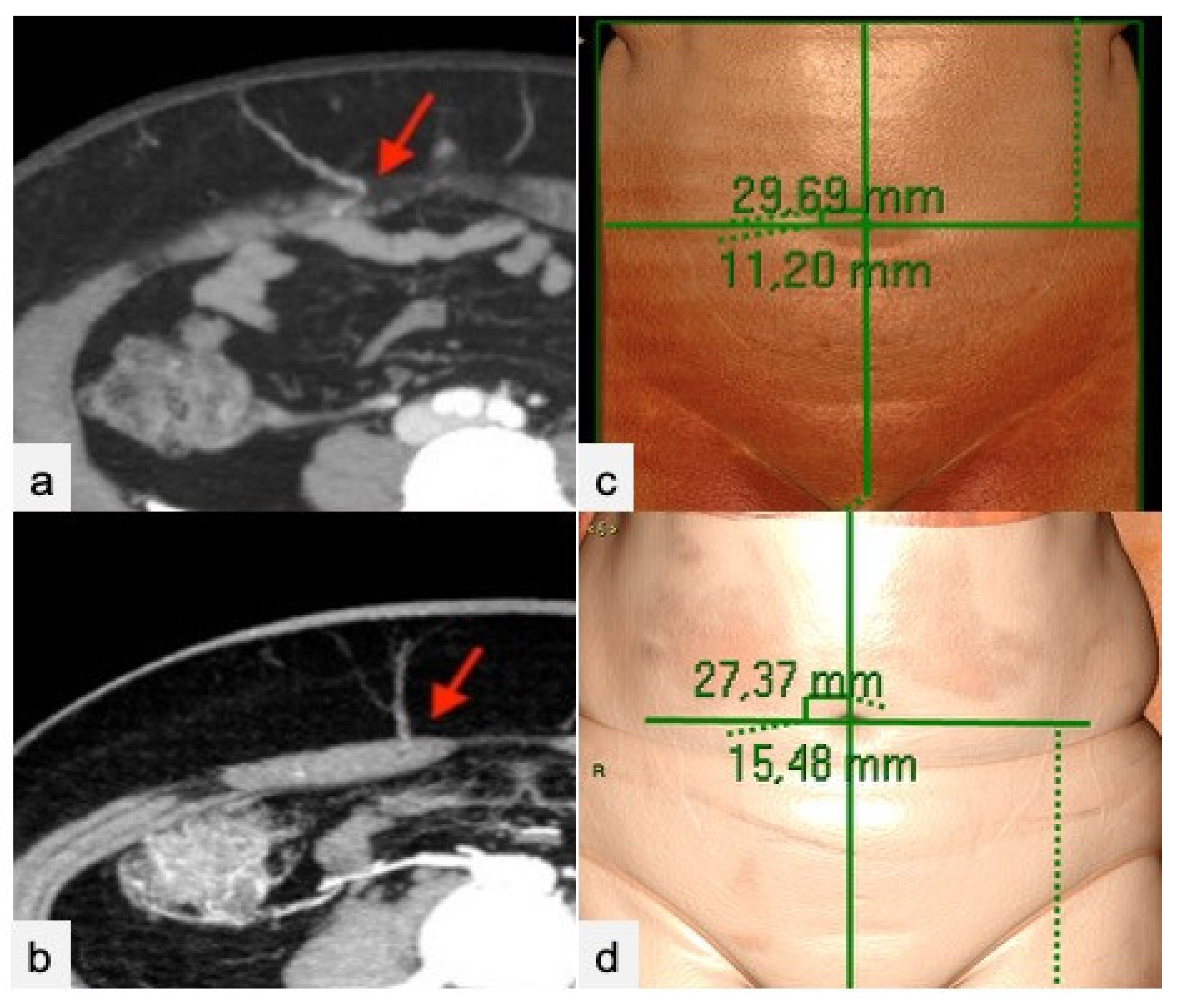
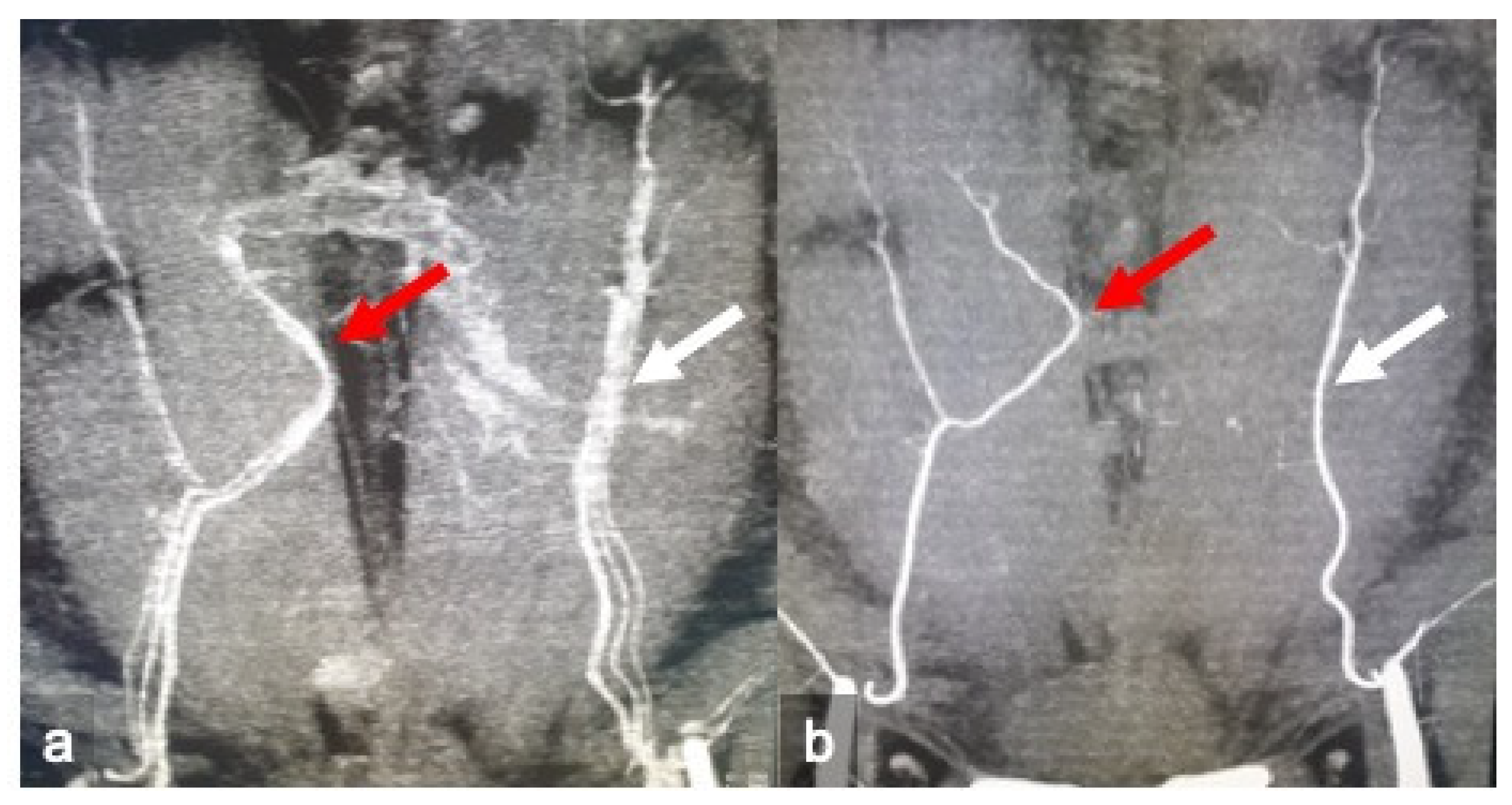
2.2. Image Analysis
2.3. Statistical Analysis
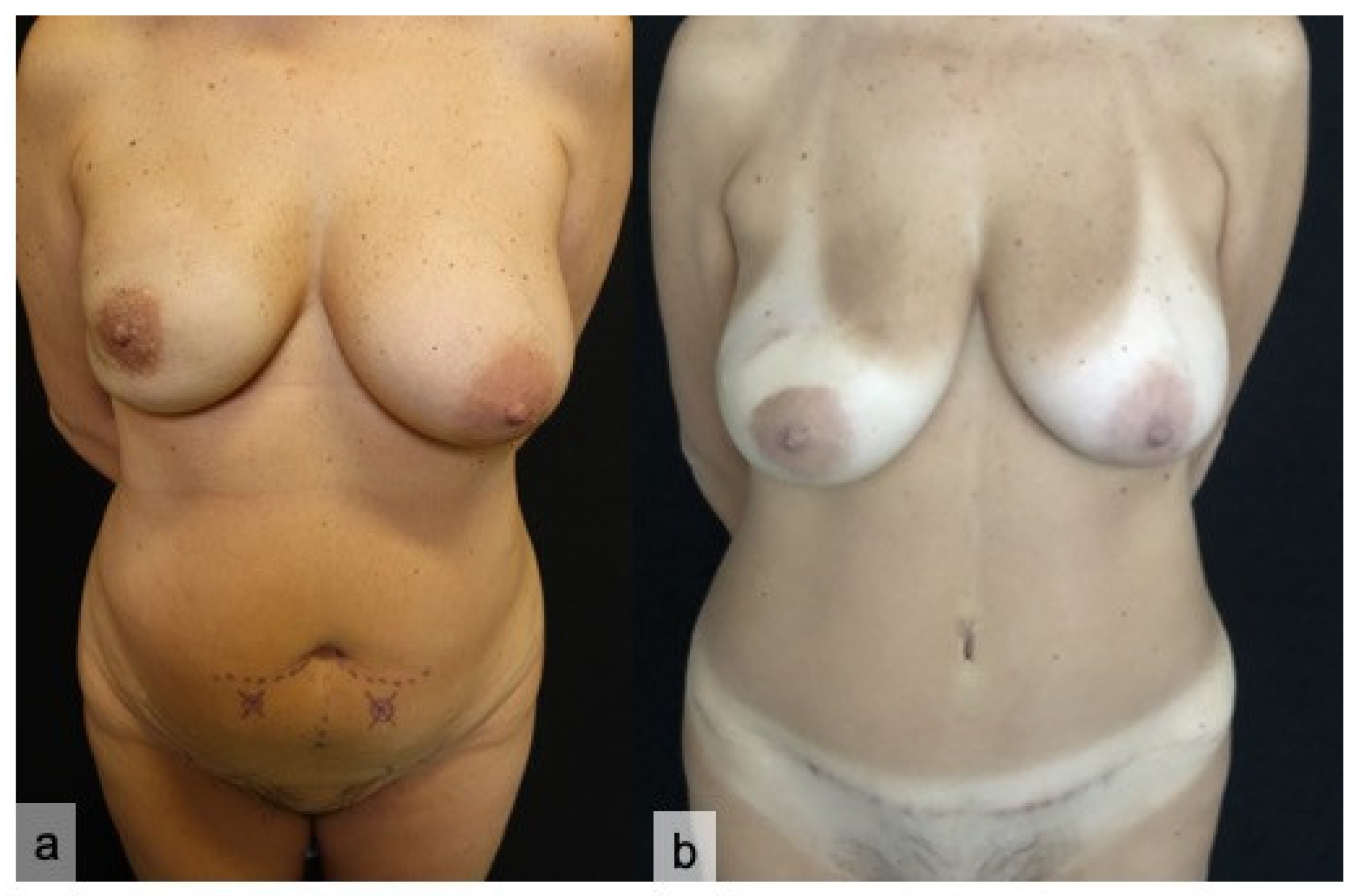
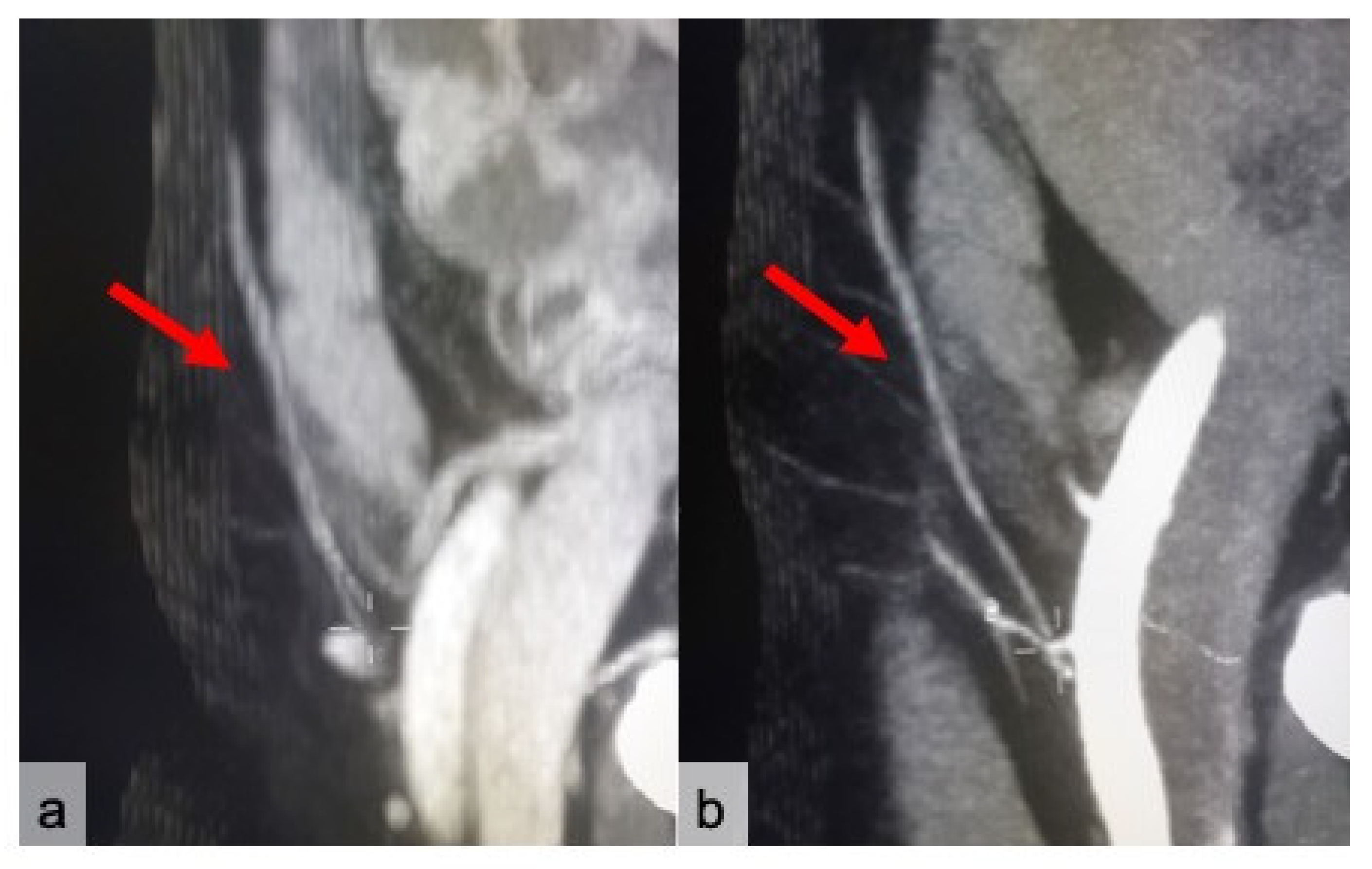
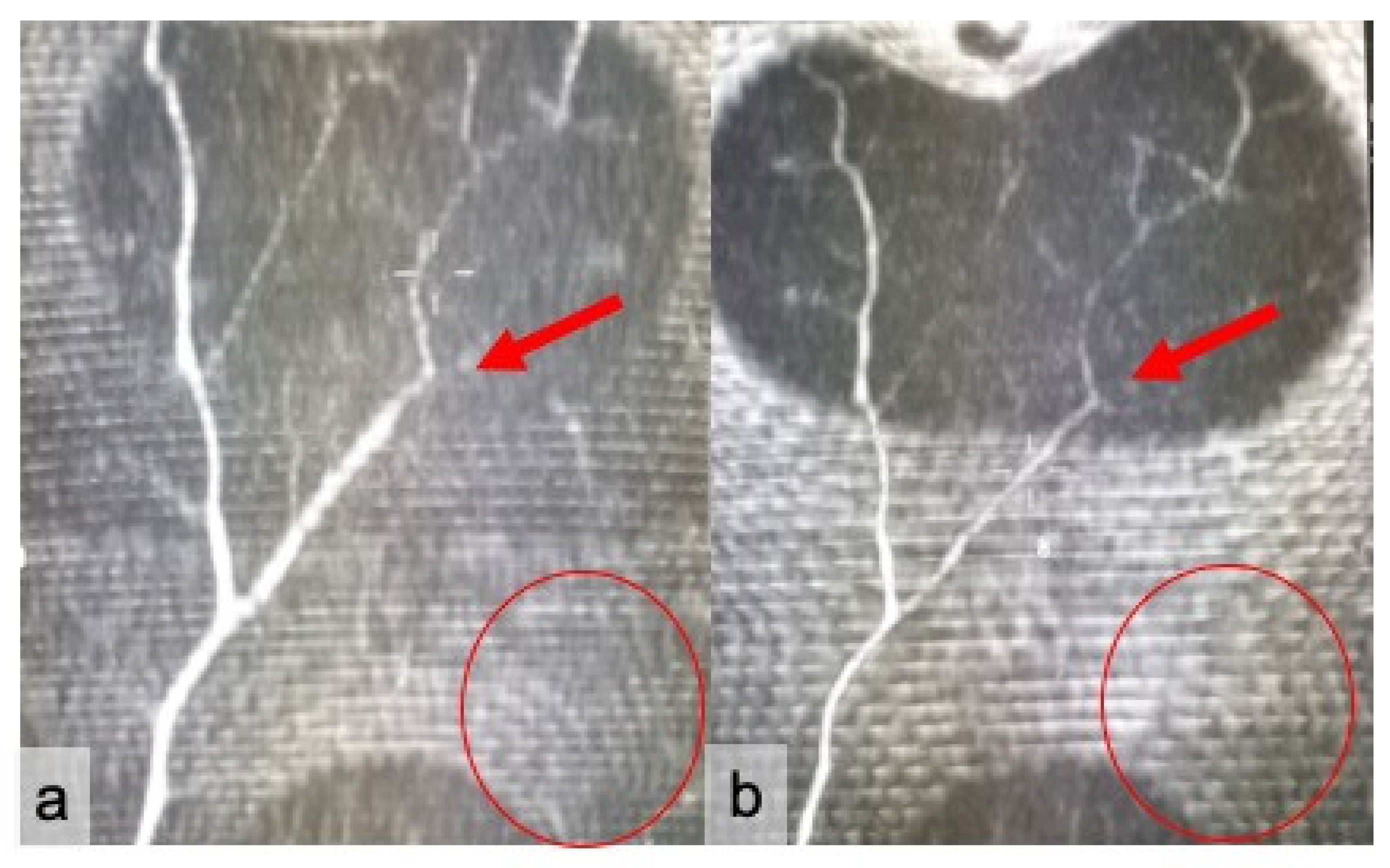
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Munhoz, A.M.; Arruda, E.; Montag, E.; Aldrighi, C.; Aldrighi, J.M.; Gemperli, R.; Ferreira, M.C. Immediate skin-sparing mastectomy reconstruction with deep inferior epigastric perforator (DIEP) flap: Technical aspects and outcome. Breast J. 2007, 13, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Ireton, J.E.; Lakhiani, C.; Saint-Cyr, M. Vascular anatomy of the deep inferior epigastric artery perforator flap: A systematic review. Plast. Reconstr. Surg. 2014, 134, 810e–821e. [Google Scholar] [CrossRef] [PubMed]
- Cina, A.; Salgarello, M.; Barone-Adesi, L.; Rinaldi, P.; Bonomo, L. Planning breast reconstruction with deep inferior epigastric artery perforating vessels: Multidetector CT angiography versus color Doppler US. Radiology 2010, 255, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.J. Comparison of the costs of DIEP and TRAM flaps. Plast. Reconstr. Surg. 2001, 108, 2165. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.L.; Allen, R.J. Cost-based comparison between perforator flaps and TRAM flaps for breast reconstruction. Plast. Reconstr. Surg. 2000, 105, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Tønseth, K.A.; Hokland, B.M.; Tindholdt, T.T.; Abyholm, F.E.; Stavem, K. Quality of life, patient satisfaction and cosmetic outcome after breast reconstruction using DIEP flap or expandable breast implant. J. Plast. Reconstr. Aesthet. Surg. 2008, 61, 1188–1194. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.I.; Hamdy, H.; El-Mrakby, H.H.; Milner, R.H. Vascular anatomy of the lower anterior abdominal wall: A microdissection study on the deep inferior epigastric vessels and the perforator branches. Plast. Reconstr. Surg. 2002, 109, 544. [Google Scholar]
- Rozen, W.M.; Ashton, M.W.; Stella, D.L.; Phillips, T.J.; Taylor, G.I. The accuracy of computed tomographic angiography for mapping the perforators of the DIEA: A cadaveric study. Plast. Reconstr. Surg. 2008, 122, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Gacto-Sánchez, P.; Sicilia-Castro, D.; Gómez-Cía, T.; Lagares, A.; Collell, T.; Suárez, C.; Parra, C.; Infante-Cossío, P.; De La Higuera, J.M. Use of a Three-Dimensional Virtual Reality Model for Preoperative Imaging in DIEP Flap Breast Reconstruction. J. Surg. Res. 2010, 162, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Giunta, R.E.; Geisweid, A.; Feller, A.M. The value of preoperative doppler sonography for planning free perforator flaps. Plast. Reconstr. Surg. 2000, 105, 2381–2386. [Google Scholar] [CrossRef] [PubMed]
- Cina, A.; Barone-Adesi, L.; Rinaldi, P.; Cipriani, A.; Salgarello, M.; Masetti, R.; Bonomo, L. Planning deep inferior epigastric perforator flaps for breast reconstruction: A comparison between multidetector computed tomography and magnetic resonance angiography. Eur. Radiol. 2013, 23, 2333–2343. [Google Scholar] [CrossRef] [PubMed]
- Casey, W.J., III; Chew, R.T.; Rebecca, A.M.; Smith, A.A.; Collins, J.M.; Pockaj, B.A. Advantages of preoperative computed tomography in deep inferior epigastric artery perforator flap breast reconstruction. Plast. Reconstr. Surg. 2009, 123, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- McMillan, K.; Bostani, M.; Cagnon, C.H.; Yu, L.; Leng, S.; McCollough, C.H.; McNitt-Gray, M.F. Estimating patient dose from CT exams that use automatic exposure control: Development and validation of methods to accurately estimate tube current values. Med. Phys. 2017, 44, 4262–4275. [Google Scholar] [CrossRef] [PubMed]
- Schaverien, M.; Ludman, C.; Neil-Dwyer, J.; Perks, G.B.; Akhtar, N.; Rodrigues, J.N.; Benetatos, K.; Raurell, A.; Rasheed, T.; McCulley, S.J.; et al. Contrast-Enhanced magnetic resonance angiography for preoperative imaging in DIEP flap breast reconstruction. Plast. Reconstr. Surg. 2011, 128, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Rozen, W.M.; Stella, D.L.; Bowden, J.; Taylor, G.I.; Ashton, M.W. Advances in the pre-operative planning of deep inferior epigastric artery perforator flaps: Magnetic resonance angiography. Microsurgery 2009, 29, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Klasson, S.; Svensson, H.; Malm, K.; Wassélius, J.; Velander, P. Preoperative CT angiography versus Doppler ultrasound mapping of abdominal perforator in DIEP breast reconstructions: A randomized prospective study. J. Plast. Reconstr. Aesthet. Surg. 2015, 68, 782–786. [Google Scholar] [CrossRef] [PubMed]
| Items | % | CI |
|---|---|---|
| 3 largest perforators | 95% | 0.80–0.98 |
| Dominant perforator | 100% | 0.89–100 |
| Perforator intramuscular course | 93% | 0.71–0.94 |
| Superficial venous communications | 90.6% | 0.79–0.98 |
| DIEA branching type | 85% | 0.69–0.93 |
| SIEA calibre | 90% | 0.84–0.99 |
| Integrity of SIEV | 96% | 0.84–0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Angelo, A.; Cina, A.; Macrì, G.; Belli, P.; Mercogliano, S.; Barbieri, P.; Grippo, C.; Franceschini, G.; D’Archi, S.; Mason, E.J.; et al. Conventional CT versus Dedicated CT Angiography in DIEP Flap Planning: A Feasibility Study. J. Pers. Med. 2021, 11, 277. https://doi.org/10.3390/jpm11040277
D’Angelo A, Cina A, Macrì G, Belli P, Mercogliano S, Barbieri P, Grippo C, Franceschini G, D’Archi S, Mason EJ, et al. Conventional CT versus Dedicated CT Angiography in DIEP Flap Planning: A Feasibility Study. Journal of Personalized Medicine. 2021; 11(4):277. https://doi.org/10.3390/jpm11040277
Chicago/Turabian StyleD’Angelo, Anna, Alessandro Cina, Giulia Macrì, Paolo Belli, Sara Mercogliano, Pierluigi Barbieri, Cristina Grippo, Gianluca Franceschini, Sabatino D’Archi, Elena Jane Mason, and et al. 2021. "Conventional CT versus Dedicated CT Angiography in DIEP Flap Planning: A Feasibility Study" Journal of Personalized Medicine 11, no. 4: 277. https://doi.org/10.3390/jpm11040277
APA StyleD’Angelo, A., Cina, A., Macrì, G., Belli, P., Mercogliano, S., Barbieri, P., Grippo, C., Franceschini, G., D’Archi, S., Mason, E. J., Visconti, G., Adesi, L. B., Salgarello, M., & Manfredi, R. (2021). Conventional CT versus Dedicated CT Angiography in DIEP Flap Planning: A Feasibility Study. Journal of Personalized Medicine, 11(4), 277. https://doi.org/10.3390/jpm11040277









