Current State of “Omics” Biomarkers in Pancreatic Cancer
Abstract
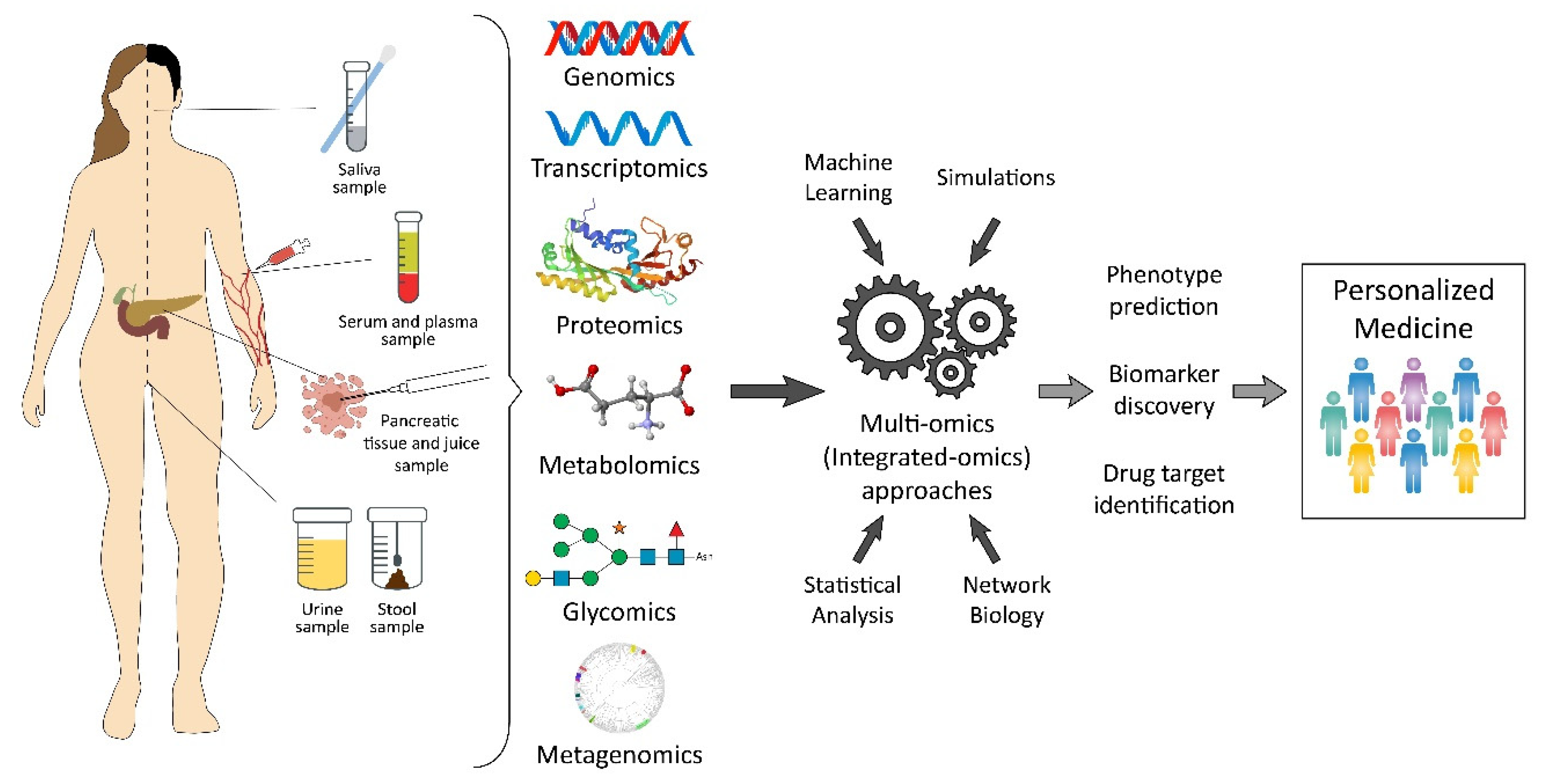
1. Introduction
2. Recent Insights from Different Omics Levels
3. Genomic Signatures
4. Coding and Noncoding RNA Signatures of Pancreatic Cancer
5. Proteomic Signatures of Pancreatic Cancer
6. Metabolomic Signature of Pancreatic Cancer
7. Glycomic Signatures of Pancreatic Cancer
8. Metagenomic Biomarkers of Pancreatic Cancer
9. Biomarkers Leading to Improved Personalized Medicine
10. Conclusions
| “Omic” Level Description | Sample Origin | Altered Molecule/Microorganism | Expression Pattern | Detection Method * | Reference Study | |
|---|---|---|---|---|---|---|
| Genomics | Mutation | Pancreatic tissue | CDKN2A, CDKN2B, TP53, SMAD4, KRAS | - | WES/WGS | [28] |
| Transcriptomics | Coding RNAs | T cell | EGLN3, PLAU | Downregulated | scRNA-seq | [72] |
| T cell | MMP9 | Dysregulated | scRNA-seq | [72] | ||
| Mouse pancreatic tissue | ONECUT2, FOXQ1, MARCKSL1, MMP7, IGFBP7 | Upregulated | scRNA-seq | [164] | ||
| Tumor tissue | hsa_circ_100782 | Upregulated | Microarray/qRT-PCR | [71] | ||
| circRNAs | Tumor tissue/plasma/cell lines | hsa_circ_0006988 | Upregulated | qRT-PCR | [165] | |
| Tumor tissue/cell lines | hsa_circ_0099999 (circZMYM2) | Upregulated | circRNA overexpression | [166] | ||
| Tumor tissue | hsa_circ_0006215 | Upregulated | circRNA overexpression | [167] | ||
| Tumor tissue, plasma exosome | circ-IARS | Upregulated | circRNA overexpression | [168] | ||
| Tumor tissue | circ-PDE8A | Upregulated | circRNA overexpression | [169] | ||
| Tumor tissue/cell | hsa_circ_0001649 | Downregulated | Microarray/qRT-PCR | [170] | ||
| Tumor tissue/cell | hsa_circ_0005397 (circ-RHOT1) | Upregulated | Microarray/qRT-PCR | [171] | ||
| Tumor tissue/cell lines | hsa_circ_0030235 | Upregulated | circRNA overexpression | [172] | ||
| Tumor tissue/cell lines | hsa_circ_0007534 | Upregulated | circRNA overexpression | [173] | ||
| Tumor tissue/cell lines | ciRS-7 (Cdr1as) | Upregulated | qRT-PCR | [174] | ||
| Tumor tissue | hsa_circ_0007334 | Upregulated | Microarray/qRT-PCR | [175] | ||
| Tumor tissue | circLDLRAD3 | Upregulated | circRNA knockdown | [176] | ||
| Tumor tissue/cell | circASH2L | Upregulated | Microarray/qRT-PCR | [177] | ||
| Tumor tissue/cell lines | circADAM9 | Upregulated | circRNA knockdown | [178] | ||
| Tumor tissue/cell | hsa_circ_001653 | Upregulated | circRNA knockdown | [179] | ||
| Tumor tissue/cell | circHIPK3 | Upregulated | circRNA knockdown | [180] | ||
| Tumor tissue/cell | circFOXK2 | Upregulated | circRNA knockdown | [181] | ||
| Tumor tissue | hsa_circ_0009065 (circBFAR) | Upregulated | circRNA overexpression | [70] | ||
| Tumor tissue | hsa_circ_0086375 (circNFIB1) | Downregulated | circRNA knockdown | [182] | ||
| Tumor tissue/cell | hsa_circ_0013912 | Upregulated | circRNA overexpression | [183] | ||
| Tumor tissue/cell lines | hsa_circ_001587 | Downregulated | circRNA knockdown | [184] | ||
| Tumor tissue | hsa_circ_0001946, hsa_circ_0005397 | Upregulated | Microarray/qRT-PCR | [67] | ||
| Tumor tissue | hsa_circ_0005785, hsa_circ_0006913, hsa_circ_0000257, hsa_circ_0041150, hsa_circ_0008719 | Downregulated | Microarray/qRT-PCR | [67] | ||
| Plasma | miR-21 | Upregulated | Microarray/qRT-PCR | [49] | ||
| Pancreatic juice | miR-155 | Upregulated | qRT-PCR | [49] | ||
| miRNAs | Tumor tissue/cell lines | miR-196a | Upregulated | Microarray/qRT-PCR | [185] | |
| Tumor tissue | miR-210 | Upregulated | qRT-PCR | [186] | ||
| Tumor tissue/cell line/serum | miR-1290 | Upregulated | Microarray/qRT-PCR | [50] | ||
| Tumor tissue/cell lines | miR-200a/miR-200b | Upregulated | Microarray/qRT-PCR | [51] | ||
| Tumor tissue/plasma/serum | miR-18a | Upregulated | qRT-PCR | [55] | ||
| Tumor tissue | miR-192 | Upregulated | Microarray/qRT-PCR | [187] | ||
| Blood | miR-22-3p/miR-642b/miR-885-5p | Upregulated | qRT-PCR | [188] | ||
| Tumor tissue | miR-23a/miR-31/miR-100/miR-143/miR-221 | Upregulated | qRT-PCR | [43] | ||
| Tumor tissue | miR-148a/miR-375/miR-217 | Downregulated | qRT-PCR | [43] | ||
| Plasma | miR-16 and miR-16 and miR-196a and CA 19-9 combination | Upregulated | qRT-PCR | [56] | ||
| Peripheral Blood Mononuclear Cells | miR-27a-3p with CA 19-9 | Upregulated | RNA-seq/qRT-PCR | [57] | ||
| Tumor tissue/cell lines | miR-221/miR-222 | Upregulated | qRT-PCR | [185] | ||
| Tumor tissue/plasma | miR-744 | Upregulated | Microarray/qRT-PCR | [62] | ||
| Tumor tissue | miR-218 | Downregulated | Microarray/qRT-PCR | [189] | ||
| Tumor tissue | miR-494 | Downregulated | Microarray/qRT-PCR | [46] | ||
| Tumor tissue | HOTAIR | Upregulated | qRT-PCR | [35] | ||
| Tumor tissue | PVT1 | Upregulated | qRT-PCR | [190] | ||
| Other ncRNAs | Tumor tissue | MALAT-1 | Upregulated | qRT-PCR | [191] | |
| Tumor tissue | Gas5 | Upregulated | qRT-PCR | [192] | ||
| Tumor tissue | MEG3 | Upregulated | qRT-PCR | [193] | ||
| Tumor tissue | HULC | Upregulated | qRT-PCR | [194] | ||
| Tumor tissue | BC008363 | Upregulated | Microarray/qRT-PCR | [195] | ||
| Tumor tissue | HSATII | Upregulated | RNA-seq | [196] | ||
| Serum/plasma | U2snRNA | Upregulated | Microarray/qRT-PCR | [197] | ||
| Pancreatic juice and cell line | REG1B/SYCN | Upregulated | ELISA | [84] | ||
| Serum | C4BPA | Upregulated | TMT labeling | [92] | ||
| Proteomics | Proteins | Plasma | IGFBP2/IGFBP3 | Upregulated | Antibody-based and LC-MS/MS-based | [93] |
| Serum | DTNBP1 | Upregulated | MS | [94] | ||
| Plasma | ctDNA with CA19-9, CEA, HGF, and osteopontin | Upregulated | Luminex bead-based immunoassays | [95] | ||
| Plasma | Combination of CA19-9, TFPI, and TNC- FNIII-B | Upregulated | ELISA | [96] | ||
| Plasma | Combination of TIMP1, LRG1, and CA19-9 | Upregulated | ELISA | [97] | ||
| Plasma | THBS-2 and CA19-9 | Upregulated | ELISA | [98] | ||
| Serum | Survivin | Upregulated | ELISA | [99] | ||
| Pancreatic ductal fluid | Mucins and S100A8 or S100A9 | Upregulated | MS | [100] | ||
| Tumor tissue | FLT3, PCBP3 | Upregulated | HDMS | [101] | ||
| Tumor tissue | Combination of hENT1 and lactic acid | GC/TOF-MS | [116] | |||
| Tumor tissue | Glucose, ascorbate, ethanolamine, and taurine | Upregulated | HRMAS-NMR | [117] | ||
| Tumor tissue | Choline, ethanolamine, glycerophosphocholine, phenylalanine, tyrosine, aspartate, threonine, succinate, glycerol, lactate, glycine, glutamate, glutamine, and creatine | Downregulated | HRMAS-NMR | [117] | ||
| Metabolomics | Metabolites | Rat tumor tissue | Kynurenate and methionine | Downregulated | NMR | [116] |
| Tumor tissue | N-glycosylation of MUC5AC, CEACAM5, IGFBP3, and LGALS3BP | Upregulated | HPLC, MS | [133] | ||
| Serum | α-linked mannose and glycan involved the Thomsen–Friedenreich antigen, fucose, and Lewis antigens affected MUC1 and MUC5AC | Upregulated | Microarray, WB | [130] | ||
| Serum | Asn-88 N-glycosylation and differential RNase-1 expression | Upregulated | ELISA, WB | [131] | ||
| Glycomics | Glycan alterations | Serum | α1-3 fucosylation in α-1-acid glycoprotein | Upregulated | ELLA, HILIC-MS, CZE | [134] |
| Serum | CA19-9 | Downregulated | Immunoassay | [139] | ||
| Tumor biopsy | CD44 antigen (CD44) | Upregulated | WB | [138] | ||
| Plasma | Glypican-1 (GPC1) | Upregulated | Flow cytometry | [137] | ||
| Glycoproteins | Serum | Mucin-5AC, MUC1, and MUC16 | Upregulated | Antibody-lectin sandwich array | [130] | |
| Metagenomics | Microbiota | Oral microbiota | Porphyromonas gingivali, Fusobacterium, Neisseria elongata, and Streptococcus mitis | High amount | plasma antibody analysis, 16S rRNA sequencing | [145] |
| Murine fecal microbiota | Proteobacteria, Actinobacteria, Fusobacteria, and Verrucomicrobia | High amount | qPCR, FISH, 16S rRNA gene sequencing | [143] | ||
| Murine gut microbiota | Proteobacteria, Bacteroidetes, and Firmicutes | High amount | qPCR, FISH, 16S rRNA gene sequencing | [143] | ||
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Sanagapalli, S.; Stoita, A. Challenges in diagnosis of pancreatic cancer. World J. Gastroenterol. 2018, 24, 2047–2060. [Google Scholar] [CrossRef]
- Maeda, S.; Unno, M.; Yu, J. Adjuvant and Neoadjuvant Therapy for Pancreatic Cancer. J. Pancreatol. 2019, 2, 100–106. [Google Scholar] [CrossRef]
- Srivastava, S.; Koay, E.J.; Borowsky, A.D.; De Marzo, A.M.; Ghosh, S.; Wagner, P.D.; Kramer, B.S. Cancer overdiagnosis: A biological challenge and clinical dilemma. Nat. Rev. Cancer 2019, 19, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Jin, G.; Wang, C.; Miao, Y.; Wang, H. The current surgical treatment of pancreatic cancer in China: A national wide cross-sectional study. J. Pancreatol. 2019, 2, 1. [Google Scholar] [CrossRef]
- Zhang, Q.; Zeng, L.; Chen, Y.; Lian, G.; Qian, C.; Chen, S.; Li, J.; Huang, K. Pancreatic Cancer Epidemiology, Detection, and Management. Gastroenterol. Res. Pract. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Wolfgang, C.L.; Herman, J.M.; Laheru, D.A.; Al, E. Recent progress in pancreatic cancer. CA Cancer J. Clin. 2013, 63, 318–348. [Google Scholar] [CrossRef] [PubMed]
- Goral, V. Pancreatic cancer: Pathogenesis and diagnosis. Asian Pac. J. Cancer Prev. 2015, 16, 5619–5624. [Google Scholar] [CrossRef]
- Klimstra, D.S.; Pitman, M.B.; Hruban, R.H. An algorithmic approach to the diagnosis of pancreatic neoplasms. Arch. Pathol. Lab. Med. 2009, 133, 454–464. [Google Scholar] [CrossRef]
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J. Gastroenterol. 2018, 24, 4846–4861. [Google Scholar] [CrossRef] [PubMed]
- Decker, G.A.; Batheja, M.J.; Collins, J.M.; Silva, A.C.; Mekeel, K.L.; Moss, A.A.; Nguyen, C.C.; Lake, D.F.; Miller, L.J. Risk factors for pancreatic adenocarcinoma and prospects for screening. Gastroenterol. Hepatol. 2010, 6, 246–254. [Google Scholar]
- Becker, A.E.; Hernandez, Y.G.; Frucht, H.; Lucas, A.L. Pancreatic ductal adenocarcinoma: Risk factors, screening, and early detection. World J. Gastroenterol. 2014. [Google Scholar] [CrossRef]
- Costache, M.I.; Costache, C.A.; Dumitrescu, C.I.; Tica, A.A.; Popescu, M.; Baluta, E.A.; Anghel, A.C.; Saftoiu, A.; Dumitrescu, D. Which is the Best Imaging Method in Pancreatic Adenocarcinoma Diagnosis and Staging—CT, MRI or EUS? Curr. Health Sci. J. 2017, 43, 132–136. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, S.; Zhang, B.; Ni, Q.; Yu, X.; Xu, J. Circulating biomarkers for early diagnosis of pancreatic cancer: Facts and hopes. Am. J. Cancer Res. 2018, 8, 332–353. [Google Scholar]
- Tjensvoll, K.; Nordgård, O.; Smaaland, R. Circulating tumor cells in pancreatic cancer patients: Methods of detection and clinical implications. Int. J. Cancer 2014, 134, 1–8. [Google Scholar] [CrossRef]
- Martini, V.; Timme-Bronsert, S.; Fichtner-Feigl, S.; Hoeppner, J.; Kulemann, B. Circulating tumor cells in pancreatic cancer: Current perspectives. Cancers 2019, 11, 1659. [Google Scholar] [CrossRef] [PubMed]
- Amantini, C.; Morelli, M.B.; Nabissi, M.; Piva, F.; Marinelli, O.; Maggi, F.; Bianchi, F.; Bittoni, A.; Berardi, R.; Giampieri, R.; et al. Expression Profiling of Circulating Tumor Cells in Pancreatic Ductal Adenocarcinoma Patients: Biomarkers Predicting Overall Survival. Front. Oncol. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Bhat, K.; Wang, F.; Ma, Q.; Li, Q.; Mallik, S.; Hsieh, T.; Wu, E. Advances in Biomarker Research for Pancreatic Cancer. Curr. Pharm. Des. 2012, 18, 2439–2451. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.H.; Wang, W.Q.; Han, X.; Gao, H.L.; Li, T.J.; Xu, S.S.; Li, S.; Xu, H.X.; Li, H.; Ye, L.Y.; et al. Advances on diagnostic biomarkers of pancreatic ductal adenocarcinoma: A systems biology perspective. Comput. Struct. Biotechnol. J. 2020, 18, 3606–3614. [Google Scholar] [CrossRef]
- Young, M.R.; Wagner, P.D.; Grosh, S.; Al, E. Validation of Biomarkers for Early Detection of Pancreatic Cancer: Summary of the Alliance of Pancreatic Cancer Consortia for Biomarkers for Early Detection Workshop. Pancreas 2019, 47, 135–141. [Google Scholar] [CrossRef]
- Adamska, A.; Domenichini, A.; Falasca, M. Pancreatic ductal adenocarcinoma: Current and evolving therapies. Int. J. Mol. Sci. 2017, 18, 1338. [Google Scholar] [CrossRef]
- Tian, Q.; Price, N.D.; Hood, L. Systems Cancer Medicine: Towards Realization of Predictive, Preventive, Personalized, and Participatory (P4) Medicine. J. Intern. Med. 2012, 271, 111–121. [Google Scholar] [CrossRef]
- Westerhoff, H.V.; Palsson, B.O. The evolution of molecular biology into systems biology. Nat. Biotechnol. 2004, 22, 1249–1252. [Google Scholar] [CrossRef]
- Sousa, C.M.; Kimmelman, A.C. The complex landscape of pancreatic cancer metabolism. Carcinogenesis 2014, 35, 1441–1450. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Qin, Y.; Zhang, B.; Ji, S.; Shi, S.; Xu, W.; Liu, J.; Xiang, J.; Liang, D.; Hu, Q.; et al. Metabolic plasticity in heterogeneous pancreatic ductal adenocarcinoma. Bioch. Biophys. Acta Rev. Cancer 2016, 1866, 177–188. [Google Scholar] [CrossRef]
- Löhr, M.; Klöppel, G.; Maisonneuve, P.; Lowenfels, A.B.; Lüttges, J. Frequency of K-ras mutations in pancreatic intraductal neoplasias associated with pancreatic ductal adenocarcinoma and chronic pancreatitis: A meta-analysis. Neoplasia 2005, 7, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Bardeesy, N.; DePinho, R.A. Pancreatic cancer biology and genetics. Nat. Rev. Cancer 2002, 2, 897–909. [Google Scholar] [CrossRef] [PubMed]
- Perera, R.M.; Bardeesy, N. Pancreatic Cancer Metabolism—Breaking it down to build it back up. Cancer Discov. 2015, 5, 1247–1261. [Google Scholar] [CrossRef] [PubMed]
- Son, J.; Lyssiotis, C.A.; Ying, H.; Wang, X.; Hua, S.; Ligorio, M.; Perera, R.M.; Ferrone, C.R.; Mullarky, E.; Shyh-, N.; et al. Glutamine supports pancreatic cancer growth through a Kras- regulated metabolic pathway. Nature 2013, 496, 101–105. [Google Scholar] [CrossRef]
- Ying, H.; Kimmelman, A.C.; Lyssiotis, C.A.; Hua, S.; Chu, G.C.; Fletcher-sananikone, E.; Locasale, J.W.; Son, J.; Zhang, H.; Coloff, J.L.; et al. Oncogenic Kras Maintains Pancreatic Tumors through Regulation of Anabolic Glucose Metabolism. Cell 2012, 149, 656–670. [Google Scholar] [CrossRef]
- Le, A.; Cooper, C.R.; Gouw, A.M.; Dinavahi, R.; Maitra, A.; Deck, L.M.; Royer, R.E.; Vander Jagt, D.L.; Semenza, G.L.; Dang, C.V. Inhibition of lactate dehydrogenase A induces oxidative stress and inhibits tumor progression. Proc. Natl. Acad. Sci. USA 2010, 107, 2037–2042. [Google Scholar] [CrossRef]
- Alian, O.; Philip, P.; Sarkar, F.; Azmi, A. Systems Biology Approaches to Pancreatic Cancer Detection, Prevention and Treatment. Curr. Pharm. Des. 2014. [Google Scholar] [CrossRef]
- Ballehaninna, U.K.; Chamberlain, R.S. Biomarkers for pancreatic cancer: Promising new markers and options beyond CA 19-9. Tumor Biol. 2013, 34, 3279–3292. [Google Scholar] [CrossRef]
- Kishikawa, T.; Otsuka, M.; Ohno, M.; Yoshikawa, T.; Takata, A.; Koike, K. Circulating RNAs as new biomarkers for detecting pancreatic cancer. World J. Gastroenterol. 2015, 21, 8527–8540. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Jacob, R.; Manne, U.; Paluri, R. Advances in pancreatic cancer biomarkers. Oncol. Rev. 2019, 13, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Pan, X.; Cobb, G.P.; Anderson, T.A. microRNAs as oncogenes and tumor suppressors. Dev. Biol. 2007, 302, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Volinia, S.; Calin, G.A.; Liu, C.-G.; Al, E. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA 2006, 103, 2257–2261. [Google Scholar] [CrossRef]
- Li, Y.; Sarkar, F.H. MicroRNA targeted therapeutic approach for pancreatic cancer. Int. J. Biol. Sci. 2016, 12, 326–337. [Google Scholar] [CrossRef]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A.; et al. MicroRNA expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar] [CrossRef]
- Rosenfeld, N.; Aharonov, R.; Meiri, E.; Rosenwald, S.; Spector, Y.; Zepeniuk, M.; Benjamin, H.; Shabes, N.; Tabak, S.; Levy, A.; et al. MicroRNAs accurately identify cancer tissue origin. Nat. Biotechnol. 2008, 26, 462–469. [Google Scholar] [CrossRef]
- Hernandez, Y.G. MicroRNA in pancreatic ductal adenocarcinoma and its precursor lesions. World J. Gastrointest. Oncol. 2016, 8, 18. [Google Scholar] [CrossRef]
- Frampton, A.E.; Giovannetti, E.; Jamieson, N.B.; Krell, J.; Gall, T.M.; Stebbing, J.; Jiao, L.R.; Castellano, L. A microRNA meta-signature for pancreatic ductal adenocarcinoma. Expert Rev. Mol. Diagn. 2014, 14, 267–271. [Google Scholar] [CrossRef]
- Bloomston, M.; Frankel, W.L.; Petrocca, F.; Volinia, S.; Alder, H.; Hagan, J.P.; Liu, C.G.; Bhatt, D.; Taccioli, C.; Croce, C.M. MicroRNA expression patterns to differentiate pancreatic adenocarcinoma from normal pancreas and chronic pancreatitis. J. Am. Med. Assoc. 2007, 297, 1901–1908. [Google Scholar] [CrossRef]
- Caponi, S.; Funel, N.; Frampton, A.E.; Mosca, F.; Santarpia, L.; Van der Velde, A.G.; Jiao, L.R.; De Lio, N.; Falcone, A.; Kazemier, G.; et al. The good, the bad and the ugly: A tale of miR-101, miR-21 and miR-155 in pancreatic intraductal papillary mucinous neoplasms. Ann. Oncol. 2013, 24, 734–741. [Google Scholar] [CrossRef]
- Szafranska, A.E.; Davison, T.S.; John, J.; Cannon, T.; Sipos, B.; Maghnouj, A.; Labourier, E.; Hahn, S.A. MicroRNA expression alterations are linked to tumorigenesis and non-neoplastic processes in pancreatic ductal adenocarcinoma. Oncogene 2007, 26, 4442–4452. [Google Scholar] [CrossRef]
- Ho, A.S.; Huang, X.; Cao, H.; Christman-Skieller, C.; Bennewith, K.; Le, Q.T.; Koong, A.C. Circulating miR-210 as a novel hypoxia marker in pancreatic cancer. Transl. Oncol. 2010, 3, 109–113. [Google Scholar] [CrossRef]
- Habbe, N.; Koorstra, J.M.; Mendell, J.T.; Offerhaus, G.J.; Kon, J.; Feldmann, G.; Mullendore, M.E.; Goggins, M.G. MicroRNA miR-155 is a biomarker of early pancreatic neoplasia. Cancer Biol. Ther. 2010, 8, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Daoud, A.Z.; Mulholland, E.J.; Cole, G.; McCarthy, H.O. MicroRNAs in Pancreatic Cancer: Biomarkers, prognostic, and therapeutic modulators. BMC Cancer 2019, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Yu, J.; Kim, H.; Wolfgang, C.L.; Al, E. MicroRNA array analysis finds elevated serum miR-1290 accurately distinguishes patients with low-stage pancreatic cancer from healthy and disease controls. Clin. Cancer Res. 2014, 19, 3600–3610. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Omura, N.; Hong, S.M.; Vincent, A.; Walter, K.; Griffith, M.; Borges, M.; Goggins, M. Pancreatic cancers epigenetically silence SIP1 and hypomethylate and overexpress miR-200a/200b in association with elevated circulating miR-200a and miR-200b levels. Cancer Res. 2010, 70, 5226–5237. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Du, Y.; Gao, J.; Liu, J.; Kong, X.; Gong, Y.; Li, Z.; Wu, H.; Chen, H. Aberrant expression mir-196a is associated with abnormal apoptosis, invasion, and proliferation of pancreatic cancer cells. Pancreas 2013, 42, 1169–1181. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, A.L.; Joergensen, M.T.; Knudsen, S.; De Muckadell, O.B.S.; Heegaard, N.H.H. Cell-free plasma microRNA in pancreatic ductal adenocarcinoma and disease controls. Pancreas 2013, 42, 1107–1113. [Google Scholar] [CrossRef]
- Ganepola, G.A. Novel blood-based microRNA biomarker panel for early diagnosis of pancreatic cancer. World J. Gastrointest. Oncol. 2014, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Morimura, R.; Komatsu, S.; Ichikawa, D.; Takeshita, H.; Tsujiura, M.; Nagata, H.; Konishi, H.; Shiozaki, A.; Ikoma, H.; Okamoto, K.; et al. Novel diagnostic value of circulating miR-18a in plasma of patients with pancreatic cancer. Br. J. Cancer 2011, 105, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gao, J.; Du, Y.; Li, Z.; Ren, Y.; Gu, J.; Wang, X.; Gong, Y.; Wang, W.; Kong, X. Combination of plasma microRNAs with serum CA19-9 for early detection of pancreatic cancer. Int. J. Cancer 2012, 131, 683–691. [Google Scholar] [CrossRef]
- Wang, W.S.; Liu, L.X.; Li, G.P.; Chen, Y.; Li, C.Y.; Jin, D.Y.; Wang, X.L. Combined serum CA19-9 and miR-27a-3p in peripheral blood mononuclear cells to diagnose pancreatic cancer. Cancer Prev. Res. 2013, 6, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Halkova, T.; Cuperkova, R.; Minarik, M.; Benesova, L. MicroRNAs in pancreatic cancer: Involvement in carcinogenesis and potential use for diagnosis and prognosis. Gastroenterol. Res. Pract. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Li, B.S.; Liu, H.; Yang, W.L. Reduced miRNA-218 expression in pancreatic cancer patients as a predictor of poor prognosis. Genet. Mol. Res. 2015, 14, 16372–16378. [Google Scholar] [CrossRef]
- Ma, Y.B.; Li, G.X.; Hu, J.X.; Liu, X.; Shi, B.M. Correlation of miR-494 expression with tumor progression and patient survival in pancreatic cancer. Genet. Mol. Res. 2015, 14, 18153–18159. [Google Scholar] [CrossRef]
- Passadouro, M.; Pedroso de Lima, M.C.; Faneca, H. MicroRNA modulation combined with sunitinib as a novel therapeutic strategy for pancreatic cancer. Int. J. Nanomed. 2014, 9, 3203–3217. [Google Scholar] [CrossRef]
- Miyamae, M.; Komatsu, S.; Ichikawa, D.; Kawaguchi, T.; Hirajima, S.; Okajima, W.; Ohashi, T.; Imamura, T.; Konishi, H.; Shiozaki, A.; et al. Plasma microRNA profiles: Identification of miR-744 as a novel diagnostic and prognostic biomarker in pancreatic cancer. Br. J. Cancer 2015, 113, 1467–1476. [Google Scholar] [CrossRef]
- Kung, J.T.Y.; Colognori, D.; Lee, J.T. Long noncoding RNAs: Past, present, and future. Genetics 2013, 193, 651–669. [Google Scholar] [CrossRef] [PubMed]
- Baraniskin, A.; Nöpel-Dünnebacke, S.; Ahrens, M.; Jensen, S.G.; Zöllner, H.; Maghnouj, A.; Wos, A.; Mayerle, J.; Munding, J.; Kost, D.; et al. Circulating U2 small nuclear RNA fragments as a novel diagnostic biomarker for pancreatic and colorectal adenocarcinoma. Int. J. Cancer 2013, 132, 1–10. [Google Scholar] [CrossRef]
- Li, J.; Yang, J.; Zhou, P.; Le, Y.; Zhou, C.; Wang, S.; Xu, D.; Lin, H.-K.; Gong, Z. Circular RNAs in cancer: Novel insights into origins, properties, functions and implications. Am. J. Cancer Res. 2015, 5, 472–480. [Google Scholar] [PubMed]
- Liu, J.; Xu, D.; Wang, Q.; Zheng, D.; Jiang, X.; Xu, L. LPS induced miR-181a promotes pancreatic cancer cell migration via targeting PTEN and MAP2K4. Dig. Dis. Sci. 2014. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hao, X.; Wang, H.; Liu, Z.; He, Y.; Pu, M.; Zhang, H.; Yu, H.; Duan, J.; Qu, S. Circular RNA Expression Profile of Pancreatic Ductal Adenocarcinoma Revealed by Microarray. Cell. Physiol. Biochem. 2016, 40, 1334–1344. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, J.Y.; Zhou, S.Y.; Yang, S.J.; Zhong, S.L. Circular RNA expression in pancreatic ductal adenocarcinoma. Oncol. Lett. 2019, 18, 2923–2930. [Google Scholar] [CrossRef]
- Seimiya, T.; Otsuka, M.; Iwata, T.; Tanaka, E.; Sekiba, K.; Shibata, C.; Moriyama, M.; Nakagawa, R.; Maruyama, R.; Koike, K. Aberrant expression of a novel circular RNA in pancreatic cancer. J. Human Genet. 2020. [Google Scholar] [CrossRef]
- Guo, X.; Zhou, Q.; Su, D.; Luo, Y.; Fu, Z.; Huang, L.; Li, Z.; Jiang, D.; Kong, Y.; Li, Z.; et al. Circular RNA circBFAR promotes the progression of pancreatic ductal adenocarcinoma via the miR-34b-5p/MET/Akt axis. Mol. Cancer 2020, 19. [Google Scholar] [CrossRef]
- Limb, C.; Liu, D.S.K.; Veno, M.T.; Rees, E.; Krell, J.; Bagwan, I.N.; Giovannetti, E.; Pandha, H.; Strobel, O.; Rockall, T.A.; et al. The role of circular rnas in pancreatic ductal adenocarcinoma and biliary-tract cancers. Cancers 2020, 12, 3250. [Google Scholar] [CrossRef]
- Peng, J.; Sun, B.F.; Chen, C.Y.; Zhou, J.Y.; Chen, Y.S.; Chen, H.; Liu, L.; Huang, D.; Jiang, J.; Cui, G.S.; et al. Single-cell RNA-seq highlights intra-tumoral heterogeneity and malignant progression in pancreatic ductal adenocarcinoma. Cell Res. 2019, 29, 725–738. [Google Scholar] [CrossRef]
- Aslam, B.; Basit, M.; Nisar, M.A.; Khurshid, M.; Rasool, M.H. Proteomics: Technologies and their applications. J. Chromatogr. Sci. 2017, 55, 182–196. [Google Scholar] [CrossRef]
- Alharbi, R.A. Proteomics approach and techniques in identification of reliable biomarkers for diseases. Saudi J. Biol. Sci. 2020, 27, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Domon, B.; Aebersold, R. Challenges and opportunities in proteomics data analysis. Mol. Cell. Proteom. 2006, 5, 1921–1926. [Google Scholar] [CrossRef]
- Iuga, C.; Seicean, A.; Iancu, C.; Buiga, R.; Sappa, P.K.; Völker, U.; Hammer, E. Proteomic identification of potential prognostic biomarkers in resectable pancreatic ductal adenocarcinoma. Proteomics 2014, 14, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Pan, S.; Brentnall, T.A.; Aebersold, R. Proteomic profiling of pancreatic cancer for biomarker discovery. Mol. Cell. Proteom. 2005, 4, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Luna, C.; Torres, C.; Ortiz, R.; Dieguez, C.; Martinez-Galan, J.; Melguizo, C.; Prados, J.C.; Caba, O. Proteomic biomarkers in body fluids associated with pancreatic cancer. Oncotarget 2018, 9, 16573–16587. [Google Scholar] [CrossRef] [PubMed]
- Sileikis, A.; Petrulionis, M.; Kurlinkus, B.; Ger, M.; Kaupinis, A.; Cicenas, J.; Valius, M.; Strupas, K. Current Role of Proteomics in Pancreatic Cancer Biomarkers Research. Curr. Proteom. 2016, 13, 68–75. [Google Scholar] [CrossRef]
- Chen, R.; Pan, S.; Yi, E.C.; Donohoe, S.; Bronner, M.P.; Potter, J.D.; Goodlett, D.R.; Aebersold, R.; Brentnall, T.A. Quantitative proteomic profiling of pancreatic cancer juice. Proteomics 2006, 6, 3871–3879. [Google Scholar] [CrossRef]
- Chen, R.; Pan, S.; Cooke, K.; Moyes, K.W.; Bronner, M.P.; Goodlett, D.R.; Aebersold, R.; Brentnall, T.A. Comparison of pancreas juice proteins from cancer versus pancreatitis using quantitative proteomic analysis. Pancreas 2007, 34, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Grønborg, M.; Bunkenborg, J.; Kristiansen, T.Z.; Jensen, O.N.; Yeo, C.J.; Hruban, R.H.; Maitra, A.; Goggins, M.G.; Pandey, A. Comprehensive proteomic analysis of human pancreatic juice. J. Proteome Res. 2004, 3, 1042–1055. [Google Scholar] [CrossRef]
- Lv, S.; Gao, J.; Al, E. Transthyretin, Identified by Proteomics, is Overabundant in Pancreatic Juice From Pancreatic Carcinoma and Originates From Pancreatic Islets. Diagn. Cytopathol. 2011, 39, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Makawita, S.; Smith, C.; Batruch, I.; Zheng, Y.; Rückert, F.; Grützmann, R.; Pilarsky, C.; Gallinger, S.; Diamandis, E.P. Integrated proteomic profiling of cell line conditioned media and pancreatic juice for the identification of pancreatic cancer biomarkers. Mol. Cell. Proteom. 2011, 10, 1–20. [Google Scholar] [CrossRef]
- Park, J.Y.; Kim, S.A.; Chung, J.W.; Bang, S.; Park, S.W.; Paik, Y.K.; Song, S.Y. Proteomic analysis of pancreatic juice for the identification of biomarkers of pancreatic cancer. J. Cancer Res. Clin. Oncol. 2011, 137, 1229–1238. [Google Scholar] [CrossRef]
- Tian, M.; Cui, Y.Z.; Song, G.H.; Zong, M.J.; Zhou, X.Y.; Chen, Y.; Han, J.X. Proteomic analysis identifies MMP-9, DJ-1 and A1BG as overexpressed proteins in pancreatic juice from pancreatic ductal adenocarcinoma patients. BMC Cancer 2008, 8, 1–11. [Google Scholar] [CrossRef]
- Kim, H.; Park, J.; Wang, J.I.; Kim, Y. Recent advances in proteomic profiling of pancreatic Ductal Adenocarcinoma and the road ahead. Expert Rev. Proteom. 2017, 14, 963–971. [Google Scholar] [CrossRef]
- Stark, A.; Donahue, T.R.; Reber, H.A.; Joe Hines, O. Pancreatic cyst disease a review. JAMA J. Am. Med. Assoc. 2016, 315, 1882–1893. [Google Scholar] [CrossRef] [PubMed]
- Eric Thomas, C.; Sexton, W.; Benson, K.; Sutphen, R.; Koomen, J. Urine collection and processing for protein biomarker discovery and quantification. Cancer Epidemiol. Biomark. Prev. 2010, 19, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Thongboonkerd, V.; Malasit, P. Renal and urinary proteomics: Current applications and challenges. Proteomics 2005, 5, 1033–1042. [Google Scholar] [CrossRef]
- Makawita, S.; Dimitromanolakis, A.; Soosaipillai, A.; Soleas, I.; Chan, A.; Gallinger, S.; Haun, R.S.; Blasutig, I.M.; Diamandis, E.P. Validation of four candidate pancreatic cancer serological biomarkers that improve the performance of CA19.9. BMC Cancer 2013, 13, 1. [Google Scholar] [CrossRef]
- Sogawa, K.; Takano, S.; Iida, F.; Satoh, M.; Tsuchida, S.; Kawashima, Y.; Yoshitomi, H.; Sanda, A.; Kodera, Y.; Takizawa, H.; et al. Identification of a novel serum biomarker for pancreatic cancer, C4b-binding protein α-chain (C4BPA) by quantitative proteomic analysis using tandem mass tags. Br. J. Cancer 2016, 115, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, T.; Ohtsuki, S.; Honda, K.; Kobayashi, M.; Iwasaki, M.; Uchida, Y.; Okusaka, T.; Nakamori, S.; Shimahara, M.; Ueno, T.; et al. Identification of IGFBP2 and IGFBP3 as compensatory biomarkers for CA19-9 in early-stage pancreatic cancer using a combination of antibody-based and LC-MS/MS-based proteomics. PLoS ONE 2016, 11, 1–23. [Google Scholar] [CrossRef]
- Guo, X.; Lv, X.; Fang, C.; Al, E. Dysbindin as a novel biomarker for pancreatic ductal adenocarcinoma identified by proteomic profiling. Int. J. Cancer 2006, 139, 1821–1829. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.D.; Javed, A.A.; Thoburn, C.; Wong, F.; Tie, J.; Gibbs, P.; Schmidt, C.M.; Yip-Schneider, M.T.; Allen, P.J.; Schattner, M.; et al. Combined circulating tumor DNA and protein biomarker-based liquid biopsy for the earlier detection of pancreatic cancers. Proc. Natl. Acad. Sci. USA 2017, 114, 10202–10207. [Google Scholar] [CrossRef]
- Balasenthil, S.; Huang, Y.; Liu, S.; Marsh, T.; Chen, J.; Stass, S.A.; KuKuruga, D.; Brand, R.; Chen, N.; Frazier, M.L.; et al. A Plasma Biomarker Panel to Identify Surgically Resectable Early-Stage Pancreatic Cancer. J. Natl. Cancer Inst. 2017, 109, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Capello, M.; Bantis, L.E.; Scelo, G.; Zhao, Y.; Li, P.; Dhillon, D.S.; Patel, N.J.; Kundnani, D.L.; Wang, H.; Abbruzzese, J.L.; et al. Sequential validation of blood-based protein biomarker candidates for early-stage pancreatic cancer. J. Natl. Cancer Inst. 2017, 109, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Bamlet, W.R.; Oberg, A.L.; Chaffee, K.G.; Donahue, G.; Cao, X.J.; Chari, S.; Garcia, B.A.; Petersen, G.M.; Zaret, K.S. Detection of early pancreatic ductal adenocarcinoma with thrombospondin-2 & CA19-9 blood markers. Sci. Transl. Med. 2017, 9, 1–14. [Google Scholar] [CrossRef]
- Ren, Y.Q.; Zhang, H.Y.; Su, T.; Wang, X.H.; Zhang, L. Clinical significance of serum survivin in patients with pancreatic ductal adenocarcinoma. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3063–3068. [Google Scholar] [PubMed]
- Chen, K.; Kim, P.D.; Jones, K.A.; Al, E. Potential Prognostic Biomarkers of Pancreatic Cancer. Pancreas 2014, 43, 1–7. [Google Scholar] [CrossRef]
- Ger, M.; Kaupinis, A.; Petrulionis, M.; Kurlinkus, B.; Cicenas, J.; Sileikis, A.; Valius, M.; Strupas, K. Proteomic identification of FLT3 and PCBP3 as potential prognostic biomarkers for pancreatic cancer. Anticancer Res. 2018, 38, 5759–5765. [Google Scholar] [CrossRef]
- Pan, S.; Chen, R.; Brand, R.E.; Hawley, S.; Tamura, Y.; Gafken, P.R.; Milless, B.P.; Goodlett, D.R.; Rush, J.; Brentnall, T.A. Multiplex targeted proteomic assay for biomarker detection in plasma: A pancreatic cancer biomarker case study. J. Proteome Res. 2012, 11, 1937–1948. [Google Scholar] [CrossRef]
- Yoneyama, T.; Ohtsuki, S.; Ono, M.; Ohmine, K.; Uchida, Y.; Yamada, T.; Tachikawa, M.; Terasaki, T. Quantitative targeted absolute proteomics-based large-scale quantification of proline-hydroxylated α-fibrinogen in plasma for pancreatic cancer diagnosis. J. Proteome Res. 2013, 12, 753–762. [Google Scholar] [CrossRef]
- Ansari, D.; Aronsson, L.; Sasor, A.; Welinder, C.; Rezeli, M.; Marko-Varga, G.; Andersson, R. The role of quantitative mass spectrometry in the discovery of pancreatic cancer biomarkers for translational science. J. Transl. Med. 2014, 12, 1–15. [Google Scholar] [CrossRef]
- Bailey, P.; Chang, D.K.; Nones, K.; Johns, A.L.; Patch, A.M.; Gingras, M.C.; Miller, D.K.; Christ, A.N.; Bruxner, T.J.C.; Quinn, M.C.; et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 2016, 531, 47–52. [Google Scholar] [CrossRef]
- Hristova, V.A.; Chan, D.W. Cancer biomarker discovery and translation: Proteomics and beyond. Expert Rev. Proteom. 2019, 16, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, S.A.; Akita, H.; Takemasa, I.; Eguchi, H.; Pastural, E.; Nagano, H.; Monden, M.; Doki, Y.; Mori, M.; Jin, W.; et al. Metabolic system alterations in pancreatic cancer patient serum: Potential for early detection. BMC Cancer 2013, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Amanam, I.; Chung, V. Current and future therapies for advanced pancreatic cancer. J. Surg. Oncol. 2017, 116, 25–34. [Google Scholar] [CrossRef]
- Fontana, A.; Copetti, M.; Di Gangi, I.M.; Mazza, T.; Tavano, F.; Gioffreda, D.; Mattivi, F.; Andriulli, A.; Vrhovsek, U.; Pazienza, V. Development of a metabolites risk score for one-year mortality risk prediction in pancreatic adenocarcinoma patients. Oncotarget 2016, 7, 8968–8978. [Google Scholar] [CrossRef] [PubMed][Green Version]
- LaConti, J.J.; Laiakis, E.C.; Mays, D.D.; Peran, I.; Kim, S.E.; Shay, J.W.; Riegel, A.T.; Fornace, A.J.; Wellstein, A. Distinct serum metabolomics profiles associated with malignant progression in the KrasG12D mouse model of pancreatic ductal adenocarcinoma. BMC Genom. 2015, 16, S1. [Google Scholar] [CrossRef] [PubMed]
- Di Gangi, I.M.; Mazza, T.; Fontana, A.; Copetti, M.; Fusilli, C.; Ippolito, A.; Mattivi, F.; Latiano, A.; Andriulli, A.; Vrhovsek, U.; et al. Metabolomic profile in pancreatic cancer patients: A consensusbased approach to identify highly discriminating metabolites. Oncotarget 2016, 7, 5815–5829. [Google Scholar] [CrossRef] [PubMed]
- Tumas, J.; Baskirova, I.; Petrenas, T.; Norkuniene, J.; Strupas, K.; Sileikis, A. Towards a personalized approach in pancreatic cancer diagnostics through plasma amino acid analysis. Anticancer Res. 2019, 39, 2035–2042. [Google Scholar] [CrossRef] [PubMed]
- Moore, H.B.; Culp-Hill, R.; Reisz, J.A.; Lawson, P.J.; Sauaia, A.; Schulick, R.D.; Del Chiaro, M.; Nydam, T.L.; Moore, E.E.; Hansen, K.C.; et al. The metabolic time line of pancreatic cancer: Opportunities to improve early detection of adenocarcinoma. Am. J. Surg. 2019, 218, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Mayerle, J.; Kalthoff, H.; Reszka, R.; Kamlage, B.; Peter, E.; Schniewind, B.; González Maldonado, S.; Pilarsky, C.; Heidecke, C.D.; Schatz, P.; et al. Metabolic biomarker signature to differentiate pancreatic ductal adenocarcinoma from chronic pancreatitis. Gut 2018, 67, 128–137. [Google Scholar] [CrossRef]
- Urayama, S. Pancreatic cancer early detection: Expanding higher-risk group with clinical and metabolomics parameters. World J. Gastroenterol. 2015, 21, 1707–1717. [Google Scholar] [CrossRef] [PubMed]
- Phua, L.C.; Goh, S.; Tai, D.W.M.; Leow, W.Q.; Alkaff, S.M.F.; Chan, C.Y.; Kam, J.H.; Lim, T.K.H.; Chan, E.C.Y. Metabolomic prediction of treatment outcome in pancreatic ductal adenocarcinoma patients receiving gemcitabine. Cancer Chemother. Pharmacol. 2018, 81, 277–289. [Google Scholar] [CrossRef]
- Battini, S.; Faitot, F.; Imperiale, A.; Cicek, A.E.; Heimburger, C.; Averous, G.; Bachellier, P.; Namer, I.J. Metabolomics approaches in pancreatic adenocarcinoma: Tumor metabolism profiling predicts clinical outcome of patients. BMC Med. 2017, 15, 1–16. [Google Scholar] [CrossRef]
- Wen, S.; Li, Z.; Feng, J.; Bai, J.; Lin, X.; Huang, H. Metabonomic changes from pancreatic intraepithelial neoplasia to pancreatic ductal adenocarcinoma in tissues from rats. Cancer Sci. 2016, 107, 836–845. [Google Scholar] [CrossRef]
- Eidelman, E.; Twum-Ampofo, J.; Ansari, J.; Siddiqui, M.M. The metabolic phenotype of prostate cancer. Front. Oncol. 2017, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Hua, Q. Applications of genome-scale metabolic models in biotechnology and systems medicine. Front. Physiol. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Mardinoglu, A.; Nielsen, J. Systems medicine and metabolic modelling. J. Intern. Med. 2012, 271, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Zhang, C.; Arif, M.; Liu, Z.; Benfeitas, R.; Bidkhori, G.; Deshmukh, S.; Al Shobky, M.; Lovric, A.; Boren, J.; et al. TCSBN: A database of tissue and cancer specific biological networks. Nucleic Acids Res. 2018, 46, D595–D600. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, P.; Mardinoglu, A.; Asplund, A.; Shoaie, S.; Kampf, C.; Uhlen, M.; Nielsen, J. Identifying anti-growth factors for human cancer cell lines through genome-scale metabolic modeling. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Yizhak, K.; Chaneton, B.; Gottlieb, E.; Ruppin, E. Modeling cancer metabolism on a genome scale. Mol. Syst. Biol. 2015, 11, 817. [Google Scholar] [CrossRef]
- López-López, Á.; López-Gonzálvez, Á.; Barker-Tejeda, T.C.; Barbas, C. A review of validated biomarkers obtained through metabolomics. Expert Rev. Mol. Diagn. 2018, 18, 557–575. [Google Scholar] [CrossRef]
- Armitage, E.G.; Ciborowski, M. Applications of Metobolomics in Cancer Studies. In Metabolomics: From Fundamentals to Clinical Applications. Advances in Experimental Medicine and Biology; Springer: Cham, Switzeland, 2017; Volume 965. [Google Scholar] [CrossRef]
- Cheung, P.K.; Ma, M.H.; Tse, H.F.; Yeung, K.F.; Tsang, H.F.; Chu, M.K.M.; Kan, C.M.; Cho, W.C.S.; Ng, L.B.W.; Chan, L.W.C.; et al. The applications of metabolomics in the molecular diagnostics of cancer. Expert Rev. Mol. Diagn. 2019, 19, 785–793. [Google Scholar] [CrossRef]
- Özdemir, V.; Arga, K.Y.; Aziz, R.K.; Bayram, M.; Conley, S.N.; Dandara, C.; Endrenyi, L.; Fisher, E.; Garvey, C.K.; Hekim, N.; et al. Digging deeper into precision/personalized medicine: Cracking the sugar code, the third alphabet of life, and sociomateriality of the cell. OMICS J. Integr. Biol. 2020, 24, 62–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, W. Validation and Development of N-glycan as Biomarker in Cancer Diagnosis. Curr. Pharmacogenom. Personal. Med. 2013, 11, 53–58. [Google Scholar] [CrossRef]
- Yue, T.; Goldstein, I.J.; Hollingsworth, M.A.; Kaul, K.; Brand, R.E.; Haab, B.B. The Prevalence and Nature of Glycan Alterations on Specific Proteins in Pancreatic Cancer Patients Revealed Using Antibody-Lectin Sandwich Arrays. Mol. Cell. Proteom. 2009, 8, 1697–1707. [Google Scholar] [CrossRef]
- Nakata, D. Increased N-glycosylation of Asn88 in serum pancreatic ribonuclease 1 is a novel diagnostic marker for pancreatic cancer. Sci. Rep. 2015, 4, 6715. [Google Scholar] [CrossRef]
- Kori, M.; Aydin, B.; Gulfidan, G.; Beklen, H.; Kelesoglu, N.; İscan, A.C.; Turanli, B.; Erzik, C.; Karademir, B.; Arga, K.Y. The Repertoire of Glycan Alterations and Glycoproteins in Human Cancers. OMICS J. Integr. Biol. 2021. [Google Scholar] [CrossRef]
- Pan, S.; Chen, R.; Tamura, Y.; Crispin, D.A.; Lai, L.A.; May, D.H.; McIntosh, M.W.; Goodlett, D.R.; Brentnall, T.A. Quantitative Glycoproteomics Analysis Reveals Changes in N-Glycosylation Level Associated with Pancreatic Ductal Adenocarcinoma. J. Proteome Res. 2014, 13, 1293–1306. [Google Scholar] [CrossRef] [PubMed]
- Balmaña, M.; Giménez, E.; Puerta, A.; Llop, E.; Figueras, J.; Fort, E.; Sanz-Nebot, V.; de Bolós, C.; Rizzi, A.; Barrabés, S.; et al. Increased α1-3 fucosylation of α-1-acid glycoprotein (AGP) in pancreatic cancer. J. Proteom. 2016, 132, 144–154. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Singh, A.P.; Chakraborty, S.; Chauhan, S.C.; Bafna, S.; Meza, J.L.; Singh, P.K.; Hollingsworth, M.A.; Mehta, P.P.; Batra, S.K. MUC4 Mucin Interacts with and Stabilizes the HER2 Oncoprotein in Human Pancreatic Cancer Cells. Cancer Res. 2008, 68, 2065–2070. [Google Scholar] [CrossRef] [PubMed]
- Simeone, D.M.; Ji, B.; Banerjee, M.; Arumugam, T.; Li, D.; Anderson, M.A.; Bamberger, A.M.; Greenson, J.; Brand, R.E.; Ramachandran, V.; et al. CEACAM1, a Novel Serum Biomarker for Pancreatic Cancer. Pancreas 2007, 34, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Tan, Y.; Zhang, Y.; Yang, Y.; Li, X. Prognostic value of glypican-1 for patients with advanced pancreatic cancer following regional intra-arterial chemotherapy. Oncol. Lett. 2018. [Google Scholar] [CrossRef]
- Li, X.P.; Zhang, X.W.; Zheng, L.Z.; Guo, W.J. Expression of CD44 in pancreatic cancer and its significance. Int. J. Clin. Exp. Pathol. 2015, 8, 6724–6731. [Google Scholar]
- Aoki, S.; Motoi, F.; Murakami, Y.; Sho, M.; Satoi, S.; Honda, G.; Uemura, K.; Okada, K.; Matsumoto, I.; Nagai, M.; et al. Decreased serum carbohydrate antigen 19–9 levels after neoadjuvant therapy predict a better prognosis for patients with pancreatic adenocarcinoma: A multicenter case-control study of 240 patients. BMC Cancer 2019, 19, 252. [Google Scholar] [CrossRef]
- Picardo, S.L.; Coburn, B.; Hansen, A.R. The microbiome and cancer for clinicians. Critical Rev. Oncol. Hematol. 2019, 141, 1–12. [Google Scholar] [CrossRef]
- Michaud, D.; Izard, J. Microbiota, Oral Microbiome, and Pancreatic Cancer. Cancer J. 2014, 20, 203–206. [Google Scholar] [CrossRef]
- Zambirinis, C.; Res, M.; Al, E. Pancreatic Cancer, Inflammation and Microbiome. Cancer J. 2014, 20, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Pushalkar, S.; Mautin, H.; Al, E. The Pancreatic Cancer Microbiome Promotes Oncogenesis by Induction of Innate and Adaptive Immune Suppression. Cancer Discov. 2018, 8, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.A. Study links periodontal disease bacteria to pancreatic cancer risk. JAMA J. Am. Med. Assoc. 2016, 315, 2653–2654. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.Y.; Shi, S.; Liang, C.; Meng, Q.C.; Hua, J.; Zhang, Y.Y.; Liu, J.; Zhang, B.; Xu, J.; Yu, X.J. The microbiota and microbiome in pancreatic cancer: More influential than expected. Mol. Cancer 2019, 18, 1–15. [Google Scholar] [CrossRef]
- Fan, X.; Alekseyenko, A.V.; Wu, J.; Jacobs, E.J.; Gapstur, S.M.; Purdue, M.P.; Abnet, C.C.; Stolzenberg-Solomon, R.; Miller, G.; Ravel, J.; et al. Human oral microbiome and prospective risk for pancreatic cancer: A population-based nested case-control study. Cancer Res. 2018, 67, 120–127. [Google Scholar] [CrossRef]
- Mitsuhashi, K.; Nosho, K.; Sukawa, Y.; Matsunaga, Y.; Ito, M.; Kurihara, H.; Kanno, S.; Igarashi, H.; Naito, T.; Adachi, Y.; et al. Association of Fusobacterium species in pancreatic cancer tissues with molecular features and prognosis. Oncotarget 2015, 6, 7209–7220. [Google Scholar] [CrossRef] [PubMed]
- Michaud, D.S.; Izard, J.; Wilhelm-benartzi, C.S.; Grote, V.A.; Tjønneland, A.; Dahm, C.C.; Overvad, K. Plasma antibodies to oral bacteria and risk of pancreatic cancer in a large European prospective cohort study. Gut 2014, 62, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; Vétizou, M.; Boneca, I.G.; Lepage, P.; Chamaillard, M.; Zitvogel, L. Enhancing the clinical coverage and anticancer efficacy of immune checkpoint blockade through manipulation of the gut microbiota. OncoImmunology 2017, 6, 10–12. [Google Scholar] [CrossRef]
- Riquelme, E.; Zhang, Y.; Zhang, L.; Montiel, M.; Zoltan, M.; Dong, W.; Quesada, P.; Sahin, I.; Chandra, V.; San Lucas, A.; et al. Tumor Microbiome Diversity and Composition Influence Pancreatic Cancer Outcomes. Cell 2019, 178, 795–806.e12. [Google Scholar] [CrossRef] [PubMed]
- Mendez, R.; Kesh, K.; Arora, N.; Di Martino, L.; McAllister, F.; Merchant, N.; Banerjee, S.; Banerjee, S. Microbial dysbiosis and polyamine metabolism as predictive markers for early detection of pancreatic cancer. Carcinogenesis 2020, 41, 561–570. [Google Scholar] [CrossRef]
- Geller, L.T.; Barzily-Rokni, M.; Danino, T.; Jonas, O.H.; Shental, N.; Nejman, D.; Gavert, N.; Zwang, Y.; Cooper, Z.A.; Shee, K.; et al. Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine. Science 2017, 15, 1156–1160. [Google Scholar] [CrossRef] [PubMed]
- Farrel, J.; Zhang, L. Variations of oral microbiota are associated with pancreatic diseases including pancreatic cancer. Gut 2013, 61, 582–588. [Google Scholar] [CrossRef]
- Ren, Z.; Jiang, J.; Xie, H.; Li, A.; Lu, H.; Xu, S.; Zhou, L.; Zhang, H.; Cui, G.; Chen, X.; et al. Gut microbial profile analysis by MiSeq sequencing of pancreatic carcinoma patients in China. Oncotarget 2017, 8, 95176–95191. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.M.; Gharaibeh, R.Z.; Gauthier, J.; Beveridge, M.; Pope, J.L.; Guijarro, M.V.; Yu, Q.; He, Z.; Ohland, C.; Newsome, R.; et al. Intestinal microbiota enhances pancreatic carcinogenesis in preclinical models. Carcinogenesis 2018, 39, 1068–1078. [Google Scholar] [CrossRef] [PubMed]
- Regel, I.; Mayerle, J.; Mukund Mahajan, U. Current strategies and future perspectives for precision medicine in pancreatic cancer. Cancers 2020, 12, 1024. [Google Scholar] [CrossRef]
- Fang, Y.; Yao, Q.; Chen, Z.; Xiang, J.; William, F.E.; Gibbs, R.A.; Chen, C. Genetic and molecular alterations in pancreatic cancer: Implications for personalized medicine. Med. Sci. Monit. 2013, 19, 916–926. [Google Scholar] [CrossRef]
- Pishvaian, M.J.; Bender, R.J.; Halverson, D.; Rahib, L.; Hendifar, A.E.; Mikhail, S.; Chung, V.; Picozzi, V.J.; Sohal, D.; Blais, E.M.; et al. Molecular Profiling of Patients with Pancreatic Cancer: Initial Results from the Know Your Tumor Initiative. Clin. Cancer Res. 2018, 24, 5018–5027. [Google Scholar] [CrossRef]
- Dreyer, S.B.; Jamieson, N.B.; Cooke, S.L.; Valle, J.W.; McKay, C.J.; Biankin, A.V.; Chang, D.K. PRECISION-Panc: The Next Generation Therapeutic Development Platform for Pancreatic Cancer. Clin. Oncol. 2020, 32, 1–4. [Google Scholar] [CrossRef]
- Dreyer, S.B.; Jamieson, N.B.; Morton, J.P.; Sansom, O.J.; Biankin, A.V.; Chang, D.K. Pancreatic Cancer: From Genome Discovery to PRECISION-Panc. Clin. Oncol. 2020, 32, 5–8. [Google Scholar] [CrossRef]
- Lomberk, G.; Dusetti, N.; Iovanna, J.; Urrutia, R. Emerging epigenomic landscapes of pancreatic cancer in the era of precision medicine. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Aung, K.L.; Fischer, S.E.; Denroche, R.E.; Jang, G.-H.; Dodd, A.; Creighton, S.; Southwood, B.; Liang, S.-B.; Chadwick, D.; Zhang, A.; et al. Genomics-Driven Precision Medicine for Advanced Pancreatic Cancer: Early Results from the COMPASS Trial. Clin. Cancer Res. 2018, 24, 1344–1354. [Google Scholar] [CrossRef] [PubMed]
- Goonetilleke, K.S.; Siriwardena, A.K. Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur. J. Surg. Oncol. EJSO 2007, 33, 266–270. [Google Scholar] [CrossRef]
- Schlesinger, Y.; Yosefov-Levi, O.; Kolodkin-Gal, D.; Granit, R.Z.; Peters, L.; Kalifa, R.; Xia, L.; Nasereddin, A.; Shiff, I.; Amran, O.; et al. Single-cell transcriptomes of pancreatic preinvasive lesions and cancer reveal acinar metaplastic cells’ heterogeneity. Nat. Commun. 2020, 11. [Google Scholar] [CrossRef]
- Yang, F.; Liu, D.Y.; Guo, J.T.; Ge, N.; Zhu, P.; Liu, X.; Wang, S.; Wang, G.X.; Sun, S.Y. Circular RNA circ-LDLRAD3 as a biomarker in diagnosis of pancreatic cancer. World J. Gastroenterol. 2017, 23, 8345–8354. [Google Scholar] [CrossRef]
- An, Y.; Cai, H.; Zhang, Y.; Liu, S.; Duan, Y.; Sun, D.; Chen, X.; He, X. CircZMYM2 competed endogenously with miR-335-5p to regulate JMJD2C in pancreatic cancer. Cell. Physiol. Biochem. 2018, 51, 2224–2236. [Google Scholar] [CrossRef]
- Zhu, P.; Ge, N.; Liu, D.; Yang, F.; Zhang, K.; Guo, J.; Liu, X.; Wang, S.; Wang, G.; Sun, S. Preliminary investigation of the function of hsa_circ_0006215 in pancreatic cancer. Oncol. Lett. 2018, 16, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Z.; Jiang, P.; Peng, M.; Zhang, X.; Chen, K.; Liu, H.; Bi, H.; Liu, X.; Li, X. Circular RNA IARS (circ-IARS) secreted by pancreatic cancer cells and located within exosomes regulates endothelial monolayer permeability to promote tumor metastasis. J. Exp. Clin. Cancer Res. 2018, 37, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yanfang, W.; Li, J.; Jiang, P.; Peng, T.; Chen, K.; Zhao, X.; Zhang, Y.; Zhen, P.; Zhu, J.; et al. Tumor-released exosomal circular RNA PDE8A promotes invasive growth via the miR-338/MACC1/MET pathway in pancreatic cancer. Cancer Lett. 2018, 432, 237–250. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, T.; Yan, L.; Qu, L. A novel prognostic biomarker for pancreatic ductal adenocarcinoma: Hsa_circ_0001649. Gene 2018, 675, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Qu, S.; Hao, X.; Song, W.; Niu, K.; Yang, X.; Zhang, X.; Shang, R.; Wang, Q.; Li, H.; Liu, Z. Circular RNA circRHOT1 is upregulated and promotes cell proliferation and invasion in pancreatic cancer. Epigenomics 2019, 11, 53–63. [Google Scholar] [CrossRef]
- Xu, Y.; Yao, Y.; Gao, P.; Cui, Y. Upregulated circular RNA circ_0030235 predicts unfavorable prognosis in pancreatic ductal adenocarcinoma and facilitates cell progression by sponging miR-1253 and miR-1294. Biochem. Biophys. Res. Commun. 2019, 509, 138–142. [Google Scholar] [CrossRef]
- Hao, L.; Rong, W.; Bai, L.; Cui, H.; Zhang, S.; Li, Y.; Chen, D.; Meng, X. Upregulated circular RNA circ_0007534 indicates an unfavorable prognosis in pancreatic ductal adenocarcinoma and regulates cell proliferation, apoptosis, and invasion by sponging miR-625 and miR-892b. J. Cell. Biochem. 2019, 120, 3780–3789. [Google Scholar] [CrossRef]
- Liu, L.; Liu, F.B.; Huang, M.; Xie, K.; Xie, Q.S.; Liu, C.H.; Shen, M.J.; Huang, Q. Circular RNA ciRS-7 promotes the proliferation and metastasis of pancreatic cancer by regulating miR-7-mediated EGFR/STAT3 signaling pathway. Hepatobiliary Pancr. Dis. Int. 2019, 18, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Cong, X.; Ren, M.; Sun, H.; Liu, T.; Chen, G.; Wang, Q.; Li, Z.; Yu, S.; Yang, Q. Circular RNA hsa-circRNA-0007334 is Predicted to Promote MMP7 and COL1A1 Expression by Functioning as a miRNA Sponge in Pancreatic Ductal Adenocarcinoma. J. Oncol. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Zhang, C.; Chen, Y.; Gao, S. Downregulation of circular RNA circ-LDLRAD3 suppresses pancreatic cancer progression through miR-137-3p/PTN axis. Life Sci. 2019, 239, 116871. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Z.; Zhang, M.; Wang, B.; Ye, J.; Zhang, Y.; Tang, D.; Ma, D.; Jin, W.; Li, X.; et al. Circ-ASH2L promotes tumor progression by sponging miR-34a to regulate Notch1 in pancreatic ductal adenocarcinoma. J. Exp. Clin. Cancer Res. 2019, 38, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Xing, C.; Ye, H.; Wang, W.; Sun, M.; Zhang, J.; Zhao, Z.; Jiang, G. Circular RNA ADAM9 facilitates the malignant behaviours of pancreatic cancer by sponging miR-217 and upregulating PRSS3 expression. Artif. Cells NanoMed. Biotechnol. 2019, 47, 3920–3928. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, H.; Zhen, T.; Dong, Y.; Pei, X.; Shi, H. hsa_circ_001653 Implicates in the Development of Pancreatic Ductal Adenocarcinoma by Regulating MicroRNA-377-Mediated HOXC6 Axis. Mol. Ther. Nucleic Acids 2020, 20, 252–264. [Google Scholar] [CrossRef]
- Liu, Y.; Xia, L.; Dong, L.; Wang, J.; Xiao, Q.; Yu, X.; Zhu, H. CircHIPK3 promotes gemcitabine (GEM) resistance in pancreatic cancer cells by sponging miR-330-5p and targets RASSF1. Cancer Manag. Res. 2020, 12, 921–929. [Google Scholar] [CrossRef]
- Wong, C.H.; Lou, U.K.; Li, Y.; Chan, S.L.; Tong, J.H.; To, K.F.; Chen, Y. CircFOXK2 Promotes Growth and Metastasis of Pancreatic Ductal Adenocarcinoma by Complexing with RNA-Binding Proteins and Sponging MiR-942. Cancer Res. 2020, 80, 2138–2149. [Google Scholar] [CrossRef]
- Kong, Y.; Li, Y.; Luo, Y.; Zhu, J.; Zheng, H.; Gao, B.; Guo, X.; Li, Z.; Chen, R.; Chen, C. CircNFIB1 inhibits lymphangiogenesis and lymphatic metastasis via the miR-486-5p/PIK3R1/VEGF-C axis in pancreatic cancer. Mol. Cancer 2020, 19, 1–17. [Google Scholar] [CrossRef]
- Guo, W.; Zhao, L.; Wei, G.; Liu, P.; Zhang, Y.; Fu, L. Blocking circ_0013912 suppressed cell growth, migration and invasion of pancreatic ductal adenocarcinoma cells in vitro and in vivo partially through sponging miR-7-5p. Cancer Manag. Res. 2020, 12, 7291–7303. [Google Scholar] [CrossRef]
- Zhang, X.; Tan, P.; Zhuang, Y.; Du, L. hsa_circRNA_001587 upregulates SLC4A4 expression to inhibit migration, invasion, and angiogenesis of pancreatic cancer cells via binding to microRNA-223. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G703–G717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, M.; Wang, H.; Fisher, W.E.; Lin, P.H.; Yao, Q.; Chen, C. Profiling of 95 MicroRNAs in pancreatic cancer cell lines and surgical specimens by real-time PCR analysis. World J. Surg. 2009, 33, 698–709. [Google Scholar] [CrossRef] [PubMed]
- Greither, T.; Grochola, L.F.; Udelnow, A.; Lautenschläger, C.; Würl, P.; Taubert, H. Elevated expression of microRNAs 155, 203, 210 and 222 in pancreatic tumors is associated with poorer survival. Int. J. Cancer 2010, 126, 73–80. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, J.; Zhang, S.; Yu, D.; Chen, Y.; Liu, Q.; Shi, M.; Ni, C.; Zhu, M. Diagnostic and biological significance of microRNA-192 in pancreatic ductal adenocarcinoma. Oncol. Rep. 2013, 30, 276–284. [Google Scholar] [CrossRef]
- Hussein, N.A.E.M.; Kholy, Z.A.E.; Anwar, M.M.; Ahmad, M.A.; Ahmad, S.M. Plasma miR-22-3p, miR-642b-3p and miR-885-5p as diagnostic biomarkers for pancreatic cancer. J. Cancer Res. Clin. Oncol. 2017, 143, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Xu, Y.; Du, J.; Tan, J.; Jiao, H. Expression of microRNA-218 in human pancreatic ductal adenocarcinoma and its correlation with tumor progression and patient survival. J. Surg. Oncol. 2014, 109, 89–94. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, P.; Yin, T.; Zhang, F.; Wang, W. Upregulation of LncRNA PVT1 facilitates pancreatic ductal adenocarcinoma cell progression and glycolysis by regulating MiR-519d-3p and HIF-1A. J. Cancer 2020, 11, 2572–2579. [Google Scholar] [CrossRef]
- Pang, E.J.; Yang, R.; Fu, X.B.; Liu, Y.F. Overexpression of long non-coding RNA MALAT1 is correlated with clinical progression and unfavorable prognosis in pancreatic cancer. Tumor Biol. 2015, 36, 2403–2407. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Fang, Y.; Wang, Z.; Xie, J.; Zhan, Q.; Deng, X.; Chen, H.; Jin, J.; Peng, C.; Li, H.; et al. Downregulation of gas5 increases pancreatic cancer cell proliferation by regulating CDK6. Cell Tissue Res. 2013, 354, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Modali, S.D.; Parekh, V.I.; Kebebew, E.; Agarwal, S.K. Epigenetic regulation of the lncRNA MEG3 and its target c-MET in pancreatic neuroendocrine tumors. Mol. Endocrinol. 2015, 29, 224–237. [Google Scholar] [CrossRef]
- Peng, W.; Gao, W.; Feng, J. Long noncoding RNA HULC is a novel biomarker of poor prognosis in patients with pancreatic cancer. Med. Oncol. 2014, 31, 1–7. [Google Scholar] [CrossRef]
- Li, J.; Liu, D.; Hua, R.; Zhang, J.; Liu, W.; Huo, Y.; Cheng, Y.; Hong, J.; Sun, Y. Long non-coding RNAs expressed in pancreatic ductal adenocarcinoma and lncRNA BC008363 an independent prognostic factor in PDAC. Pancreatology 2014, 14, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Ting, D.T.; Lipson, D. Aberrant Overexpression of Satellite Repeats in Pancreatic and Other Epithelial Cancers. Science 2011, 331, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, J.D.; Baraniskin, A.; Hahn, S.A.; Mosel, F.; Bredemeier, M.; Wimberger, P.; Kimmig, R.; Kasimir-Bauer, S. Circulating U2 small nuclear RNA fragments as a novel diagnostic tool for patients with epithelial ovarian cancer. Clin. Chem. 2014, 60, 206–213. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turanli, B.; Yildirim, E.; Gulfidan, G.; Arga, K.Y.; Sinha, R. Current State of “Omics” Biomarkers in Pancreatic Cancer. J. Pers. Med. 2021, 11, 127. https://doi.org/10.3390/jpm11020127
Turanli B, Yildirim E, Gulfidan G, Arga KY, Sinha R. Current State of “Omics” Biomarkers in Pancreatic Cancer. Journal of Personalized Medicine. 2021; 11(2):127. https://doi.org/10.3390/jpm11020127
Chicago/Turabian StyleTuranli, Beste, Esra Yildirim, Gizem Gulfidan, Kazim Yalcin Arga, and Raghu Sinha. 2021. "Current State of “Omics” Biomarkers in Pancreatic Cancer" Journal of Personalized Medicine 11, no. 2: 127. https://doi.org/10.3390/jpm11020127
APA StyleTuranli, B., Yildirim, E., Gulfidan, G., Arga, K. Y., & Sinha, R. (2021). Current State of “Omics” Biomarkers in Pancreatic Cancer. Journal of Personalized Medicine, 11(2), 127. https://doi.org/10.3390/jpm11020127







