The Influence of Baseline Clinical Status and Surgical Strategy on Early Good to Excellent Result in Spinal Lumbar Arthrodesis: A Machine Learning Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical and Demographic Data
2.2. Clinical Outcomes
2.3. Machine Learning Approach
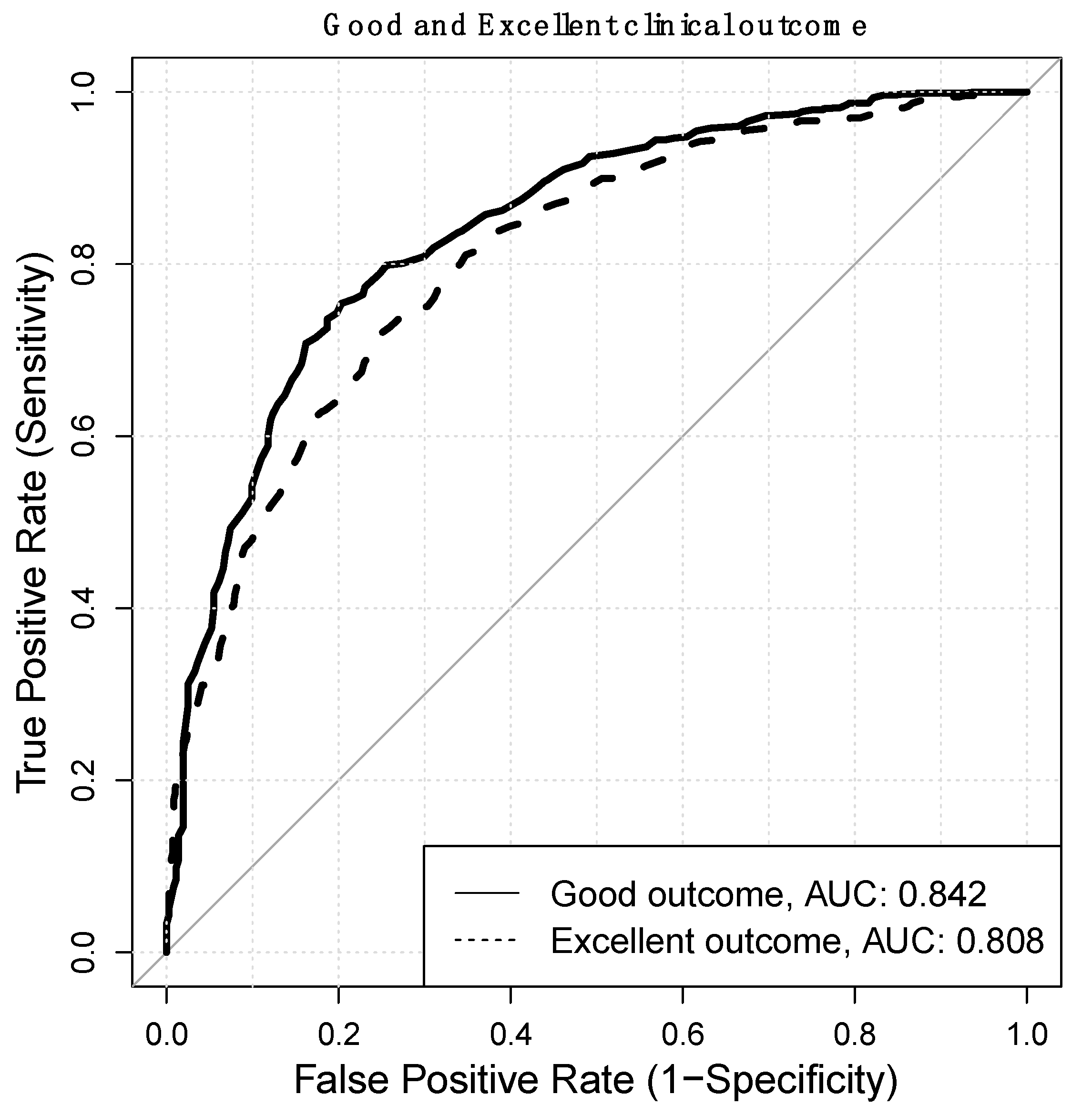
3. Results
Machine Learning Model
4. Discussion
4.1. Predictive Models of Surgical Improvement Based on Clinical Data
4.2. Predictive Models of Surgical Improvement Based on PROMs
4.3. Predictive Models’ Performances
4.4. Influence of ML Predictions on Therapeutic Strategy
4.5. Methodological Consideration and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schwab, F.; Dubey, A.; Gamez, L.; El Fegoun, A.B.; Hwang, K.; Pagala, M.; Farcy, J.-P. Adult Scoliosis: Prevalence, SF-36, and Nutritional Parameters in an Elderly Volunteer Population. Spine 2005, 30, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Pellisé, F.; Vila-Casademunt, A.; Ferrer, M.; Domingo-Sàbat, M.; Bago, J.; Pérez-Grueso, F.J.S.; Alanay, A.; Mannion, A.F.; Acaroglu, E. Impact on health related quality of life of adult spinal deformity (ASD) compared with other chronic conditions. Eur. Spine J. 2014, 24, 3–11. [Google Scholar] [CrossRef]
- Weinstein, J.N.; Lurie, J.D.; Tosteson, T.D.; Zhao, W.; Blood, E.A.; Tosteson, A.N.; Birkmeyer, N.J.O.; Herkowitz, H.N.; Longley, M.C.; Lenke, L.G.; et al. Surgical Compared with Nonoperative Treatment for Lumbar Degenerative Spondylolisthesis. four-year results in the Spine Patient Outcomes Research Trial (SPORT) randomized and observational cohorts. J. Bone Jt. Surg. Am. Vol. 2009, 91, 1295–1304. [Google Scholar] [CrossRef] [Green Version]
- Dagenais, S.; Caro, J.; Haldeman, S. A systematic review of low back pain cost of illness studies in the United States and internationally. Spine J. 2008, 8, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Zanirato, A.; Damilano, M.; Formica, M.; Piazzolla, A.; Lovi, A.; Villafañe, J.H.; Berjano, P. Complications in adult spine deformity surgery: A systematic review of the recent literature with reporting of aggregated incidences. Eur. Spine J. 2018, 27, 2272–2284. [Google Scholar] [CrossRef]
- Ferguson, T.B., Jr. The Institute of Medicine Committee Report “Best Care at Lower Cost: The Path to Continuously Learning Health Care”. Circ. Cardiovasc. Qual. Outcomes 2012, 5, e93–e94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGirt, M.J.; Sivaganesan, A.; Asher, A.L.; Devin, C.J. Prediction model for outcome after low-back surgery: Individualized likelihood of complication, hospital readmission, return to work, and 12-month improvement in functional disability. Neurosurg. Focus 2015, 39, E13. [Google Scholar] [CrossRef] [Green Version]
- Sinikallio, S.; Aalto, T.; Airaksinen, O.; Lehto, S.M.; Kröger, H.; Viinamäki, H. Depression Is Associated with a Poorer Outcome of Lumbar Spinal Stenosis Surgery: A two-year prospective follow-up study. Spine 2011, 36, 677–682. [Google Scholar] [CrossRef]
- Archer, K.R.; Devin, C.J.; Vanston, S.W.; Koyama, T.; Phillips, S.E.; George, S.Z.; McGirt, M.J.; Spengler, D.M.; Aaronson, O.S.; Cheng, J.S.; et al. Cognitive-Behavioral–Based Physical Therapy for Patients with Chronic Pain Undergoing Lumbar Spine Surgery: A Randomized Controlled Trial. J. Pain 2016, 17, 76–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGirt, M.J.; Bydon, M.; Archer, K.R.; Devin, C.J.; Chotai, S.; Parker, S.L.; Nian, H.; Harrell, F.E.; Speroff, T.; Dittus, R.S.; et al. An analysis from the Quality Outcomes Database, Part 1. Disability, quality of life, and pain outcomes following lumbar spine surgery: Predicting likely individual patient outcomes for shared decision-making. J. Neurosurg. Spine 2017, 27, 357–369. [Google Scholar] [CrossRef] [Green Version]
- Langella, F.; Barletta, P.; Baroncini, A.; Agarossi, M.; Scaramuzzo, L.; Luca, A.; Bassani, R.; Peretti, G.M.; Lamartina, C.; Villafañe, J.H.; et al. The use of electronic PROMs provides same outcomes as paper version in a spine surgery registry. Results from a prospective cohort study. Eur. Spine J. 2021, 30, 2645–2653. [Google Scholar] [CrossRef]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Glassman, S.D.; Carreon, L.Y.; Anderson, P.A.; Resnick, D.K. A diagnostic classification for lumbar spine registry development. Spine J. 2011, 11, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Monticone, M.; Baiardi, P.; Vanti, C.; Ferrari, S.; Pillastrini, P.; Mugnai, R.; Foti, C. Responsiveness of the Oswestry Disability Index and the Roland Morris Disability Questionnaire in Italian subjects with sub-acute and chronic low back pain. Eur. Spine J. 2011, 21, 122–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copay, A.G.; Glassman, S.D.; Subach, B.R.; Berven, S.; Schuler, T.C.; Carreon, L.Y. Minimum clinically important difference in lumbar spine surgery patients: A choice of methods using the Oswestry Disability Index, Medical Outcomes Study questionnaire Short Form 36, and Pain Scales. Spine J. 2008, 8, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Mannion, A.F.; Porchet, F.; Kleinstück, F.S.; Lattig, F.; Jeszenszky, D.; Bartanusz, V.; Dvorak, J.; Grob, D. The quality of spine surgery from the patient’s perspective: Part 2. Minimal clinically important difference for improvement and deterioration as measured with the Core Outcome Measures Index. Eur. Spine J. 2009, 18, 374–379. [Google Scholar] [CrossRef] [Green Version]
- Harel, O.; Mitchell, E.M.; Perkins, N.; Cole, S.R.; Tchetgen, E.J.T.; Sun, B.; Schisterman, E. Multiple Imputation for Incomplete Data in Epidemiologic Studies. Am. J. Epidemiol. 2018, 187, 576–584. [Google Scholar] [CrossRef] [Green Version]
- Van Buuren, S.; Groothuis-Oudshoorn, K. mice: Multivariate Imputation by Chained Equations inR. J. Stat. Softw. 2011, 45, 1–67. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Delgado, M.; Cernadas, E.; Barro, S.; Amorim, D. Do we need hundreds of classifiers to solve real world classification problems? J. Mach. Learn. Res. 2014, 15, 3133–3181. Available online: http://hdl.handle.net/10347/17792 (accessed on 9 December 2021).
- Bagnall, A.; Cawley, G.C. On the Use of Default Parameter Settings in the Empirical Evaluation of Classification Algorithms. arXiv 2017, arXiv:1703.06777. in preprint. [Google Scholar]
- Bunn, M.K.A. An Introduction to dplR. Ind. Commer. Train. 2008, 10, 11–18. [Google Scholar]
- Collins, G.S.; Reitsma, J.B.; Altman, D.G.; Moons, K.G.M. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD Statement. BMC Med. 2015, 13, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reid, D.B.; Daniels, A.H.; Ailon, T.; Miller, E.; Sciubba, D.M.; Smith, J.S.; Shaffrey, C.I.; Schwab, F.; Burton, D.; Hart, R.A.; et al. Frailty and Health-Related Quality of Life Improvement Following Adult Spinal Deformity Surgery. World Neurosurg. 2018, 112, e548–e554. [Google Scholar] [CrossRef]
- Berjano, P.; Lamartina, C. Far lateral approaches (XLIF) in adult scoliosis. Eur. Spine J. 2012, 22, 242–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langella, F.; Villafañe, J.H.; Damilano, M.; Cecchinato, R.; Pejrona, M.; Ismael, M.; Berjano, P. Predictive Accuracy of Surgimap Surgical Planning for Sagittal Imbalance: A Cohort Study. Spine 2017, 42, E1297–E1304. [Google Scholar] [CrossRef] [PubMed]
- Berjano, P.; Langella, F.; Ismael, M.-F.; Damilano, M.; Scopetta, S.; Lamartina, C. Successful correction of sagittal imbalance can be calculated on the basis of pelvic incidence and age. Eur. Spine J. 2014, 23, 587–596. [Google Scholar] [CrossRef]
- Yamato, Y.; Hasegawa, T.; Kobayashi, S.; Yasuda, T.; Togawa, D.; Arima, H.; Oe, S.; Iida, T.; Matsumura, A.; Hosogane, N.; et al. Calculation of the Target Lumbar Lordosis Angle for Restoring an Optimal Pelvic Tilt in Elderly Patients with Adult Spinal Deformity. Spine 2016, 41, E211–E217. [Google Scholar] [CrossRef]
- Mulvaney, G.; Rice, O.M.; Rossi, V.; Peters, D.; Smith, M.; Patt, J.; Pfortmiller, D.; Asher, A.L.; Kim, P.; Bernard, J.; et al. Mild and Severe Obesity Reduce the Effectiveness of Lumbar Fusions: 1-Year Patient-Reported Outcomes in 8171 Patients. Neurosurgery 2020, 88, 285–294. [Google Scholar] [CrossRef]
- Lingutla, K.K.; Pollock, R.; Benomran, E.; Purushothaman, B.; Kasis, A.; Bhatia, C.K.; Krishna, M.; Friesem, T. Outcome of lumbar spinal fusion surgery in obese patients: A systematic review and meta-analysis. Bone Jt. J. 2015, 97-B, 1395–1404. [Google Scholar] [CrossRef]
- Tan, G.H.; Goss, B.G.; Thorpe, P.J.; Williams, R.P. CT-based classification of long spinal allograft fusion. Eur. Spine J. 2007, 16, 1875–1881. [Google Scholar] [CrossRef] [Green Version]
- Berjano, P.; Langella, F.; Damilano, M.; Pejrona, M.; Buric, J.; Ismael, M.; Villafañe, J.H.; Lamartina, C. Fusion rate following extreme lateral lumbar interbody fusion. Eur. Spine J. 2015, 24, 369–371. [Google Scholar] [CrossRef]
- Cecchinato, R.; Langella, F.; Bassani, R.; Sansone, V.; Lamartina, C.; Berjano, P. Variations of cervical lordosis and head alignment after pedicle subtraction osteotomy surgery for sagittal imbalance. Eur. Spine J. 2014, 23, 644–649. [Google Scholar] [CrossRef]
- Carragee, E.J.; Cheng, I. Minimum acceptable outcomes after lumbar spinal fusion. Spine J. 2010, 10, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Staartjes, V.E.; de Wispelaere, M.P.; Vandertop, W.P.; Schröder, M.L. Deep learning-based preoperative predictive analytics for patient-reported outcomes following lumbar discectomy: Feasibility of center-specific modeling. Spine J. 2019, 19, 853–861. [Google Scholar] [CrossRef]
- Karnuta, J.M.; Golubovsky, J.L.; Haeberle, H.; Rajan, P.V.; Navarro, S.; Kamath, A.F.; Schaffer, J.L.; Krebs, V.E.; Pelle, D.W.; Ramkumar, P.N. Can a machine learning model accurately predict patient resource utilization following lumbar spinal fusion? Spine J. 2020, 20, 329–336. [Google Scholar] [CrossRef]
- Siccoli, A.; De Wispelaere, M.P.; Schröder, M.L.; Staartjes, V.E. Machine learning–based preoperative predictive analytics for lumbar spinal stenosis. Neurosurg. Focus 2019, 46, E5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.S.; Grabowski, M.M.; Habboub, G.; Mroz, T.E. The Impact of Artificial Intelligence on Quality and Safety. Glob. Spine J. 2020, 10, 99S–103S. [Google Scholar] [CrossRef]
- Khan, O.; Badhiwala, J.H.; Witiw, C.D.; Wilson, J.R.; Fehlings, M.G. Machine learning algorithms for prediction of health-related quality-of-life after surgery for mild degenerative cervical myelopathy. Spine J. 2021, 21, 1659–1669. [Google Scholar] [CrossRef] [PubMed]
- Merali, Z.G.; Witiw, C.D.; Badhiwala, J.H.; Wilson, J.R.; Fehlings, M.G. Using a machine learning approach to predict outcome after surgery for degenerative cervical myelopathy. PLoS ONE 2019, 14, e0215133. [Google Scholar] [CrossRef] [PubMed]
- Ames, C.P.; European Spine Study Group; Smith, J.S.; Pellisé, F.; Kelly, M.; Gum, J.L.; Alanay, A.; Acaroğlu, E.; Pérez-Grueso, F.J.S.; Kleinstück, F.S.; et al. Development of predictive models for all individual questions of SRS-22R after adult spinal deformity surgery: A step toward individualized medicine. Eur. Spine J. 2019, 28, 1998–2011. [Google Scholar] [CrossRef] [PubMed]
- Obeid, I.; Berjano, P.; Lamartina, C.; Chopin, D.; Boissière, L.; Bourghli, A. Classification of coronal imbalance in adult scoliosis and spine deformity: A treatment-oriented guideline. Eur. Spine J. 2019, 28, 94–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Glassman | Equipe | Age | Gender | BMI | ASA | ODIPre |
|---|---|---|---|---|---|---|
| 18.8% | 0.0% | 0.0% | 0.0% | 1.0% | 0.0% | 0.0% |
| COMIPre | SFPPre | SFMPre | Levels | From Level | To Level | CCI |
| 0.0% | 0.0% | 0.0% | 20.3% | 20.4% | 20.4% | 19.0% |
| Outcome + | Outcome − | Total | |||
|---|---|---|---|---|---|
| Prediction + | 653 | 75 | 728 | Sensitivity | 74.3% |
| Prediction − | 226 | 289 | 515 | Specificity | 79.4% |
| Total | 879 | 364 | 1243 | PPV | 89.7% |
| NPV | 56.1% | ||||
| Accuracy | 75.8% | ||||
| AUC ROC | 0.842 |
| Outcome + | Outcome − | Total | |||
|---|---|---|---|---|---|
| Prediction + | 399 | 199 | 598 | Sensitivity | 74.2% |
| Prediction − | 139 | 506 | 645 | Specificity | 71.8% |
| Total | 538 | 705 | 1243 | PPV | 66.7% |
| NPV | 78.4% | ||||
| Accuracy | 72.8% | ||||
| AUC ROC | 0.808 |
| Predictive Variables | Good Clinical Outcome | Predictive Variables | Excellent Clinical Outcome |
|---|---|---|---|
| SFMPre | 73.20 | SFPPre | 90.34 |
| SFPPre | 70.80 | SFMPre | 87.13 |
| ODIPre | 66.77 | BMI | 78.61 |
| BMI | 62.97 | Age | 69.92 |
| Age | 61.12 | ODIPre | 66.21 |
| COMIPre | 31.00 | COMIPre | 43.74 |
| Glassman | 28.30 | Glassman | 29.90 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berjano, P.; Langella, F.; Ventriglia, L.; Compagnone, D.; Barletta, P.; Huber, D.; Mangili, F.; Licandro, G.; Galbusera, F.; Cina, A.; et al. The Influence of Baseline Clinical Status and Surgical Strategy on Early Good to Excellent Result in Spinal Lumbar Arthrodesis: A Machine Learning Approach. J. Pers. Med. 2021, 11, 1377. https://doi.org/10.3390/jpm11121377
Berjano P, Langella F, Ventriglia L, Compagnone D, Barletta P, Huber D, Mangili F, Licandro G, Galbusera F, Cina A, et al. The Influence of Baseline Clinical Status and Surgical Strategy on Early Good to Excellent Result in Spinal Lumbar Arthrodesis: A Machine Learning Approach. Journal of Personalized Medicine. 2021; 11(12):1377. https://doi.org/10.3390/jpm11121377
Chicago/Turabian StyleBerjano, Pedro, Francesco Langella, Luca Ventriglia, Domenico Compagnone, Paolo Barletta, David Huber, Francesca Mangili, Ginevra Licandro, Fabio Galbusera, Andrea Cina, and et al. 2021. "The Influence of Baseline Clinical Status and Surgical Strategy on Early Good to Excellent Result in Spinal Lumbar Arthrodesis: A Machine Learning Approach" Journal of Personalized Medicine 11, no. 12: 1377. https://doi.org/10.3390/jpm11121377
APA StyleBerjano, P., Langella, F., Ventriglia, L., Compagnone, D., Barletta, P., Huber, D., Mangili, F., Licandro, G., Galbusera, F., Cina, A., Bassani, T., Lamartina, C., Scaramuzzo, L., Bassani, R., Brayda-Bruno, M., Villafañe, J. H., Monti, L., & Azzimonti, L. (2021). The Influence of Baseline Clinical Status and Surgical Strategy on Early Good to Excellent Result in Spinal Lumbar Arthrodesis: A Machine Learning Approach. Journal of Personalized Medicine, 11(12), 1377. https://doi.org/10.3390/jpm11121377







