Association between Retinal Thickness Variability and Visual Acuity Outcome during Maintenance Therapy Using Intravitreal Anti-Vascular Endothelial Growth Factor Agents for Neovascular Age-Related Macular Degeneration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Optical Coherence Tomography Imaging and Measurements
2.3. Statistical Analysis
3. Results
3.1. Baseline Patient Demographics
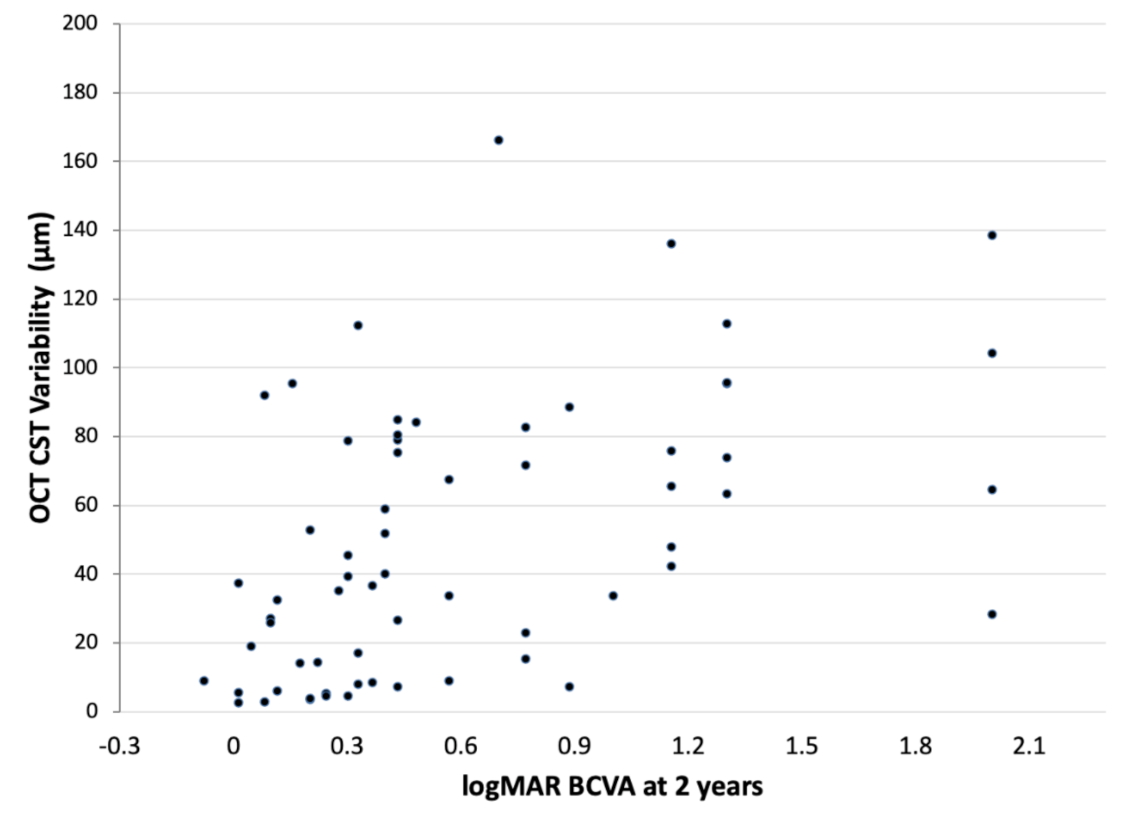
3.2. Correlation of OCT CST Variability Values and BCVA at 2 Years
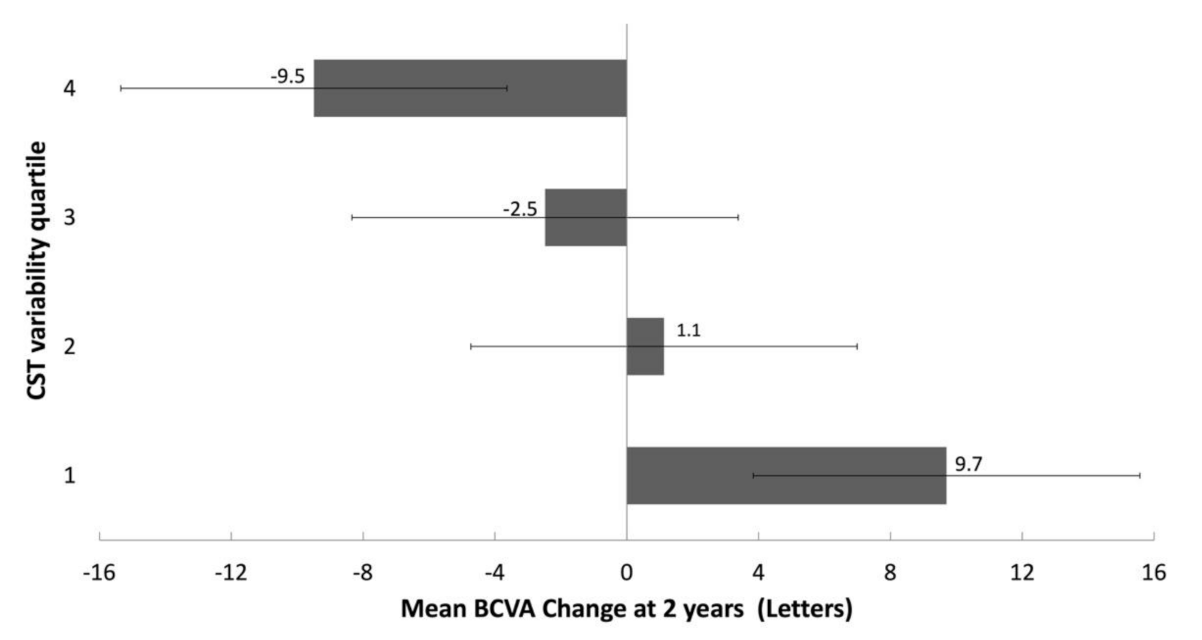
3.3. Correlation of OCT CST Variability Quartiles and Change in BCVA at 2 Years
3.4. Anti-VEGF Injections and OCT CST Variability Quartiles
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Flaxman, S.R.; Bourne, R.; Resnikoff, S.; Ackland, P.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, e1221–e1234. [Google Scholar] [CrossRef] [Green Version]
- Adamis, A.P.; Brittain, C.J.; Dandekar, A.; Hopkins, J.J. Building on the success of anti-vascular endothelial growth factor therapy: A vision for the next decade. Eye 2020, 34, 1966–1972. [Google Scholar] [CrossRef] [PubMed]
- Sloan, F.A.; Hanrahan, B.W. The effects of technological advances on outcomes for elderly persons with exudative age-related macular degeneration. JAMA Ophthalmol. 2014, 132, 456–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finger, R.P.; Daien, V.; Eldem, B.M.; Talks, J.S.; Korobelnik, J.F.; Mitchell, P.; Sakamoto, T.; Wong, T.Y.; Pantiri, K.; Carrasco, J. Anti-vascular endothelial growth factor in neovascular age-related macular degeneration-a systematic review of the impact of anti-VEGF on patient outcomes and healthcare systems. BMC Ophthalmol. 2020, 20, 294. [Google Scholar] [CrossRef] [PubMed]
- Heath Jeffery, R.C.; Mukhtar, S.A.; Lopez, D.; Preen, D.B.; McAllister, I.L.; Mackey, D.A.; Morlet, N.; Morgan, W.H.; Chen, F.K. Incidence of newly registered blindness from age-related macular degeneration in Australia over a 21-Year Period: 1996–2016. Asia Pac. J. Ophthalmol. 2021, 10, 442–449. [Google Scholar] [CrossRef]
- Mantel, I. Optimizing the anti-VEGF treatment strategy for neovascular age-related macular degeneration: From clinical trials to real-life requirements. Transl. Vis. Sci. Technol. 2015, 4, 6. [Google Scholar] [CrossRef]
- García-Layana, A.; Figueroa, M.S.; Arias, L.; Araiz, J.; Ruiz-Moreno, J.M.; García-Arumí, J.; Gómez-Ulla, F.; López-Gálvez, M.I.; Cabrera-López, F.; García-Campos, J.M.; et al. Individualized therapy with ranibizumab in wet age-related macular degeneration. J. Ophthalmol. 2015, 2015, 412903. [Google Scholar] [CrossRef]
- Garweg, J.G.; Niderprim, S.A.; Russ, H.M.; Pfister, I.B. Comparison of strategies of treatment with ranibizumab in newly-diagnosed cases of neovascular age-related macular degeneration. J. Ocul. Pharmacol. Ther. 2017, 33, 773–778. [Google Scholar] [CrossRef]
- Hatz, K.; Prünte, C. Treat and extend versus pro re nata regimens of ranibizumab in neovascular age-related macular degeneration: A comparative 12 month study. Acta Opphthalmol. 2017, 95, e67–e72. [Google Scholar] [CrossRef] [Green Version]
- Chaikitmongkol, V.; Sagong, M.; Lai, T.Y.Y.; Tan, G.S.W.; Fariza, N.; Ohji, M.; Mitchell, P.; Yang, C.H.; Ruamviboonsuk, P.; Wong, I.; et al. Treat-and-extend regimens for the management of neovascular age-related macular degeneration and polypoidal choroidal vasculopathy: Consensus and recommendations from the Asia-Pacific Vitreo-Retina Society. Asia Pac. J. Ophthalmol. 2021, in press. [Google Scholar]
- Okada, M.; Kandasamy, R.; Chong, E.W.; McGuiness, M.; Guymer, R.H. The treat-and-extend injection regimen versus alternate dosing strategies in age-related macular degeneration: A systematic review and meta-analysis. Am. J. Ophthalmol. 2018, 192, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.H.; Downey, L.; Devonport, H.; Gale, R.P.; Kotagiri, A.; Mahmood, S.; Mehta, H.; Narendran, N.; Patel, P.J.; Parmar, N.; et al. Recommendations by a UK expert panel on an aflibercept treat-and-extend pathway for the treatment of neovascular age-related macular degeneration. Eye 2020, 34, 1825–1834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spaide, R.F. Antivascular endothelial growth factor dosing and expected acuity outcome at 1 year. Retina 2021, 41, 1153–1163. [Google Scholar]
- Phadikar, P.; Saxena, S.; Ruia, S.; Lai, T.Y.; Meyer, C.H.; Eliott, D. The potential of spectral domain optical coherence tomography imaging based retinal biomarkers. Int. J. Retin. Vitr. 2017, 3, 1. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Lai, T.Y.Y. Baseline predictors of visual acuity outcome in patients with wet age-related macular degeneration. BioMed Res. Int. 2018, 2018, 9640131. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, V.; Puzo, M.; Sanchez-Monroy, J.; Gabrielle, P.H.; Garcher, C.C.; Baudin, F.; Wolff, B.; Castelnovo, L.; Michel, G.; O’Toole, L.; et al. Association between anatomical and clinical outcomes of neovascular age-related macular degeneration treated with antivascular endothelial growth factor. Retina 2021, 41, 1446–1454. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.N.; Reeves, B.C.; Maguire, M.G.; Martin, D.F.; Muldrew, A.; Peto, T.; Rogers, C.; Chakravarthy, U. Associations of variation in retinal thickness with visual acuity and anatomic outcomes in eyes with neovascular age-related macular degeneration lesions treated with anti-vascular endothelial growth factor agents. JAMA Ophthalmol. 2020, 138, 1043–1051. [Google Scholar] [CrossRef]
- Kaiser, P.K.; Wykoff, C.C.; Singh, R.P.; Khanani, A.M.; Do, D.V.; Patel, H.; Patel, N. Retinal fluid and thickness as measures of disease activity in neovascular age-related macular degeneration. Retina 2021, 41, 1579–1586. [Google Scholar] [CrossRef]
- Eldem, B.; Lai, T.Y.Y.; Ngah, N.F.; Vote, B.; Yu, H.G.; Fabre, A.; Backer, A.; Clunas, N.J. An analysis of ranibizumab treatment and visual outcomes in real-world settings: The UNCOVER study. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 963–973. [Google Scholar] [CrossRef] [Green Version]
- Lai, T.Y.Y.; Cheung, C.M.G.; Mieler, W.F. Ophthalmic application of anti-VEGF therapy. Asia Pac. J. Ophthalmol. 2017, 6, 479–480. [Google Scholar] [CrossRef]
- Chandra, S.; Arpa, C.; Menon, D.; Khalid, H.; Hamilton, R.; Nicholson, L.; Pal, B.; Fasolo, S.; Hykin, P.; Keane, P.A.; et al. Ten-year outcomes of antivascular endothelial growth factor therapy in neovascular age-related macular degeneration. Eye 2020, 34, 1888–1896. [Google Scholar] [CrossRef] [PubMed]
- Ciulla, T.A.; Hussain, R.M.; Pollack, J.S.; Williams, D.F. Visual acuity outcomes and anti-vascular endothelial growth factor therapy intensity in neovascular age-related macular degeneration patients: A real-world analysis of 49 485 Eyes. Ophthalmol. Retin. 2020, 4, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Rasheed, R.; Menon, D.; Patrao, N.; Lamin, A.; Gurudas, S.; Balaskas, K.; Patel, P.J.; Ali, N.; Sivaprasad, S. Impact of injection frequency on 5-year real-world visual acuity outcomes of aflibercept therapy for neovascular age-related macular degeneration. Eye 2021, 35, 409–417. [Google Scholar] [CrossRef]
- Nicolò, M.; Ferro Desideri, L.; Vagge, A.; Traverso, C.E. Faricimab: An investigational agent targeting the Tie-2/angiopoietin pathway and VEGF-A for the treatment of retinal diseases. Expert Opin. Investig. Drugs 2021, 30, 193–200. [Google Scholar] [CrossRef]
- Khanani, A.M.; Russell, M.W.; Aziz, A.A.; Danzig, C.J.; Weng, C.Y.; Eichenbaum, D.A.; Singh, R.P. Angiopoietins as potential targets in management of retinal disease. Clin. Ophthalmol. 2021, 15, 3747–3755. [Google Scholar] [CrossRef]
- Iglicki, M.; González, D.P.; Loewenstein, A.; Zur, D. Longer-acting treatments for neovascular age-related macular degeneration-present and future. Eye 2021, 35, 1111–1116. [Google Scholar] [CrossRef]
- Dugel, P.U.; Singh, R.P.; Koh, A.; Ogura, Y.; Weissgerber, G.; Gedif, K.; Jaffe, G.J.; Tadayoni, R.; Schmidt-Erfurth, U.; Holz, F.G. HAWK and HARRIER: Ninety-Six-Week Outcomes from the Phase 3 Trials of Brolucizumab for Neovascular Age-Related Macular Degeneration. Ophthalmology 2021, 128, 89–99. [Google Scholar] [CrossRef] [PubMed]
| Quartile 1
(Least Variability) | Quartile 2 | Quartile 3 | Quartile 4 (Greatest Variability) |
|---|---|---|---|
| <14.4 µm | 14.4–39.4 µm | 39.4–78.7 µm | ≥78.7 µm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, T.Y.Y.; Lai, R.Y.K. Association between Retinal Thickness Variability and Visual Acuity Outcome during Maintenance Therapy Using Intravitreal Anti-Vascular Endothelial Growth Factor Agents for Neovascular Age-Related Macular Degeneration. J. Pers. Med. 2021, 11, 1024. https://doi.org/10.3390/jpm11101024
Lai TYY, Lai RYK. Association between Retinal Thickness Variability and Visual Acuity Outcome during Maintenance Therapy Using Intravitreal Anti-Vascular Endothelial Growth Factor Agents for Neovascular Age-Related Macular Degeneration. Journal of Personalized Medicine. 2021; 11(10):1024. https://doi.org/10.3390/jpm11101024
Chicago/Turabian StyleLai, Timothy Y. Y., and Ricky Y. K. Lai. 2021. "Association between Retinal Thickness Variability and Visual Acuity Outcome during Maintenance Therapy Using Intravitreal Anti-Vascular Endothelial Growth Factor Agents for Neovascular Age-Related Macular Degeneration" Journal of Personalized Medicine 11, no. 10: 1024. https://doi.org/10.3390/jpm11101024
APA StyleLai, T. Y. Y., & Lai, R. Y. K. (2021). Association between Retinal Thickness Variability and Visual Acuity Outcome during Maintenance Therapy Using Intravitreal Anti-Vascular Endothelial Growth Factor Agents for Neovascular Age-Related Macular Degeneration. Journal of Personalized Medicine, 11(10), 1024. https://doi.org/10.3390/jpm11101024






