Radiation Biomarkers in Large Scale Human Health Effects Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Blood Sampling
2.2. Ex Vivo Irradiation of Separated Lymphocytes and Whole Blood
2.3. Gamma H2AX Assay for Initial and Residual Radiation-Induced Foci
2.4. Dicentric and Micronucleus Assays
2.5. DCA Calibration Curve Construction
2.6. Statistical Methods
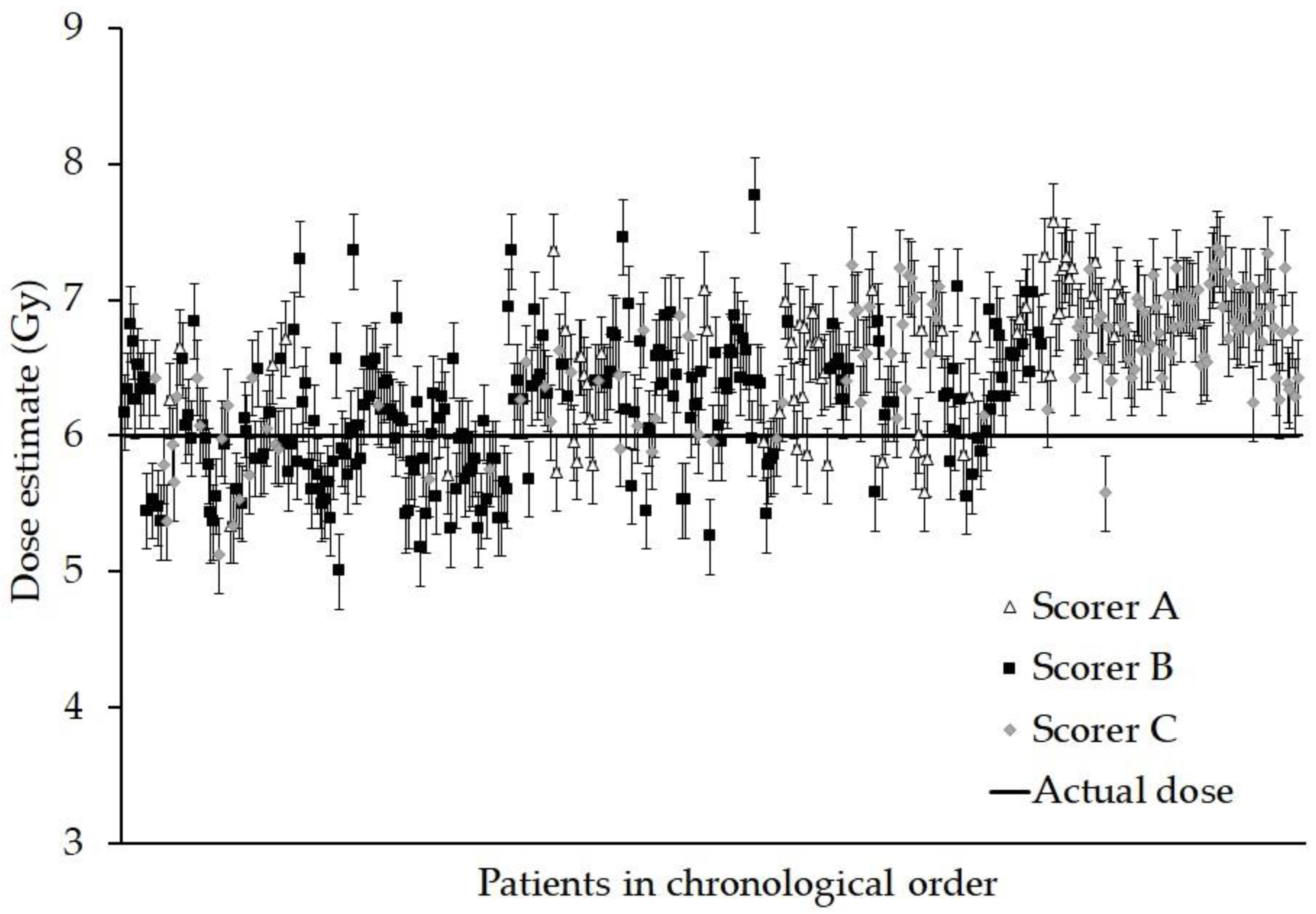
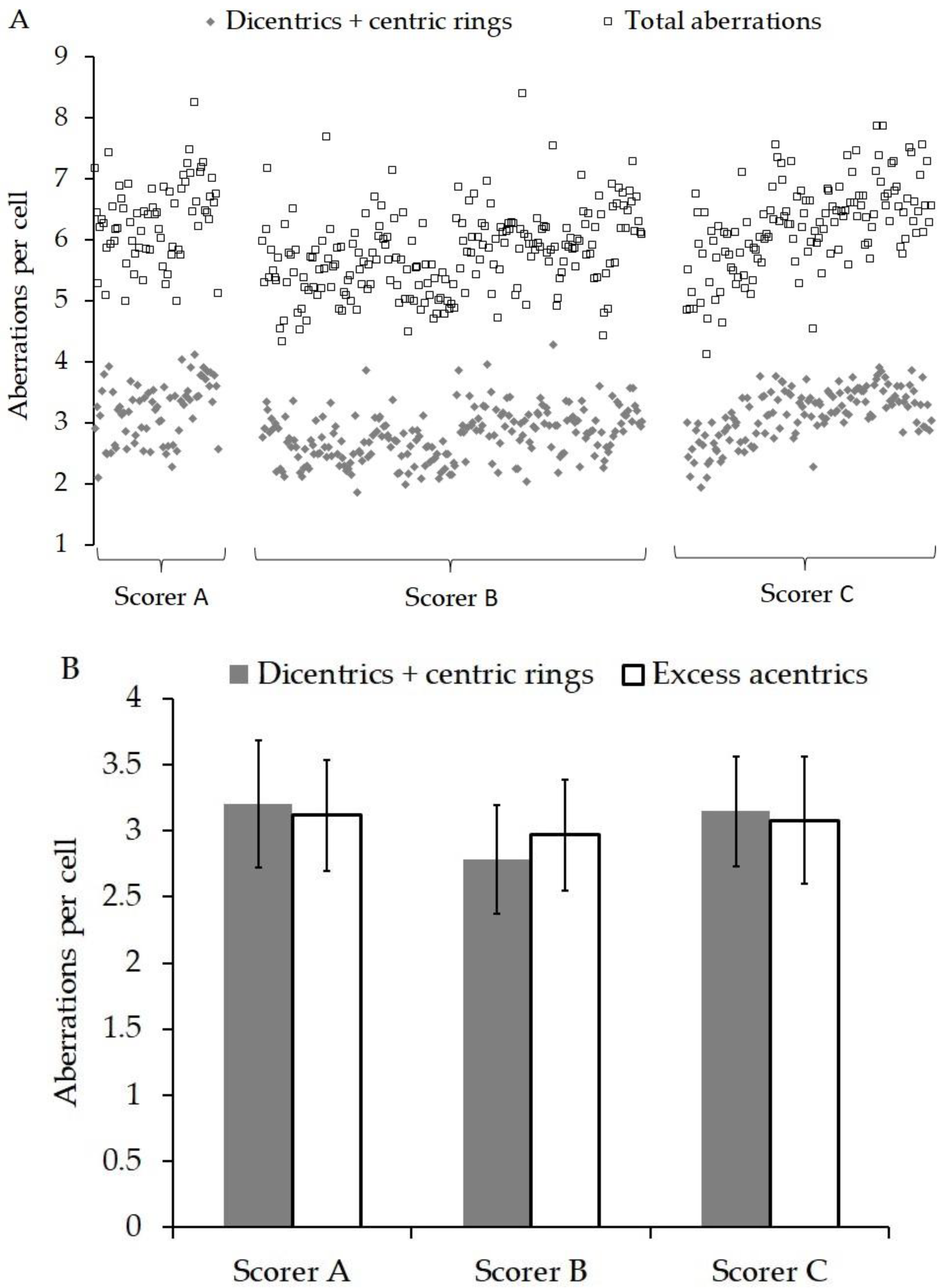
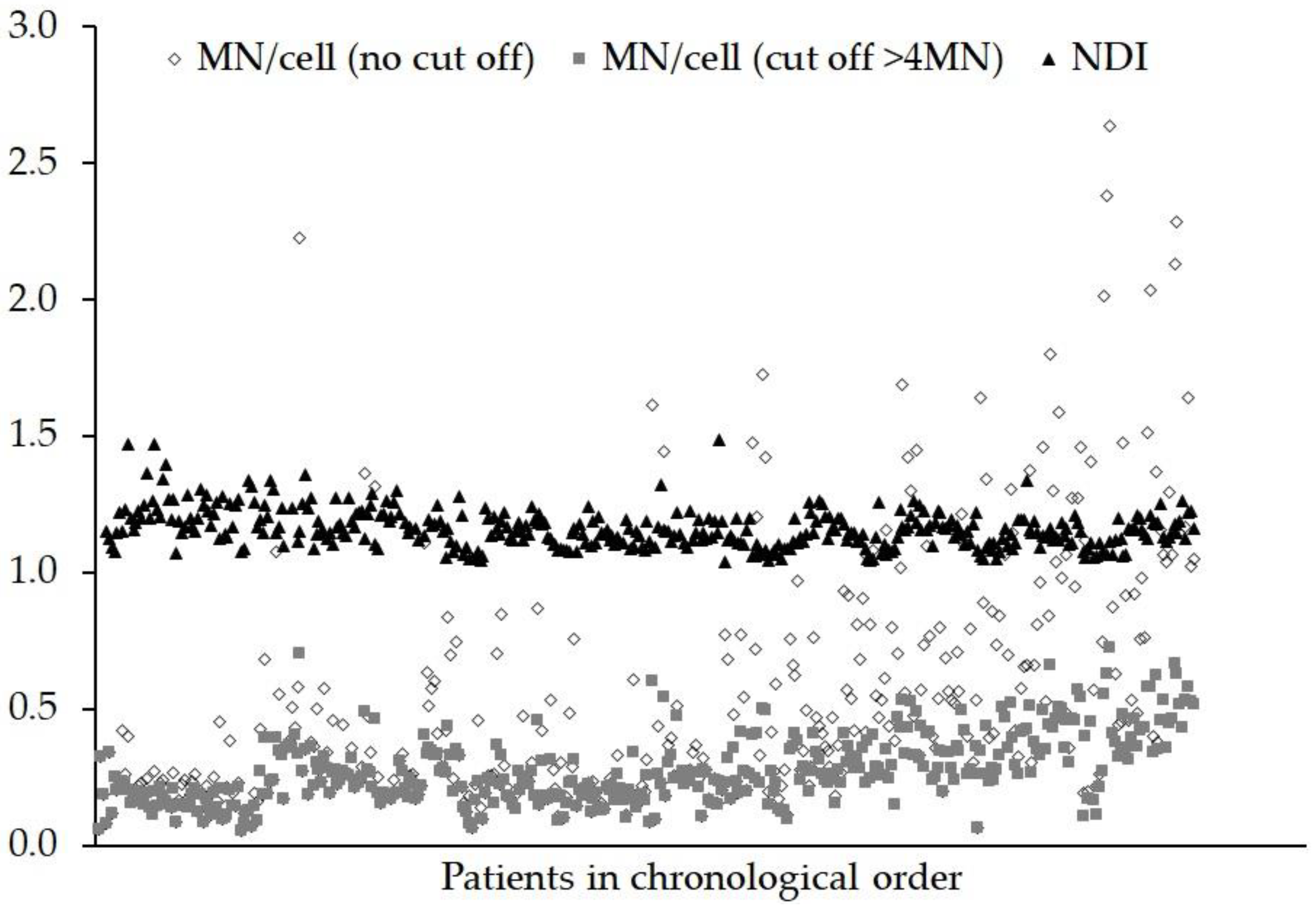
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Obe, G.; Natarajan, A.T. (Eds.) Chromosome Aberrations; Karger: Basel, Switzerland, 2004. [Google Scholar]
- Rothkamm, K.; Barnard, S.; Moquet, J.; Ellender, M.; Rana, Z.; Burdak-Rothkamm, S. DNA damage foci Meaning and significance. Environ. Mol. Mutagen. 2015, 56, 491–504. [Google Scholar] [CrossRef]
- International Atomic Energy Agency (IAEA). Cytogenetic Dosimetry: Applications in Preparedness for and Response to Radiation Emergencies; IAEA: Vienna, Austria, 2011. [Google Scholar]
- Sun, M.; Moquet, J.E.; Barnard, S.; Lloyd, D.C.; Rothkamm, K.; Ainsbury, E.A. Doses in Radiation Accidents Investigated by Chromosomal Aberration Analysis XXV. Review of Cases Investigated, 2006–2015. PHE-CRCE-025. 2016; PHE Publication Gateway Number: 2015730. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/515260/PHE-CRCE-025.pdf (accessed on 12 May 2016).
- Sotnik, N.V.; Azizova, T.V.; Darroudi, F.; Ainsbury, E.A.; Moquet, J.E.; Fomina, J.; Lloyd, D.C.; Edwards, A.A. Verification by the FISH translocation assay of historic doses to Mayak workers from external gamma radiation. Radiat. Environ. Biophys. 2015, 5494, 445–451. [Google Scholar] [CrossRef]
- Chua, M.L.K.; Rothkamm, K. Biomarkers of radiation exposure: Can they predict normal tissue radiosensitivity? Clin. Oncol. 2013, 25, 610–616. [Google Scholar] [CrossRef]
- Rothkamm, K.; Lloyd, D. Established and emerging methods of biological dosimetry. In Comprehensive Biomedical Physics; Brahme, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 7, pp. 289–310. [Google Scholar]
- Borgmann, K.; Röper, B.; El-Awady, R.A.; Brackrock, S.; Bigalke, M.; Dörk, T.; Alberti, W.; Dikomey, E.; Dahm-Daphi, J. Indicators of late normal tissue response after radiotherapy for head and neck cancer: Fibroblasts, lymphocytes, genetics, DNA repair and chromosome aberrations. Radiother. Oncol. 2002, 64, 141–152. [Google Scholar] [CrossRef]
- Chua, M.L.K.; Somaiah, N.; A’Hern, R.; Davies, S.; Gothard, L.; Yarnold, J.; Rothkamm, K. Residual DNA and chromosomal damage in ex vivo irradiated blood lymphocytes correlated with late normal tissue response to breast radiotherapy. Radiother. Oncol. 2011, 99, 362–366. [Google Scholar] [CrossRef]
- Beaton, L.A.; Marro, L.; Samiee, S.; Malone, S.; Grimes, S.; Malone, K.; Wilkins, R.C. Investigating chromosome damage using fluorescent in situ hybridization to identify biomarkers of radiosensitivity in prostate cancer patients. Int. J. Radiat. Biol. 2013, 89, 1087–1093. [Google Scholar] [CrossRef]
- Marková, E.; Somsedíková, A.; Vasilyev, S.; Pobijaková, M.; Lacková, A.; Lukačko, P.; Belyaev, I. DNA repair foci and late apoptosis/necrosis in peripheral blood lymphocytes of breast cancer patients undergoing radiotherapy. Int. J. Radiat. Biol. 2015, 91, 934–945. [Google Scholar]
- Qvarnström, F.; Simonsson, M.; Nyman, J.; Hermansson, I.; Book, M.; Johansson, K.-A.; Turesson, I. Double strand break induction and kinetics indicate preserved hypersensitivity in keratinocytes to subtherapeutic doses for 7 weeks of radiotherapy. Radiother. Oncol. 2017, 122, 163–169. [Google Scholar] [CrossRef]
- Talbot, C.J.; Veldwijk, M.R.; Azria, D.; Batini, C.; Bierbaum, M.; Brengues, M.; Chang-Claude, J.; Johnson, K.; Keller, A.; Smith, S.; et al. Multi-centre technical evaluation of the radiation-induced lymphocyte apoptosis assay as a predictive test for radiotherapy toxicity. Clin. Transl. Radiat. Oncol. 2019, 18, 1–8. [Google Scholar] [CrossRef]
- Kulka, U.; Abend, M.; Ainsbury, E.; Badie, C.; Barquinero, J.F.; Barrios, L.; Beinke, C.; Bortolin, E.; Cucu, A.; De Amicis, A.; et al. RENEB–Running the European Network of biological dosimetry and physical retrospective dosimetry. Int. J. Radiat. Biol. 2017, 93, 2–14. [Google Scholar] [CrossRef]
- RENEB Strategic Research Agenda. November 2015. Available online: http://www.reneb.net/wp-content/uploads/2017/10/reneb-sra_november-2015_2.pdf (accessed on 5 July 2016).
- Gregoire, E.; Ainsbury, E.; Barrios, L.; Bassinet, C.; Fattibene, P.; Kulka, U.; Oestreicher, U.; Pantelias, G.; Terzoudi, G.; Trompier, F.; et al. The harmonization process to set up and maintain an operational biological and physical retrospective dosimetry network: QA QM applied to the RENEB network. Int. J. Radiat. Biol. 2017, 93, 81–86. [Google Scholar] [CrossRef]
- Ainsbury, E.; Badie, C.; Barnard, S.; Manning, G.; Moquet, J.; Abend, M.; Antunes, A.C.; Barrios, L.; Bassinet, C.; Beinke, C.; et al. Integration of new biological and physical retrospective dosimetry methods into EU emergency response plans–joint RENEB and EURADOS inter-laboratory comparisons. Int. J. Radiat. Biol. 2017, 93, 99–109. [Google Scholar] [CrossRef]
- FAST Trialists Group; Agrawal, R.K.; Alhasso, A.; Barrett-Lee, P.J.; Bliss, P.; Bloomfield, D.; Bowen, J.; Brunt, A.M.; Donovan, E.; Emson, M.; et al. First results of the randomised UK FAST Trial of radiotherapy hypofractionation for treatment of early breast cancer (CRUKE/04/015). Radiother. Oncol. 2011, 100, 93–100. [Google Scholar]
- Dearnaley, D.; Syndikus, I.; Mossop, H.; Khoo, V.; Birtle, A.; Bloomfield, D.; Graham, J.; Kirkbride, P.; Logue, J.; Malik, Z.; et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016, 17, 1047–1060. [Google Scholar] [CrossRef]
- World Health Organisation. Guidance on Regulations for the Transport of Infectious Substances; 2009–2010 WHO/HSE/EPR/2008.10; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- Guogytė, K.; Plieskienė, A.; Ladygienė, R.; Vaisiūnas, Ž.; Sevriukova, O.; Janušonis, V.; Žilukas, J. Assessment of correlation between chromosomal radiosensitivity of peripheral blood lymphocytes after in vitro irradiation and normal tissue side effects for cancer patients undergoing radiotherapy. Genome Integr. 2017, 8, 1. [Google Scholar]
- Rothkamm, K.; Barnard, S.; Ainsbury, E.A.; Al-Hafidh, J.; Barquinero, J.-F.; Lindholm, C.; Moquet, J.; Perälä, M.; Roch-Lefèvre, S.; Scherthan, H.; et al. Manual versus automated γ-H2AX foci analysis across five European laboratories: Can this assay be used for rapid biodosimetry in a large scale radiation accident? Mutat. Res. 2013, 756, 170–173. [Google Scholar] [CrossRef]
- Horn, S.; Barnard, S.; Rothkamm, K. Gamma-H2AX-based dose estimation for whole and partial body radiation exposure. PLoS ONE 2011, 6, e25113. [Google Scholar] [CrossRef]
- Lloyd, D.C.; Purrott, R.J.; Dolphin, G.W.; Bolton, D.; Edwards, A.A.; Corp, M.J. The relationship between chromosome aberrations and low LET radiation dose in human lymphocytes. Int. J. Radiat. Biol. 1975, 28, 75–90. [Google Scholar] [CrossRef]
- Thierens, H.; Vral, A.; Vandevoorde, C.; Vandersickle, V.; de Gelder, V.; Romm, H.; Oestreicher, U.; Rothkamm, K.; Barnard, S.; Ainsbury, E.; et al. Is a semi-automated approach indicated in the application of the automated micronucleus assay for triage purposes? Radiat. Prot. Dosimetry 2014, 159, 87–94. [Google Scholar] [CrossRef]
- Ainsbury, E.A.; Lloyd, D.C. Dose estimation software for radiation biodosimetry. Health Phys. 2010, 98, 290–295. [Google Scholar] [CrossRef]
- Willems, P.; August, L.; Slabbert, J.; Romm, H.; Oestreicher, U.; Thierens, H.; Vral, A. Automated micronucleus (MN) scoring for population triage in case of large scale radiation events. Int. J. Radiat. Biol. 2010, 86, 2–11. [Google Scholar] [CrossRef]
- Lloyd, D.C.; Edward, A.A.; Prosser, J.S. Chromosome aberrations induced in human lymphocytes by in-vitro acute X and gamma radiation. Radiat. Prot. Dosimetry 1986, 15, 83–88. [Google Scholar] [CrossRef]
- Moquet, J.; Barnard, S.; Staynova, A.; Lindholm, C.; Monteiro Gil, O.; Martins, V.; Rößler, U.; Vral, A.; Vandevoorde, C.; Wojewódzka, M.; et al. The second gamma-H2AX assay inter-comparison exercise carried out in the framework of the European biodosimetry network (RENEB). Int. J. Radiat. Biol. 2017, 93, 58–64. [Google Scholar] [CrossRef]
- Oestreicher, U.; Samaga, D.; Ainsbury, E.; Antunes, A.C.; Baeyens, A.; Barrios, L.; Beinke, C.; Beukes, P.; Blakely, W.F.; Cucu, A.; et al. RENEB intercomparisons applying the conventional dicentric chromosome assay (DCA). Int. J. Radiat. Biol. 2017, 93, 20–29. [Google Scholar] [CrossRef]
- Lloyd, D.C.; Purrott, R.J.; Reeder, E.J. The incidence of unstable chromosomes aberrations in peripheral blood lymphocytes from unirradiated and occupationally exposed people. Mutat. Res. 1980, 72, 523–532. [Google Scholar] [CrossRef]
- Hoffmann, G.R.; Sayer, A.M.; Littlefield, L.G. Higher frequency of chromosome aberrations in late-arising first-division metaphases than in early-arising metaphases after exposure of human lymphocytes to X-rays in G0. Int. J. Radiat. Biol. 2002, 78, 765–772. [Google Scholar] [CrossRef]
- Hone, P.A.; Edwards, A.A.; Lloyd, D.C.; Moquet, J.E. The yield of radiation-induced chromosomal aberrations in first division human lymphocytes depends on the culture time. Int. J. Radiat. Biol. 2005, 81, 523–529. [Google Scholar] [CrossRef]
- Livingston, G.K.; Wilkins, R.C.; Ainsbury, E.A. Pilot website to support international collaboration for dose assessments in a radiation emergency. Radiat. Meas. 2011, 46, 912–915. [Google Scholar] [CrossRef]
- Romm, H.; Ainsbury, E.; Bajinskis, A.; Barnard, S.; Barquinero, J.F.; Barrios, L.; Beinke, C.; Puig-Casanovas, R.; Deperas-Kaminska, M.; Gregoire, E.; et al. Web-based scoring of the dicentric assay, a collaborative biodosimetric scoring strategy for population triage in large scale radiation accidents. Radiat. Environ. Biophys. 2014, 53, 241–254. [Google Scholar] [CrossRef]
- Romm, H.; Ainsbury, E.A.; Barquinero, J.F.; Barrios, L.; Beinke, C.; Cucu, A.; Moreno Domene, M.; Filippi, S.; Monteiro Gil, O.; Gregoire, E.; et al. Web based scoring is useful for validation and harmonisation of scoring criteria within RENEB. Int. J. Radiat. Biol. 2017, 93, 110–117. [Google Scholar] [CrossRef]
- Depuydt, J.; Baeyens, A.; Barnard, S.; Beinke, C.; Benedek, A.; Beukes, P.; Buraczewska, I.; Darroudi, F.; De Sanctis, S.; Dominguez, I.; et al. RENEB intercomparison exercises analyzing micronuclei (Cytokinesis-block Micronucleus Assay). Int. J. Radiat. Biol. 2017, 93, 36–47. [Google Scholar] [CrossRef]
- Fenech, M. Cytokinesis-block micronucleus cytome assay. Nat. Protoc. 2007, 2, 1084–1104. [Google Scholar] [CrossRef]
- Rothkamm, K.; Horn, S. gamma-H2AX as protein biomarker for radiation exposure. Ann. 1st Super Sanità. 2009, 45, 265–271. [Google Scholar]
- Ainsbury, E.A.; Al-Hafidh, J.; Bajinskis, A.; Barnard, S.; Barquinero, J.F.; Beinke, C.; de Gelder, V.; Gregoire, E.; Jaworska, A.; Lindholm, C.; et al. Inter- and intra-laboratory comparison of a multibiodosimetric approach to triage in a simulated, large scale radiation emergency. Int. J. Radiat. Biol. 2014, 90, 193–202. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moquet, J.; Rothkamm, K.; Barnard, S.; Ainsbury, E. Radiation Biomarkers in Large Scale Human Health Effects Studies. J. Pers. Med. 2020, 10, 155. https://doi.org/10.3390/jpm10040155
Moquet J, Rothkamm K, Barnard S, Ainsbury E. Radiation Biomarkers in Large Scale Human Health Effects Studies. Journal of Personalized Medicine. 2020; 10(4):155. https://doi.org/10.3390/jpm10040155
Chicago/Turabian StyleMoquet, Jayne, Kai Rothkamm, Stephen Barnard, and Elizabeth Ainsbury. 2020. "Radiation Biomarkers in Large Scale Human Health Effects Studies" Journal of Personalized Medicine 10, no. 4: 155. https://doi.org/10.3390/jpm10040155
APA StyleMoquet, J., Rothkamm, K., Barnard, S., & Ainsbury, E. (2020). Radiation Biomarkers in Large Scale Human Health Effects Studies. Journal of Personalized Medicine, 10(4), 155. https://doi.org/10.3390/jpm10040155





