Using Machine Learning to Predict 30-Day Hospital Readmissions in Patients with Atrial Fibrillation Undergoing Catheter Ablation
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
2.2. Outcome
2.3. Demographics
2.4. Data Processing
2.5. Variable Selection
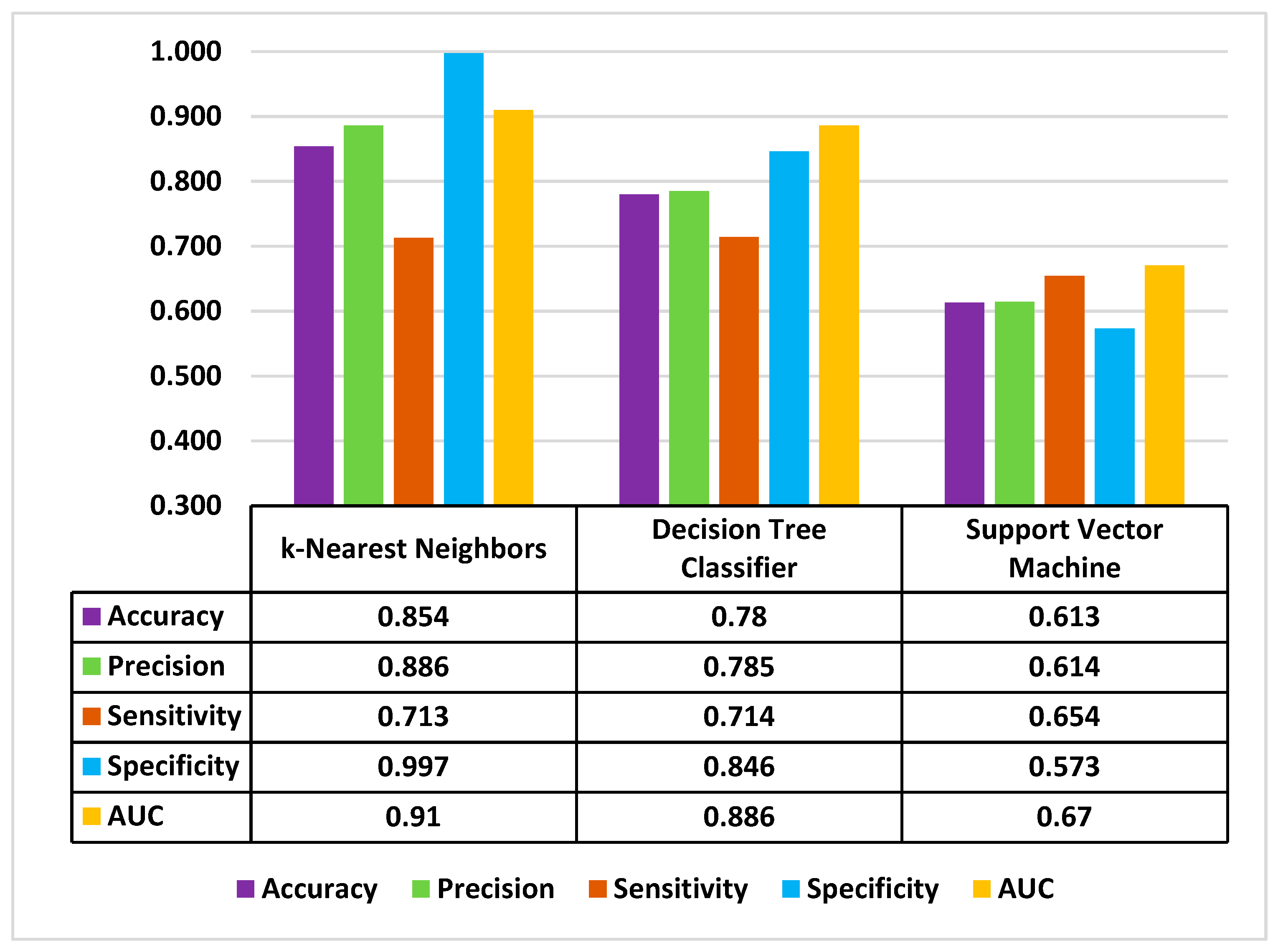
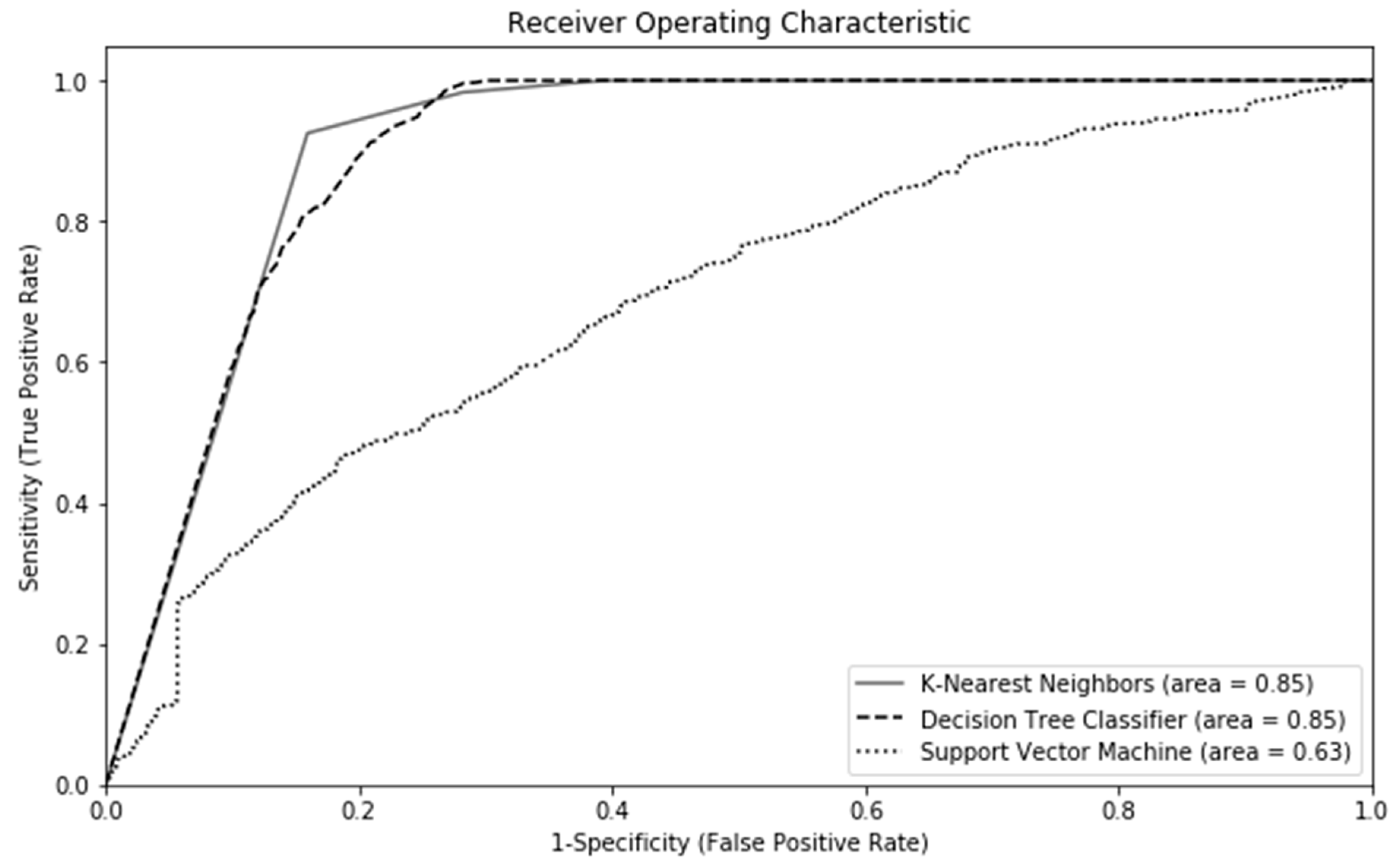
2.6. Machine Learning Algorithims
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thrall, G.; Lane, D.; Carroll, D.; Lip, G.Y. Quality of life in patients with atrial fibrillation: A systematic review. Am. J. Med. 2006, 119, e1–e19. [Google Scholar] [CrossRef]
- Allan, K.S.; Henry, S.; Aves, T.; Banfield, L.; Victor, J.C.; Dorian, P.; Healey, J.S.; Andrade, J.; Carroll, S.; McGillion, M. Comparison of health-related quality of life in patients with atrial fibrillation treated with catheter ablation or antiarrhythmic drug therapy: A systematic review and meta-analysis protocol. BMJ Open 2017, 7, e017577. [Google Scholar] [CrossRef]
- Ball, J.; Carrington, M.J.; McMurray, J.J.; Stewart, S. Atrial fibrillation: Profile and burden of an evolving epidemic in the 21st century. Int. J. Cardiol. 2013, 167, 1807–1824. [Google Scholar] [CrossRef]
- Go, A.S.; Hylek, E.M.; Phillips, K.A.; Chang, Y.; Henault, L.E.; Selby, J.V.; Singer, D.E. Prevalence of diagnosed atrial fibrillation in adults: National implications for rhythm management and stroke prevention: The AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. J. Amer. Med. Assoc. 2001, 285, 2370–2375. [Google Scholar] [CrossRef]
- Tsang, T.S.; Gersh, B.J. Atrial fibrillation: An old disease, a new epidemic. Am. J. Med. 2002, 113, 432–435. [Google Scholar] [CrossRef]
- Coyne, K.S.; Paramore, C.; Grandy, S.; Mercader, M.; Reynolds, M.; Zimetbaum, P. Assessing the direct costs of treating nonvalvular atrial fibrillation in the United States. Value Health 2006, 9, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Piccini, J.P.; Sinner, M.F.; Greiner, M.A.; Hammill, B.G.; Fontes, J.D.; Daubert, J.P.; Ellinor, P.T.; Hernandez, A.F.; Walkey, A.J.; Heckbert, S.R.; et al. Outcomes of Medicare beneficiaries undergoing catheter ablation for atrial fibrillation. Circulation 2012, 126, 2200–2207. [Google Scholar] [CrossRef] [PubMed]
- Thrall, G.; Lip, G.Y.; Carroll, D.; Lane, D. Depression, anxiety, and quality of life in patients with atrial fibrillation. Chest 2007, 132, 1259–1264. [Google Scholar] [CrossRef] [PubMed]
- Weerasooriya, R.; Khairy, P.; Litalien, J.; Macle, L.; Hocini, M.; Sacher, F.; Lellouche, N.; Knecht, S.; Wright, M.; Nault, I.; et al. Catheter ablation for atrial fibrillation: Are results maintained at 5 years of follow-up? J. Am. Coll. Cardiol. 2011, 57, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.U.; Freeman, J.V.; Shilane, D.; Wang, P.J.; Go, A.S.; Hlatky, M.A. Procedural complications, rehospitalizations, and repeat procedures after catheter ablation for atrial fibrillation. J. Am. Coll. Cardiol. 2012, 59, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Bunch, T.J.; Crandall, B.G.; Weiss, J.P.; May, H.T.; Bair, T.L.; Osborn, J.S.; Anderson, J.L.; Muhlestein, J.B.; Horne, B.D.; Lappe, D.L.; et al. Patients treated with catheter ablation for atrial fibrillation have long-term rates of death, stroke, and dementia similar to patients without atrial fibrillation. J. Cardiovasc. Electr. 2011, 22, 839–845. [Google Scholar] [CrossRef]
- Zhuang, J.; Lu, Y.; Tang, K.; Peng, W.; Xu, Y. Influence of body mass index on recurrence and quality of life in atrial fibrillation patients after catheter ablation: A meta-analysis and systematic review. Clin. Cardiol. 2013, 36, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Shameer, K.; Johnson, K.W.; Yahi, A.; Miotto, R.; Li, L.I.; Ricks, D.; Jebakaran, J.; Kovatch, P.; Sengupta, P.P.; Gelijns, S.; et al. Predictive modeling of hospital readmission rates using electronic medical record-wide machine learning: A case-study using Mount Sinai Heart Failure Cohort. Pac. Symp. Biocomput. 2017, 22, 276–287. [Google Scholar] [PubMed]
- Boccuti, C.; Casillas, G. Aiming for Fewer Hospital U-turns: The Medicare Hospital Readmission Reduction Program. Medicare 2017. Available online: http://files.kff.org/attachment/Issue-Brief-Fewer-Hospital-U-turns-The-Medicare-Hospital-Readmission-Reduction-Program (accessed on 6 May 2020).
- McIlvennan, C.K.; Eapen, Z.J.; Allen, L.A. Hospital readmissions reduction program. Circulation 2015, 131, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Opolski, G.; Januszkiewicz, L.; Szczerba, E.; Osińska, B.; Rutkowski, D.; Kalarus, Z.; Kaźmierczak, J. Readmissions and repeat procedures after catheter ablation for atrial fibrillation. Cardiol. J. 2015, 22, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Krumholz, H.M.; Lin, Z.; Keenan, P.S.; Chen, J.; Ross, J.S.; Drye, E.E.; Bernheim, S.M.; Wang, Y.; Bradley, E.H.; Han, L.F.; et al. Relationship between hospital readmission and mortality rates for patients hospitalized with acute myocardial infarction, heart failure, or pneumonia. J. Amer. Med. Assoc. 2013, 309, 587–593. [Google Scholar] [CrossRef]
- Kim, M.H.; Johnston, S.S.; Chu, B.C.; Dalal, M.R.; Schulman, K.L. Estimation of total incremental health care costs in patients with atrial fibrillation in the United States. Circ–Cardiovasc. Qual. 2011, 4, 313–320. [Google Scholar] [CrossRef]
- Hosseinzadeh, A.; Izadi, M.T.; Verma, A.; Precup, D.; Buckeridge, D.L. Assessing the Predictability of Hospital Readmission Using Machine Learning. Presented at the IAAI; 2013. Available online: https://www.researchgate.net/publication/307938384_Assessing_the_predictability_of_hospital_readmission_using_machine_learning (accessed on 6 May 2020).
- Kuo, C.Y.; Yu, L.C.; Chen, H.C.; Chan, C.L. Comparison of Models for the Prediction of Medical Costs of Spinal Fusion in Taiwan Diagnosis-Related Groups by Machine Learning Algorithms. Healthc. Inform. Res. 2018, 24, 29–37. [Google Scholar] [CrossRef]
- Project HCUP Overview of the Nationwide Readmissions Database (NRD). HCUP. 2017.
- Arora, S.; Lahewala, S.; Tripathi, B.; Mehta, V.; Kumar, V.; Chandramohan, D.; Lemor, A.; Dave, M.; Patel, N.; Patel, N.V.; et al. Causes and Predictors of Readmission in Patients With Atrial Fibrillation Undergoing Catheter Ablation: A National Population-Based Cohort Study. J. Am. Heart. Assoc. 2018, 7, e009294. [Google Scholar] [CrossRef] [PubMed]
- Guyon, I.; Elisseeff, A. An introduction to variable and feature selection. J. Mach. Learn. Res. 2003, 3, 1157–1182. [Google Scholar]
- Genuer, R.; Poggi, J.M.; Tuleau-Malot, C. Variable selection using random forests. Pattern. Recogn. Lett. 2010, 31, 2225–2236. [Google Scholar] [CrossRef]
- Wiens, J.; Shenoy, E.S. Machine Learning for Healthcare: On the Verge of a Major Shift in Healthcare Epidemiology. Clin. Infect. Dis. 2018, 66, 149–153. [Google Scholar] [CrossRef]
- Raghupathi, W.; Raghupathi, V. Big data analytics in healthcare: Promise and potential. Health. Inf. Sci. Syst. 2014, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Kotsiantis, S.B.; Zaharakis, I.; Pintelas, P. Supervised machine learning: A review of classification techniques. Informatica 2007, 160, 3–24. [Google Scholar]
- Safavian, S.R.; Landgrebe, D. A survey of decision tree classifier methodology. IEEE T. Syst. Man. Cyb. 1991, 21, 660–674. [Google Scholar] [CrossRef]
- Wu, Y.; Ianakiev, K.; Govindaraju, V. Improved k-nearest neighbor classification. Pattern. Recogn. 2002, 35, 2311–2318. [Google Scholar] [CrossRef]
- Chapelle, O.; Haffner, P.; Vapnik, V.N. Support vector machines for histogram-based image classification. IEEE T. Neural. Networ. 1999, 10, 1055–1064. [Google Scholar] [CrossRef]
- Furey, T.S.; Cristianini, N.; Duffy, N.; Bednarski, D.W.; Schummer, M.; Haussler, D. Support vector machine classification and validation of cancer tissue samples using microarray expression data. Bioinformatics 2000, 16, 906–914. [Google Scholar] [CrossRef]
- Noseworthy, P.A.; Kapa, S.; Haas, L.R.; Van Houten, H.; Deshmuk, A.J.; Mulpuru, S.K.; McLeod, C.J.; Asirvatham, S.J.; Friedman, P.A.; Shah, N.D.; et al. Trends and predictors of readmission after catheter ablation for atrial fibrillation, 2009-2013. Am. Heart. J. 2015, 170, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Ziaeian, B.; Fonarow, G.C. The Prevention of Hospital Readmissions in Heart Failure. Prog. Cardiovasc. Dis. 2016, 58, 379–385. [Google Scholar] [CrossRef] [PubMed]
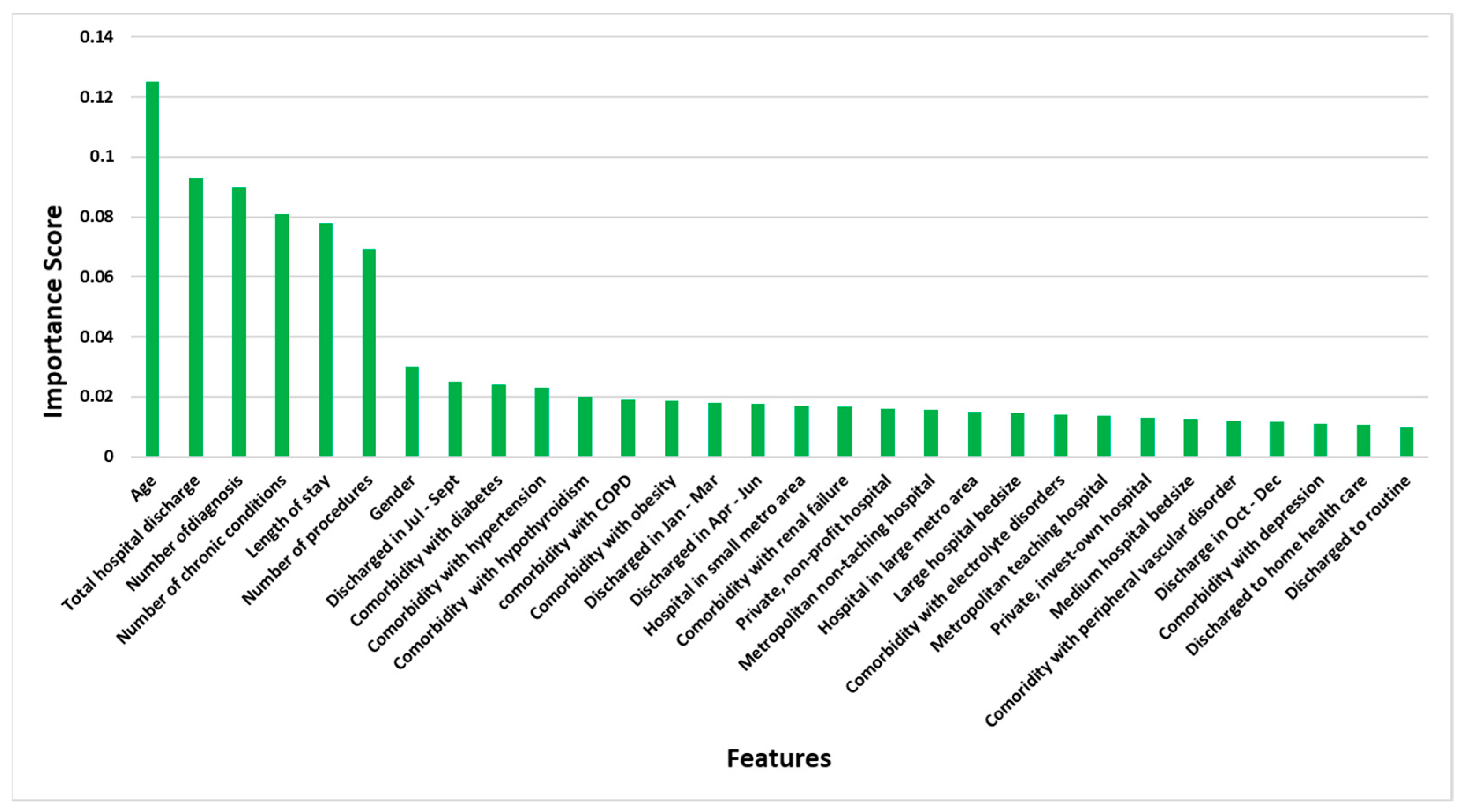
| Variable | Description |
|---|---|
| Age | Age in years at admission |
| Total Hospital Discharges | Total number of hospital discharges the patient has experienced |
| Number of Diagnosis | Total number of diagnosed conditions for the patient |
| Number of Chronic Conditions | Total number of chronic conditions |
| Length of Stay | Length of stay (days) |
| Number of Procedures | Number of procedures on this discharge |
| Gender | Gender (male or female) |
| Discharged in Jul-Sept | Date of discharge was between July and September |
| Comorbidity with Diabetes | Patient has comorbidity with diagnosed diabetes |
| Comorbidity with Hypertension | Patient has comorbidity with diagnosed hypertension |
| Comorbidity with Hypothroidism | Patient has comorbidity with diagnosed hypothyroidism |
| Comorbidity with COPD | Patient has comorbidity with diagnosed COPD |
| Comorbidity with Obesity | Patient has comorbidity with diagnosed obesity |
| Discharged Jan-Mar | Date of discharge was between January and March |
| Discharged in Apr-Jun | Date of discharge was between April and June |
| Hospitality in Small Metro Area | Hospital is located in a small metro area |
| Comorbidity with Renal Failure | Patient has comorbidity with diagnosed renal failure |
| Private, Non-Profit Hospital | Hospital is categorized as a private, non-profit hospital |
| Metropolitan Non-teaching Hospital | Hospital is categorized as a metropolitan non-teaching hospital |
| Hospital in Large Metro Area | Hospital is located in a large metro area |
| Large Hospital Bedsize | Size of hospital beds is large |
| Comorbidity with Electrolyte Disorder | Patient has comorbidity with diagnosed electrolyte disorder |
| Metropolitan Teaching Hospital | Hospital is categorized as a metropolitan teaching hospital |
| Private, Invest-Own Hospital | Hospital is categorized as private, invest-own hospital |
| Medium Hospital Bedsize | Size of hospital beds is medium |
| Comorbidity with Peripheral Vascular Disorder | Patient has comorbidity with peripheral vascular disorder |
| Discharged in Oct-Dec | Date of discharge was between October and December |
| Comorbidity with Depression | Patient has comorbidity with diagnosed depression |
| Discharged to Health Home Care | Patient was discharged from hospital to go home health care. |
| Discharged to Routine | Patient was discharged from hospital to go home |
| Variables | Mean | SD | Median | N | % |
|---|---|---|---|---|---|
| Age (years) | 64.3(64.9) | 11.6(11.4) | 66(66) | 12,634(5872) | 100 |
| Number of Chronic Conditions | 5.2 (5.2) | 2.7(2.7) | 5(5) | 12,634(5872) | 100 |
| Number of Diagnosis | 8.2(8.1) | 4.8(4.7) | 7(7) | 12,634(5872) | 100 |
| Number of Procedures | 3.6(3.6) | 1.6(1.6) | 3(3) | 12,634(5872) | 100 |
| Length of Stay (days) | 2.5(2.4) | 3.0(2.9) | 1(1) | 12,634(5872) | 100 |
| Gender | |||||
| Male | 7906 (3652) | 62.6(62.2) | |||
| Female | 4728(2220) | 37.4(37.8) | |||
| Income | |||||
| 0–25th percentile | 2453(1152) | 19.7(19.9) | |||
| 26th to 50th percentile | 3040(1389) | 24.4(24.0) | |||
| 51st to 75th percentile | 3308(1498) | 26.6(25.9) | |||
| 76th to 100th percentile | 3650(1741) | 29.3(30.1) | |||
| Expected Primary Payer | |||||
| Medicare | 7029(3290) | 55.6(56.0) | |||
| Medicaid | 391(188) | 3.1(3.2) | |||
| Private Insurance | 4726(2201) | 38.0(37.5) | |||
| Self-pay | 77(40) | 0.6(0.7) | |||
| No charge | 26(13) | 0.2(0.2) | |||
| Other | 311(139) | 2.5(2.4) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, M.; Lauren, E.; Hon, E.; Xu, J.; Ruiz-Negrón, B.; Rosales, M.; Li, W.; Barton, T.; O’Brien, J.; Su, W. Using Machine Learning to Predict 30-Day Hospital Readmissions in Patients with Atrial Fibrillation Undergoing Catheter Ablation. J. Pers. Med. 2020, 10, 82. https://doi.org/10.3390/jpm10030082
Hung M, Lauren E, Hon E, Xu J, Ruiz-Negrón B, Rosales M, Li W, Barton T, O’Brien J, Su W. Using Machine Learning to Predict 30-Day Hospital Readmissions in Patients with Atrial Fibrillation Undergoing Catheter Ablation. Journal of Personalized Medicine. 2020; 10(3):82. https://doi.org/10.3390/jpm10030082
Chicago/Turabian StyleHung, Man, Evelyn Lauren, Eric Hon, Julie Xu, Bianca Ruiz-Negrón, Megan Rosales, Wei Li, Tanner Barton, Jacob O’Brien, and Weicong Su. 2020. "Using Machine Learning to Predict 30-Day Hospital Readmissions in Patients with Atrial Fibrillation Undergoing Catheter Ablation" Journal of Personalized Medicine 10, no. 3: 82. https://doi.org/10.3390/jpm10030082
APA StyleHung, M., Lauren, E., Hon, E., Xu, J., Ruiz-Negrón, B., Rosales, M., Li, W., Barton, T., O’Brien, J., & Su, W. (2020). Using Machine Learning to Predict 30-Day Hospital Readmissions in Patients with Atrial Fibrillation Undergoing Catheter Ablation. Journal of Personalized Medicine, 10(3), 82. https://doi.org/10.3390/jpm10030082





