HIV Viral Load Estimation Using Hematocrit Corrected Dried Blood Spot Results on a BioMerieux NucliSENS® Platform
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Setting
2.2.1. National VL Program and its Microbiological Reference Laboratory
2.2.2. VL Testing Procedure
VL Specimen Collection Procedure
Processing
2.3. Study Population
2.4. Variables, Sources of Data, and Data Collection
2.5. Data Analysis
2.6. Ethics Approval
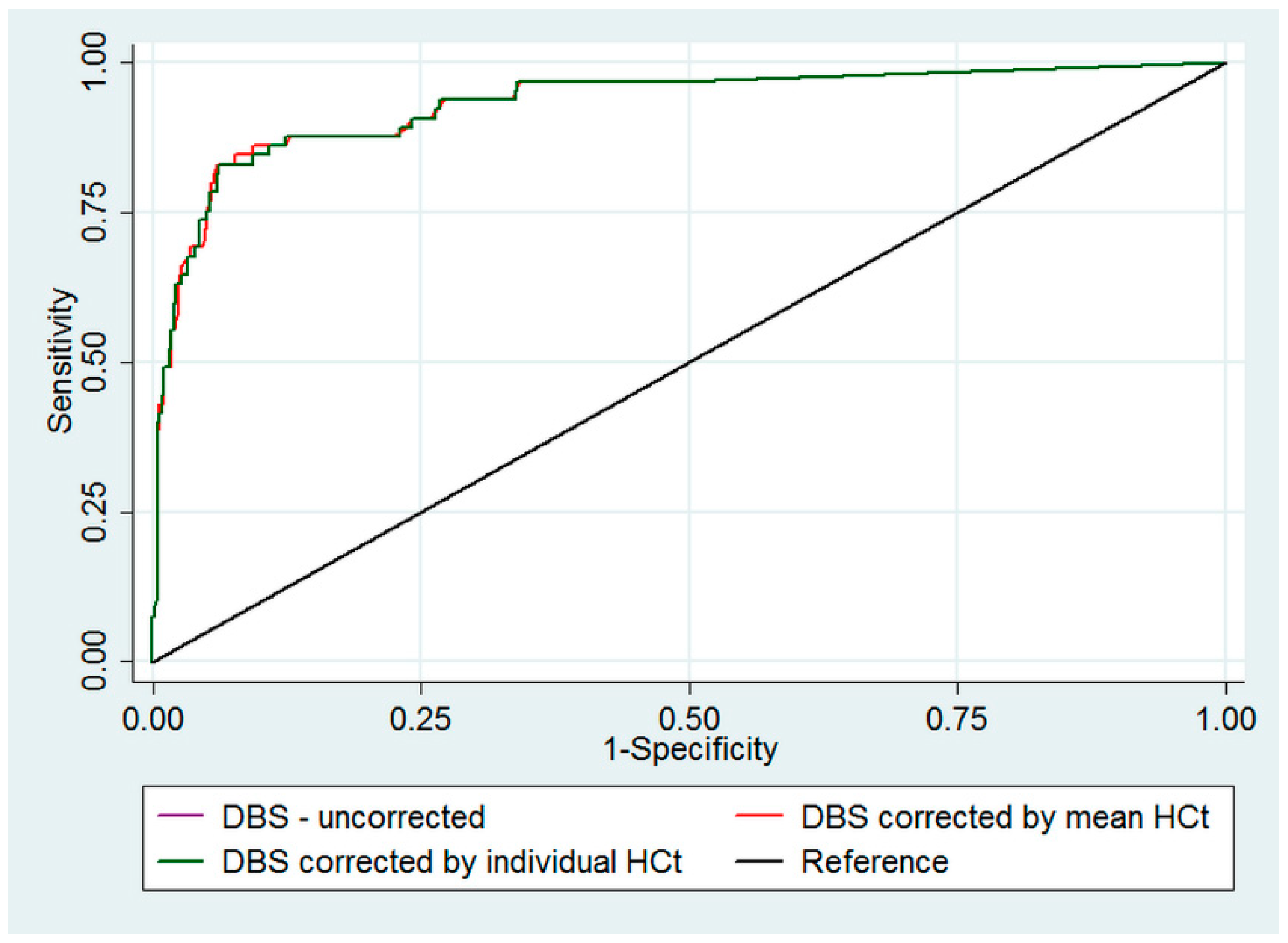
3. Results
4. Discussion
4.1. Limitations
4.2. Key Findings and Implications
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- UNAIDS. 90-90-90 An Ambitious Treatment Target to Help End the AIDS Epidemic; UNAIDS: Geneva, Switzerland, 2014. [Google Scholar]
- World Health Organization (WHO). Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection: Recommendations for a Public Health Approach; WHO: Geneva, Switzerland, 2016; ISBN 9789241549684. [Google Scholar]
- World Health Organization (WHO). Technical and Operational Considerations for Implementing HIV Viral Load Testing: Interim Technical Update; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Roberts, T.; Cohn, J.; Bonner, K.; Hargreaves, S. Scale-up of Routine Viral Load Testing in Resource-Poor Settings: Current and Future Implementation Challenges. Clin. Infect. Dis. 2016, 62, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Johannessen, A.; Trøseid, M.; Calmy, A. Dried blood spots can expand access to virological monitoring of HIV treatment in resource-limited settings. J. Antimicrob. Chemother. 2009, 64, 1126–1129. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Monleau, M.; Butel, C.; Delaporte, E.; Boillot, F.; Peeters, M. Effect of storage conditions of dried plasma and blood spots on HIV-1 RNA quantification and PCR amplification for drug resistance genotyping. J. Antimicrob. Chemother. 2010, 65, 1562–1566. [Google Scholar] [CrossRef] [PubMed]
- Pannus, P.; Claus, M.; Gonzalez, M.M.P.; Ford, N.; Fransen, K. Sensitivity and specificity of dried blood spots for HIV-1 viral load quantification: A laboratory assessment of 3 commercial assays. Medicine 2016, 95, e5475. [Google Scholar] [CrossRef] [PubMed]
- Smit, P.W.; Sollis, K.A.; Fiscus, S.; Ford, N.; Vitoria, M.; Essajee, S.; Barnett, D.; Cheng, B.; Crowe, S.M.; Denny, T.; et al. Systematic review of the use of dried blood spots for monitoring HIV viral load and for early infant diagnosis. PLoS ONE 2014, 9, e86461. [Google Scholar] [CrossRef] [PubMed]
- Erba, F.; Brambilla, D.; Ceffa, S.; Ciccacci, F.; Luhanga, R.; Sidumo, Z.; Palombi, L.; Mancinelli, S.; Marazzi, M.C.; Andreotti, M.; et al. Measurement of viral load by the automated Abbott real-time HIV-1 assay using dried blood spots collected and processed in Malawi and Mozambique. South African Med. J. 2015, 105, 1036–1038. [Google Scholar] [CrossRef] [PubMed]
- Zmbabwe National Statistical Agency (ZIMSTAT). Inter-censal Demographic Survey 2017; Zmbabwe National Statistical Agency (ZIMSTAT): Harare, Zimbabwe, 2017. [Google Scholar]
- Ministry of Health and Child Care (MOHCC) Zimbabwe. Zimbabwe Population-Based HIV Impact Assessment; Ministry of Health and Child Care (MOHCC) Zimbabwe: Harare, Zimbabwe, 2017.
- Biomerieux diagnostics NucliSENS easyMAG. Available online: https://www.biomerieux-diagnostics.com/nuclisensr-easymagr (accessed on 16 July 2019).
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.; Arnold, M.; Bryan, P.; Duggan, J.; James, C.A.; Li, W.; Lowes, S.; Matassa, L.; Olah, T.; Timmerman, P.; et al. Implementing dried blood spot sampling for clinical pharmacokinetic determinations: considerations from the IQ Consortium Microsampling Working Group. AAPS J. 2015, 17, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Garrido, C.; Zahonero, N.; Corral, A.; Arredondo, M.; Soriano, V.; de Mendoza, C. Correlation between Human Immunodeficiency Virus Type 1 (HIV-1) RNA Measurements Obtained with Dried Blood Spots and Those Obtained with Plasma by Use of Nuclisens EasyQ HIV-1 and Abbott RealTime HIV Load Tests. J. Clin. Microbiol. 2009, 47, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- van Deursen, P.; Oosterlaken, T.; Andre, P.; Verhoeven, A.; Bertens, L.; Anne, M.; Ligeon, V.; De Jong, J. Measuring human immunodeficiency virus type 1 RNA loads in dried blood spot specimens using NucliSENS EasyQ HIV-1 v2.0. J. Clin. Virol. 2010, 47, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, M.; Pirillo, M.; Guidotti, G.; Ceffa, S.; Paturzo, G.; Germano, P.; Luhanga, R.; Chimwaza, D.; Mancini, M.G.; Marazzi, M.C.; et al. Correlation between HIV-1 viral load quantification in plasma, dried blood spots, and dried plasma spots using the Roche COBAS Taqman assay. J. Clin. Virol. 2010, 47, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Laprise, C.; de Pokomandy, A.; Baril, J.-G.; Dufresne, S.; Trottier, H. Virologic Failure Following Persistent Low-level Viremia in a Cohort of HIV-Positive Patients: Results From 12 Years of Observation. Clin. Infect. Dis. 2013, 57, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Elvstam, O.; Medstrand, P.; Yilmaz, A.; Isberg, P.-E.; Gisslén, M.; Björkman, P. Virological failure and all-cause mortality in HIV-positive adults with low-level viremia during antiretroviral treatment. PLoS ONE 2017, 12, e0180761. [Google Scholar] [CrossRef] [PubMed]
| Intraclass Correlation (ICC) of Plasma with | ICC * | (95% CI) | p-Value |
|---|---|---|---|
| All specimen (n = 517) | |||
| Uncorrected DBS | 0.640 | (0.586, 0.688) | <0.001 |
| Mean hematocrit corrected DBS | 0.859 | (0.835, 0.809) | <0.001 |
| Individual hematocrit corrected DBS | 0.809 | (0.777, 0.837) | <0.001 |
| Subgroup-plasma result ≥ 1000 (n = 65) | |||
| Uncorrected DBS | 0.635 | (0.462, 0.761) | <0.001 |
| Mean hematocrit corrected DBS | 0.857 | (0.769, 0.912) | <0.001 |
| Individual hematocrit corrected DBS | 0.805 | (0.694, 0.878) | <0.001 |
| Subgroup-plasma result < 1000 (n = 452) ^ | |||
| Uncorrected DBS | 0.000 ^ | (−0.091, 0.091) | 0.500 |
| Mean hematocrit corrected DBS | 0.000 ^ | (−0.091, 0.091) | 0.500 |
| Individual hematocrit corrected DBS | 0.000 ^ | (−0.091, 0.091) | 0.500 |
| DBS HIV-RNA Result | Total | Plasma HIV-RNA | Sensitivity (95% CI) | Specificity (95% CI) | PPV (95% CI) | NPV (95% CI) | ||
|---|---|---|---|---|---|---|---|---|
| ≥1000 | <1000 | Total | ||||||
| 65 | 452 | 517 | ||||||
| Uncorrected DBS | ≥1000 | 44 | 16 | 60 | 67.7 (54.8–78.5) | 96.5 (94.2–97.9) | 73.3 (60.1–83.5) | 95.4 (92.9–97.1) |
| <1000 | 21 | 436 | 457 | |||||
| Mean hematocrit corrected DBS | ≥1000 | 52 | 26 | 78 | 80.0 (67.9–88.5) | 94.3 (91.6–96.1) | 66.7 (55.0–76.7) | 97.0 (94.9–98.3) |
| <1000 | 13 | 426 | 439 | |||||
| Individual hematocrit corrected DBS | ≥1000 | 52 | 27 | 79 | 80.0 (67.9–88.5) | 94.0 (91.3–95.0) | 65.8 (54.2–74.9) | 97.0 (94.8–98.3) |
| <1000 | 13 | 425 | 438 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nyagupe, C.; Shewade, H.D.; Ade, S.; Timire, C.; Tweya, H.; Vere, N.; Chipuka, S.; Sisya, L.; Gumbo, H.; Ditima, E.; et al. HIV Viral Load Estimation Using Hematocrit Corrected Dried Blood Spot Results on a BioMerieux NucliSENS® Platform. Diagnostics 2019, 9, 86. https://doi.org/10.3390/diagnostics9030086
Nyagupe C, Shewade HD, Ade S, Timire C, Tweya H, Vere N, Chipuka S, Sisya L, Gumbo H, Ditima E, et al. HIV Viral Load Estimation Using Hematocrit Corrected Dried Blood Spot Results on a BioMerieux NucliSENS® Platform. Diagnostics. 2019; 9(3):86. https://doi.org/10.3390/diagnostics9030086
Chicago/Turabian StyleNyagupe, Charles, Hemant Deepak Shewade, Serge Ade, Collins Timire, Hannock Tweya, Norah Vere, Sandra Chipuka, Lucia Sisya, Hlanai Gumbo, Ezekiel Ditima, and et al. 2019. "HIV Viral Load Estimation Using Hematocrit Corrected Dried Blood Spot Results on a BioMerieux NucliSENS® Platform" Diagnostics 9, no. 3: 86. https://doi.org/10.3390/diagnostics9030086
APA StyleNyagupe, C., Shewade, H. D., Ade, S., Timire, C., Tweya, H., Vere, N., Chipuka, S., Sisya, L., Gumbo, H., Ditima, E., & Zinyowera, S. (2019). HIV Viral Load Estimation Using Hematocrit Corrected Dried Blood Spot Results on a BioMerieux NucliSENS® Platform. Diagnostics, 9(3), 86. https://doi.org/10.3390/diagnostics9030086





