Current and Emerging Methods of Antibiotic Susceptibility Testing
Abstract
1. Introduction
2. Phenotypic AST Methods
2.1. Diffusion
2.2. Dilution
2.3. Etest
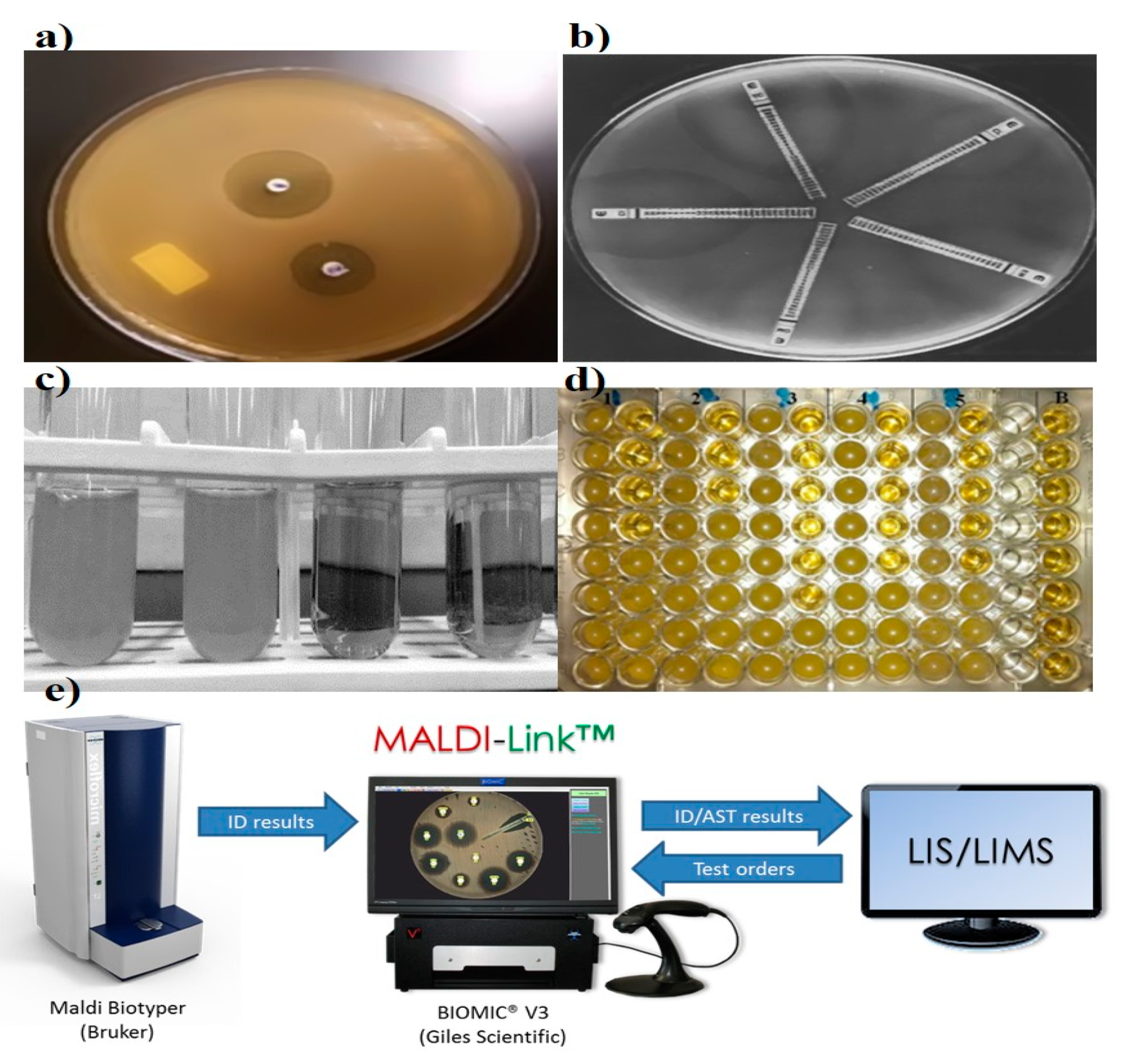
2.4. Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS)
2.5. Automated Systems
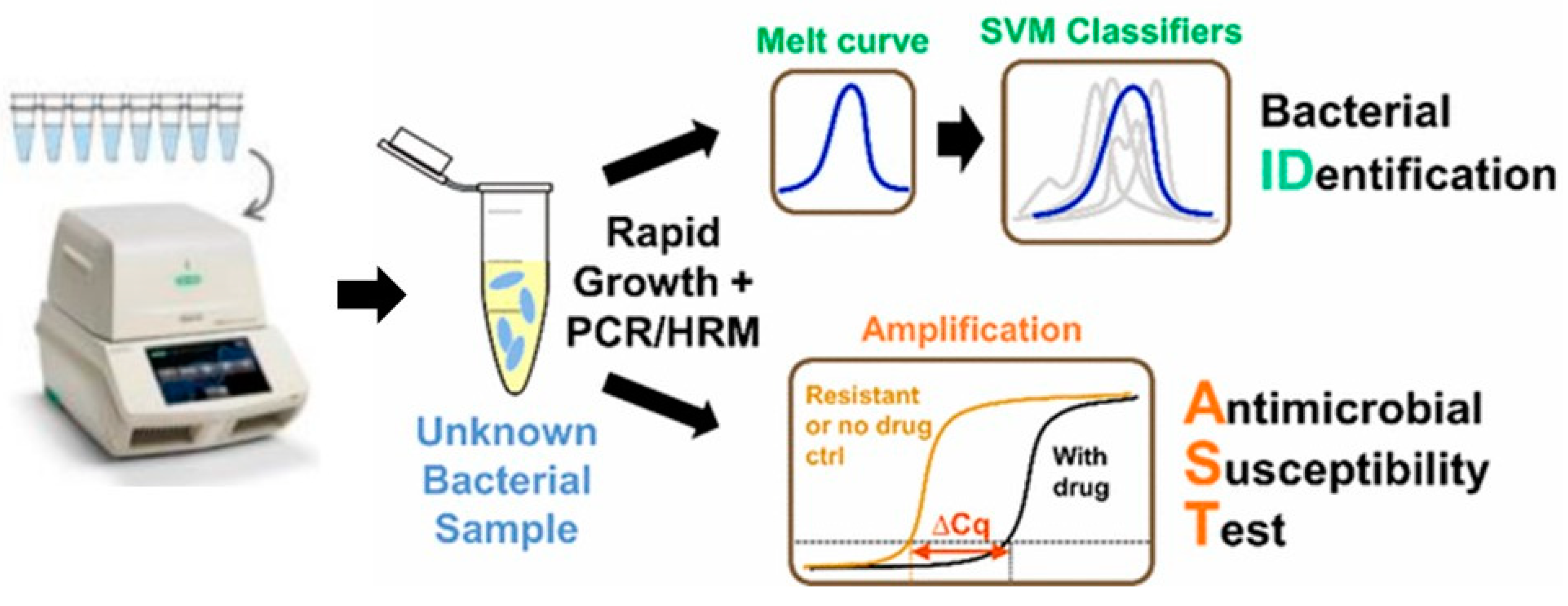
3. Genotypic AST Methods
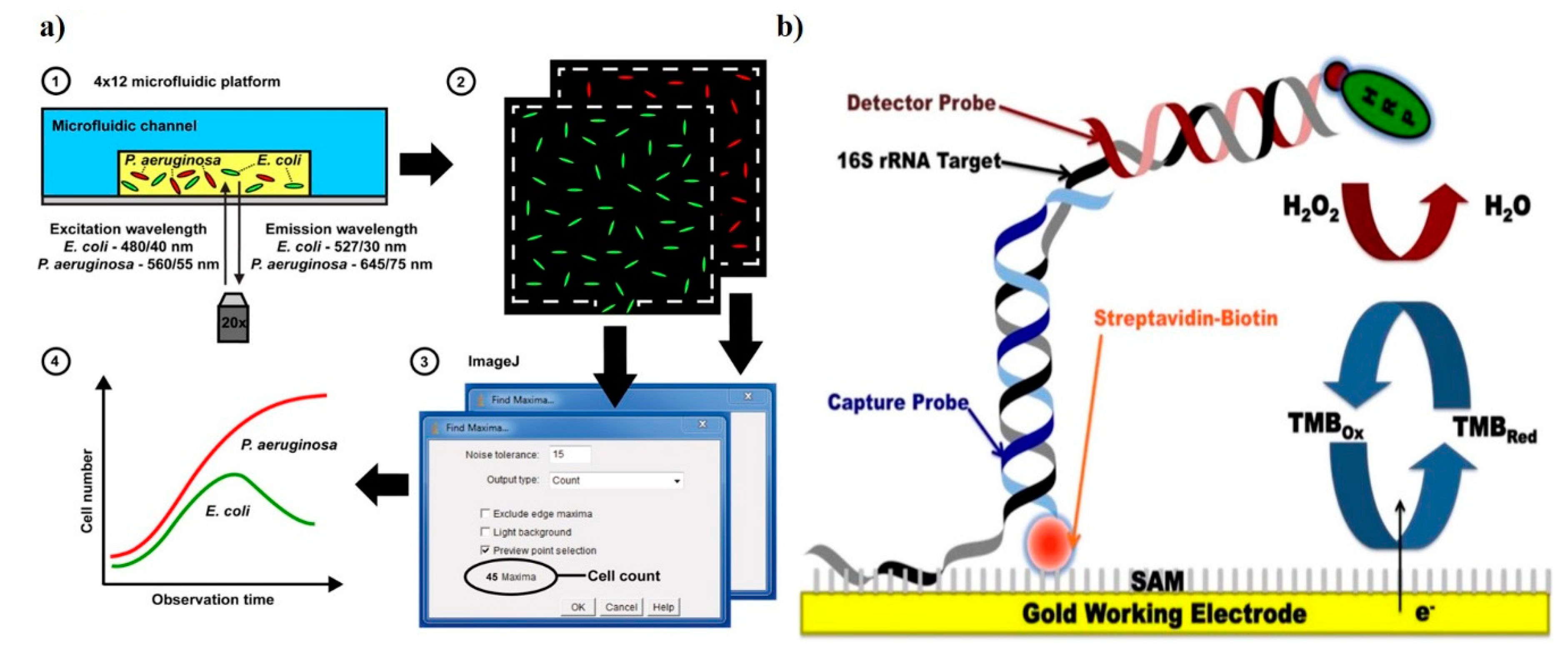
4. Emerging Methods for AST
5. Challenges and Future Perspective
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Blair, J.M.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015, 13, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Martínez, J.L. Antibiotics and Antibiotic Resistance Genes in Natural Environments. Science 2008, 321, 365–367. [Google Scholar] [CrossRef] [PubMed]
- Lalitha, M. Manual on antimicrobial susceptibility testing. In Performance Standards for Antimicrobial Testing: Twelfth Informational Supplement; CLSI: Wayne, PA, USA, 2004; Volume 56238, pp. 454–456. [Google Scholar]
- Van Boeckel, T.P.; Gandra, S.; Ashok, A.; Caudron, Q.; Grenfell, B.T.; Levin, S.A.; Laxminarayan, R. Global antibiotic consumption 2000 to 2010: An analysis of national pharmaceutical sales data. Lancet Infect. Dis. 2014, 14, 742–750. [Google Scholar] [CrossRef]
- Lee, C.-R.; Cho, I.H.; Jeong, B.C.; Lee, S.H. Strategies to Minimize Antibiotic Resistance. Int. J. Environ. Res. Public Health 2013, 10, 4274–4305. [Google Scholar] [CrossRef]
- Ayukekbong, J.A.; Ntemgwa, M.; Atabe, A.N. The threat of antimicrobial resistance in developing countries: Causes and control strategies. Antimicrob. Resist. Infect. Control 2017, 6, 47. [Google Scholar] [CrossRef]
- Brogan, D.M.; Mossialos, E. A critical analysis of the review on antimicrobial resistance report and the infectious disease financing facility. Glob. Health 2016, 12, 8. [Google Scholar] [CrossRef]
- Viens, A.M.; Littmann, J. Is Antimicrobial Resistance a Slowly Emerging Disaster? Public Health Ethics 2015, 8, 255–265. [Google Scholar] [CrossRef]
- Kassim, A.; Omuse, G.; Premji, Z.; Revathi, G. Comparison of Clinical Laboratory Standards Institute and European Committee on Antimicrobial Susceptibility Testing guidelines for the interpretation of antibiotic susceptibility at a University teaching hospital in Nairobi, Kenya: A cross-sectional study. Ann. Clin. Microbiol. Antimicrob. 2016, 15, 21. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
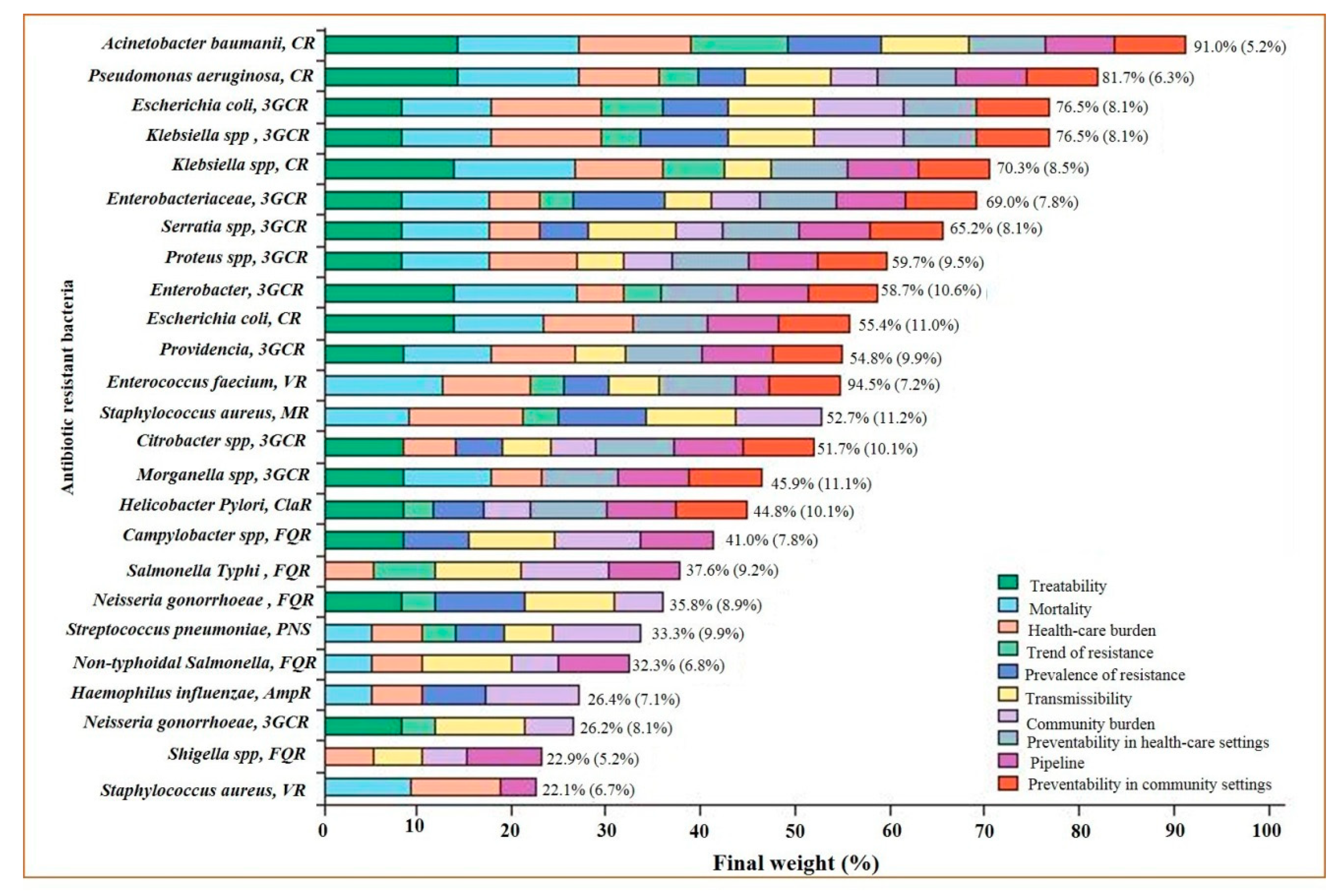
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Reller, L.B.; Weinstein, M.; Jorgensen, J.H.; Ferraro, M.J. Antimicrobial Susceptibility Testing: A Review of General Principles and Contemporary Practices. Clin. Infect. Dis. 2009, 49, 1749–1755. [Google Scholar] [CrossRef]
- Fleming, A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of B. influenzae. Br. J. Exp. Pathol. 1929, 10, 226. [Google Scholar] [CrossRef]
- Abraham, E.P.; Chain, E.; Fletcher, C.M.; Gardner, A.D.; Heatley, N.G.; Jennings, M.A.; Florey, H.W. Further observations on penicillin. Lancet 1941, 238, 177–189. [Google Scholar] [CrossRef]
- Foster, J.W.; Woodruff, H.B. Microbiological aspects of penicillin: I. Methods of assay. J. Bacteriol. 1943, 46, 187. [Google Scholar]
- Vincent, J.G.; Vincent, H.W.; Morton, J. Filter Paper Disc Modification of the Oxford Cup Penicillin Determination. Proc. Soc. Exp. Biol. Med. 1944, 55, 162–164. [Google Scholar] [CrossRef]
- Gavin, J.J. Analytical Microbiology: II. The Diffusion Methods. Appl. Microbiol. 1957, 5, 25. [Google Scholar]
- Hoyt, R.E.; Levine, M.G. A Method for determining Sensitivity to Penicillin and Streptomycin. Science (Washington) 1947, 171. [Google Scholar] [CrossRef]
- Bondi, A., Jr.; Spaulding, E.H.; Smith, D.E.; Dietz, C.C. A routine method for the rapid determination of susceptibility to penicillin and other antibiotics. Am. J. Med. Sci. 1947, 213, 221–225. [Google Scholar] [CrossRef]
- Gould, J.C. The determination of bacterial sensitivity to antibiotics. Edinb. Med. J. 1952, 59, 178–199. [Google Scholar] [PubMed]
- Stokes, E.J. Antibiotic sensitivity testing. Br. Med. J. 1971, 2, 707. [Google Scholar] [CrossRef][Green Version]
- Mayrhofer, S.; Domig, K.J.; Mair, C.; Zitz, U.; Huys, G.; Kneifel, W. Comparison of broth microdilution, Etest, and agar disk diffusion methods for antimicrobial susceptibility testing of Lactobacillus acidophilus group members. Appl. Environ. Microbiol. 2008, 74, 3745–3748. [Google Scholar] [CrossRef]
- World Health Organization. Standardization of Methods for Conducting Microbic Sensitivity Tests: Second Report of the Expert Committee on Antibiotics [Meeting Held in Geneva from 11 to 16 July 1960]; WHO: Geneva, Switzerland, 1961. [Google Scholar]
- Patel, J.B.; Tenover, F.C.; Turnidge, J.D.; Jorgensen, J.H. Susceptibility test methods: Dilution and disk diffusion methods. In Manual of Clinical Microbiology, 10th ed.; American Society of Microbiology: Sterling, VA, USA, 2011; pp. 1122–1143. [Google Scholar]
- Hombach, M.; Zbinden, R.; Bottger, E.C. Standardisation of disk diffusion results for antibiotic susceptibility testing using the sirscan automated zone reader. BMC Microbiol. 2013, 13, 225. [Google Scholar] [CrossRef]
- Murray, P.R. Antibiotic Susceptibility Testing. Part I. Lab. Med. 1983, 14, 345–350. [Google Scholar] [CrossRef][Green Version]
- Felmingham, D.; Brown, D.F. Instrumentation in antimicrobial susceptibility testing. J. Antimicrob. Chemother. 2001, 48, 81–85. [Google Scholar] [CrossRef][Green Version]
- Sageerabanoo, S.; Malini, A.; Mangaiyarkarasi, T.; Hemalatha, G. Phenotypic detection of extended spectrum β-lactamase and Amp-C β-lactamase producing clinical isolates in a Tertiary Care Hospital: A preliminary study. J. Nat. Sci. Biol. Med. 2015, 6, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Sader, H.S.; Pignatari, A.C. E test: A novel technique for antimicrobial susceptibility testing. Sao Paulo Med. J. Rev. Paul. Med. 1994, 112, 635–638. [Google Scholar] [CrossRef] [PubMed]
- Guardino, R.F. Early History of Microbiology and Microbiological Methods; Parenteral Drug Association: Wilmington, DE, USA, 2005. [Google Scholar]
- Rittenberg, S.C. Three Centuries of Microbiology. Hubert, A. Lechevalier and Morris Solotorovsky. McGraw-Hill, New York, 1965. viii + 536 pp. Paper, $4.95. Science 1965, 149, 530–531. [Google Scholar] [CrossRef]
- Poupard, J.A.; Rittenhouse, S.F.; Walsh, L.R. The evolution of antimicrobial susceptibility testing methods. In Antimicrobial Susceptibility Testing; Springer, Plenum Publishing: New York, NY, USA, 1994; pp. 3–14. [Google Scholar]
- Ericsson, H.M.; Sherris, J.C. Antibiotic sensitivity testing. Report of an international collaborative study. Acta Pathol. Microbiol. Scand. 1971, 90. [Google Scholar]
- Jorgensen, J.H.; Turnidge, J.D.; Washington, J.A.; Murray, P.R.; Pfaller, M.A.; Tenover, F.C.; Baron, E.J.; Yolken, R.H. Antibacterial susceptibility tests: Dilution and disk diffusion Methods. In Manual of Clinical Microbiology; Geo. F. Brooks Publisher: Washington, DC, USA, 1999. [Google Scholar]
- Phelps, E.B. A method of calculating the numbers of B. coli from the results of dilution tests. Am. J. Public Hyg. 1908, 18, 141. [Google Scholar]
- McCrady, M. Tables for rapid interpretation of fermentation-tube results. Public Health J. 1918, 9, 201–220. [Google Scholar]
- Reed, J. Report of Advisory Committee on Official Water Standards; Public Health Reports; Sage Publications, Inc.: Teller Road, AK, USA, 1925; Volume 40. [Google Scholar]
- Greenwood, M.; Yule, G.U. On the statistical interpretation of some bacteriological methods employed in water analysis. J. Hyg. 1917, 16, 36. [Google Scholar] [CrossRef]
- Fisher, R.A. Statistical methods for research workers. In Breakthroughs in Statistics; Springer, Oliver & Boyd: New York, NY, USA, 1992; pp. 66–70. [Google Scholar]
- Wheat, P.F. History and development of antimicrobial susceptibility testing methodology. J. Antimicrob. Chemother. 2001, 48, 1–4. [Google Scholar] [CrossRef]
- Tang, Y.-W.; Stratton, C.W. Advanced Techniques in Diagnostic Microbiology; Springer: Nashville, TN, USA, 2012. [Google Scholar]
- Waites, K.B.; Duffy, L.B.; Bébéar, C.M.; Matlow, A.; Talkington, D.F.; Kenny, G.E.; Totten, P.A.; Bade, D.J.; Zheng, X.; Davidson, M.K. Standardized methods and quality control limits for agar and broth microdilution susceptibility testing of Mycoplasma pneumoniae, Mycoplasma hominis, and Ureaplasma urealyticum. J. Clin. Microbiol. 2012, 50, 3542–3547. [Google Scholar] [CrossRef]
- Collins, A.M.; Craig, G.; Zaiman, E.; Roy, T.E. A comparison between disk-plate and tube-dilution methods for antibiotic sensitivity testing of bacteria. Can. J. Public Health/Rev. Can. Sante’e Publique 1954, 45, 430–439. [Google Scholar]
- Picard, J. Applied Veterinary Bacteriology and Mycology: Bacteriological Techniques; University of Pretoria, Afrivip: Pretoria, South Africa, 1990. [Google Scholar]
- Sanchez, M.L.; Jones, R.N. E test, an antimicrobial susceptibility testing method with broad clinical and epidemiologic application. Antimicrob. Newsl. 1992, 8, 1–7. [Google Scholar] [CrossRef]
- Joyce, L.F.; Downes, J.; Stockman, K.; Andrew, J.H. Comparison of five methods, including the PDM Epsilometer test (E test), for antimicrobial susceptibility testing of Pseudomonas aeruginosa. J. Clin. Microbiol. 1992, 30, 2709–2713. [Google Scholar]
- Baker, C.N.; Stocker, S.A.; Culver, D.H.; Thornsberry, C. Comparison of the E Test to agar dilution, broth microdilution, and agar diffusion susceptibility testing techniques by using a special challenge set of bacteria. J. Clin. Microbiol. 1991, 29, 533–538. [Google Scholar]
- Glupczynski, Y.; Labbé, M.; Hansen, W.; Crokaert, F.; Yourassowsky, E. Evaluation of the E test for quantitative antimicrobial susceptibility testing of Helicobacter pylori. J. Clin. Microbiol. 1991, 29, 2072–2075. [Google Scholar]
- Van Dyck, E.; Smet, H.; Piot, P. Comparison of E test with agar dilution for antimicrobial susceptibility testing of Neisseria gonorrhoeae. J. Clin. Microbiol. 1994, 32, 1586–1588. [Google Scholar]
- Schulz, J.E.; Sahm, D.F. Reliability of the E test for detection of ampicillin, vancomycin, and high-level aminoglycoside resistance in Enterococcus spp. J. Clin. Microbiol. 1993, 31, 3336–3339. [Google Scholar]
- Cantón, R.; Livermore, D.M.; Morosini, M.I.; Díaz-Regañón, J.; Rossolini, G.M. Etest® versus broth microdilution for ceftaroline MIC determination with Staphylococcus aureus: Results from PREMIUM, a European multicentre study. J. Antimicrob. Chemother. 2017, 72, 431–436. [Google Scholar] [CrossRef]
- Skov, R.; Smyth, R.; Larsen, A.R.; Bolmstrom, A.; Karlsson, A.; Mills, K.; Frimodt-Moller, N.; Kahlmeter, G. Phenotypic detection of methicillin resistance in Staphylococcus aureus by disk diffusion testing and Etest on Mueller-Hinton agar. J. Clin. Microbiol. 2006, 44, 4395–4399. [Google Scholar] [CrossRef] [PubMed]
- Hariharan, H.; Sharma, S.; Chikweto, A.; Matthew, V.; DeAllie, C. Antimicrobial drug resistance as determined by the E-test in Campylobacter jejuni, C. coli, and C. lari isolates from the ceca of broiler and layer chickens in Grenada. Comp. Immunol. Microbiol. Infect. Dis. 2009, 32, 21–28. [Google Scholar] [CrossRef]
- Falagas, M.E.; Karageorgopoulos, D.E. Extended-spectrum β-lactamase-producing organisms. J. Hosp. Infect. 2009, 73, 345–354. [Google Scholar] [CrossRef]
- Frosch, M.; Maiden, M.C.J. Handbook of Meningococcal Disease: Infection Biology, Vaccination, Clinical Management; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Bernardo, K.; Pakulat, N.; Macht, M.; Krut, O.; Seifert, H.; Fleer, S.; Hünger, F.; Krönke, M. Identification and discrimination of Staphylococcus aureus strains using matrix-assisted laser desorption/ionization-time of flight mass spectrometry. Proteomics 2002, 2, 747–753. [Google Scholar] [CrossRef]
- Hrabák, J.; Chudáčková, E.; Walková, R. Matrix-assisted laser desorption ionization–time of flight (MALDI-TOF) mass spectrometry for detection of antibiotic resistance mechanisms: From research to routine diagnosis. Clin. Microbiol. Rev. 2013, 26, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Eberl, T.; Sparbier, K.; Lange, C.; Kostrzewa, M.; Schubert, S.; Wieser, A. Rapid detection of antibiotic resistance based on mass spectrometry and stable isotopes. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 949–955. [Google Scholar] [CrossRef]
- Zimmermann, S.; Burckhardt, I. Development and Application of MALDI-TOF for Detection of Resistance Mechanisms. In MALDI-TOF and Tandem MS for Clinical Microbiology; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 231–248. [Google Scholar]
- Richter, S.S.; Ferraro, M.J. Susceptibility testing instrumentation and computerized expert systems for data analysis and interpretation. In Manual of Clinical Microbiology, 10th ed.; American Society of Microbiology: Sterling, VA, USA, 2011; pp. 1144–1154. [Google Scholar]
- Sellenriek, P.; Holmes, J.; Ferrett, R.; Drury, R.; Storch, G.A. Comparison of MicroScan Walk-Away®, Phoenix™ and VITEK-TWO® Microbiology systems used in the identification and susceptibility testing of bacteria. In Proceedings of the Abstr 105th General Meeting of the American Society for Microbiology, Atlanta, GA, USA, 5–9 June 2005. [Google Scholar]
- Wright, D.N.; Matsen, J.M.; DiPersio, J.R.; Kirk, M.; Saxon, B.; Ficorilli, S.M.; Spencer, H.J. Evaluation of four newer antimicrobial agents in the Avantage susceptibility test system. J. Clin. Microbiol. 1989, 27, 2381–2383. [Google Scholar]
- Wellinghausen, N.; Pietzcker, T.; Poppert, S.; Belak, S.; Fieser, N.; Bartel, M.; Essig, A. Evaluation of the Merlin MICRONAUT System for Rapid Direct Susceptibility Testing of Gram-Positive Cocci and Gram-Negative Bacilli from Positive Blood Cultures. J. Clin. Microbiol. 2007, 45, 789–795. [Google Scholar] [CrossRef] [PubMed]
- Karlowsky, J.A.; Richter, S.S. Antimicrobial susceptibility testing systems. In Manual of Clinical Microbiology, 11th ed.; American Society of Microbiology: Sterling, VA, USA, 2015; pp. 1274–1285. [Google Scholar]
- Cockerill, F.R. Genetic methods for assessing antimicrobial resistance. Antimicrob. Agents Chemother. 1999, 43, 199–212. [Google Scholar] [CrossRef]
- Fluit, A.C.; Visser, M.R.; Schmitz, F.-J. Molecular detection of antimicrobial resistance. Clin. Microbiol. Rev. 2001, 14, 836–871. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.B.; Tang, Y.-W. Basic Concepts of Microarrays and Potential Applications in Clinical Microbiology. Clin. Microbiol. Rev. 2009, 22, 611–633. [Google Scholar] [CrossRef]
- Li, Y.; Fan, P.; Zhou, S.; Zhang, L. Loop-mediated isothermal amplification (LAMP): A novel rapid detection platform for pathogens. Microb. Pathog. 2017, 107, 54–61. [Google Scholar] [CrossRef]
- Athamanolap, P.; Hsieh, K.; Chen, L.; Yang, S.; Wang, T.H. Integrated Bacterial Identification and Antimicrobial Susceptibility Testing Using PCR and High-Resolution Melt. Anal. Chem. 2017, 89, 11529–11536. [Google Scholar] [CrossRef]
- Frye, J.G.; Lindsey, R.L.; Rondeau, G.; Porwollik, S.; Long, F.; McClelland, M.; Jackson, C.R.; Englen, M.D.; Meinersmann, R.J.; Berrang, M.E.; et al. Development of a DNA microarray to detect antimicrobial resistance genes identified in the National Center for Biotechnology Information database. Microb. Drug Resist. 2010, 16, 9–19. [Google Scholar] [CrossRef]
- Gryadunov, D.; Mikhailovich, V.; Lapa, S.; Roudinskii, N.; Donnikov, M.; Pan’kov, S.; Markova, O.; Kuz’min, A.; Chernousova, L.; Skotnikova, O.; et al. Evaluation of hybridisation on oligonucleotide microarrays for analysis of drug-resistant Mycobacterium tuberculosis. Clin. Microbiol. Infect. 2005, 11, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.L.; Hsu, Z.J.; Chang, T.C.; Jou, R. Rapid and accurate detection of rifampin and isoniazid-resistant Mycobacterium tuberculosis using an oligonucleotide array. Clin. Microbiol. Infect. 2014, 20, O542–O549. [Google Scholar] [CrossRef] [PubMed]
- Sackmann, E.K.; Fulton, A.L.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef]
- Park, S.; Zhang, Y.; Lin, S.; Wang, T.H.; Yang, S. Advances in microfluidic PCR for point-of-care infectious disease diagnostics. Biotechnol. Adv. 2011, 29, 830–839. [Google Scholar] [CrossRef]
- Ibrahim, W.A.; Marouf, S.A.; Erfan, A.M.; Nasef, S.A.; Jakee, J.K.E. The occurrence of disinfectant and antibiotic-resistant genes in Escherichia coli isolated from chickens in Egypt. Vet. World 2019, 12, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Chin, C.D.; Laksanasopin, T.; Cheung, Y.K.; Steinmiller, D.; Linder, V.; Parsa, H.; Wang, J.; Moore, H.; Rouse, R.; Umviligihozo, G.; et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 2011, 17, 1015–1019. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Whitesides, G.M.; Carrilho, E. Diagnostics for the Developing World: Microfluidic Paper-Based Analytical Devices; ACS Publications: Sterling, VA, USA, 2009. [Google Scholar]
- Chen, C.H.; Lu, Y.; Sin, M.L.Y.; Mach, K.E.; Zhang, D.D.; Gau, V.; Liao, J.C.; Wong, P.K. Antimicrobial susceptibility testing using high surface-to-volume ratio microchannels. Anal. Chem. 2010, 82, 1012–1019. [Google Scholar] [CrossRef]
- Baltekin, Ö.; Boucharin, A.; Andersson, D.I.; Elf, J. Fast Antibiotic Susceptibility Testing (FASTest) based on single cell growth rate measurements. bioRxiv 2016. [Google Scholar] [CrossRef]
- Webb, J.S.; Barratt, S.R.; Sabev, H.; Nixon, M.; Eastwood, I.M.; Greenhalgh, M.; Handley, P.S.; Robson, G.D. Green fluorescent protein as a novel indicator of antimicrobial susceptibility in Aureobasidium pullulans. Appl. Environ. Microbiol. 2001, 67, 5614–5620. [Google Scholar] [CrossRef]
- Mohan, R.; Sanpitakseree, C.; Desai, A.V.; Sevgen, S.E.; Schroeder, C.M.; Kenis, P.J.A. A microfluidic approach to study the effect of bacterial interactions on antimicrobial susceptibility in polymicrobial cultures. RSC Adv. 2015, 5, 35211–35223. [Google Scholar] [CrossRef]
- Kalashnikov, M.; Lee, J.C.; Campbell, J.; Sharon, A.; Sauer-Budge, A.F. A microfluidic platform for rapid, stress-induced antibiotic susceptibility testing of Staphylococcus aureus. Lab Chip 2012, 12, 4523–4532. [Google Scholar] [CrossRef]
- Elavarasan, T.; Chhina, S.K.; Ash, M.P.; Sankaran, K. Resazurin reduction based colorimetric antibiogram in microfluidic plastic chip. Sens. Actuators B Chem. 2013, 176, 174–180. [Google Scholar] [CrossRef]
- Liu, T.; Lu, Y.; Gau, V.; Liao, J.C.; Wong, P.K. Rapid antimicrobial susceptibility testing with electrokinetics enhanced biosensors for diagnosis of acute bacterial infections. Ann. Biomed. Eng. 2014, 42, 2314–2321. [Google Scholar] [CrossRef]
- Marques, S.M.; Esteves da Silva, J.C. Firefly bioluminescence: A mechanistic approach of luciferase catalyzed reactions. IUBMB Life 2009, 61, 6–17. [Google Scholar] [CrossRef]
- Dong, T.; Zhao, X. Rapid identification and susceptibility testing of uropathogenic microbes via immunosorbent ATP-bioluminescence assay on a microfluidic simulator for antibiotic therapy. Anal. Chem. 2015, 87, 2410–2418. [Google Scholar] [CrossRef]
- Ivančić, V.; Mastali, M.; Percy, N.; Gornbein, J.; Babbitt, J.T.; Li, Y.; Landaw, E.M.; Bruckner, D.A.; Churchill, B.M.; Haake, D.A. Rapid antimicrobial susceptibility determination of uropathogens in clinical urine specimens by use of ATP bioluminescence. J. Clin. Microbiol. 2008, 46, 1213–1219. [Google Scholar] [CrossRef]
- Webster, T.A.; Sismaet, H.J.; Goluch, E.D. Electrochemically monitoring the antibiotic susceptibility of Pseudomonas aeruginosa biofilms. Analyst 2015, 140, 7195–7201. [Google Scholar] [CrossRef]
- Safavieh, M.; Pandya, H.J.; Venkataraman, M.; Thirumalaraju, P.; Kanakasabapathy, M.K.; Singh, A.; Prabhakar, D.; Chug, M.K.; Shafiee, H. Rapid Real-Time Antimicrobial Susceptibility Testing with Electrical Sensing on Plastic Microchips with Printed Electrodes. ACS Appl. Mater. Interfaces 2017, 9, 12832–12840. [Google Scholar] [CrossRef]
- Dailey, P.J.; Osborn, J.; Ashley, E.A.; Baron, E.J.; Dance, D.A.B.; Fusco, D.; Fanello, C.; Manabe, Y.C.; Mokomane, M.; Newton, P.N.; et al. Defining System Requirements for Simplified Blood Culture to Enable Widespread Use in Resource-Limited Settings. Diagnostics 2019, 9, 10. [Google Scholar] [CrossRef]
- Rodriguez, F.D.; Simonsson, P.; Alling, C. A method for maintaining constant ethanol concentrations in cell culture media. Alcohol Alcohol. 1992, 27, 309–313. [Google Scholar]
- Dawson, A.I. Bacterial Variations Induced by Changes in the Composition of Culture Media. J. Bacteriol. 1919, 4, 133–148. [Google Scholar] [PubMed]
- Schreckenberger, P.C.; Binnicker, M.J. Optimizing Antimicrobial Susceptibility Test Reporting. J. Clin. Microbiol. 2011, 49, S15–S19. [Google Scholar] [CrossRef]
- Martinez, J.L.; Baquero, F. Mutation Frequencies and Antibiotic Resistance. Antimicrob. Agents Chemother. 2000, 44, 1771–1777. [Google Scholar] [CrossRef]
- Khan, Z.A.; Siddiqui, M.F.; Park, S. Progress in antibiotic susceptibility tests: A comparative review with special emphasis on microfluidic methods. Biotechnol. Lett. 2019, 41, 221–230. [Google Scholar] [CrossRef]
- van Belkum, A.; Dunne, W.M., Jr. Next-generation antimicrobial susceptibility testing. J. Clin. Microbiol. 2013, 51, 2018–2024. [Google Scholar] [CrossRef]
- Kim, S.; Masum, F.; Jeon, J.S. Recent Developments of Chip-based Phenotypic Antibiotic Susceptibility Testing. BioChip J. 2019, 13, 43–52. [Google Scholar] [CrossRef]
- Huang, X.; Xu, D.; Chen, J.; Liu, J.; Li, Y.; Song, J.; Ma, X.; Guo, J. Smartphone-based analytical biosensors. Analyst 2018, 143, 5339–5351. [Google Scholar] [CrossRef]
- Vergara, A.; Zboromyrska, Y.; Mosqueda, N.; Morosini, M.I.; García-Fernández, S.; Roca, I.; Cantón, R.; Marco, F.; Vila, J. Evaluation of a Loop-Mediated Isothermal Amplification-Based Methodology To Detect Carbapenemase Carriage in Acinetobacter Clinical Isolates. Antimicrob. Agents Chemother. 2014, 58, 7538–7540. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, Z.A.; Siddiqui, M.F.; Park, S. Current and Emerging Methods of Antibiotic Susceptibility Testing. Diagnostics 2019, 9, 49. https://doi.org/10.3390/diagnostics9020049
Khan ZA, Siddiqui MF, Park S. Current and Emerging Methods of Antibiotic Susceptibility Testing. Diagnostics. 2019; 9(2):49. https://doi.org/10.3390/diagnostics9020049
Chicago/Turabian StyleKhan, Zeeshan A., Mohd F. Siddiqui, and Seungkyung Park. 2019. "Current and Emerging Methods of Antibiotic Susceptibility Testing" Diagnostics 9, no. 2: 49. https://doi.org/10.3390/diagnostics9020049
APA StyleKhan, Z. A., Siddiqui, M. F., & Park, S. (2019). Current and Emerging Methods of Antibiotic Susceptibility Testing. Diagnostics, 9(2), 49. https://doi.org/10.3390/diagnostics9020049





