Can 3D RVEF be Prognostic for the Non-Ischemic Cardiomyopathy Patient but Not the Ischemic Cardiomyopathy Patient? A Cardiovascular MRI Study
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miszalski-Jamka, T.; Klimeczek, P.; Tomala, M.; Krupiński, M.; Zawadowski, G.; Noelting, J.; Lada, M.; Sip, K.; Banyś, R.; Mazur, W.; et al. Extent of RV dysfunction and myocardial infarction assessed by CMR are independent outcome predictors early after STEMI treated with primary angioplasty. JACC Cardiovasc. Imaging 2010, 3, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Di Salvo, T.G.; Mathier, M.; Semigran, M.J.; Dec, G.W. Preserved right ventricular ejection fraction predicts exercise capacity and survival in advanced heart failure. J. Am. Coll. Cardiol. 1995, 25, 1143–1153. [Google Scholar] [CrossRef]
- De Groote, P.; Millaire, A.; Foucher-Hossein, C.; Nugue, O.; Marchandise, X.; Ducloux, G.; Lablanche, J.M. Right ventricular ejection fraction is an independent predictor of survival in patients with moderate heart failure. J. Am. Coll. Cardiol. 1998, 32, 948–954. [Google Scholar] [CrossRef]
- Gavazzi, A.; Berzuini, C.; Campana, C.; Inserra, C.; Ponzetta, M.; Sebastiani, R.; Ghio, S.; Recusani, F. Value of right ventricular ejection fraction in predicting short-term prognosis of patients with severe chronic heart failure. J. Heart Lung Transplant. 1997, 16, 774–785. [Google Scholar] [PubMed]
- Beygui, F.; Furber, A.; Delépine, S.; Helft, G.; Metzger, J.-P.; Geslin, P.; Le Jeune, J.J. Routine breath-hold gradient echo MRI-derived right ventricular mass, volumes and function: Accuracy, reproducibility and coherence study. Int. J. Cardiovasc. Imaging 2004, 20, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Bourantas, C.V.; Loh, H.P.; Bragadeesh, T.; Rigby, A.S.; Lukaschuk, E.I.; Garg, S.; Tweddel, A.C.; Alamgir, F.M.; Nikitin, N.P.; Clark, A.L.; et al. Relationship between right ventricular volumes measured by cardiac magnetic resonance imaging and prognosis in patients with chronic heart failure. Eur. J. Heart Fail. 2011, 13, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.A.; Picard, M.H.; Sleeper, L.A.; Thompson, C.R.; Jacobs, A.K.; White, H.D.; Hochman, J.S.; Davidoff, R. Cardiogenic shock: Predictors of outcome based on right and left ventricular size and function at presentation. Coron. Artery Dis. 2005, 16, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Meluzín, J.; Spinarová, L.; Dusek, L.; Toman, J.; Hude, P.; Krejcí, J. Prognostic importance of the right ventricular function assessed by Doppler tissue imaging. Eur. J. Echocardiogr. 2003, 4, 262–271. [Google Scholar] [CrossRef]
- Iglesias-Garriz, I.; Olalla-Gómez, C.; Garrote, C.; López-Benito, M.; Martín, J.; Alonso, D.; Rodríguez, M.A. Contribution of right ventricular dysfunction to heart failure mortality: A meta-analysis. Rev. Cardiovasc. Med. 2012, 13, e62–e69. [Google Scholar] [PubMed]
- Catalano, O.; Antonaci, S.; Opasich, C.; Moro, G.; Mussida, M.; Perotti, M.; Calsamiglia, G.; Frascaroli, M.; Baldi, M.; Cobelli, F. Intra-observer and interobserver reproducibility of right ventricle volumes, function and mass by cardiac magnetic resonance. J. Cardiovasc. Med. 2007, 8, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Grothues, F.; Moon, J.C.; Bellenger, N.G.; Smith, G.S.; Klein, H.U.; Pennell, D.J. Interstudy reproducibility of right ventricular volumes, function, and mass with cardiovascular magnetic resonance. Am. Heart J. 2004, 147, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Rominger, M.B.; Bachmann, G.F.; Pabst, W.; Rau, W.S. Right ventricular volumes and ejection fraction with fast cine MR imaging in breath-hold technique: Applicability, normal values from 52 volunteers, and evaluation of 325 adult cardiac patients. J. Magn. Reson. Imaging 1999, 10, 908–918. [Google Scholar] [CrossRef]
- Richardson, P.; McKenna, W.; Bristow, M.; Maisch, B.; Mautner, B.; O’Connell, J.; Olsen, E.; Thiene, G.; Goodwin, J.; Gyarfas, I.; et al. Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of Cardiomyopathies. Circulation 1996, 93, 841–842. [Google Scholar] [PubMed]
- Juillière, Y.; Barbier, G.; Feldmann, L.; Grentzinger, A.; Danchin, N.; Cherrier, F. Additional predictive value of both left and right ventricular ejection fractions on long-term survival in idiopathic dilated cardiomyopathy. Eur. Heart J. 1999, 18, 276–280. [Google Scholar] [CrossRef]
- Damy, T.; Kallvikbacka-Bennett, A.; Goode, K.; Khaleva, O.; Lewinter, C.; Hobkirk, J.; Nikitin, N.P.; Dubois-Randé, J.L.; Hittinger, L.; Clark, A.L.; et al. Prevalence of, associations with, and prognostic value of tricuspid annular plane systolic excursion (TAPSE) among out-patients referred for the evaluation of heart failure. J. Card. Fail. 2012, 18, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Meluzin, J.; Spinarová, L.; Hude, P.; Krejcí, J.; Kincl, V.; Panovský, R.; Dusek, L. Prognostic importance of various echocardiographic right ventricular functional parameters in patients with symptomatic heart failure. J. Am. Soc. Echocardiogr. 2005, 18, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Sakata, K.; Yoshino, H.; Kurihara, H.; Iwamori, K.; Houshaku, H.; Yanagisawa, A.; Ishikawa, K. Prognostic significance of persistent right ventricular dysfunction as assessed by radionuclide angiocardiography in patients with inferior wall acute myocardial infarction. Am. J. Cardiol. 2000, 85, 939–944. [Google Scholar] [CrossRef]
- Skali, H.; Zornoff, L.A.; Pfeffer, M.A.; Arnold, M.O.; Lamas, G.A.; Moyé, L.A.; Plappert, T.; Rouleau, J.L.; Sussex, B.A.; St John Sutton, M.; et al. Prognostic use of echocardiography 1 year after a myocardial infarction. Am. Heart J. 2005, 150, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Ghio, S.; Recusani, F.; Klersy, C.; Sebastiani, R.; Laudisa, M.L.; Campana, C.; Gavazzi, A.; Tavazzi, L. Prognostic usefulness of the tricuspid annular plane systolic excursion in patients with congestive heart failure secondary to idiopathic or ischemic dilated cardiomyopathy. Am. J. Cardiol. 2000, 85, 837–842. [Google Scholar] [CrossRef]
- La Vecchia, L.; Zanolla, L.; Varotto, L.; Bonanno, C.; Spadaro, G.L.; Ometto, R.; Fontanelli, A. Reduced right ventricular ejection fraction as a marker for idiopathic dilated cardiomyopathy compared with ischemic left ventricular dysfunction. Am. Heart J. 2001, 142, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Kjaergaard, J.; Akkan, D.; Iversen, K.K.; Køber, L.; Torp-Pedersen, C.; Hassager, C. Right ventricular dysfunction as an independent predictor of short- and long-term mortality in patients with heart failure. Eur. J. Heart Fail. 2007, 9, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.D.C.P.; Rocha, M.O.C.; Ribeiro, A.L.P.; Colosimo, E.A.; Rezende, R.A.; Carmo, G.A.; Barbosa, M.M. Right ventricular dysfunction is an independent predictor of survival in patients with dilated chronic Chagas’ cardiomyopathy. Int. J. Cardiol. 2008, 127, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Polak, J.F.; Holman, B.L.; Wynne, J.; Colucci, W.S. Right ventricular ejection fraction: An indicator of increased mortality in patients with congestive heart failure associated with coronary artery disease. J. Am. Coll. Cardiol. 1983, 2, 217–224. [Google Scholar] [CrossRef]
- Juillière, Y. Right ventricular failure in terminale cardiac failure: Prognostic value and evaluation before transplantation assessment. Arch. Mal. Coeur Vaiss. 1996, 89, 23–25. [Google Scholar] [PubMed]
- Sayer, G.T.; Semigran, M.J. Right ventricular performance in chronic congestive heart failure. Cardiol. Clin. 2012, 30, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Otasević, P.; Popović, Z.; Pratali, L.; Vlahović, A.; Vasiljević, J.D.; Nesković, A.N. Right vs. left ventricular contractile reserve in one-year prognosis of patients with idiopathic dilated cardiomyopathy: Assessment by dobutamine stress echocardiography. Eur. J. Echocardiogr. 2005, 6, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Antoniou, C.-K.; Kotrogiannis, I.; Metallinos, G.; Aggelis, A.; Andreou, I.; Brili, S.; Pitsavos, C.; Stefanadis, C. Role of right ventricular systolic function on long-term outcome in patients with newly diagnosed systolic heart failure. Circ. J. 2011, 75, 2176–2181. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.; Filippatos, G.S.; Ahmed, M.I.; Iskandrian, A.E.; Bittner, V.; Perry, G.J.; White, M.; Aban, I.B.; Mujib, M.; Dell’Italia, L.J.; et al. Effects of right ventricular ejection fraction on outcomes in chronic systolic heart failure. Circulation 2010, 121, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Kenchaiah, S.; Larson, M.G.; Benjamin, E.J.; Kupka, M.J.; Ho, K.K.; Murabito, J.M.; Vasan, R.S. Long-term trends in the incidence of and survival with heart failure. N. Engl. J. Med. 2002, 347, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Roger, V.L.; Weston, S.A.; Redfield, M.M.; Hellermann-Homan, J.P.; Killian, J.; Yawn, B.P.; Jacobsen, S.J. Trends in heart failure incidence and survival in a community-based population. J. Am. Med. Assoc. 2004, 292, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sellés, M.; Martínez, E.; Cortés, M.; Prieto, R.; Gallego, L.; Fernández-Avilés, F. Determinants of long-term survival in patients hospitalized for heart failure. J. Cardiovasc. Med. 2010, 11, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Zornoff, L.A.M.; Skali, H.; Pfeffer, M.A.; St John Sutton, M.; Rouleau, J.L.; Lamas, G.A.; Plappert, T.; Rouleau, J.R.; Moyé, L.A.; Lewis, S.J.; et al. Right ventricular dysfunction and risk of heart failure and mortality after myocardial infarction. J. Am. Coll. Cardiol. 2002, 39, 1450–1455. [Google Scholar] [CrossRef]
- Larose, E.; Ganz, P.; Reynolds, H.G.; Dorbala, S.; Di Carli, M.F.; Brown, K.A.; Kwong, R.Y. Right ventricular dysfunction assessed by cardiovascular magnetic resonance imaging predicts poor prognosis late after myocardial infarction. J. Am. Coll. Cardiol. 2007, 49, 855–862. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Ischemic CMX | Non-Ischemic CMX | Significance |
|---|---|---|---|
| Number of patients | 43 | 44 | |
| Mean age ± SD (years) | 65 ± 11 | 51 ± 12 | p < 0.05 |
| Percentage of males | 90% | 68% | |
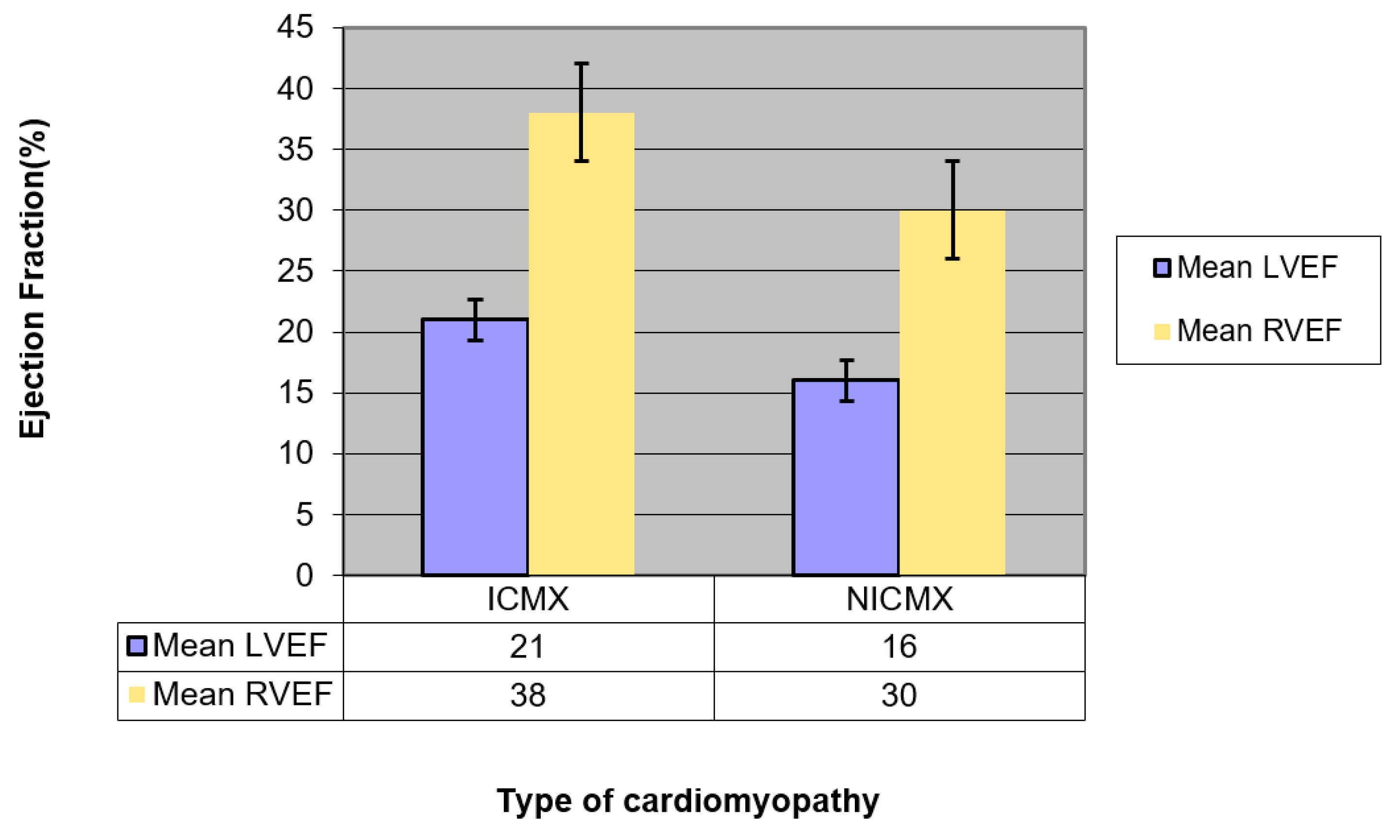
| Mean LVEF (95% confidence interval (CI)) | 21 (19.2–22.8) | 13 (11.2–14.8) | p = 0.001 (LVEF ICMX >> LVEF NICMX) |
| Mean RVEF (95% CI) | 38 (33.8–42.2) | 30 (25.8–34.1) | p = 0.007 (RVEF ICMX >> RVEF NICMX) |
| Parameter | Ischemic CMX | Non-ischemic CMX | Significance |
|---|---|---|---|
| Patients lost from MACE analysis | 10 (24%) | 4 (10%) | p = ns |
| Median, range of follow-up for MACE (days) | 440, 2–2615 | 574, 3–2139 | p = ns |
| Median, interquartile range (IQR) between CMR scan date and first MACE (days) | 41, 2–91 | 64, 19–127 | p = ns |
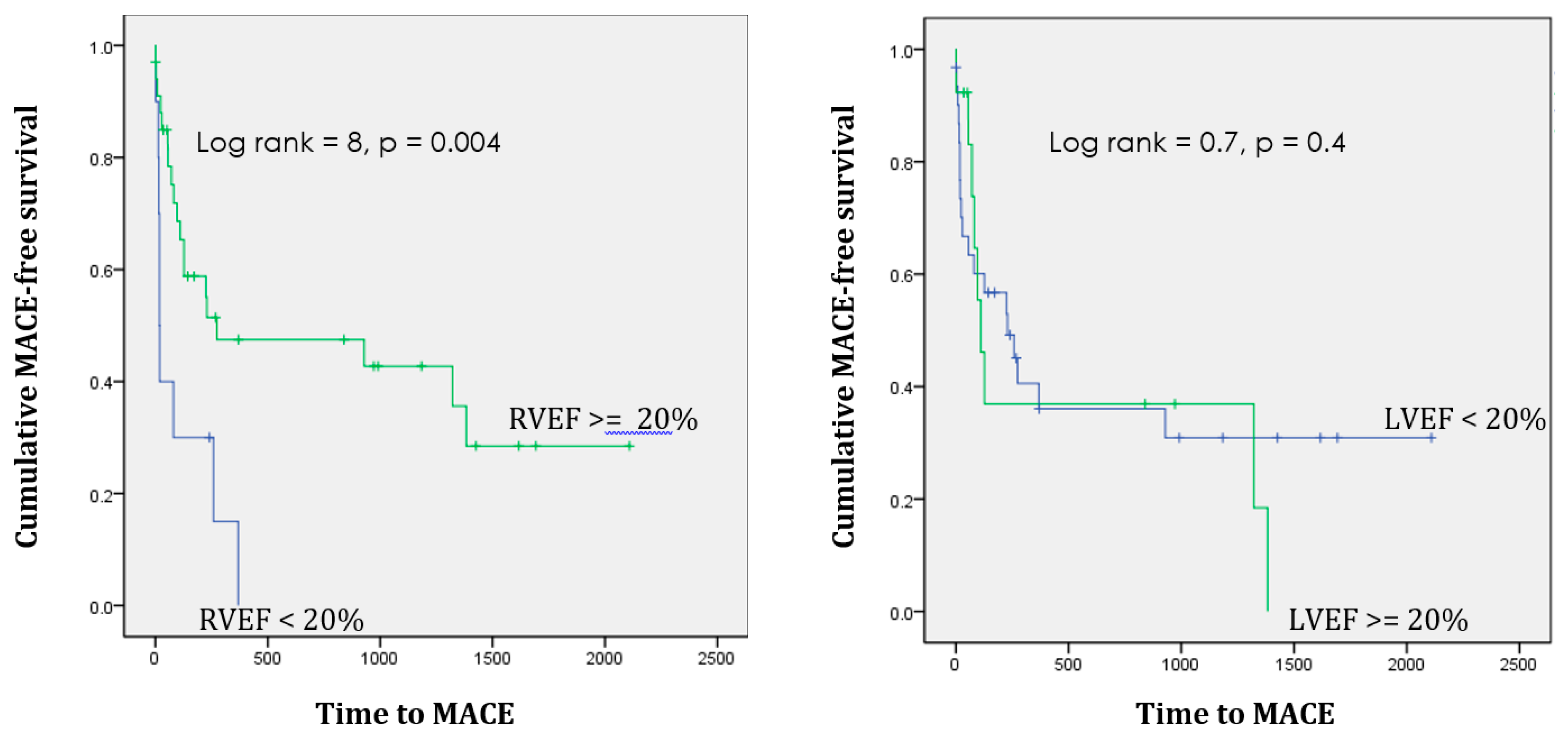
| Number of patients who had MACE | 18 (42%) | 12 (27%) | p = ns |
| Pts lost from death analysis (% of total) | 3 (7%) | 1 (2%) | p = ns |
| Median, range of death follow-up (days) | 1235, 39–2612 | 1098, 58–2238 | p = ns |
| Number of patients who died | 12/40 (30%) | 7/43 (16%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gill, S.S.; Doyle, M.; Thompson, D.V.; Williams, R.; Yamrozik, J.; Grant, S.B.; Biederman, R.W.W. Can 3D RVEF be Prognostic for the Non-Ischemic Cardiomyopathy Patient but Not the Ischemic Cardiomyopathy Patient? A Cardiovascular MRI Study. Diagnostics 2019, 9, 16. https://doi.org/10.3390/diagnostics9010016
Gill SS, Doyle M, Thompson DV, Williams R, Yamrozik J, Grant SB, Biederman RWW. Can 3D RVEF be Prognostic for the Non-Ischemic Cardiomyopathy Patient but Not the Ischemic Cardiomyopathy Patient? A Cardiovascular MRI Study. Diagnostics. 2019; 9(1):16. https://doi.org/10.3390/diagnostics9010016
Chicago/Turabian StyleGill, Sartaj S., Mark Doyle, Diane V. Thompson, Ronald Williams, June Yamrozik, Saundra B. Grant, and Robert W. W. Biederman. 2019. "Can 3D RVEF be Prognostic for the Non-Ischemic Cardiomyopathy Patient but Not the Ischemic Cardiomyopathy Patient? A Cardiovascular MRI Study" Diagnostics 9, no. 1: 16. https://doi.org/10.3390/diagnostics9010016
APA StyleGill, S. S., Doyle, M., Thompson, D. V., Williams, R., Yamrozik, J., Grant, S. B., & Biederman, R. W. W. (2019). Can 3D RVEF be Prognostic for the Non-Ischemic Cardiomyopathy Patient but Not the Ischemic Cardiomyopathy Patient? A Cardiovascular MRI Study. Diagnostics, 9(1), 16. https://doi.org/10.3390/diagnostics9010016





