The Effect of Microdialysis Catheter Insertion on Glutamate and Serotonin Levels in Masseter Muscle in Patients with Myofascial Temporomandibular Disorders and Healthy Controls
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
2.3. Microdialysis
2.4. Analyses of Algesics Substances and Metabolites
2.5. Statistics
3. Results
3.1. Serotonin
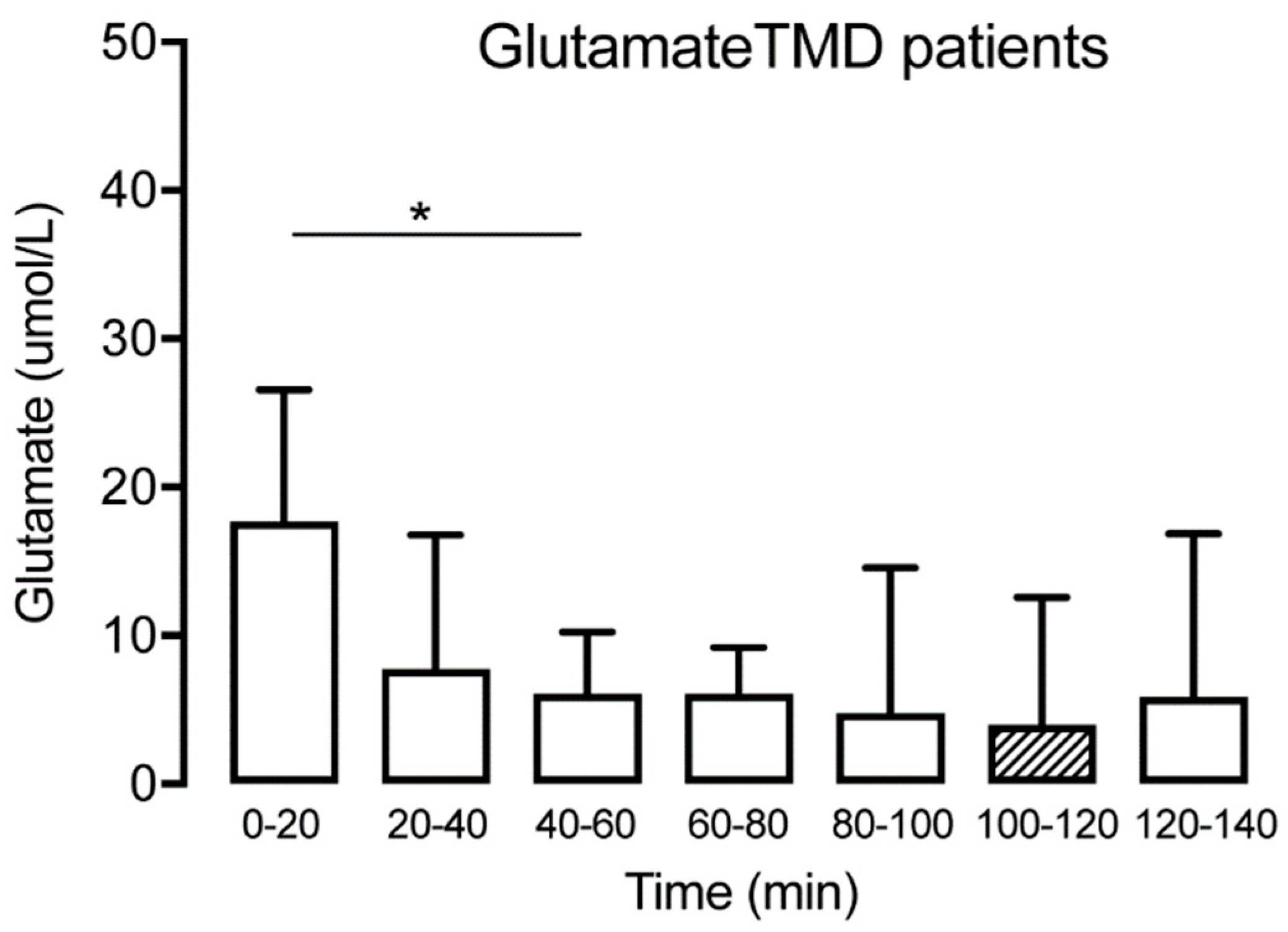
3.2. Glutamate
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- LeResche, L. Epidemiology of temporomandibular disorders: Implications for the investigation of etiologic factors. Crit. Rev. Oral Biol. Med. 1997, 8, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Breivik, H.; Collett, B.; Ventafridda, V.; Cohen, R.; Gallacher, D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur J Pain 2006, 10, 287–333. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.K.; MacBarb, R.F.; Wong, M.E.; Athanasiou, K.A. Temporomandibular Joint Disorders: A Review of Etiology, Clinical Management, and Tissue Engineering Strategies. Int. J. Oral Maxillofac. Implant. 2013, 28, e393–e414. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, S.F.; Huggins, K.H.; LeResche, L.; Von Korff, M.; Howard, J.; Truelove, E.; Sommers, E. Epidemiology of signs and symptoms in temporomandibular disorders: Clinical signs in cases and controls. J. Am. Dent. Assoc. 1990, 120, 273–281. [Google Scholar] [CrossRef]
- Dworkin, S.F.; Le Resche, L. Research diagnostic criteria for temporomandibular disorders: Review, criteria, examinations and specifications, critique. J. Craniomandib. Disord. 1992, 6, 301–355. [Google Scholar]
- Svensson, P.; Graven-Nielsen, T. Craniofacial muscle pain: Review of mechanisms and clinical manifestations. J. Orofac. Pain 2001, 15, 117–145. [Google Scholar]
- Svensson, P.; Jadidi, F.; Arima, T.; Baad-Hansen, L.; Sessle, B.J. Relationships between craniofacial pain and bruxism. J. Oral Rehabil. 2008, 35, 524–547. [Google Scholar] [CrossRef]
- Sessle, B.J. Peripheral and central mechanisms of orofacial pain and their clinical correlates. Minerva Anestesiol. 2005, 71, 117–136. [Google Scholar]
- Dawson, A. Experimental tooth clenching. A model for studying mechanisms of muscle pain. Swed. Dent. J. Suppl. 2013, 228, 9–94. [Google Scholar]
- Dawson, A.; Ghafouri, B.; Gerdle, B.; List, T.; Svensson, P.; Ernberg, M. Effects of experimental tooth clenching on pain and intramuscular release of 5-HT and glutamate in patients with myofascial TMD. Clin. J. Pain 2015, 31, 740–749. [Google Scholar] [CrossRef]
- Ernberg, M.; Lundeberg, T.; Kopp, S. Pain and allodynia/hyperalgesia induced by intramuscular injection of serotonin in patients with fibromyalgia and healthy individuals. Pain 2000, 85, 31–39. [Google Scholar] [CrossRef]
- Kehr, J. Chapter 2.1 New methodological aspects of microdialysis. In Handbook of Behavioral Neuroscience; Westerink, B.H.C., Cremers, T.I.F.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 111–129. [Google Scholar]
- Dawson, A.; Ghafouri, B.; Gerdle, B.; List, T.; Svensson, P.; Ernberg, M. Pain and intramuscular release of algesic substances in the masseter muscle after experimental tooth-clenching exercises in healthy subjects. J. Orofac. Pain 2013, 27, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Ernberg, M.; Hedenberg-Magnusson, B.; Alstergren, P.; Kopp, S. The level of serotonin in the superficial masseter muscle in relation to local pain and allodynia. Life Sci. 1999, 65, 313–325. [Google Scholar] [CrossRef]
- Gerdle, B.; Söderberg, K.; Salvador Puigvert, L.; Rosendal, L.; Larsson, B. Increased interstitial concentrations of pyruvate and lactate in the trapezius muscle of patients with fibromyalgia: A microdialysis study. J. Rehabil. Med. 2010, 42, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri, B.; Larsson, B.K.; Sjörs, A.; Leandersson, P.; Gerdle, B.U. Interstitial concentration of serotonin is increased in myalgic human trapezius muscle during rest, repetitive work and mental stress—An in vivo microdialysis study. Scand. J. Clin. Lab. Investig. 2010, 70, 478–486. [Google Scholar] [CrossRef]
- Rosendal, L.; Blangsted, A.K.; Kristiansen, J.; Søgaard, K.; Langberg, H.; Sjøgaard, G.; Kjaer, M. Interstitial muscle lactate, pyruvate and potassium dynamics in the trapezius muscle during repetitive low-force arm movements, measured with microdialysis. Acta Physiol. Scand. 2004, 182, 379–388. [Google Scholar] [CrossRef]
- Rosendal, L.; Larsson, B.; Kristiansen, J.; Peolsson, M.; Søgaard, K.; Kjaer, M.; Sørensen, J.; Gerdle, B. Increase in muscle nociceptive substances and anaerobic metabolism in patients with trapezius myalgia: Microdialysis in rest and during exercise. Pain 2004, 112, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Peck, C.C.; Murray, G.M.; Gerzina, T.M. How does pain affect jaw muscle activity? The Integrated Pain Adaptation Model. Aust. Dent. J. 2008, 53, 201–207. [Google Scholar] [CrossRef]
- Lund, J.P.; Donga, R.; Widmer, C.G.; Stohler, C.S. The pain-adaptation model: A discussion of the relationship between chronic musculoskeletal pain and motor activity. Can. J. Physiol. Pharmacol. 1991, 69, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Sahara, Y.; Noro, N.; Iida, Y.; Soma, K.; Nakamura, Y. Glutamate receptor subunits GluR5 and KA-2 are coexpressed in rat trigeminal ganglion neurons. J. Neurosci. 1997, 17, 6611–6620. [Google Scholar] [CrossRef]
- Sato, K.; Kiyama, H.; Park, H.T.; Tohyama, M. AMPA, KA and NMDA receptors are expressed in the rat DRG neurones. Neuroreport 1993, 4, 1263–1265. [Google Scholar] [CrossRef] [PubMed]
- Louca, S.; Christidis, N.; Ghafouri, B.; Gerdle, B.; Svensson, P.; List, T.; Ernberg, M. Serotonin, glutamate and glycerol are released after the injection of hypertonic saline into human masseter muscles—A microdialysis study. J. Headache Pain 2014, 15, 89. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Flodgren, G.M.; Crenshaw, A.G.; Alfredson, H.; Fahlström, M.; Hellström, F.B.; Bronemo, L.; Djupsjöbacka, M. Glutamate and prostaglandin E2 in the trapezius muscle of female subjects with chronic muscle pain and controls determined by microdialysis. Eur. J. Pain 2005, 9, 511. [Google Scholar] [CrossRef] [PubMed]
- Ernberg, M.; Castrillon, E.E.; Ghafouri, B.; Larsson, B.; Gerdle, B.; List, T.; Svensson, P. Experimental myalgia induced by repeated infusion of acidic saline into the human masseter muscle does not cause the release of algesic substances. Eur. J. Pain 2013, 17, 539–550. [Google Scholar] [CrossRef]
- Gerdle, B.; Lemming, D.; Kristiansen, J.; Larsson, B.; Peolsson, M.; Rosendal, L. Biochemical alterations in the trapezius muscle of patients with chronic whiplash associated disorders (WAD)—A microdialysis study. Eur. J. Pain 2008, 12, 82–93. [Google Scholar] [CrossRef]
- Larsson, B.; Rosendal, L.; Kristiansen, J.; Sjøgaard, G.; Søgaard, K.; Ghafouri, B.; Abdiu, A.; Kjaer, M.; Gerdle, B. Responses of algesic and metabolic substances to 8 h of repetitive manual work in myalgic human trapezius muscle. Pain 2008, 140, 479–490. [Google Scholar] [CrossRef]
- Cairns, B.E.; Svensson, P.; Wang, K.; Hupfeld, S.; Graven-Nielsen, T.; Sessle, B.J.; Berde, C.B.; Arendt-Nielsen, L. Activation of peripheral NMDA receptors contributes to human pain and rat afferent discharges evoked by injection of glutamate into the masseter muscle. J. Neurophysiol. 2003, 90, 2098–2105. [Google Scholar] [CrossRef]
- Hu, W.P.; Li, X.M.; Wu, J.L.; Zheng, M.; Li, Z.W. Bradykinin potentiates 5-HT3 receptor-mediated current in rat trigeminal ganglion neurons. Acta Pharmacol. Sin. 2005, 26, 428–434. [Google Scholar] [CrossRef][Green Version]
- Ernberg, M. Serotonergic receptor involvement in muscle pain and hyperalgesia. In Fundamentals of Musculoskeletal Pain; Graven-Nielsen, T., Arendt-Nielsen, L., Mense, S., Eds.; IASP Press: Seattle, WA, USA, 2008; pp. 139–155. [Google Scholar]
- Kumar, M.; Chawla, R.; Goyal, M. Topical anesthesia. J. Anaesthesiol. Clin. Pharmacol. 2015, 31, 450–456. [Google Scholar] [CrossRef]
- Monteiro, A.A.; Kopp, S. Estimation of blood flow by 133Xe clearance in human masseter muscle during rest, endurance of isometric contraction, and recovery. Arch. Oral Biol. 1988, 33, 561–565. [Google Scholar] [CrossRef]
- Oliveira-Fusaro, M.C.G.; Clemente-Napimoga, J.T.; Teixeira, J.M.; Torres-Chávez, K.E.; Parada, C.A.; Tambeli, C.H. 5-HT induces temporomandibular joint nociception in rats through the local release of inflammatory mediators and activation of local β adrenoceptors. Pharmacol. Biochem. Behav. 2012, 102, 458–464. [Google Scholar] [CrossRef] [PubMed]
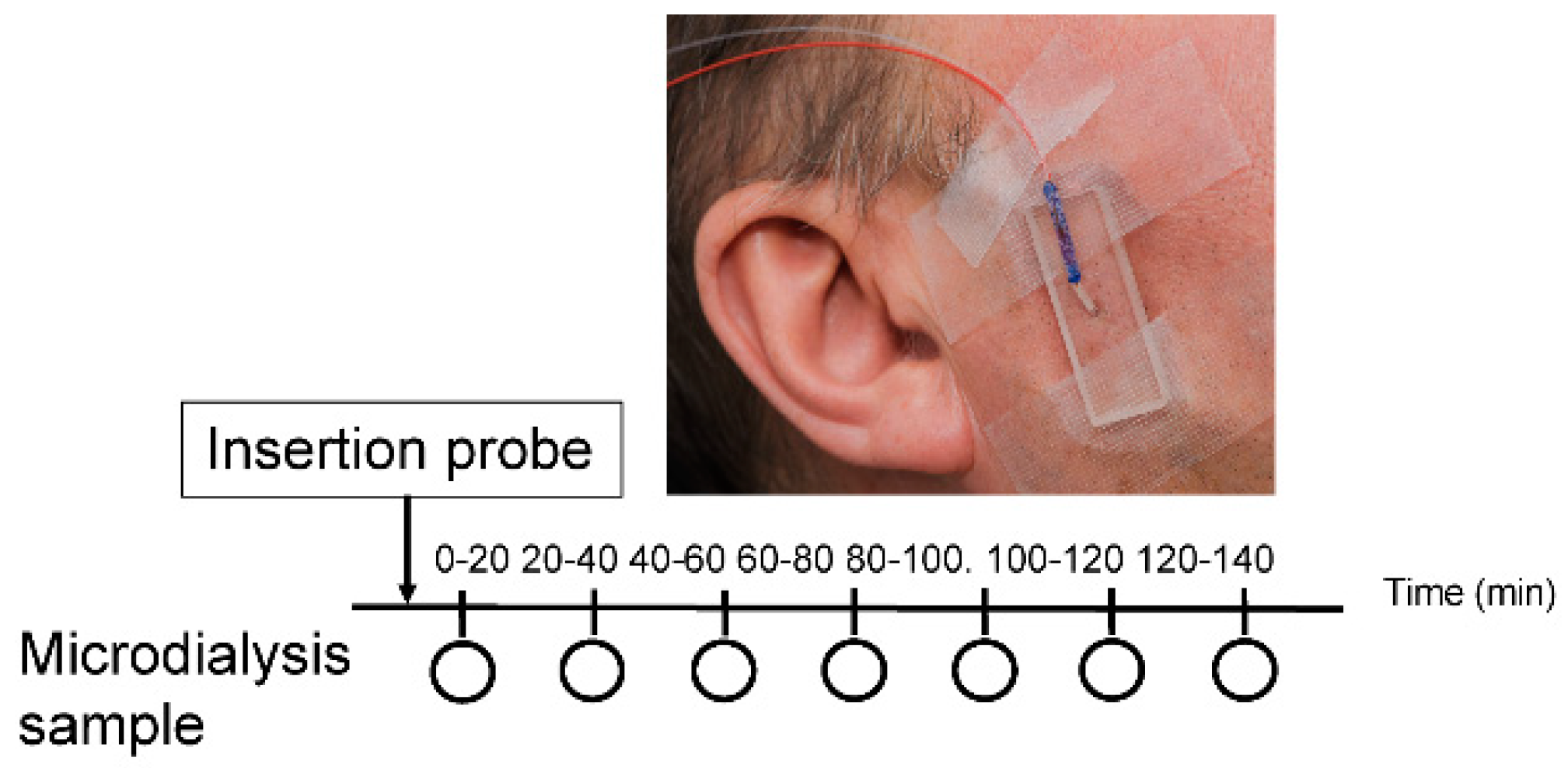
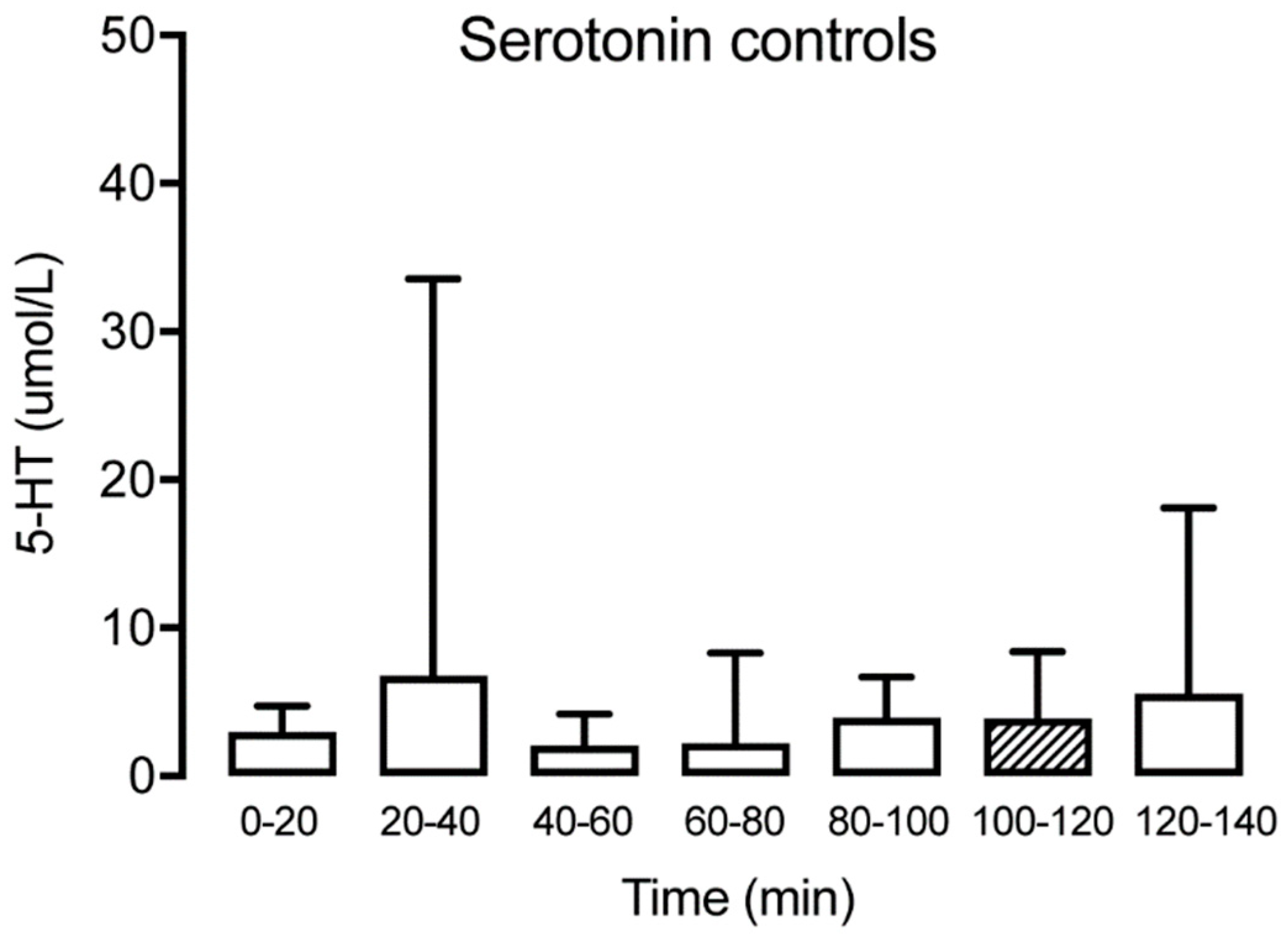
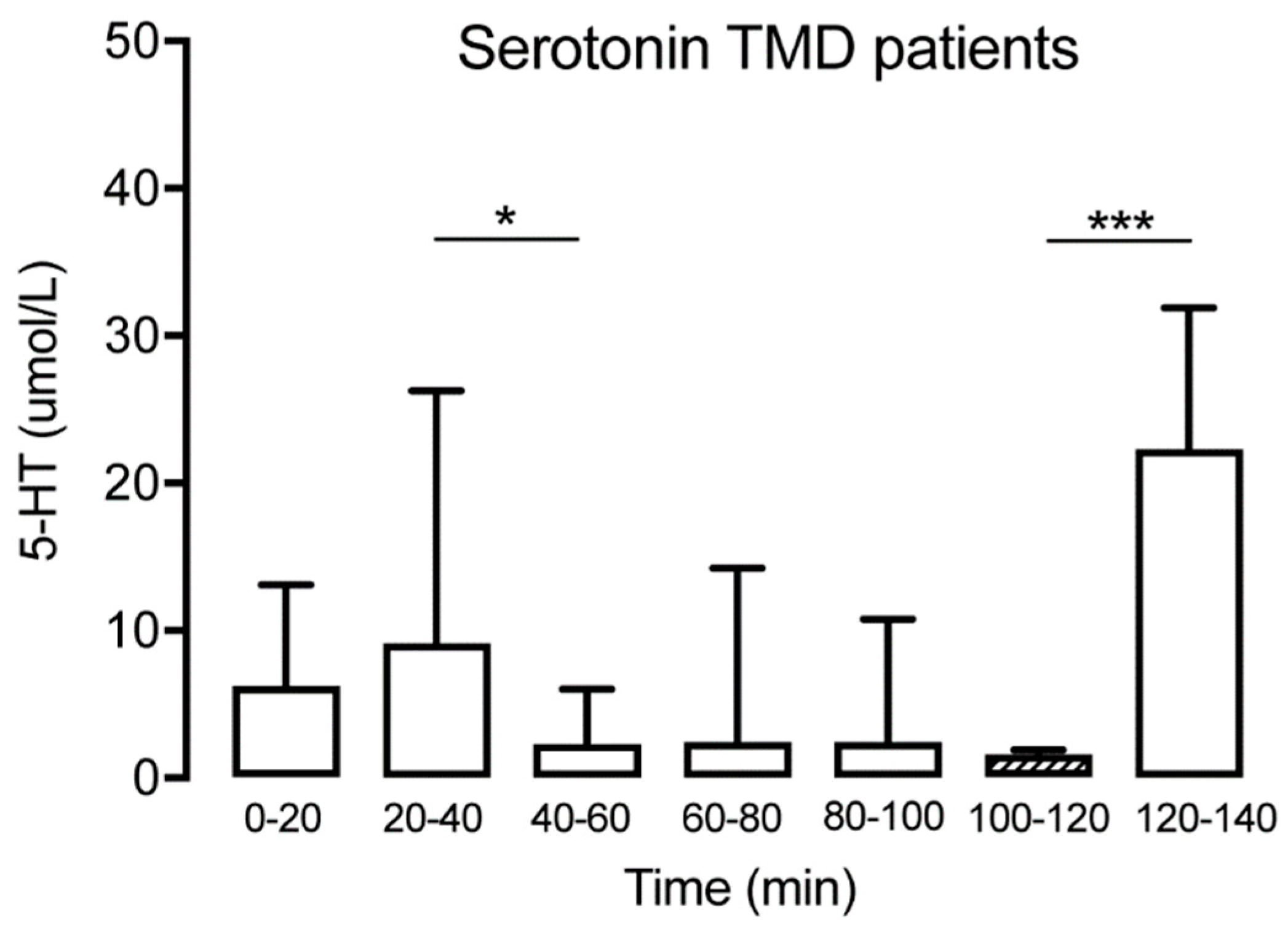
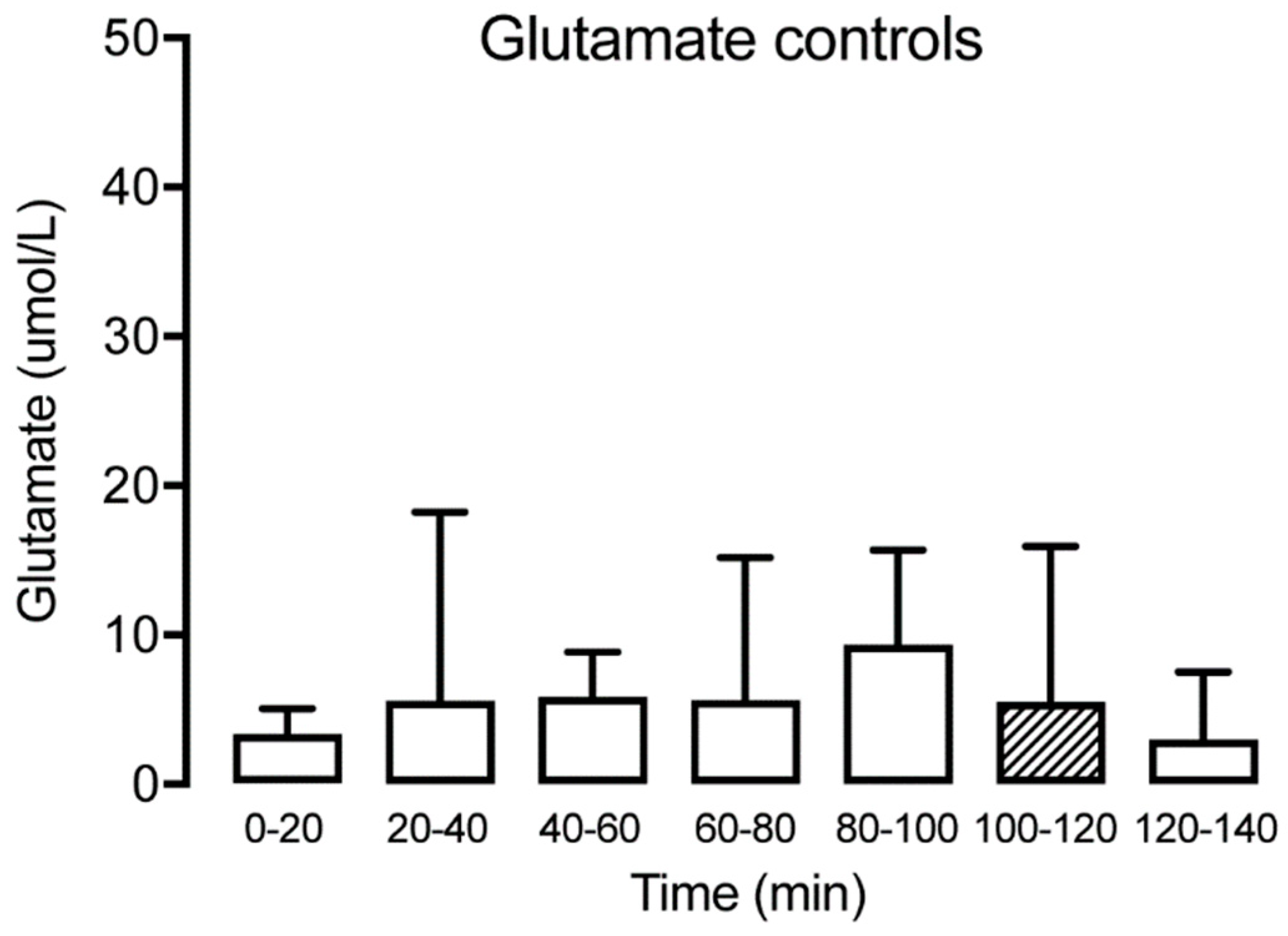
| Substances | Subjects | T0–20 | T20–40 | T40–60 | T60–80 | T80–100 | T100–120 | T120–140 |
|---|---|---|---|---|---|---|---|---|
| Serotonin | Control | 3.6 (1.7) | 30.0 (59.1) | 5.8 (8.2) | 8.5 (12.8) | 6.0 (7.4) | 5.5 (5.0) | 8.0 (7.8) |
| TMD | 8.3 (6.1) | 22.7 (36.2) | 4.9 (6.1) | 12.1 (19.4) | 12.1 (23.2) | 3.0 (4.5) | 27.9 (35.4) | |
| Glutamate | Control | 5.8 (6.2) | 12.6 (17.5) | 7.8 (8.0) | 18.0 (40.3) | 12.9 (13.7) | 12.7 (13.0) | 5.2 (4.5) |
| TMD | 12.0 (3.1) | 13.8 (3.6) | 4.6 (1.2) | 5.6 (1.4) | 11.3 (2.9) | 5.6 (1.5) | 7.2 (1.9) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bajramaj, E.; Häggman-Henrikson, B.; Dawson, A.; Gerdle, B.; Ghafouri, B. The Effect of Microdialysis Catheter Insertion on Glutamate and Serotonin Levels in Masseter Muscle in Patients with Myofascial Temporomandibular Disorders and Healthy Controls. Diagnostics 2019, 9, 14. https://doi.org/10.3390/diagnostics9010014
Bajramaj E, Häggman-Henrikson B, Dawson A, Gerdle B, Ghafouri B. The Effect of Microdialysis Catheter Insertion on Glutamate and Serotonin Levels in Masseter Muscle in Patients with Myofascial Temporomandibular Disorders and Healthy Controls. Diagnostics. 2019; 9(1):14. https://doi.org/10.3390/diagnostics9010014
Chicago/Turabian StyleBajramaj, Ermira, Birgitta Häggman-Henrikson, Andreas Dawson, Björn Gerdle, and Bijar Ghafouri. 2019. "The Effect of Microdialysis Catheter Insertion on Glutamate and Serotonin Levels in Masseter Muscle in Patients with Myofascial Temporomandibular Disorders and Healthy Controls" Diagnostics 9, no. 1: 14. https://doi.org/10.3390/diagnostics9010014
APA StyleBajramaj, E., Häggman-Henrikson, B., Dawson, A., Gerdle, B., & Ghafouri, B. (2019). The Effect of Microdialysis Catheter Insertion on Glutamate and Serotonin Levels in Masseter Muscle in Patients with Myofascial Temporomandibular Disorders and Healthy Controls. Diagnostics, 9(1), 14. https://doi.org/10.3390/diagnostics9010014





