Gestational Age-Specific Biometric and Estimated Fetal Weight Curves in Gastroschisis: A Brazilian Multicenter Cohort Study
Abstract
1. Introduction
2. Methods
2.1. Study Design and Setting
2.2. Inclusion Criteria
2.3. Follow-Up Protocol
2.4. Ultrasound Parameters and Weight Estimation
2.5. Statistical Analysis
3. Results
3.1. Study Population
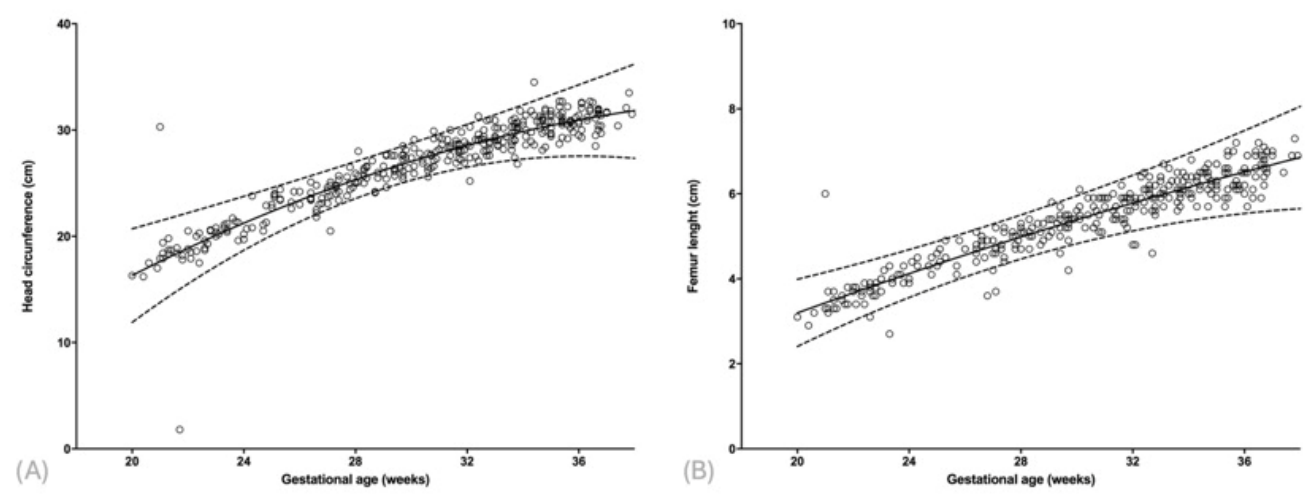
3.2. Gestational Age-Specific Curves
3.3. Comparison with Normative Growth Charts
3.4. Agreement Between EFW and Birth Weight
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Girsen, A.I.; Do, S.; Davis, A.S.; Hintz, S.R.; Desai, A.K.; Mansour, T.; Merritt, T.A.; Oshiro, B.T.; El-Sayed, Y.Y.; Blumenfeld, Y.J. Peripartum and neonatal outcomes of small-for-gestational-age infants with gastroschisis. Prenat. Diagn. 2015, 35, 477–482. [Google Scholar] [CrossRef]
- Perry, H.; Healy, C.; Wellesley, D.; Hall, N.J.; Drewett, M.; Burge, D.M.; Howe, D.T. Intrauterine death rate in gastroschisis following the introduction of an antenatal surveillance program: Retrospective observational study. J. Obstet. Gynaecol. Res. 2017, 43, 492–497. [Google Scholar] [CrossRef]
- Lap, C.C.M.M.; Pistorius, L.R.; Mulder, E.J.H.; Aliasi, M.; Kramer, W.L.M.; Bilardo, C.M.; Cohen-Overbeek, T.E.; Pajkrt, E.; Tibboel, D.; Wijnen, R.M.H.; et al. Ultrasound markers for prediction of complex gastroschisis and adverse outcome: Longitudinal prospective nationwide cohort study. Ultrasound Obstet. Gynecol. 2020, 55, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Netta, D.A.; Wilson, R.D.; Visintainer, P.; Johnson, M.P.; Hedrick, H.L.; Flake, A.W.; Adzick, N.S. Gastroschisis: Growth patterns and a proposed prenatal surveillance protocol. Fetal Diagn. Ther. 2007, 22, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, M.M.; Bennini, J.R.; Nomura, M.L.; Morais, S.S.; Surita, F.G. Fetal growth standards in gastroschisis: Reference values for ultrasound measurements. Prenat. Diagn. 2017, 37, 1327–1334. [Google Scholar] [CrossRef] [PubMed]
- Boute, T.; Rizzo, G.; Mappa, I.; Makatsariya, A.; Toneto, B.R.; Moron, A.F.; Rolo, L.C. Correlation between estimated fetal weight and weight at birth in infants with gastroschisis and omphalocele. J. Matern-Fetal Neonatal Med. 2022, 35, 3070–3075. [Google Scholar] [CrossRef]
- Fisher, J.E.; Tolcher, M.C.; Shamshirsaz, A.A.; Espinoza, J.; Cortes, M.S.; Donepudi, R.; Belfort, M.A.; Nassr, A.A. Accuracy of Ultrasound to Predict Neonatal Birth Weight Among Fetuses with Gastroschisis: Impact on Timing of Delivery. J. Ultrasound Med. 2021, 40, 1383–1389. [Google Scholar] [CrossRef]
- Salomon, L.J.; Alfirevic, Z.; Da Silva Costa, F.; Deter, R.L.; Figueras, F.; Ghi, T.; Glanc, P.; Khalil, A.; Lee, W.; Napolitano, R.; et al. ISUOG Practice Guidelines: Ultrasound assessment of fetal biometry and growth. Ultrasound Obstet. Gynecol. 2019, 53, 715–723. [Google Scholar] [CrossRef]
- Hadlock, F.P.; Harrist, R.B.; Sharman, R.S.; Deter, R.L.; Park, S.K. Estimation of fetal weight with the use of head, body, and femur measurements--a prospective study. Am. J. Obstet. Gynecol. 1985, 151, 333–337. [Google Scholar] [CrossRef]
- Siemer, J.; Hilbert, A.; Hart, N.; Hoopmann, M.; Schneider, U.; Girschick, G.; Müller, A.; Schild, R.L. Specific weight formula for fetuses with abdominal wall defects. Ultrasound Obstet. Gynecol. 2008, 31, 397–400. [Google Scholar] [CrossRef]
- Peixoto, A.B.; da Cunha Caldas, T.M.R.; Dulgheroff, F.F.; Martins, W.P.; Araujo Júnior, E. Fetal biometric parameters: Reference charts for a non-selected risk population from Uberaba, Brazil. J. Ultrason. 2017, 17, 23–29. [Google Scholar]
- Hadlock, F.P.; Harrist, R.B.; Martinez-Poyer, J. In utero analysis of fetal growth: A sonographic weight standard. Radiology 1991, 181, 129–133. [Google Scholar] [CrossRef]
- Altman, D.G.; Chitty, L.S. Charts of fetal size: 1. Methodology. Br. J. Obstet. Gynaecol. 1994, 101, 29–34. [Google Scholar] [CrossRef]
- Martins, W.P.; Nastri, C.O. Interpreting reproducibility results for ultrasound measurements. Ultrasound Obstet. Gynecol. 2014, 43, 479–480. [Google Scholar] [CrossRef] [PubMed]
- Centofanti, S.F.; Brizot, M.d.L.; Liao, A.W.; Francisco, R.P.; Zugaib, M. Fetal Growth Pattern and Prediction of Low Birth Weight in Gastroschisis. Fetal Diagn. Ther. 2015, 38, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Payne, N.R.; Simonton, S.C.; Olsen, S.; Arnesen, M.A.; Pfleghaar, K.M. Growth restriction in gastroschisis: Quantification of its severity and exploration of a placental cause. BMC Pediatr. 2011, 11, 90. [Google Scholar] [CrossRef] [PubMed]
- D’antonio, F.; Virgone, C.; Rizzo, G.; Khalil, A.; Baud, D.; Cohen-Overbeek, T.E.; Kuleva, M.; Salomon, L.J.; Flacco, M.E.; Manzoli, L.; et al. Prenatal risk factors and outcomes in gastroschisis: A meta-analysis. Pediatriccs 2015, 136, e159–e169. [Google Scholar] [CrossRef]
- O’kEeffe, R.; Mulligan, K.; McParland, P.; McAuliffe, F.M.; Mahony, R.; Corcoran, S.; O’cOnnor, C.; Carroll, S.; Walsh, J. Estimating fetal weight in gastroschisis: A 10 year audit of outcomes at the National Maternity Hospital. Int. J. Gynaecol. Obstet. 2024, 166, 1198–1202. [Google Scholar] [CrossRef]
- Zaki, M.N.; A Lusk, L.; Overcash, R.T.; Rao, R.; Truong, Y.N.; Liebowitz, M.; Porto, M. Predicting birth weight in fetuses with gastroschisis. J. Perinatol. 2018, 38, 122–126. [Google Scholar] [CrossRef]
- Spaulding, P.; Edwards, A.; Coombs, P.; Davies-Tuck, M.; Robinson, A. Accuracy of sonographic estimation of weight in fetuses with abdominal wall defects. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, 766–772. [Google Scholar] [CrossRef]
- Di Filippo, D.; Henry, A.; Patel, J.; Jiwane, A.; Welsh, A.W. Fetal abdominal wall defects in an Australian tertiary setting: Contemporary characteristics, ultrasound accuracy, and outcome. J. Matern-Fetal Neonatal Med. 2021, 34, 1269–1276. [Google Scholar] [CrossRef] [PubMed]
- Chaudhury, P.; Haeri, S.; Horton, A.L.; Wolfe, H.M.; Goodnight, W.H. Ultrasound prediction of birthweight and growth restriction in fetal gastroschisis. Am. J. Obstet. Gynecol. 2010, 203, 395.e1–395.e5. [Google Scholar] [CrossRef]
- Nicholas, S.; Tuuli, M.G.; Dicke, J.; Macones, G.A.; Stamilio, D.; Odibo, A.O. Estimation of fetal weight in fetuses with abdominal wall defects: Comparison of 2 recent sonographic formulas to the Hadlock formula. J. Ultrasound Med. 2010, 29, 1069–1074. [Google Scholar] [CrossRef]
- Adams, S.R.; Durfee, S.; Pettigrew, C.; Katz, D.; Jennings, R.; Ecker, J.; House, M.; Benson, C.B.; Wolfberg, A. Accuracy of Sonography to Predict Estimated Weight in Fetuses with Gastroschisis. J. Ultrasound Med. 2012, 31, 1753–1758. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.L.; Powell, M.S.; Wolfe, H.M. Intrauterine growth patterns in fetal gastroschisis. Am. J. Perinatol. 2010, 27, 211–217. [Google Scholar] [CrossRef] [PubMed]




| Variable | Value (n = 116) |
|---|---|
| Maternal age (years) | 21.0 (19.0–38.0) |
| Body mass index (kg/m2) | 24.1 (4.1) |
| Number of pregnancies | 1.0 (1.0–9.0) |
| Number of deliveries | 2.0 (1.0–2.0) |
| Gestational age at first US evaluation (weeks) | 25.3 (20.0–34.8) |
| Biparietal diameter (cm) | 6.1 (4.1–9.0) |
| Occipitofrontal diameter (cm) | 8.3 (1.5) |
| Head circumference (cm) | 22.7 (1.8–31.0) |
| Abdominal circumference (cm) | 19.7 (4.1) |
| Femur length (cm) | 4.5 (2.7–6.7) |
| Amniotic fluid index (cm) | 11.9 (8.8–20.5) |
| Estimated fetal weight (Siemer) (g) | 705.3 (192.3–2441.0) |
| Estimated fetal weight (Hadlock IV) (g) | 712.8 (92.1–2329.0) |
| Gestational age at delivery (weeks) | 37.3 (24.8–40.1) |
| Birth weight (g) | 2434.0 (559.1) |
| Mode of delivery | |
| Vaginal | 14.9% (13/87) |
| Cesarean section | 85.1% (74/87) |
| Sex | |
| Male | 45.1% (37/82) |
| Female | 54.9% (45/82) |
| APGAR score at 1 min | 8.0 (1.0–9.0) |
| APGAR score at 5 min | 9.0 (3.0–10.0) |
| Variable | P5 Equation | P50 Equation | P95 Equation | r | R2 | p-Value |
|---|---|---|---|---|---|---|
| Siemer EFW (g) | Y = 98.57 × GA − 1955 | Y = 135.4 × GA − 2698 | Y = 172.2 × GA − 3441 | 0.91 | 0.837 | <0.0001 |
| Hadlock EFW (g) | Y = 72.51 × GA − 1330 | Y = 119.6 × GA − 2309 | Y = 166.7 × GA − 3289 | 0.86 | 0.728 | <0.0001 |
| Head circumference (cm) | Y = 0.8041 × GA − 0.2119 | Y = 0.8388 × GA + 1.286 | Y = 0.8674 × GA + 2.891 | 0.93 | 0.849 | <0.0001 |
| Femur length (cm) | Y = 0.1733 × GA − 0.548 | Y = 0.2014 × GA − 0.6989 | Y = 0.2294 × GA − 0.8426 | 0.92 | 0.877 | <0.0001 |
| Formula | CCC | SE | 95% CI | Relative Difference (Bias/LoA) (%) | Absolute Difference (Bias/LoA) (g) |
|---|---|---|---|---|---|
| Siemer formula | 0.55 | 0.10 | 0.35–0.75 | −4.6/−42.0 to 32.8 | −105.9/−949.5 to 737.7 |
| Hadlock formula | 0.44 | 0.11 | 0.23–0.65 | −12.2/−52.8 to 28.4 | −259.4/−1184.6 to 665.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Pontes, K.F.M.; Rolo, L.C.; Callado, G.Y.; Peixoto, A.B.; Sarno, M.; Souza, A.S.R.; Carvalho, F.H.C.; Braga, A.; Araujo Júnior, E. Gestational Age-Specific Biometric and Estimated Fetal Weight Curves in Gastroschisis: A Brazilian Multicenter Cohort Study. Diagnostics 2026, 16, 1402. https://doi.org/10.3390/diagnostics16091402
Pontes KFM, Rolo LC, Callado GY, Peixoto AB, Sarno M, Souza ASR, Carvalho FHC, Braga A, Araujo Júnior E. Gestational Age-Specific Biometric and Estimated Fetal Weight Curves in Gastroschisis: A Brazilian Multicenter Cohort Study. Diagnostics. 2026; 16(9):1402. https://doi.org/10.3390/diagnostics16091402
Chicago/Turabian StylePontes, Karina Felippe Monezi, Liliam Cristine Rolo, Gustavo Yano Callado, Alberto Borges Peixoto, Manoel Sarno, Alex Sandro Rolland Souza, Francisco Herlânio Costa Carvalho, Antonio Braga, and Edward Araujo Júnior. 2026. "Gestational Age-Specific Biometric and Estimated Fetal Weight Curves in Gastroschisis: A Brazilian Multicenter Cohort Study" Diagnostics 16, no. 9: 1402. https://doi.org/10.3390/diagnostics16091402
APA StylePontes, K. F. M., Rolo, L. C., Callado, G. Y., Peixoto, A. B., Sarno, M., Souza, A. S. R., Carvalho, F. H. C., Braga, A., & Araujo Júnior, E. (2026). Gestational Age-Specific Biometric and Estimated Fetal Weight Curves in Gastroschisis: A Brazilian Multicenter Cohort Study. Diagnostics, 16(9), 1402. https://doi.org/10.3390/diagnostics16091402








