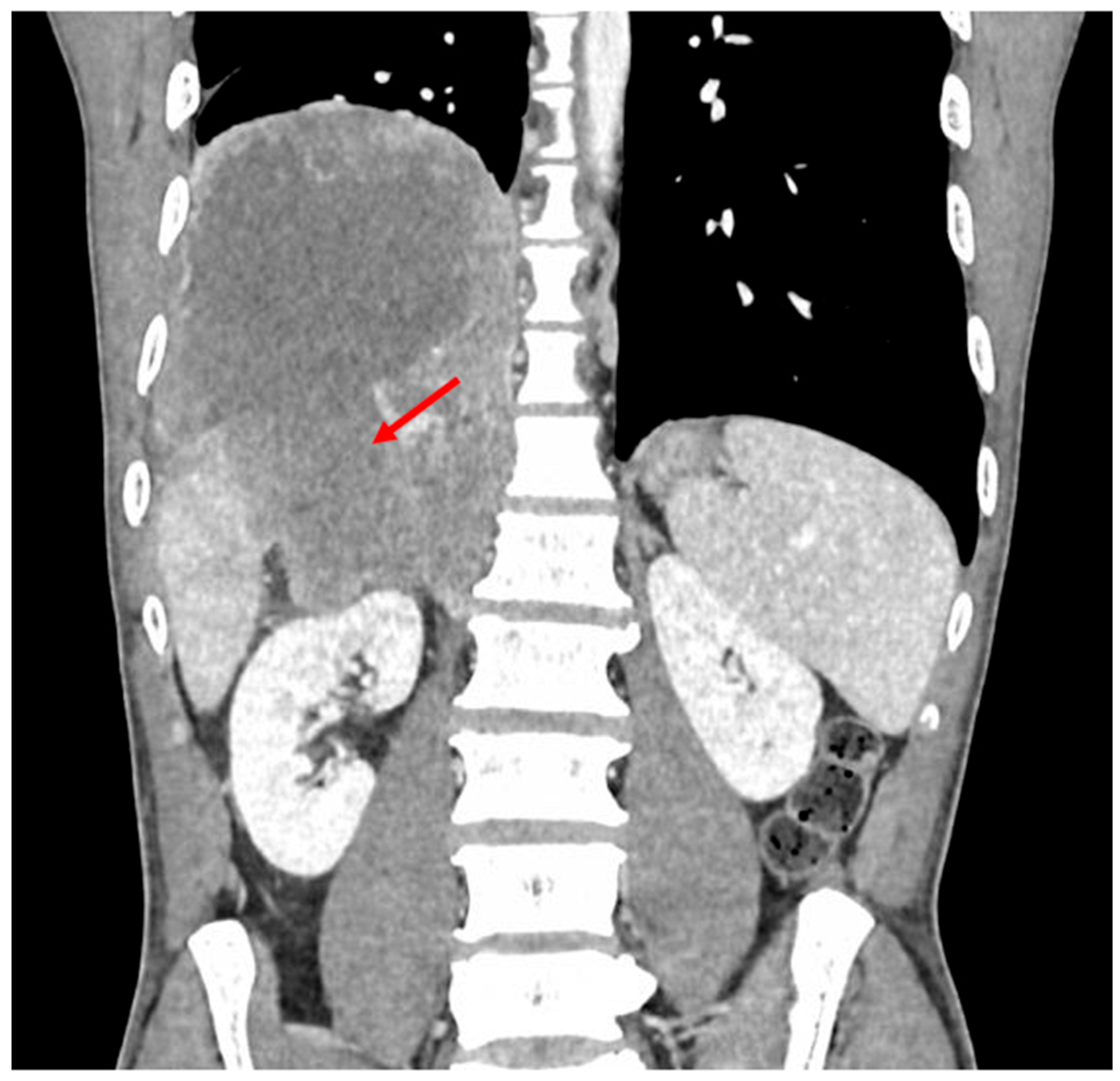
Cerebral Metastasis in a Fatal Adrenocortical Carcinoma: A Rare Presentation of an Aggressive Tumor
Abstract



Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, E.; Dahal, S.; Sharma, P.; Bhandari, A.; Gupta, V.; Amgai, B.; Dahal, S. The characteristics and trends in adrenocortical carcinoma: A United States popu-lation based study. J. Clin. Med. Res. 2018, 10, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, A.; Taieb, A.; Hattab, F.; Ghachem, A.; Slama, N.B.H.; Halloul, I.; Saafi, W.; Marzouk, H.; Elfekih, H.; Saad, G.; et al. Characterization of Unilateral Adrenal Incidentalomas: Hormonal Analysis, Computed Tomography, and Magnetic Resonance Imaging Correlation. Cureus 2025, 17, e100542. [Google Scholar] [CrossRef] [PubMed]
- Lerario, A.M.; Moraitis, A.; Hammer, G.D. Genetics and epigenetics of adrenocortical tumors. Mol. Cell. Endocrinol. 2014, 386, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Gatta-Cherifi, B.; Chabre, O.; Murat, A.; Niccoli, P.; Cardot-Bauters, C.; Rohmer, V.; Young, J.; Delemer, B.; Du Boullay, H.; Verger, M.F.; et al. Adrenal involvement in MEN1. Analysis of 715 cases from the Groupe d’etude des Tumeurs Endocrines database. Eur. J. Endocrinol. 2012, 166, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Thampi, A.; Shah, E.; Elshimy, G.; Correa, R. Adrenocortical carcinoma: A literature review. Transl. Cancer Res. 2020, 9, 1253–1264. [Google Scholar] [CrossRef] [PubMed]
- Shariq, O.A.; McKenzie, T.J. Adrenocortical carcinoma: Current state of the art, ongoing controversies, and future directions in diagnosis and treatment. Ther. Adv. Chronic Dis. 2021, 12, 20406223211033103. [Google Scholar] [CrossRef] [PubMed]
- Fassnacht, M.; Assie, G.; Baudin, E.; Eisenhofer, G.; de la Fouchardiere, C.; Haak, H.; de Krijger, R.; Porpiglia, F.; Terzolo, M.; Berruti, A.; et al. Adrenocortical carcinomas and malignant phaeochromocytomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2020, 31, 1476–1490. [Google Scholar] [CrossRef] [PubMed]
- Fassnacht, M.; Dekkers, O.M.; Else, T.; Baudin, E.; Berruti, A.; de Krijger, R.R.; Haak, H.R.; Mihai, R.; Assie, G.; Terzolo, M. European Society of Endocrinology Clinical Practice Guidelines on the management of adrenocortical carcinoma in adults, in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 2018, 179, G1–G46. [Google Scholar] [CrossRef] [PubMed]
- Chukkalore, D.; MacDougall, K.; Master, V.; Bilen, M.A.; Nazha, B. Adrenocortical Carcinomas: Molecular Patho-genesis, Treatment Options, and Emerging Immunotherapy and Targeted Therapy Approaches. Oncologist 2024, 29, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Szyszka, P.; Grossman, A.B.; Diaz-Cano, S.; Sworczak, K.; Dworakowska, D. Molecular pathways of human adrenocortical carcinoma—Translating cell signalling knowledge into diagnostic and treatment options. Endokrynol. Pol. 2016, 67, 427–450. [Google Scholar] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Taieb, A.; Yanes, A.; Fredj, R.B.; Barkache, M.; Zarrouk, O.; Saafi, W.; Halloul, I.; El Fekih, H.; Lajmi, Z.; Ben Romdhane, Y.; et al. Cerebral Metastasis in a Fatal Adrenocortical Carcinoma: A Rare Presentation of an Aggressive Tumor. Diagnostics 2026, 16, 1143. https://doi.org/10.3390/diagnostics16081143
Taieb A, Yanes A, Fredj RB, Barkache M, Zarrouk O, Saafi W, Halloul I, El Fekih H, Lajmi Z, Ben Romdhane Y, et al. Cerebral Metastasis in a Fatal Adrenocortical Carcinoma: A Rare Presentation of an Aggressive Tumor. Diagnostics. 2026; 16(8):1143. https://doi.org/10.3390/diagnostics16081143
Chicago/Turabian StyleTaieb, Ach, Amira Yanes, Rihab Ben Fredj, Majdouline Barkache, Oumaima Zarrouk, Wiem Saafi, Imen Halloul, Hamza El Fekih, Zeineb Lajmi, Yasmine Ben Romdhane, and et al. 2026. "Cerebral Metastasis in a Fatal Adrenocortical Carcinoma: A Rare Presentation of an Aggressive Tumor" Diagnostics 16, no. 8: 1143. https://doi.org/10.3390/diagnostics16081143
APA StyleTaieb, A., Yanes, A., Fredj, R. B., Barkache, M., Zarrouk, O., Saafi, W., Halloul, I., El Fekih, H., Lajmi, Z., Ben Romdhane, Y., Saad, G., & Hasni, Y. (2026). Cerebral Metastasis in a Fatal Adrenocortical Carcinoma: A Rare Presentation of an Aggressive Tumor. Diagnostics, 16(8), 1143. https://doi.org/10.3390/diagnostics16081143






